HL7 FHIR Compliance Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
The HL7 FHIR Compliance Market is segmented by compliance focus (regulatory compliance and certification support, API conformance and validation testing, security, consent, and privacy compliance), customer type (payers and health insurers, providers and hospital systems, Health IT vendors and digital health companies), offering type (software tools and platforms, implementation and integration services, managed compliance and monitoring services) and region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
HL7 FHIR Compliance Market Forecast and Outlook By FMI
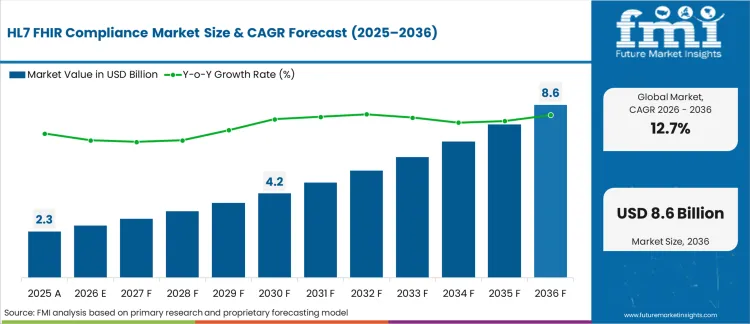
In 2025, the HL7 FHIR compliance market was valued at USD 2.3 billion. Based on Future Market Insights' analysis, demand for HL7 FHIR compliance solutions is estimated to grow to USD 2.6 billion in 2026 and USD 8.6 billion by 2036. FMI projects a CAGR of 12.7% during the forecast period.
Absolute dollar growth of USD 6.0 billion over 2026 to 2036 points to steady conversion of interoperability work from ad hoc integration projects into repeatable compliance programs. Growth holds because payers, providers, and vendors must keep pace with shifting implementation guides, certification pathways, security controls, and conformance testing, yet timelines stretch where legacy interfaces, identity systems, and data governance create long validation cycles.
Summary of HL7 FHIR Compliance Market
- Market Definition:
- HL7 FHIR compliance comprises testing, certification support, security validation, and monitoring used to prove that FHIR APIs and profiles behave as required by published guides.
- Demand Drivers in the Market:
- Federal agencies in the United States are aligning FHIR adoption through a shared action plan, which increases demand for conformance programs and repeatable validation. [1]
- Health systems and payers are shifting from portal-driven exchange to API exchange, raising the cost of poor conformance because failures appear as workflow downtime and audit risk.
- National digital health rails in India expand the base of ABDM-enabled software, which increases the number of products needing implementation guide compliance work. [10]
- Key Segments Analyzed in the FMI Report:
- Compliance focus: Regulatory compliance and certification support leads with 41% share in 2026 because procurement often requires proof of conformance and documented sign-off.
- Customer type: Providers and hospital systems lead with 40% share in 2026 because they carry integration workload across many endpoints and must pass supplier assurance.
- Offering type: Implementation and integration services lead with 45% share in 2026 as buyers pay for mapping, testing, remediation, and deployment governance.
- Analyst Opinion at FMI:
- Analysts at Future Market Insights note that this report helps executives connect implementation guide change cycles to operating cost and plan the testing, certification, and monitoring capacity needed to stay compliant.
- Strategic Implications / Executive Takeaways:
- Budget for ongoing re-validation, not one-time certification, since implementation guides and security control expectations keep changing after go-live.
- Separate API conformance testing from workflow testing so failures can be traced to profile rules, auth scopes, or client behavior without long triage loops.
- Use automated monitoring on production endpoints to detect drift in profiles, search parameters, and security controls before it becomes a regulatory or contract issue.
- Methodology:
- Forecast sizing combines adoption timelines with observed spend patterns for testing, certification support, and remediation work.
- Country growth rates reflect public program milestones and implementation guide activity rather than generic digitization signals.
- Validation uses public guidance, implementation guide metadata, and documented supplier requirements to triangulate segment shares.
As Ewout Kramer, CTO of Firely and co-founder of the FHIR standard, noted regarding the practical challenges of implementation guide compliance, "We have a lot of implementation guides, but the real question is how many are compatible, or even implementable? Even native FHIR servers like ours need to perform mapping between FHIR data transferred using different versions of IGs to maintain compatibility." [11]
India leads the growth set at 15.2% CAGR as ABDM-aligned implementation guidance and exchange rails push more products into conformance work. The United States follows at 13.6% CAGR as federal coordination and agency-led FHIR programs drive more testing, security, and certification demand. Germany posts 12.9% CAGR as gematik-linked implementation guides widen the scope of structured validation. The United Kingdom tracks 12.6% CAGR as NHS API standards and FHIR-aligned integration expectations expand supplier obligations. Canada rises at 12.1% CAGR as pan-Canadian FHIR implementation activity increases conformance testing and ongoing monitoring.
| Metric | Value |
|---|---|
| Market Size (2026) | USD 2.6 Bn |
| Forecast Value (2036) | USD 8.6 Bn |
| CAGR (2026 to 2036) | 12.7% |
HL7 FHIR Compliance Market Definition
HL7 FHIR compliance covers the tools and services used to prove that health data systems follow FHIR specifications and the local implementation guides that govern real-world exchange. Buyers use these offerings to validate API behavior, confirm resource profiles, test conformance against published guides, and document security, consent, and privacy controls required for regulated data sharing. The market includes certification support, conformance testing platforms, security and access control validation, and implementation work that brings payer, provider, and vendor systems into verifiable alignment with FHIR exchange requirements.
Market Inclusions
This report covers global and regional market sizing for 2025, 2026, and 2036 with a 2026 to 2036 forecast. It provides segmentation by compliance focus, customer type, and offering type, with country growth profiling for priority markets. Coverage includes conformance testing, certification pathways, implementation guide alignment, security and privacy validation for FHIR APIs, and recurring monitoring requirements that arise after go-live.
Market Exclusions
The scope excludes general system integration that does not involve FHIR conformance, profile validation, certification activity, or API compliance testing. It also excludes core EHR licensing, claims platform replacement, and analytics tooling unless the work directly supports FHIR compliance, validation, certification, or ongoing conformance monitoring tied to published FHIR specifications or implementation guides.
HL7 FHIR Compliance Market Research Methodology
- Primary Research: FMI conducted interviews with payer interoperability leads, hospital integration directors, digital health vendor product owners, testing-lab specialists, and implementation partners responsible for conformance and security sign-off.
- Desk Research: Inputs included HL7 publications, implementation guide repositories, government interoperability programs, NHS API and standards pages, gematik implementation guide metadata, ABDM implementation guidance, and vendor technical documentation for FHIR services and controls.
- Market-sizing and Forecasting: A hybrid model combined bottom-up demand for testing, certification, and implementation work with top-down adoption timelines linked to program, policy, and standards cycles.
- Data Validation and Update Cycle: Forecasts were checked against implementation guide version cadence, program milestone updates, and documented changes to security and compliance control expectations, then refreshed when major public guidance changes.
Segmental Analysis
HL7 FHIR Compliance Market Analysis by Compliance Focus
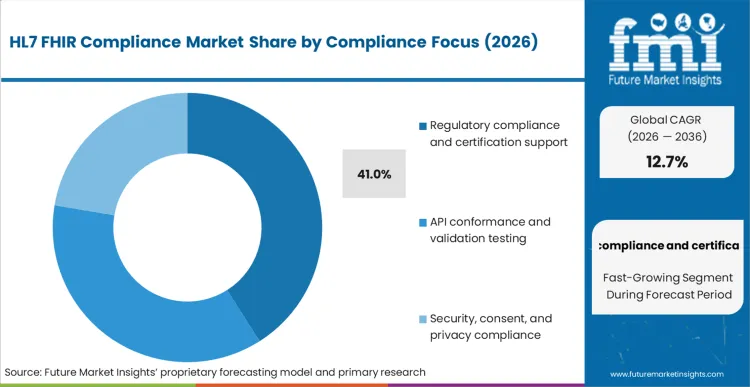
Based on FMI's HL7 FHIR compliance market report, consumption of regulatory compliance and certification support is estimated to hold 41% share in 2026. This sub-segment leads because many buyers treat certification, audit readiness, and documented conformance as a procurement gate, not an optional quality activity, especially when exchanging regulated health data across organizations.
- Certification pathways: Drummond Group positions FHIR certification programs for payer APIs and client applications as a way to prove conformance in line with mandate roadmaps, which keeps demand high for certification preparation and testing. [5]
- Program-driven conformance: The United States Draft Federal FHIR Action Plan is designed to guide consistent agency use of FHIR, which increases the number of systems that must pass repeatable conformance checks. [1]
- Adoption expectations: Firely's State of FHIR survey results highlight broad expectations of rising FHIR adoption, with a significant majority of respondents anticipating increased adoption, which supports continued spending on compliance tooling as endpoint counts grow. [6]
HL7 FHIR Compliance Market Analysis by Customer Type
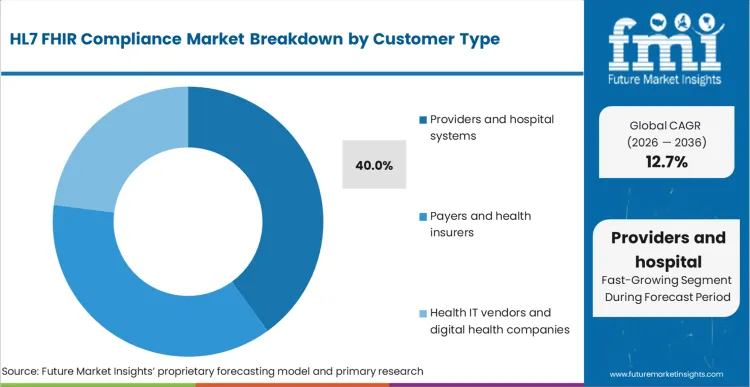
Based on FMI's HL7 FHIR compliance market report, consumption by providers and hospital systems is estimated to hold 40% share in 2026. Providers lead because they must integrate across multiple payers, national services, and vendor endpoints, and they carry operational risk when conformance issues block workflows such as referrals, medication processes, and record exchange.
- Supplier assurance: NHS England maintains an API catalogue and standards ecosystem that shapes how suppliers integrate, increasing compliance work for provider-facing deployments. [8]
- Security and access controls: Microsoft documents compliance boundaries and security controls for FHIR services in Azure, which reflects the control-layer buyers expect when deploying FHIR endpoints that handle PHI. [4]
- Multi-endpoint burden: Large hospital groups often manage many inbound and outbound connections, which increases recurring test cycles whenever implementation guides or authentication practices change.
HL7 FHIR Compliance Market Analysis by Offering Type
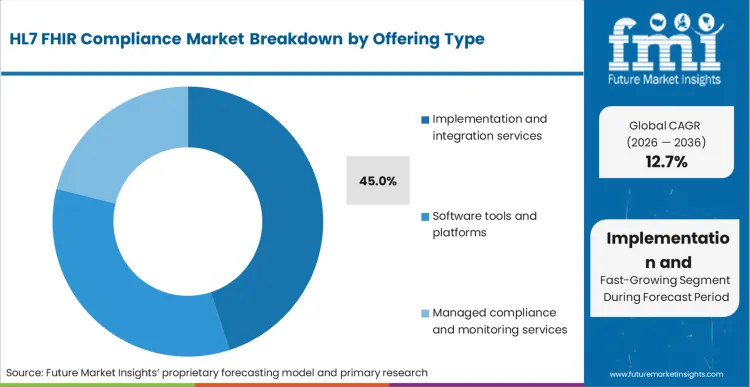
Based on FMI's HL7 FHIR compliance market report, consumption of implementation and integration services is estimated to hold 45% share in 2026. Services lead because buyers pay for the practical work of mapping data to profiles, remediating gaps, validating endpoints, and coordinating go-live governance across multiple parties.
- Managed FHIR endpoints: Microsoft describes the FHIR service as a managed, standards-based API for clinical data, which increases downstream services demand for integration, profile alignment, and operational readiness. [3]
- IG-linked validation: gematik publishes implementation guide metadata for ePA medication services, which illustrates the kind of versioned guidance that implementers must track and validate against. [9]
- Operational monitoring: Compliance work persists after launch because drift in profiles, scopes, and client behavior requires monitoring and remediation cycles, not just initial testing.
HL7 FHIR Compliance Market Drivers, Restraints, Opportunities
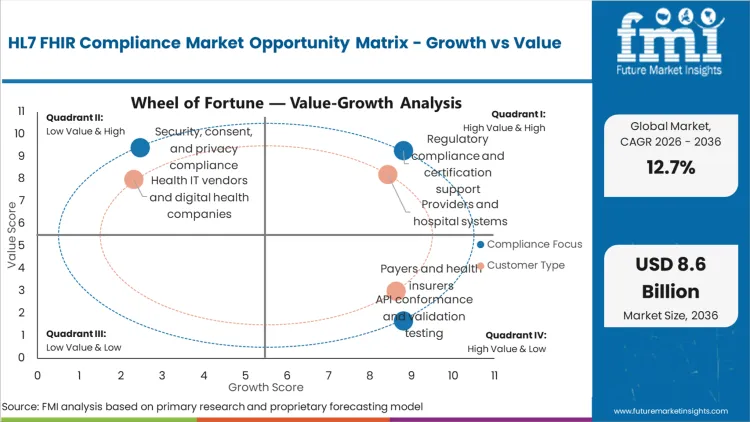
Future Market Insights analysis finds that FHIR compliance spending rises when interoperability moves from pilot exchange to scaled exchange, because scale exposes conformance gaps quickly. The market sits at its current size because payers, hospitals, and vendors must keep systems aligned with evolving implementation guides, security expectations, and testing requirements, while avoiding downtime, audit risk, and contract disputes tied to non-conforming endpoints.
FMI analysts observe a shift from integration work that ends at go-live to a model where compliance is treated as an operating function. Demand grows where public programs, national API standards, and implementation guide ecosystems create a consistent basis for testing. Demand is slower where endpoints are fragmented across legacy systems and where governance slows profile adoption, even when budgets exist, because validation and clinical safety assurance take time.
- Government alignment pressure: The 2024 Draft Federal FHIR Action Plan is intended to strengthen consistent agency use of FHIR, which increases compliance workload across systems that touch federal exchange programs. [1]
- National standards ecosystems: NHS England's interoperability and API standards direction supports FHIR-aligned integration, increasing supplier obligations for conformance and assurance. [7]
- India scale-up dynamic: India's ABDM program shows rapid growth in registered entities and ABDM-enabled software usage, which expands the pool of products requiring implementation guide compliance. [10]
Regional Analysis
North America, Europe, and the Asia Pacific are assessed as the core regions for HL7 FHIR compliance demand, with country performance shaped by government coordination, implementation guide ecosystems, and buyer assurance requirements. The full report offers market attractiveness analysis by region and country.
.webp)
| Country | CAGR (2026-2036) |
|---|---|
| India | 15.2% |
| United States | 13.6% |
| Germany | 12.9% |
| United Kingdom | 12.6% |
| Canada | 12.1% |
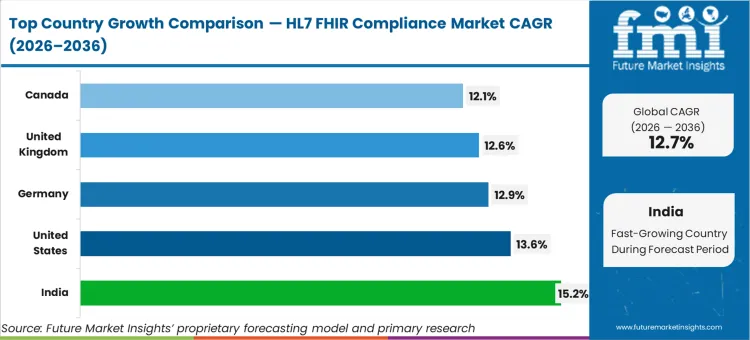
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Market Analysis
North America acts as the policy-and-procurement engine, where public coordination and large buyer requirements push repeatable testing, certification readiness, and security validation. Epic Systems and Oracle Health influence provider-side implementation patterns through platform capabilities, while Microsoft and InterSystems shape managed data platform approaches that still require conformance and control-layer validation.
- United States: The HL7 FHIR compliance market in the United States is projected to grow at a CAGR of 13.6% through 2036. Growth is linked to federal coordination that increases the scope of systems expected to implement FHIR consistently. In September 2024, the Office of the National Coordinator published an introduction to the 2024 Draft Federal FHIR Action Plan, describing its goal to strengthen consistent agency use of FHIR across federal health IT. [1] This creates recurring compliance work because systems must align to shared capabilities and reuse patterns rather than one-off exchanges. Microsoft documents regulatory compliance controls and security control expectations for Azure Health Data Services FHIR, which reflects the control categories buyers must map to their own risk and assurance requirements. [4]
- Canada: The HL7 FHIR compliance market in Canada is expected to grow at a CAGR of 12.1% from 2026 to 2036. Growth is tied to steady expansion of FHIR implementation activity across jurisdictions and national collaboration on profiles and exchange patterns. Canada's demand is shaped by procurement expectations from health authorities and large provider networks, where suppliers must show that APIs conform to published guidance and that security controls meet regional privacy and consent expectations.
FMI's analysis of HL7 FHIR compliance market in North America consists of country-wise assessment that includes the United States and Canada. Readers can find program-led adoption signals, assurance requirements, and buyer testing priorities tied to procurement and operational risk.
Europe Market Analysis
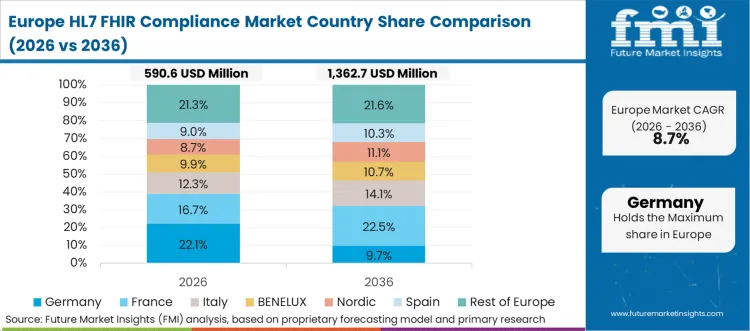
Europe acts as the implementation-guide governance engine, where national bodies publish guidance that shapes conformance expectations and increases the scope of structured validation. Oracle Health, InterSystems, and regional integration specialists influence program delivery, while testing platforms and tooling vendors support repeatable validation against country-linked guides.
- United Kingdom: The HL7 FHIR compliance market for the United Kingdom is expected to grow at a CAGR of 12.6% from 2026 to 2036. Growth is linked to NHS-driven API and standards infrastructure that shapes how suppliers build and test integrations. NHS England's API catalogue lists APIs and standards available or in development, which increases the number of supplier endpoints that must meet published requirements and pass assurance checks. [8] Compliance work rises because suppliers must keep pace with standards direction and handle versioned updates without breaking dependent workflows.
- Germany: The HL7 FHIR compliance market in Germany is projected to grow at a CAGR of 12.9% from 2026 to 2036. Growth is supported by the presence of published, versioned implementation guides linked to national infrastructure programs. For example, implementation guide metadata for gematik ePA medication services shows an active versioned guide with a published date and lifecycle status, illustrating the structured guidance that implementers must validate against. [9] Compliance activity grows because systems must align to national profiles, validate resource constraints, and document behavior under certification and assurance regimes.
FMI's analysis of HL7 FHIR compliance market in Europe consists of country-wise assessment that includes the United Kingdom and Germany. Readers can find country-level guide ecosystems, assurance constraints, and the compliance focus areas that attract the most spend.
Asia Pacific Market Analysis
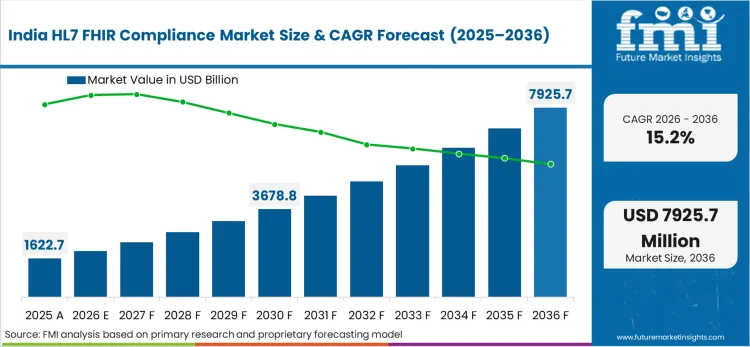
Asia Pacific acts as the scale-up engine, where national digital health programs and rapid growth in connected entities widen the base of systems needing implementation guide compliance, security validation, and ongoing monitoring.
- India: The HL7 FHIR compliance market in India is projected to grow at a CAGR of 15.2% from 2026 to 2036. Growth is linked to ABDM's push to standardize exchange and expand the installed base of ABDM-enabled software. The ABDM Health Data Interchange implementation guide describes minimum conformance requirements based on FHIR R4 for ABDM health data interchange specifications, which increases the need for formal compliance work among vendors and implementers. [10] The expansion of registered facilities and ABHA creation drives growing compliance workload as products connect into exchange rails.
FMI's analysis of HL7 FHIR compliance market in Asia Pacific consists of country-wise assessment that includes India. Readers can find adoption pacing signals, implementation guide implications, and the compliance workload drivers tied to scale and endpoint growth.
Competitive Aligners for Market Players
Competition in HL7 FHIR compliance is shaped by testing credibility, implementation guide coverage, and the ability to keep pace with versioned guidance. The market is moderately concentrated among interoperability platforms, managed FHIR data services, testing and certification ecosystems, and specialist vendors focused on conformance tooling. Competitive variables center on the breadth of supported profiles, the strength of validation engines, security and consent controls, and the ability to automate regression testing across frequent changes.
Structural advantages accrue to firms that sit close to production deployments and can observe real-world failure modes, since they can translate those patterns into better validators, profilers, and monitoring rules. Managed platform providers benefit from existing enterprise security and operational tooling that can be adapted to healthcare compliance controls, while interoperability platforms and EHR vendors benefit from embedded workflow and data model access that reduces mapping effort.
Buyer behavior reflects risk management and assurance. Large payers and hospital systems run multi-vendor stacks and avoid dependency by separating tooling from implementation services. Procurement teams prefer evidence of conformance and repeatable validation, since failures appear as disrupted workflows and audit exposure. Pricing power tends to sit with vendors that reduce time-to-approval in assurance processes, shorten remediation cycles, and offer scalable monitoring that prevents drift after go-live.
Recent Developments:
- In June 2025, Microsoft published Azure Policy regulatory compliance controls documentation for the FHIR service in Azure Health Data Services, listing compliance domains and security controls used to support standards alignment. [4]
- InterSystems has developed solutions such as the InterSystems Payer Connector to integrate with platforms like the Epic Payer Platform, supporting health plans with governed integration hubs.
Key Players in the HL7 FHIR Compliance Market
- Epic Systems
- Oracle Health
- InterSystems
- Microsoft (Azure Health Data Services FHIR)
- Redox
- Smile Digital Health (HAPI FHIR)
- Firely (Firely Server)
- AEGIS.net (Touchstone)
- Drummond Group (FHIRplace testing & certification)
- Health Gorilla
Scope of the Report
| Item | Value |
|---|---|
| Quantitative Units | USD 2.6 billion (2026) to USD 8.6 billion (2036), at a CAGR of 12.7% |
| Market Definition | The HL7 FHIR compliance market comprises software tools, testing platforms, certification support, and services used to validate FHIR API conformance, adherence to implementation guides, and security, consent, and privacy compliance for regulated health data exchange. |
| Compliance Focus Segmentation | Regulatory compliance and certification support, API conformance and validation testing, Security, consent, and privacy compliance |
| Customer Type Segmentation | Payers and health insurers, Providers and hospital systems, Health IT vendors and digital health companies |
| Offering Type Segmentation | Software tools and platforms, Implementation and integration services, Managed compliance and monitoring services |
| Regions Covered | North America, Europe, Asia Pacific |
| Countries Covered | India, United States, Germany, United Kingdom, Canada |
| Key Companies Profiled | Epic Systems, Oracle Health, InterSystems, Microsoft (Azure Health Data Services FHIR), Redox, Smile Digital Health (HAPI FHIR), Firely (Firely Server), AEGIS.net (Touchstone), Drummond Group (FHIRplace testing & certification), Health Gorilla |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up market modeling anchored on implementation guide cadence and public program milestones, validated through primary interviews with payers, provider integration teams, and compliance testing specialists. |
HL7 FHIR Compliance Market Analysis by Segments
By Compliance Focus:
- Regulatory compliance and certification support
- API conformance and validation testing
- Security, consent, and privacy compliance
By Customer Type:
- Payers and health insurers
- Providers and hospital systems
- Health IT vendors and digital health companies
By Offering Type:
- Software tools and platforms
- Implementation and integration services
- Managed compliance and monitoring services
By Region:
- North America
- Europe
- Asia Pacific
Bibliography
- [1] Office of the National Coordinator for Health IT (ONC). (2024). Introducing the 2024 Draft Federal FHIR Action Plan. Health IT Buzz Blog, September 23, 2024.
- [2] HL7 International. (2024). HL7 Civitas 2024 Conference. HL7 International Events, October 2024.
- [3] Microsoft. (2024). What is the FHIR service in Azure Health Data Services? Microsoft Learn Documentation, November 2024.
- [4] Microsoft. (2024). Azure Policy Regulatory Compliance controls for FHIR service in Azure Health Data Services. Microsoft Learn Documentation, June 2025.
- [5] Drummond Group LLC. (2025). Payer and Patient Access FHIR Certification Program. Drummond Group FHIRplace Services.
- [6] Firely. (2024). 8 Key Insights from the 2024 State of FHIR Survey. Firely Blog, July 2024.
- [7] NHS England. (2025). Interoperability (including FHIR UK Core reference). NHS England Long Read.
- [8] NHS England Digital. (2026). API and integration catalogue. NHS England Developer Portal.
- [9] gematik GmbH. (2025). Implementation Guide ePA Medication Service. ePA FHIR Implementation Guide, Version 1.1.5, May 2025.
- [10] National Resource Centre for EHR Standards (NRCeS). (2024). FHIR Implementation Guide for ABDM Health Data Interchange Specifications 1.0. Government of India, Ministry of Health and Family Welfare.
- [11] Firely. (2024). FHIR DevDays 2024 Highlights. Firely Blog, July 1, 2024.
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
This Report Addresses
- Market sizing for HL7 FHIR compliance from 2026 to 2036 with 2026 baseline and 2036 forecast aligned to master values.
- Growth opportunity mapping by compliance focus, customer type, and offering type with leading sub-segment share benchmarks.
- Country-level CAGR profiling for priority markets to support investment planning and regional go-to-market sequencing.
- Competitive assessment across testing platforms, managed FHIR services, interoperability platforms, and certification ecosystems.
- Coverage of implementation guide cadence, validation needs, and operational monitoring requirements that drive recurring compliance spend.
- Security, consent, and privacy compliance analysis for regulated exchange, including control-layer considerations for production endpoints.
- Regional analysis linking compliance demand to public programs, standards ecosystems, and buyer assurance requirements.
- Deliverables packaged for strategy, product, and implementation teams across narrative report and table-ready outputs.
Frequently Asked Questions
How large is the HL7 FHIR compliance market in 2026?
It is USD 2.6 billion in 2026, reflecting demand for conformance testing, certification readiness, and security validation across deployed FHIR endpoints.
What will the market size be by 2036?
It is projected to reach USD 8.6 billion by 2036 as compliance becomes an operating function for API exchange and implementation guide updates.
What CAGR is expected from 2026 to 2036?
FMI projects a 12.7% CAGR for 2026 to 2036, driven by rising endpoint counts and recurring validation cycles tied to evolving guidance.
How much absolute dollar growth is expected over the forecast period?
Absolute growth is USD 6.0 billion from 2026 to 2036, as repeatable conformance programs replace one-time integration validation approaches.
Which compliance focus segment leads the market in 2026?
Regulatory compliance and certification support leads with 41% share in 2026 because many buyers require documented conformance and sign-off for procurement.
Which customer type leads the market in 2026?
Providers and hospital systems lead with 40% share in 2026 because they manage multi-endpoint integration risk and must pass supplier assurance processes.
Why is India the fastest-growing market in this set?
India grows at 15.2% CAGR as ABDM-linked implementation guidance and expanding connected software bases increase the number of systems needing conformance work.
What drives growth in the United States market?
The United States grows at 13.6% CAGR as federal coordination and agency-led FHIR programs increase the scope of endpoints that require repeatable compliance.
What explains Germany's growth profile in FHIR compliance?
Germany grows at 12.9% CAGR as versioned implementation guides increase validation needs, while governance and certification cycles extend remediation timelines.
Why does the United Kingdom track close to the global growth rate?
The United Kingdom grows at 12.6% CAGR as NHS API standards expand supplier obligations, but change control and assurance processes moderate ramp speed.
What supports Canada's steady growth in compliance services?
Canada grows at 12.1% CAGR as jurisdiction participation increases implementation activity, raising ongoing test and monitoring needs across more endpoints.
What is the main restraint that slows compliance programs?
Legacy interfaces and fragmented identity systems increase exception handling, making conformance remediation slower and raising the cost of repeated regression testing.
How do buyers measure ROI in FHIR compliance investments?
ROI is tracked through fewer integration incidents, faster onboarding cycles, reduced failed transactions, and lower remediation effort after implementation guide updates.
Why does API conformance testing remain a recurring spend line?
API behavior can drift with upgrades, profile changes, and client differences, so buyers keep regression suites to prevent workflow outages and audit exposure.
How significant is security, consent, and privacy compliance in purchasing decisions?
It is a gating requirement because regulated exchange needs access control, auditability, and privacy compliance evidence before endpoints can be approved for use.
What role do certification programs play in vendor selection?
Certification reduces buyer risk by providing documented proof of conformance, which shortens procurement review cycles and increases trust in supplier endpoint behavior.
Where do implementation and integration services create the most value?
They create value in data mapping to profiles, closing conformance gaps, validating auth scopes, and coordinating multi-party testing for production readiness.
How should executives plan for implementation guide change cycles?
Executives should fund ongoing re-validation capacity, automate regression tests, and maintain monitoring to detect profile drift before it impacts operations.
What should buyers prioritize when selecting compliance tooling?
Buyers should prioritize implementation guide coverage, automated validation depth, audit-ready reporting, security control support, and proven ability to scale testing.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Compliance Focus
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Compliance Focus , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Compliance Focus , 2026 to 2036
- Regulatory compliance and certification support
- API conformance and validation testing
- Security, consent, and privacy compliance
- Regulatory compliance and certification support
- Y-o-Y Growth Trend Analysis By Compliance Focus , 2021 to 2025
- Absolute $ Opportunity Analysis By Compliance Focus , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Customer Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Customer Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Customer Type, 2026 to 2036
- Providers and hospital systems
- Payers and health insurers
- Health IT vendors and digital health companies
- Providers and hospital systems
- Y-o-Y Growth Trend Analysis By Customer Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Customer Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Offering Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Offering Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Offering Type, 2026 to 2036
- Implementation and integration services
- Software tools and platforms
- Managed compliance and monitoring services
- Implementation and integration services
- Y-o-Y Growth Trend Analysis By Offering Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Offering Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Compliance Focus
- By Customer Type
- By Offering Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Compliance Focus
- By Customer Type
- By Offering Type
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Compliance Focus
- By Customer Type
- By Offering Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Compliance Focus
- By Customer Type
- By Offering Type
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Compliance Focus
- By Customer Type
- By Offering Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Compliance Focus
- By Customer Type
- By Offering Type
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Compliance Focus
- By Customer Type
- By Offering Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Compliance Focus
- By Customer Type
- By Offering Type
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Compliance Focus
- By Customer Type
- By Offering Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Compliance Focus
- By Customer Type
- By Offering Type
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Compliance Focus
- By Customer Type
- By Offering Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Compliance Focus
- By Customer Type
- By Offering Type
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Compliance Focus
- By Customer Type
- By Offering Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Compliance Focus
- By Customer Type
- By Offering Type
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Compliance Focus
- By Customer Type
- By Offering Type
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Compliance Focus
- By Customer Type
- By Offering Type
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Compliance Focus
- By Customer Type
- By Offering Type
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Compliance Focus
- By Customer Type
- By Offering Type
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Compliance Focus
- By Customer Type
- By Offering Type
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Compliance Focus
- By Customer Type
- By Offering Type
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Compliance Focus
- By Customer Type
- By Offering Type
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Compliance Focus
- By Customer Type
- By Offering Type
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Compliance Focus
- By Customer Type
- By Offering Type
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Compliance Focus
- By Customer Type
- By Offering Type
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Compliance Focus
- By Customer Type
- By Offering Type
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Compliance Focus
- By Customer Type
- By Offering Type
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Compliance Focus
- By Customer Type
- By Offering Type
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Compliance Focus
- By Customer Type
- By Offering Type
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Compliance Focus
- By Customer Type
- By Offering Type
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Compliance Focus
- By Customer Type
- By Offering Type
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Compliance Focus
- By Customer Type
- By Offering Type
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Compliance Focus
- By Customer Type
- By Offering Type
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Compliance Focus
- By Customer Type
- By Offering Type
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Compliance Focus
- By Customer Type
- By Offering Type
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Compliance Focus
- By Customer Type
- By Offering Type
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Compliance Focus
- By Customer Type
- By Offering Type
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Compliance Focus
- By Customer Type
- By Offering Type
- Competition Analysis
- Competition Deep Dive
- Epic Systems
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Oracle Health
- InterSystems
- Microsoft (Azure Health Data Services FHIR)
- Redox
- Smile Digital Health (HAPI FHIR)
- Firely (Firely Server)
- AEGIS.net (Touchstone)
- Drummond Group (FHIRplace testing & certification)
- Health Gorilla
- Epic Systems
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Compliance Focus , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Offering Type, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Compliance Focus , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Offering Type, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Compliance Focus , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Offering Type, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Compliance Focus , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Offering Type, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Compliance Focus , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by Offering Type, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Compliance Focus , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by Offering Type, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Compliance Focus , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by Offering Type, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Compliance Focus , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by Offering Type, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Compliance Focus , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Compliance Focus , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Compliance Focus
- Figure 6: Global Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Customer Type
- Figure 9: Global Market Value Share and BPS Analysis by Offering Type, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Offering Type, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Offering Type
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Compliance Focus , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Compliance Focus , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Compliance Focus
- Figure 26: North America Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Customer Type
- Figure 29: North America Market Value Share and BPS Analysis by Offering Type, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Offering Type, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Offering Type
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Compliance Focus , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Compliance Focus , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Compliance Focus
- Figure 36: Latin America Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Customer Type
- Figure 39: Latin America Market Value Share and BPS Analysis by Offering Type, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Offering Type, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Offering Type
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Compliance Focus , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Compliance Focus , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Compliance Focus
- Figure 46: Western Europe Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Customer Type
- Figure 49: Western Europe Market Value Share and BPS Analysis by Offering Type, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by Offering Type, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by Offering Type
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Compliance Focus , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Compliance Focus , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Compliance Focus
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Customer Type
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by Offering Type, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by Offering Type, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by Offering Type
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Compliance Focus , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Compliance Focus , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Compliance Focus
- Figure 66: East Asia Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Customer Type
- Figure 69: East Asia Market Value Share and BPS Analysis by Offering Type, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by Offering Type, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by Offering Type
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Compliance Focus , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Compliance Focus , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Compliance Focus
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Customer Type
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by Offering Type, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by Offering Type, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by Offering Type
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Compliance Focus , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Compliance Focus , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Compliance Focus
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Customer Type
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by Offering Type, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by Offering Type, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by Offering Type
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

