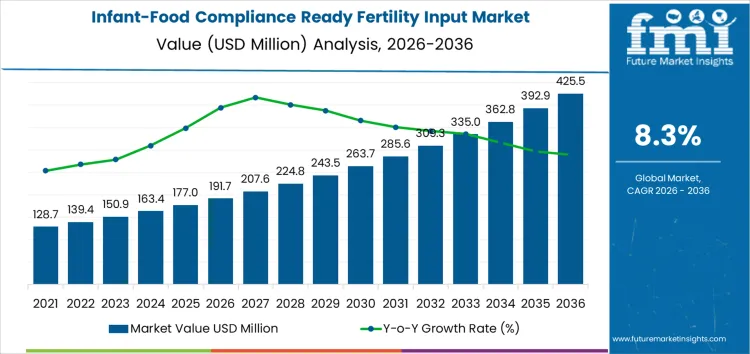
The infant-food compliance ready fertility input market is valued at USD 191.7 million in 2026 and is projected to reach USD 425.5 million by 2036, registering a CAGR of 8.3%. Performance across this period is governed by regulatory exposure rather than agricultural scale expansion. Fertility inputs used upstream in infant nutrition supply chains operate under materially tighter contaminant thresholds, where a single non-compliant batch can disqualify finished ingredients from high-value channels. Cost structures reflect this risk profile. Value concentrates around suppliers able to control impurity levels, document sourcing, and sustain repeat audit performance, allowing them to command stable pricing even when overall fertilizer pricing remains volatile. Margin capture aligns with compliance assurance and documentation depth rather than volume throughput.
Buyer behavior reinforces structural stickiness. Infant food manufacturers and ingredient processors display high risk aversion, favoring long-term input relationships once fertility programs demonstrate consistent control of heavy metals, nitrates, and trace residues. Switching suppliers introduces exposure to requalification delays, certification resets, and brand liability, which discourages procurement churn. Capital allocation therefore favors purification capability, batch testing, and traceability systems rather than capacity expansion. Demand quality remains strongest where infant nutrition brands impose upstream accountability on growers, linking fertilizer selection directly to contract eligibility. Growth remains measured and selective, anchored in regulated supply chains where compliance reliability determines access, continuity, and long-term revenue visibility.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 191.7 million |
| Market Forecast Value (2036) | USD 425.5 million |
| Forecast CAGR (2026-2036) | 8.3% |
Demand for infant-food compliance ready fertility inputs is growing globally due to increasing regulatory scrutiny and stakeholder expectations around micronutrient safety, contaminant limits, and traceability in crops destined for baby food supply chains. Governments and food safety authorities impose strict maximum levels for heavy metals, nitrates, and other agricultural residues in ingredients used for infant nutrition, which motivates growers to adopt fertility inputs formulated and documented to support compliance with infant food standards. Cereals, vegetables, and pulses targeted for baby food formulations require nutrient programs that deliver balanced growth while minimizing uptake of undesirable elements. Growers integrate compliance ready fertility inputs that are tested for low impurity profiles and designed to improve soil nutrient balance without contributing risk factors linked to infant exposure concerns.
Expansion of infant nutrition markets worldwide increases demand for reliable supply of compliant raw materials, which strengthens adoption of documented fertility solutions with robust traceability. Regulatory frameworks in multiple regions emphasize ingredient specification, documentation, and quality management, which reinforces use of fertility inputs aligned with restricted substance lists and safety criteria. Advances in formulation technology improve nutrient availability, reduce leaching, and support consistent crop performance under varied soil and climatic conditions. Growers and supply chain partners prioritize fertility programs that reduce compliance risk, support certification pathways, and deliver predictable outcomes in infant food feedstock production systems.
The global infant-food compliance ready fertility input market is defined by strict safety thresholds, traceability requirements, and nutritional consistency standards imposed across infant and early-life nutrition value chains. Fertility inputs used upstream in crop and dairy systems supplying infant food manufacturers must meet ultra-low contaminant limits, controlled residue profiles, and predictable nutrient bioavailability outcomes. Demand is shaped less by volume optimization and more by regulatory alignment, audit readiness, and compatibility with infant nutrition formulation standards. Adoption is concentrated among vertically integrated food producers, ingredient suppliers, and contract manufacturers supplying infant formula, baby food, and early-stage nutritional products across regulated global markets.
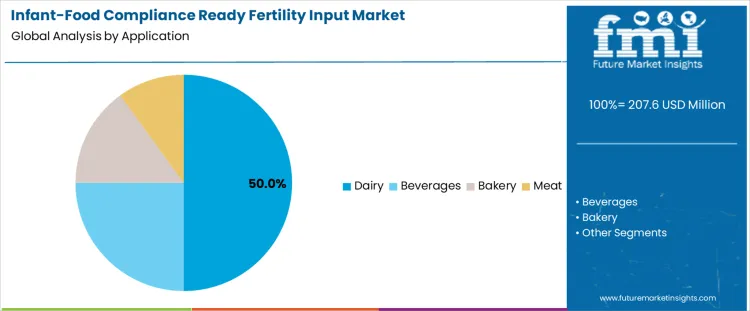
Dairy applications account for the largest share at 50.0%, reflecting the central role of milk-derived ingredients in infant formula and early nutrition products. Fertility inputs supporting dairy feed and forage systems are selected for their ability to influence protein quality, mineral balance, and contaminant control in milk outputs. Beverage applications represent 25.0%, driven by cereal-based infant drinks, fortified liquids, and reconstitution-ready nutrition formats requiring compliant upstream inputs. Bakery applications hold 15.0%, primarily supporting infant cereals, rusks, and soft-baked products where grain quality and nutrient consistency are critical. Meat-related applications account for 10.0%, focused on specialized infant protein preparations and blended nutrition products.
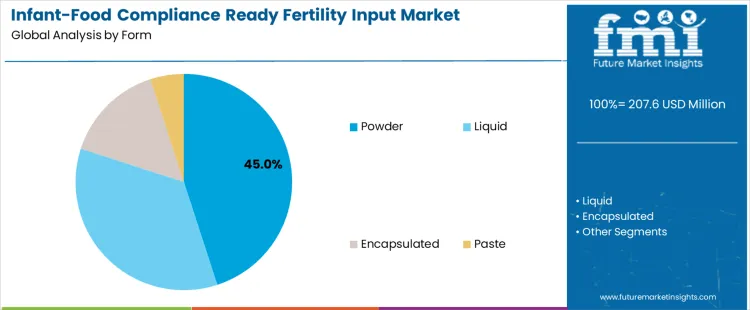
Powdered fertility inputs dominate the market with a 45.0% share, as they offer superior stability, dosing precision, and contamination control during storage and transport. Powder formats align well with audited supply chains that require batch traceability and minimal microbial risk. Liquid formulations account for 35.0%, favored where rapid nutrient uptake and uniform application are required, particularly in controlled agricultural systems supplying infant-grade raw materials. Encapsulated formats represent 15.0%, used to protect sensitive nutrients and enable controlled release while minimizing interaction with soil or feed matrices. Paste formulations account for 5.0%, remaining limited to specialized applications due to handling and shelf-life constraints.
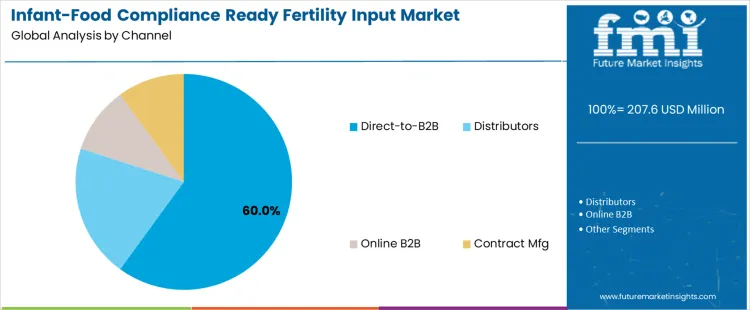
Direct-to-B2B channels dominate global distribution with a 60.0% share, reflecting the preference of infant food producers for tightly controlled procurement relationships. Direct sourcing enables alignment on compliance documentation, formulation customization, and long-term quality assurance protocols. Distributors account for 20.0%, supporting regional infant food suppliers and smaller manufacturers requiring localized regulatory expertise. Online B2B platforms represent 10.0%, primarily facilitating standardized replenishment orders and documentation-driven transactions. Contract manufacturing also accounts for 10.0%, enabling proprietary fertility input development aligned with brand-specific infant nutrition standards.
Global demand rises as agricultural input suppliers and cereal, vegetable, and pulse growers adopt fertility products tailored to meet stringent contaminant limits and nutrient standards required for crops destined for infant food production. Infant-food compliance ready fertility inputs are formulated to minimize heavy metals, ensure balanced macro- and micronutrient availability, and support traceability from field to finished product. Adoption aligns with rising regulatory requirements for infant food safety, quality assurance protocols by manufacturers, and growing consumer expectation for transparent ingredient sourcing across North America, Europe, Asia-Pacific, and other major agricultural regions.
How do regulatory frameworks and food safety standards shape adoption?
Infant food processors enforce rigorous compliance criteria for raw materials to meet infant nutrition regulations that restrict contaminants such as cadmium, lead, and arsenic and define acceptable nutrient profiles. Growers supplying infant-food ingredient supply chains integrate compliance-ready fertility inputs that help reduce risk of contaminant uptake while enhancing crop nutritional attributes. Fertility programs align nutrient delivery with soil tests, crop requirements, and documented application records necessary for certification and audit trails demanded by infant-formula manufacturers and ingredient buyers. Retailers and brand owners emphasize infant food safety credentials that trace fertilizer inputs, reinforcing adoption in quality-driven production systems.
How do cost barriers, input complexity, and supply chain requirements influence market scalability?
Infant-food compliance ready fertility inputs involve advanced formulation, dedicated sourcing of low-risk feedstocks, and testing protocols that increase production cost relative to conventional fertilizers. Higher input costs influence grower decisions, particularly in commodity crop systems with narrow profit margins. Complexity of product specifications, testing documentation, and traceability requirements adds operational overhead for producers and input suppliers. Variable regional regulatory frameworks governing contaminant limits and nutrient claims affect compliance interpretation and global distribution strategies. Integration with existing agronomy practices requires education, monitoring systems, and record-keeping that can strain smaller farms’ resources. Long-term growth depends on continued refinement of cost-effective compliance-ready inputs, harmonized regulatory guidelines, and agronomic support that enhances confidence in meeting infant food safety criteria across global crop production networks.
Demand for infant-food compliance ready fertility inputs is increasing globally due to stricter contaminant limits, traceability requirements, and rising production of baby-grade cereals, fruits, and vegetables. India leads with a 15.2% CAGR, supported by expansion of infant nutrition manufacturing and export-oriented agriculture. China follows at 14.8%, driven by tightened food safety regulations and rebuilding of consumer trust in infant products. USA records a 14.0% CAGR, shaped by regulatory oversight and procurement standards from infant food brands. France posts 13.1%, reflecting EU compliance frameworks and premium baby food positioning. Growth reflects transition toward fertilizers engineered for ultra-low heavy metals, controlled nitrate levels, and verified compliance with infant food safety thresholds.
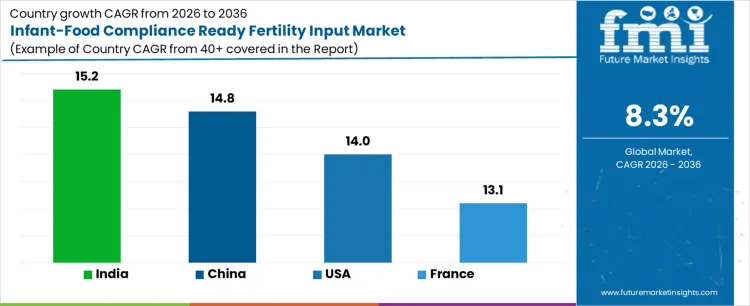
| Country | CAGR (%) |
|---|---|
| India | 15.2% |
| China | 14.8% |
| USA | 14.0% |
| France | 13.1% |
India demand is driven by growth of domestic infant food manufacturing and increasing exports of baby-grade cereals and fruits. CAGR of 15.2% reflects adoption of fertility inputs formulated to minimize heavy metals, nitrates, and unwanted residues. Producers supplying rice, wheat, fruits, and vegetables for infant foods require strict compliance with global safety specifications. Government oversight and private certification programs influence input selection at farm level. Export buyers impose documented traceability from soil to harvest. Domestic fertilizer suppliers invest in purification, batch testing, and compliance documentation. Growth remains compliance-led and agriculture-driven, aligned with infant nutrition expansion and international quality expectations.
China demand reflects heightened regulatory enforcement and long-term rebuilding of trust in infant nutrition supply chains. CAGR of 14.8% is supported by adoption of fertility inputs meeting strict limits for cadmium, lead, and nitrate accumulation. Infant food processors require verified upstream compliance from agricultural suppliers. Large farming operations supplying grains and produce adopt standardized compliant fertility programs. Government agencies increase monitoring across production stages. Domestic input manufacturers upgrade purification and quality assurance processes. Growth remains regulation-driven and scale-aligned, supported by centralized oversight and structured infant food sourcing programs.
USA demand is shaped by strict oversight from food safety authorities and liability sensitivity among infant nutrition brands. CAGR of 14.0% reflects increased use of fertility inputs designed to control nitrate uptake and eliminate heavy metal risks. Growers supplying baby food manufacturers follow detailed nutrient protocols and testing regimes. Retailers and brands require documented compliance and traceability. Precision nutrient management supports consistent outcomes. Domestic suppliers align formulations with regulatory thresholds and audit requirements. Growth remains compliance-focused and risk-driven, aligned with regulatory enforcement and brand protection strategies.
France demand reflects strong positioning in premium infant nutrition and alignment with European Union safety frameworks. CAGR of 13.1% is supported by use of fertility inputs meeting strict contaminant and residue limits for baby food crops. Growers supplying fruits, vegetables, and cereals prioritize compliance with EU infant nutrition standards. Advisory-led agriculture promotes careful nutrient planning. Certification schemes reinforce adoption of compliant inputs. Domestic suppliers offer specialized fertility products aligned with infant food regulations. Growth remains quality-driven and regulation-aligned, supported by premium branding and high consumer trust in baby food products.
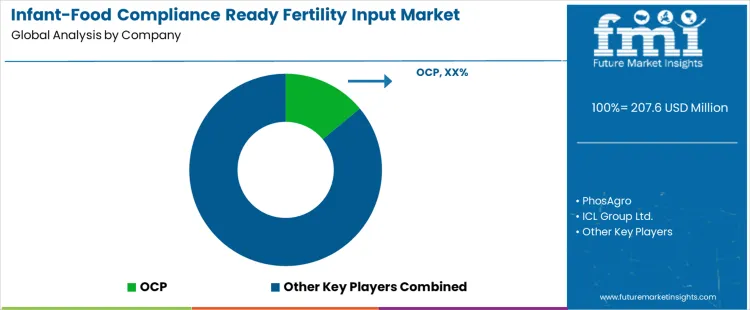
Demand for infant-food compliance ready fertility inputs is driven by strict limits on heavy metals, nitrate accumulation, and contaminant transfer from soil to crops used in baby food. These inputs are applied in cereals, fruits, vegetables, and raw materials destined for infant and toddler nutrition, where regulatory thresholds are materially tighter than conventional food production. Buyers evaluate cadmium and lead content, nutrient purity, solubility behavior, and consistency across production batches. Procurement teams prioritize suppliers with controlled phosphate rock sourcing, advanced purification processes, traceable supply chains, and documentation aligned with infant nutrition standards. Trend in the global market reflects tighter enforcement by food brands, retailer audits, and growing linkage between upstream fertilizer quality and downstream infant food certification.
OCP maintains leading positioning through large-scale access to low-contaminant phosphate resources and refined processing tailored for food-grade and sensitive crop applications. PhosAgro benefits from naturally low-cadmium phosphate rock, enabling consistent compliance for crops entering infant food supply chains. ICL Group Ltd. supports demand with specialty fertilizers and controlled nutrient solutions designed for quality-critical agricultural production. EuroChem contributes through vertically integrated fertilizer production and selective sourcing supporting regulated food markets. The Mosaic Company participates with premium phosphate fertilizers produced under stringent quality and monitoring frameworks. Competitive differentiation depends on contaminant control capability, regulatory documentation depth, supply reliability, and ability to meet infant-food grade specifications across regions.
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Application | Dairy; Beverages; Bakery; Meat |
| Form | Powder; Liquid; Encapsulated; Paste |
| Channel | Direct-to-B2B; Distributors; Online B2B; Contract Mfg |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, China, USA, France, and 40+ countries |
| Key Companies Profiled | OCP; PhosAgro; ICL Group Ltd.; EuroChem; The Mosaic Company; Nutrien |
| Additional Attributes | Dollar sales by application and form; adoption trends for infant-food compliance-ready fertility inputs across regulated supply chains; heavy metal thresholds, contaminant control, and traceability performance benchmarks; compatibility with food-grade crop programs and residue management protocols; formulation consistency, solubility, and handling considerations; regulatory alignment with infant nutrition standards and food safety compliance influencing fertility input selection. |
How big is the infant-food compliance ready fertility input market in 2026?
The global infant-food compliance ready fertility input market is estimated to be valued at USD 191.7 million in 2026.
What will be the size of infant-food compliance ready fertility input market in 2036?
The market size for the infant-food compliance ready fertility input market is projected to reach USD 425.5 million by 2036.
How much will be the infant-food compliance ready fertility input market growth between 2026 and 2036?
The infant-food compliance ready fertility input market is expected to grow at a 8.3% CAGR between 2026 and 2036.
What are the key application types in the infant-food compliance ready fertility input market?
The key application types in infant-food compliance ready fertility input market are dairy, beverages, bakery and meat.
Which form segment will contribute significant share in the infant-food compliance ready fertility input market in 2026?
In terms of form, powder segment to command 45.0% share in the infant-food compliance ready fertility input market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.