Prior Authorization Workflow Orchestration Market
The Prior Authorization Workflow Orchestration Market is segmented by Solution Type (Orchestration, Attachment Automation, Rules Discovery, Status Tracking, Appeals Routing), Deployment Model (Cloud, Hybrid, On-Premise), Buyer Type (Providers, Payers, Intermediaries), Clinical Scope (Medical Services, Pharmacy, Specialty Drugs, Imaging), Core Function (Submission Automation, Criteria Matching, Document Intake, Decision Support, Workqueue Analytics), Integration Layer (FHIR APIs, Portal Connectors, EHR Embeds, Clearinghouse Links), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Prior Authorization Workflow Orchestration Market Size, Market Forecast and Outlook By FMI
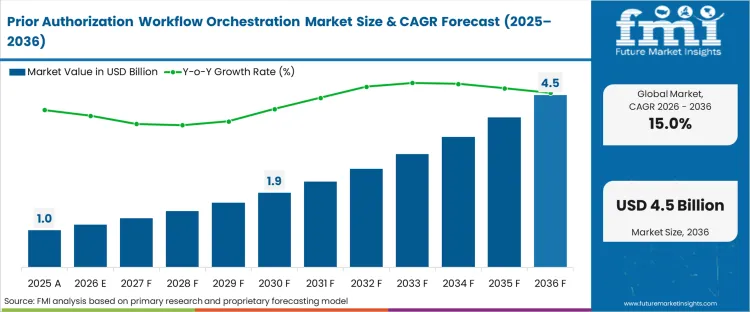
The prior authorization workflow orchestration market surpassed a value of USD 0.9 billion in 2025 leading the industry revenue expansion to be expected to hit USD 1.1 billion in 2026 at a CAGR of 15.0% during this forecast period. Persistent increase in the market growth propels total valuation to USD 4.4 billion through 2036 as health systems abandon manual web portal scraping for bidirectional electronic health record integration pathways.
Summary of Prior Authorization Workflow Orchestration Market
- Prior Authorization Workflow Orchestration Market Definition
- Software infrastructure automating clinical criteria matching, documentation gathering, and electronic payer submissions through bidirectional data exchange.
- Demand Drivers in the Market
- Federal interoperability mandates compel revenue cycle directors to evaluate prior authorization software vendors immediately.
- Rising denial rates force utilization management heads to adopt prior authorization denial prevention software.
- Clinical staff shortages push hospital administrators to eliminate manual payer portal navigation.
- Key Segments Analyzed in the FMI Report
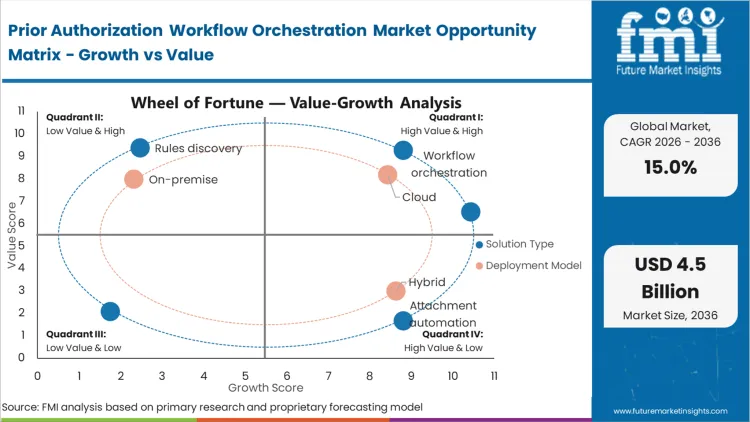
- Workflow Orchestration: 31.0% share in 2026, driven by demand for end-to-end integration.
- Cloud: 62.0% share in 2026 due to rapid API update capabilities.
- Providers: 44.0% share in 2026 as hospitals target denial reduction.
- Medical Services: 53.0% share in 2026 based on complex clinical charting requirements.
- Submission Automation: 28.0% share in 2026 since web scraping elimination remains paramount.
- FHIR APIs: 36.0% share in 2026 dictated by federal interoperability standards.
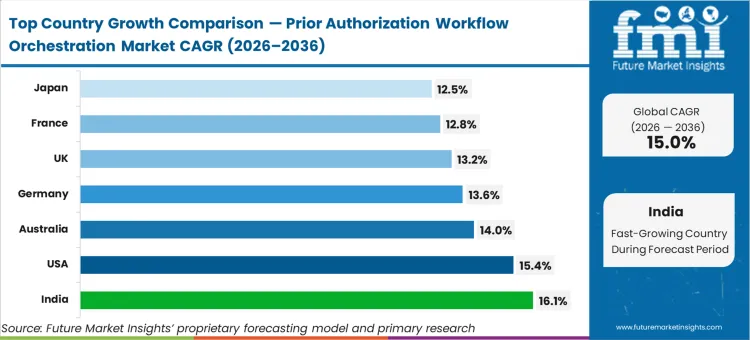
- India: A CAGR of 16.1%, expanding as large corporate hospital chains digitize operations.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Analyst, Healthcare, at FMI, points out, "Healthcare executives assume federal interoperability mandates drive automated authorization adoption uniformly. Reality dictates a different adoption curve. Revenue cycle directors at mid-size hospital networks do not purchase orchestration engines for compliance; they buy them to stop uncompensated labor hemorrhaging. Basic transaction standards solve data transport, yet payer criteria remain notoriously opaque and constantly shifting. Successful orchestration platforms differentiate not on connectivity, but on maintaining highly accurate, continuously updated payer rules engines that predict denials before clinicians submit documentation."
- Strategic Implications / Executive Takeaways
- Hospital CIOs must prioritize bidirectional EHR integration to avoid severe physician workflow disruption.
- Revenue cycle vendors risk obsolescence unless they adopt native FHIR API standards rapidly.
- Utilization management directors gain significant operational efficiency by replacing manual portal checks with automated status tracking.
- Methodology
- Connects technical interoperability requirements with hospital revenue cycle operational realities.
- Evaluates vendor capabilities against emerging federal implementation guidelines.
- Correlates API transaction volumes with overall automation adoption trajectories.
- Anchors demand projections in actual hospital IT budget allocation shifts.
Revenue cycle directors face escalating administrative denials tied to medical necessity documentation gaps. Eliminating manual prior authorization bottlenecks requires sophisticated prior authorization automation rather than basic screen scraping utilities. Hospitals must deploy automated clinical data extraction engines or risk severe cash flow degradation. Compliance timelines surrounding the CMS prior authorization API deadline force immediate action from utilization management heads. Institutions delaying FHIR prior authorization upgrades suffer immediate accounts receivable deterioration. Staff burnout drives urgent technology acquisition to prevent widespread operational failure.
Once bidirectional HL7 integration achieves reliable prior authorization attachment automation, adoption scales exponentially across health networks. Application programming interface connectors replace brittle web scrapers, eliminating transcription errors completely. Health systems clear authorization backlogs significantly faster after deploying native electronic health record embeds, driving rapid expansion across the prior authorization workflow footprint.
India advances at 16.1% as large corporate provider networks digitize revenue cycles rapidly. United States facilities track at 15.4% on back of strict federal interoperability mandates. Australia advances at 14.0% due to private health insurance integration efforts. Germany grows at 13.6%, United Kingdom tracks at 13.2%, France hits 12.8%, and Japan registers 12.5% compound growth as national health systems standardize data exchange frameworks. European adoption splits structurally between statutory fund requirements and private hospital modernization pushes.
Prior Authorization Workflow Orchestration Market Definition
The prior authorization workflow orchestration encompasses software infrastructure designed to automate clinical criteria matching, documentation gathering, and payer submission processes. Systems establish bidirectional data exchange between provider electronic health records and payer utilization management platforms. Core architecture replaces manual web portal navigation with automated rules engines executing complex clinical logic sequences to secure necessary medical approvals.
Prior Authorization Workflow Orchestration Market Inclusions
Core software platforms managing end-to-end authorization lifecycles fall strictly within scope. Capabilities cover automated clinical chart extraction, real-time status tracking, rules-based triage, and electronic attachments formatting. Systems facilitating integration via standard interoperability protocols belong here. Modules interacting with payer policy intelligence platforms constitute critical functional components evaluated within this analysis, explicitly including dedicated healthcare prior authorization software designed for specialized institutional use.
Prior Authorization Workflow Orchestration Market Exclusions
General electronic health record systems lacking dedicated authorization automation modules fall outside our analysis. Basic revenue cycle billing software without clinical criteria matching capabilities remains excluded. Outsourced human-in-loop service operations without proprietary automation technology do not qualify. Generic document management software fails to meet clinical logic requirements necessary for automated payer decision support.
Prior Authorization Workflow Orchestration Market Research Methodology
- Primary Research: Revenue cycle directors, utilization management heads, chief medical informatics officers.
- Desk Research: CMS interoperability rule dockets, HL7 Da Vinci implementation guides, CAQH index reports.
- Market-Sizing and Forecasting: Hospital IT budget allocations for revenue cycle automation software baseline this model.
- Data Validation and Update Cycle: Annual vendor API transaction volume reports cross-validate adoption curves.
Segmental Analysis
Prior Authorization Workflow Orchestration Market Analysis by Solution Type
Fragmented point solutions fail when payer rules change mid-cycle. Workflow orchestration commands 31.0% share because it centralizes discrete automation tasks into unified command centers. Revenue cycle directors abandon standalone utilities in favor of comprehensive orchestration suites to maintain visibility across complex multi-step approvals. Integrated platforms eliminate manual hand-offs between nurses and administrative staff. FMI analysis indicates isolated rules engines often trigger false confidence; true orchestration systems validate clinical documentation against clinical documentation integrity automation datasets prior to transmission. Facilities relying on fragmented tools experience higher downstream denial rates due to mismatched attachments.
- Pre-Submission Validation: Integrated engines cross-reference patient charts against real-time payer policies. Utilization management directors avoid submitting incomplete clinical dossiers.
- Bidirectional Status Syncing: Advanced prior authorization status tracking software continuously polls payer endpoints for decision updates. Billing teams eliminate manual portal checking entirely.
- Automated Exception Routing: Systems intelligently route complex cases through a designated prior authorization appeals workflow directly to physician workqueues. Medical directors bypass administrative triaging delays.
Prior Authorization Workflow Orchestration Market Analysis By Deployment Model
On-premise servers cannot update payer rules engines fast enough. Cloud architecture dominates with 62.0% share as vendors push daily policy updates across entire client bases simultaneously. Chief information officers at large health systems demand cloud-native platforms to ensure continuous prior authorization platform compliance readiness. Legacy systems require manual patch management, creating unacceptable compliance risks during federal mandate transitions. According to FMI's estimates, multitenant cloud architectures provide vendors with aggregated payer behavior data, allowing predictive denial models that single-tenant systems cannot replicate. Hospitals clinging to local deployments suffer escalating IT maintenance costs.
- Centralized Policy Distribution: Cloud vendors update clinical criteria matrices centrally. Compliance officers ensure all facility locations operate under identical rule sets instantly.
- Scalable Compute Provisioning: High-volume batch processing scales dynamically during peak admission hours. IT directors avoid expensive on-premise hardware over-provisioning.
- Aggregated Machine Learning: Multitenant environments pool anonymized denial data across hundreds of hospitals. Revenue cycle vice presidents access predictive intelligence unavailable locally.
Prior Authorization Workflow Orchestration Market Analysis By Buyer Type
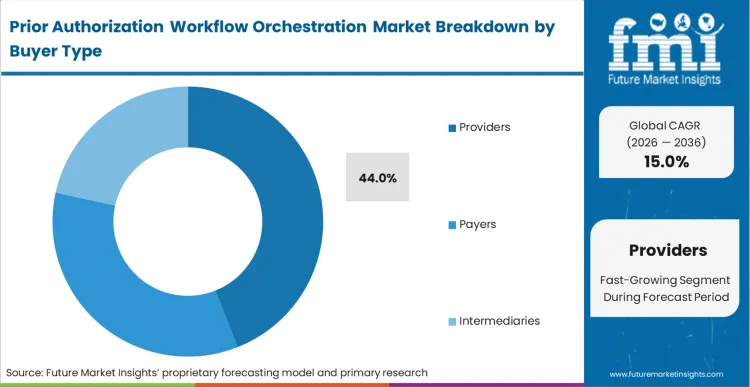
Margin compression forces immediate action from hospital administrators as clinical staff shortages make manual authorization processes financially unsustainable. Patient access directors bear primary responsibility for securing approvals prior to elective procedures, requiring advanced provider prior authorization workflow tools. Manual workflows generate unacceptable surgery cancellation rates. Based on FMI's assessment, payer-side automation investments historically focused on auto-denying claims, leading the providers to capture a 44.0% share, forcing providers to deploy the best prior authorization software for hospitals as defense mechanisms. Health systems failing to automate front-end clearance face severe revenue leakage from services rendered without proper authorization.
- Surgical Clearance Acceleration: Automated systems extract necessary clinical markers directly from operative notes. Patient access managers secure approvals days faster.
- Retrospective Denial Defense: Platforms maintain immutable audit trails of all payer communications. Appeals coordinators win disputes by proving timely submission of required documentation.
- Physician Workflow Protection: Systems extract criteria directly from natural clinical documentation. Chief medical officers prevent administrative burdens from degrading clinical productivity.
Prior Authorization Workflow Orchestration Market Analysis By Clinical Scope
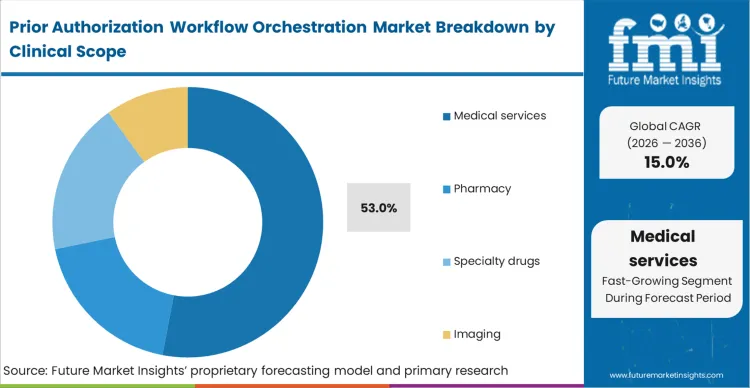
Complex procedural coding drives massive administrative friction as surgical directors face constant shifting payer guidelines regarding operative prerequisites. Automated systems parse extensive surgical histories to validate criteria fulfillment, with medical services holding a 53.0% share because surgical and diagnostic interventions require extensive clinical documentation to prove medical necessity. In FMI's view, specialty drug prior authorization automation solved simple structured data exchange years ago; medical service authorization remains structurally difficult because it relies heavily on unstructured clinical narrative text requiring advanced clinical workflow solutions. Facilities attempting manual medical service authorizations experience highest peer-to-peer review ratios.
- Unstructured Text Parsing: Natural language processing algorithms extract vital signs from physician notes. Utilization nurses save hours of chart review during complex oncology prior authorization automation cases.
- Conservative Therapy Verification: Specialized radiology prior authorization workflow software automatically verifies whether patients failed required preliminary imaging treatments. Clinical pathways directors ensure compliance with step-therapy mandates.
- Inpatient Admission Justification: Algorithms cross-reference presenting symptoms against standardized admission criteria. Bed management directors avoid costly retrospective downgrades to observation status.
Prior Authorization Workflow Orchestration Market Analysis By Core Function
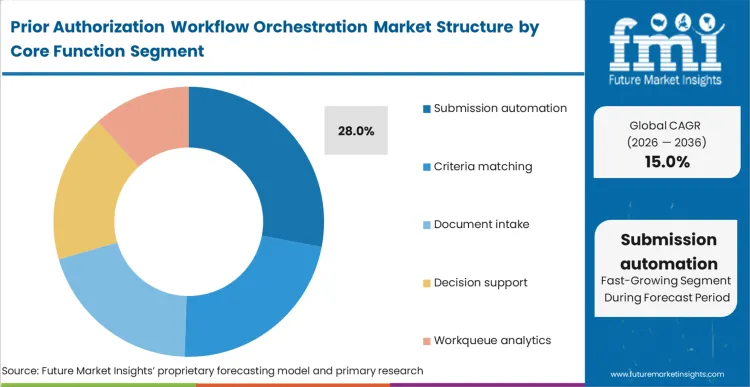
Submission automation leads with 28.0% share as health systems prioritize eliminating brute-force portal scraping, replacing human data entry forms fundamental automation baseline. Revenue cycle vice presidents target submission automation first because it yields immediate, measurable headcount reduction and accelerates prior authorization turnaround time automation. Software robots log into payer portals and populate forms using data extracted directly from electronic health records. FMI observes that generalists assume criteria matching drives most value, but hospital IT directors know perfectly matched criteria fail if provider directory accuracy management issues cause submission rejections at portal endpoints. Hospitals skipping submission automation layers find their advanced clinical rules engines stranded without delivery mechanisms.
- Portal Navigation Elimination: Scripts bypass user interfaces entirely via direct server connections. Billing clerks reallocate time to high-value denial appeals.
- Payload Formatting Standardization: Engines automatically format clinical attachments to meet specific payer file constraints. IT administrators avoid custom integration builds per payer.
- Receipt Confirmation Logging: Platforms capture cryptographic timestamps of successful submissions. Audit directors utilize these logs to contest arbitrary timely-filing denials.
Prior Authorization Workflow Orchestration Market Analysis By Integration Layer
Federal interoperability regulations dictate technical transport standards because CMS mandates strictly enforce this protocol for payer-provider data exchange. Chief informatics officers mandate a standardized FHIR prior authorization API for all new revenue cycle vendor purchases. Standardized resource mapping enables vendors to deploy pre-built connectors rather than custom interfaces. FMI analysts note that generalists assume HL7 FHIR compliance guarantees smooth data exchange, as FHIR APIs commands 36.0% share; interoperability directors know payer endpoints frequently return unstructured PDF payloads masquerading as structured data, breaking automated workflows completely. Health systems failing to secure deep prior authorization workflow integration with EHR face increasing data translation errors.
- Standardized Resource Mapping: Connectors translate proprietary EHR data models into standardized clinical resources. Interface engineers deploy new payer connections in days rather than months.
- Real-Time Payload Validation: API gateways check data completeness before transmission. Data governance officers prevent malformed requests from triggering automatic technical denials.
- Version Control Management: Platforms automatically handle backward compatibility when payers upgrade API versions. System administrators avoid workflow disruptions during payer maintenance windows.
Prior Authorization Workflow Orchestration Market Drivers, Restraints, and Opportunities
Enforcement mechanisms attached to CMS 0057-F mandate strict compliance timelines for payer API availability. Revenue cycle directors examining how does CMS 0057-F affect prior authorization software demand realize this regulatory pressure forces payers into accepting automated electronic submissions. Hospitals face escalating uncompensated care costs when manual processes delay care delivery beyond medically advisable windows. Executives utilize advanced revenue cycle denials intelligence to demonstrate return on investment for orchestration platforms. Delaying technology adoption guarantees mounting operational losses as payer algorithms become increasingly sophisticated at finding technical reasons to deny claims.
Payer proprietary portal fragmentation slows comprehensive automation adoption even among highly motivated health systems. Regional payers often lack technical infrastructure required to support bidirectional API data exchange. Chief information officers struggle integrating specialized orchestration platforms when smaller regional insurers insist on manual fax submissions or proprietary web forms. This technological disparity forces hospitals to maintain parallel manual workflows alongside automated systems, severely diluting expected efficiency gains. Vendors attempting universal coverage encounter hard technical limits when payers refuse to open endpoint access.
Opportunities in the Prior Authorization Workflow Orchestration Market
- Predictive Denial Modeling: Integrating AI in prior authorization workflows analyzes historical payer decisions to highlight high-risk submissions. Revenue cycle directors correct documentation deficiencies prior to transmission.
- Specialty-Specific Rules Engines: Vendors develop deep clinical logic customized for complex cardiology treatments. Service line directors capture exact clinical markers required for specialized care pathway variance analytics.
- Payer-Side Workflow Alignment: Emerging payer prior authorization automation tools alert physicians during order entry if selected treatments require authorization. Chief medical officers prevent unauthorized procedures before patients leave examination rooms.
Regional Analysis
Adoption across the prior authorization workflow orchestration market diverges sharply among global regions, dictated by specific national regulatory deadlines and the maturity of local health information exchanges. While North America reacts urgently to federal interoperability mandates, European and Asia-Pacific markets are shaped primarily by statutory funding requirements and private hospital corporatization respectively. Based on regional analysis, the market is segmented into North America, Europe, Asia-Pacific & ASEAN across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| United States | 15.4% |
| India | 16.1% |
| Australia | 14.0% |
| Germany | 13.6% |
| United Kingdom | 13.2% |
| France | 12.8% |
| Japan | 12.5% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Prior Authorization Workflow Orchestration Market Analysis
The velocity of adoption across North American health systems is defined largely by federal mandates enforcing strict technical interoperability standards. While Centers for Medicare & Medicaid Services regulations compel payers to construct standardized APIs to provide hospital IT departments with reliable endpoints, commercial insurers frequently implement these standards with extreme inconsistency. The vendors as a result are forced to maintain massive exception-handling libraries, complicating the automated data exchange landscape significantly.
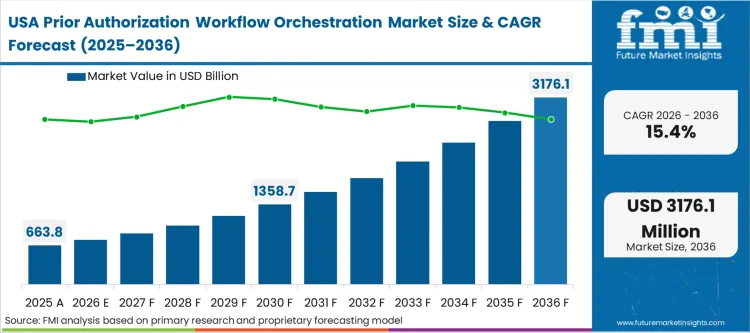
- United States: Massive technology procurement cycles are sweeping across hospital networks as the CMS prior authorization API deadline approaches. Reflecting an urgent push to replace obsolete manual scraping tools and eliminate manual prior authorization bottlenecks, the United States prior authorization software market is expanding at a 15.4% CAGR, as revenue cycle vice presidents scramble to deploy certified orchestration platforms capable of handling mandated API data flows. Significant financial returns accrue to facilities that act early by aggressively reducing administrative overhead per patient encounter.
The broader North American landscape beyond the United States will closely monitor these initial API deployments, utilizing early integration failures to refine subsequent implementation strategies and bypass expensive architectural missteps.
Europe Prior Authorization Workflow Orchestration Market Analysis
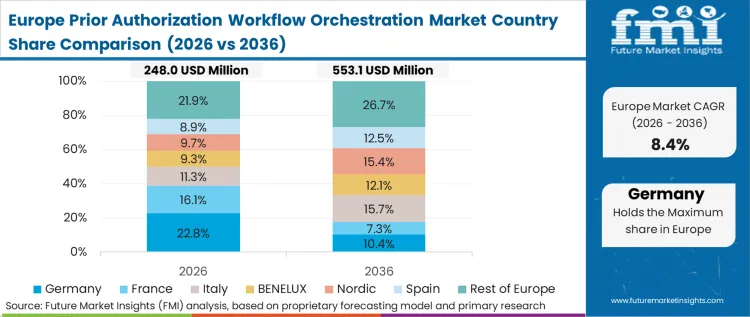
Across European hospital networks, structural upgrades shaping the Europe prior authorization workflow market are primarily driven by statutory health fund digitization initiatives. To control escalating administrative costs within nationalized systems, government ministries are actively pushing centralized data exchange frameworks. However, international vendors face severe complications during cross-border platform deployment due to highly fragmented regional data privacy regulations.
- Germany: Digital revenue cycle infrastructure acquisitions are heavily subsidized directly through Hospital Future Act funding. Hospital administrators are actively purchasing orchestration engines to ensure they meet stringent statutory funding requirements. With a 13.6% CAGR anticipated across the Germany prior authorization software market, adoption curves mirror federal funding disbursement schedules closely, allowing early platform vendors to establish long-term vendor lock-in.
- United Kingdom: Expanding at a steady 13.2% CAGR, regional networks see trusts migrating away from legacy paper-based referral management. Unified clinical clearance workflows across merged regional facilities are now a hard requirement driven by NHS trust consolidation projects. To standardize clinical criteria evaluation effectively, digital transformation directors are rapidly utilizing advanced orchestration tools.
- France: Secure bidirectional data exchange between clinics and statutory funds is a direct mandate of the Segur de la Sante digital health investments. Steady replacement of outdated manual billing systems with automated clearance engines drives a 12.8% CAGR across the country. To guarantee strict funding compliance, IT directors must deploy these certified modules without delay.
FMI's report includes adjacent European markets where nationalized healthcare infrastructure dictates distinct, localized integration timelines completely separate from broader European Union mandates.
Asia-Pacific & ASEAN Prior Authorization Workflow Orchestration Market Analysis
Aggressive revenue cycle modernization is heavily driven by rapid corporatization within private healthcare sectors throughout this geography. To standardize billing operations across newly acquired regional clinics, large hospital chains are deploying enterprise-wide orchestration platforms. Extreme variation in the technical maturity of private health insurers forces these platforms to simultaneously support both advanced APIs and legacy document transmission protocols, often utilizing imaging interoperability middleware as an initial entry point for broader automation.
- India: Massive administrative bottlenecks when processing complex private insurance claims plague corporate hospital chains across the subcontinent. To reduce severe accounts receivable days outstanding, revenue cycle heads are deploying automated extraction engines at scale. Recording an explosive 16.1% CAGR, the India prior authorization automation market highlights a massive unmet need for scalable billing infrastructure that ultimately yields higher operating margins than regional competitors.
- Australia: Tracking at a 14.0% CAGR adoption velocity, the Australia prior authorization platform market relies entirely on large private insurers successfully opening API endpoints to hospital IT departments. Tighter prior authorization workflow integration with EHR platforms and fund assessment engines is a direct result of ongoing private health insurance reforms. The chief informatics officers as a result heavily prioritize platforms capable of true real-time clinical data exchange.
- Japan: Extreme pressure on clinical staff due to aging population demographics makes administrative automation a critical operational necessity rather than a luxury. Generating a 12.5% CAGR, Japanese facility investments focus heavily on systems that effectively remove clerical burdens directly from nursing staff. Hospital directors therefore prioritize platforms featuring deep native language processing capabilities necessary to handle highly complex clinical narratives.
Examining the broader Asia-Pacific ecosystem reveals that markets outside these primary hubs are beginning to evaluate lightweight orchestration tools, prioritizing basic portal automation before committing to full API integration workflows.
Competitive Aligners for Market Players
Legacy clearinghouses still have a clear advantage at the start of many orchestration deployments because they already sit inside large provider networks. Availity and Waystar benefit from those long-standing portal relationships and can extend advanced API automation into hospital accounts that are already familiar with their systems. For revenue cycle teams, adding new functionality through an existing vendor often feels easier than bringing in a completely new platform and going through another round of security and compliance review. That installed presence makes it harder for pure-play automation startups to break into large hospital environments early.
Specialist technology vendors are trying to compete in a different way. Companies such as Cohere Health are focusing more deeply on clinical logic, especially where prior authorization depends on information buried in unstructured notes rather than standard claims fields. That gives them a stronger position in workflows where medical necessity is harder to interpret. Health systems testing these platforms often look closely at how well they can handle unusual clinical scenarios and department-specific requirements. Vendors that combine stronger natural language processing with tools such as radiology reporting automation can gain traction in narrower use cases before expanding into a broader enterprise role.
Even with those differences, large health systems are careful about giving too much control to any single vendor. CIOs and digital leaders usually want platforms that can work across existing systems instead of locking data into one orchestration layer. Buyers reviewing prior authorization automation platforms are paying close attention to interoperability, especially bidirectional HL7 and FHIR support. In the final evaluation stage, comparisons such as Availity, Waystar, and Cohere Health often come down to how open the architecture feels, how well the platform fits into current workflows, and whether it leaves the organization with enough flexibility later.
Key Players in Prior Authorization Workflow Orchestration Market
- Availity
- Cohere Health
- Waystar
- Surescripts
- CoverMyMeds
- EviCore by Evernorth
- Oracle Health
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 1.1 billion to USD 4.4 billion, at a CAGR of 15.0% |
| Market Definition | Software infrastructure automating clinical criteria matching, documentation gathering, and electronic payer submissions through bidirectional data exchange. |
| Segmentation | Solution type, Deployment model, Buyer type, Clinical scope, Core function, Integration layer, Region |
| Regions Covered | North America, Europe, Asia-Pacific & ASEAN |
| Countries Covered | United States, Germany, United Kingdom, France, Japan, India, Australia |
| Key Companies Profiled | Availity, Cohere Health, Waystar, Surescripts, CoverMyMeds, EviCore by Evernorth, Oracle Health |
| Forecast Period | 2026 to 2036 |
| Approach | Hospital IT budget allocations for revenue cycle automation software baseline this model. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Segments
Solution Type:
- Workflow orchestration
- Attachment automation
- Rules discovery
- Status tracking
- Appeals routing
Deployment Model:
- Cloud
- Hybrid
- On-premise
Buyer Type:
- Providers
- Payers
- Intermediaries
Clinical Scope:
- Medical services
- Pharmacy
- Specialty drugs
- Imaging
Core Function:
- Submission automation
- Criteria matching
- Document intake
- Decision support
- Workqueue analytics
Integration Layer:
- FHIR APIs
- Portal connectors
- EHR embeds
- Clearinghouse links
Regions:
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Bibliography
- Centers for Medicare & Medicaid Services. (2024, January 17). CMS interoperability and prior authorization final rule (CMS-0057-F) fact sheet. U.S. Department of Health and Human Services.
- CAQH. (2024). 2024 CAQH Index Report Key Takeaways.
- CAQH CORE. (2024, June 21). Navigating The CMS 0057 Final Rule: A Guide For Implementing Prior Authorization Requirements.
- American Medical Association. (2024). 2024 AMA Prior Authorization Physician Survey.
- Health Level Seven International. (2024). 2024 Da Vinci Implementation Guide Roadmap.
- Chen, W. C., Kim, J., D’Souza, D., Miller, R. C., Lowenstein, P. R., Crane, C. H., & Song, D. Y. (2025). Integrating prior authorization into clinical workflows for care delivery at an academic cancer center. JAMA Network Open, 8(1), e2843121.
- Osmundson, E. C. (2025). Technology-enabled reform of prior authorization. JAMA Network Open, 8(1), e2843124.
- Sidiqi, B. U., Chen, W. C., Song, D. Y., Miller, R. C., & Crane, C. H. (2024). Implementation of a clinically integrated authorization system to streamline cancer care. International Journal of Radiation Oncology, Biology, Physics, 120(Supplement), article S0360-3016(24)02204-1.
- Workgroup for Electronic Data Interchange. (2025, December 21). WEDI survey finds payers and providers continue to face challenges in implementing the CMS interoperability and prior authorization final rule.
- America’s Health Insurance Plans. (2025, June 23). Health plans take action to simplify prior authorization.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Identifies exact implementation timelines for CMS 0057-F compliance standards.
- Evaluates vendor capabilities parsing unstructured clinical data for medical services clearance.
- Analyzes hospital revenue cycle IT budget shifts toward FHIR API connectors.
- Details operational differences between cloud architectures versus legacy on-premise engines.
- Benchmarks adoption velocity across distinct European statutory health fund frameworks.
- Measures financial impact when manual web scraping causes downstream denial events.
- Maps transition dynamics moving from simple pharmacy rules toward complex surgical criteria.
- Outlines competitive barriers facing pure-play automation startups against established clearinghouses.
Frequently Asked Questions
What is prior authorization workflow orchestration?
Prior authorization workflow orchestration is software infrastructure designed to automate clinical criteria matching, documentation gathering, and payer submission processes through bidirectional data exchange.
How large is the prior authorization software sector?
Projections indicate total valuation hits USD 4.4 billion through 2036 as hospital networks realize manual clearance processes create unsustainable uncompensated care volumes.
What is the expected prior authorization workflow forecast rate?
Revenue expands at 15.0% compound growth as severe margin compression forces facility administrators to eliminate expensive administrative labor dedicated purely to navigating proprietary payer web portals.
Why is CMS 0057-F increasing demand for prior authorization software?
Revenue cycle directors leverage the strict compliance timelines of CMS 0057-F to deploy systems that force payers into accepting automated electronic submissions.
Which companies currently lead prior authorization automation?
Established entities like Availity and Waystar leverage massive existing provider networks, while innovators like Cohere Health focus entirely on extracting complex medical necessity markers from unstructured clinical notes.
How should buyers compare prior authorization software vendors?
Chief information officers must evaluate bidirectional HL7 FHIR compliance during initial procurement evaluations, immediately disqualifying platform vendors that fail to demonstrate open architecture data liquidity.
What ROI do hospitals get from prior authorization automation?
Facilities secure significant financial returns by reducing administrative overhead per patient encounter and eliminating costly retrospective denials to drastically reduce accounts receivable days outstanding.
How do FHIR APIs change payer-provider prior authorization workflows?
Standardized resource mapping enables vendors to deploy pre-built connectors rather than custom interfaces, allowing interface engineers to establish new payer connections in days and effectively eliminating brittle web scraping tools.
Which solution architecture dominates current deployment?
Workflow orchestration captures 31.0% share currently because revenue cycle vice presidents require comprehensive oversight platforms rather than fragmented point solutions that lose visibility during complex peer-to-peer review escalation cycles.
Why does cloud deployment capture majority share?
Cloud architectures hold 62.0% share because single-tenant local servers simply cannot download and integrate complex daily payer policy updates fast enough to prevent technical denials.
Who constitutes the primary buyer demographic?
Providers command 44.0% share right now as administrators deploy these systems as defensive revenue protection mechanisms against unauthorized elective procedures resulting in complete non-payment.
Which clinical scope segment requires most automation?
Medical services secure 53.0% share because surgical clearance requires complex unstructured text parsing from operative notes, making manual chart review excessively slow and prone to human transcription errors.
Why does submission automation command significant focus?
Submission automation holds 28.0% share because IT directors value engines that bypass user interfaces entirely via direct secure server connections to yield immediate headcount reduction.
How fast do United States facilities adopt this technology?
United States infrastructure grows at 15.4% as Centers for Medicare & Medicaid Services regulations compel payers to build standardized APIs, creating reliable endpoints for automated hospital data exchange.
What drives Indian hospital automation growth?
India advances at 16.1% because corporate hospital chains face massive administrative bottlenecks processing complex private insurance claims, forcing rapid deployment of automated extraction engines.
Why do German hospitals invest heavily in orchestration?
Germany tracks at 13.6% as Hospital Future Act funding subsidizes digital revenue cycle infrastructure acquisitions, pushing hospital administrators to purchase orchestration engines to meet stringent statutory funding requirements.
How does Australian private health influence adoption?
Australia hits 14.0% because private health insurance reforms require tighter integration between hospital electronic medical records and fund assessment engines, driving demand for real-time clinical data exchange capabilities.
What operational friction slows comprehensive automation?
Payer proprietary portal fragmentation slows adoption severely because regional payers often lack the technical infrastructure required for bidirectional API data exchange, forcing hospitals to maintain parallel manual workflows.
How do systems prevent retrospective denials?
Appeals coordinators utilize definitive timestamp logs from immutable audit trails capturing all payer communications cryptographically to contest arbitrary timely-filing denials successfully.
What advantage do legacy clearinghouses hold over startups?
Established clearinghouses leverage massive existing provider network connectivity, allowing hospital IT directors to activate new modules within existing vendor contracts rather than enduring lengthy security reviews for new startups.
How do pure-play technology vendors compete successfully?
Innovators outperform basic claims clearinghouses limited strictly to structured data parsing during specialized departmental contracts by focusing entirely on extracting complex medical necessity markers from unstructured clinical notes.
What role does predictive denial modeling play?
Machine learning analyzes historical payer decisions to highlight high-risk submissions, allowing revenue cycle directors to correct documentation deficiencies prior to transmission and avoid costly back-end rework entirely.
How do specialized rules engines benefit service lines?
Service line directors capture exact clinical markers required for specialized care pathway compliance automatically as vendors develop deep clinical logic customized for complex treatments.
Why is point-of-care decision support critical?
Chief medical officers prevent unauthorized procedures before patients ever leave examination rooms because systems alert physicians during order entry if selected treatments require authorization.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Solution Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Solution Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Solution Type , 2026 to 2036
- Workflow orchestration
- Attachment automation
- Rules discovery
- Status tracking
- Appeals routing
- Workflow orchestration
- Y to o to Y Growth Trend Analysis By Solution Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Solution Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Deployment Model
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Deployment Model, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Deployment Model, 2026 to 2036
- Cloud
- Hybrid
- On-premise
- Cloud
- Y to o to Y Growth Trend Analysis By Deployment Model, 2021 to 2025
- Absolute $ Opportunity Analysis By Deployment Model, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Buyer Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Buyer Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Buyer Type, 2026 to 2036
- Providers
- Payers
- Intermediaries
- Providers
- Y to o to Y Growth Trend Analysis By Buyer Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Buyer Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Clinical Scope
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Clinical Scope, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Clinical Scope, 2026 to 2036
- Medical services
- Pharmacy
- Specialty drugs
- Imaging
- Medical services
- Y to o to Y Growth Trend Analysis By Clinical Scope, 2021 to 2025
- Absolute $ Opportunity Analysis By Clinical Scope, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Core Function
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Core Function, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Core Function, 2026 to 2036
- Submission automation
- Criteria matching
- Document intake
- Decision support
- Workqueue analytics
- Submission automation
- Y to o to Y Growth Trend Analysis By Core Function, 2021 to 2025
- Absolute $ Opportunity Analysis By Core Function, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Integration Layer
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Integration Layer, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Integration Layer, 2026 to 2036
- FHIR APIs
- Portal connectors
- EHR embeds
- Clearinghouse links
- FHIR APIs
- Y to o to Y Growth Trend Analysis By Integration Layer, 2021 to 2025
- Absolute $ Opportunity Analysis By Integration Layer, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- By Country
- Market Attractiveness Analysis
- By Country
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- By Country
- Market Attractiveness Analysis
- By Country
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- By Country
- Market Attractiveness Analysis
- By Country
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- By Country
- Market Attractiveness Analysis
- By Country
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- By Country
- Market Attractiveness Analysis
- By Country
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- By Country
- Market Attractiveness Analysis
- By Country
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- By Country
- Market Attractiveness Analysis
- By Country
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Solution Type
- By Deployment Model
- By Buyer Type
- By Clinical Scope
- By Core Function
- By Integration Layer
- Competition Analysis
- Competition Deep Dive
- Availity
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Cohere Health
- Waystar
- Surescripts
- CoverMyMeds
- EviCore by Evernorth
- Oracle Health
- Availity
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Solution Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Buyer Type, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Clinical Scope, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Core Function, 2021 to 2036
- Table 7: Global Market Value (USD Million) Forecast by Integration Layer, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Solution Type , 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Buyer Type, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Clinical Scope, 2021 to 2036
- Table 13: North America Market Value (USD Million) Forecast by Core Function, 2021 to 2036
- Table 14: North America Market Value (USD Million) Forecast by Integration Layer, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Solution Type , 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Buyer Type, 2021 to 2036
- Table 19: Latin America Market Value (USD Million) Forecast by Clinical Scope, 2021 to 2036
- Table 20: Latin America Market Value (USD Million) Forecast by Core Function, 2021 to 2036
- Table 21: Latin America Market Value (USD Million) Forecast by Integration Layer, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Solution Type , 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 25: Western Europe Market Value (USD Million) Forecast by Buyer Type, 2021 to 2036
- Table 26: Western Europe Market Value (USD Million) Forecast by Clinical Scope, 2021 to 2036
- Table 27: Western Europe Market Value (USD Million) Forecast by Core Function, 2021 to 2036
- Table 28: Western Europe Market Value (USD Million) Forecast by Integration Layer, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Solution Type , 2021 to 2036
- Table 31: Eastern Europe Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 32: Eastern Europe Market Value (USD Million) Forecast by Buyer Type, 2021 to 2036
- Table 33: Eastern Europe Market Value (USD Million) Forecast by Clinical Scope, 2021 to 2036
- Table 34: Eastern Europe Market Value (USD Million) Forecast by Core Function, 2021 to 2036
- Table 35: Eastern Europe Market Value (USD Million) Forecast by Integration Layer, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: East Asia Market Value (USD Million) Forecast by Solution Type , 2021 to 2036
- Table 38: East Asia Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 39: East Asia Market Value (USD Million) Forecast by Buyer Type, 2021 to 2036
- Table 40: East Asia Market Value (USD Million) Forecast by Clinical Scope, 2021 to 2036
- Table 41: East Asia Market Value (USD Million) Forecast by Core Function, 2021 to 2036
- Table 42: East Asia Market Value (USD Million) Forecast by Integration Layer, 2021 to 2036
- Table 43: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: South Asia and Pacific Market Value (USD Million) Forecast by Solution Type , 2021 to 2036
- Table 45: South Asia and Pacific Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 46: South Asia and Pacific Market Value (USD Million) Forecast by Buyer Type, 2021 to 2036
- Table 47: South Asia and Pacific Market Value (USD Million) Forecast by Clinical Scope, 2021 to 2036
- Table 48: South Asia and Pacific Market Value (USD Million) Forecast by Core Function, 2021 to 2036
- Table 49: South Asia and Pacific Market Value (USD Million) Forecast by Integration Layer, 2021 to 2036
- Table 50: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 51: Middle East & Africa Market Value (USD Million) Forecast by Solution Type , 2021 to 2036
- Table 52: Middle East & Africa Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 53: Middle East & Africa Market Value (USD Million) Forecast by Buyer Type, 2021 to 2036
- Table 54: Middle East & Africa Market Value (USD Million) Forecast by Clinical Scope, 2021 to 2036
- Table 55: Middle East & Africa Market Value (USD Million) Forecast by Core Function, 2021 to 2036
- Table 56: Middle East & Africa Market Value (USD Million) Forecast by Integration Layer, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Solution Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Solution Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Solution Type
- Figure 6: Global Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Deployment Model
- Figure 9: Global Market Value Share and BPS Analysis by Buyer Type, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Buyer Type, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Buyer Type
- Figure 12: Global Market Value Share and BPS Analysis by Clinical Scope, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Clinical Scope, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Clinical Scope
- Figure 15: Global Market Value Share and BPS Analysis by Core Function, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Core Function, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Core Function
- Figure 18: Global Market Value Share and BPS Analysis by Integration Layer, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Integration Layer, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Integration Layer
- Figure 21: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 22: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 23: Global Market Attractiveness Analysis by Region
- Figure 24: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 29: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 30: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 31: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 32: North America Market Value Share and BPS Analysis by Solution Type , 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Solution Type , 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Solution Type
- Figure 35: North America Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Deployment Model
- Figure 38: North America Market Value Share and BPS Analysis by Buyer Type, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Buyer Type, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Buyer Type
- Figure 41: North America Market Value Share and BPS Analysis by Clinical Scope, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Clinical Scope, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Clinical Scope
- Figure 44: North America Market Value Share and BPS Analysis by Core Function, 2026 and 2036
- Figure 45: North America Market Y-o-Y Growth Comparison by Core Function, 2026-2036
- Figure 46: North America Market Attractiveness Analysis by Core Function
- Figure 47: North America Market Value Share and BPS Analysis by Integration Layer, 2026 and 2036
- Figure 48: North America Market Y-o-Y Growth Comparison by Integration Layer, 2026-2036
- Figure 49: North America Market Attractiveness Analysis by Integration Layer
- Figure 50: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 51: Latin America Market Value Share and BPS Analysis by Solution Type , 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Solution Type , 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Solution Type
- Figure 54: Latin America Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Deployment Model
- Figure 57: Latin America Market Value Share and BPS Analysis by Buyer Type, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Buyer Type, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Buyer Type
- Figure 60: Latin America Market Value Share and BPS Analysis by Clinical Scope, 2026 and 2036
- Figure 61: Latin America Market Y-o-Y Growth Comparison by Clinical Scope, 2026-2036
- Figure 62: Latin America Market Attractiveness Analysis by Clinical Scope
- Figure 63: Latin America Market Value Share and BPS Analysis by Core Function, 2026 and 2036
- Figure 64: Latin America Market Y-o-Y Growth Comparison by Core Function, 2026-2036
- Figure 65: Latin America Market Attractiveness Analysis by Core Function
- Figure 66: Latin America Market Value Share and BPS Analysis by Integration Layer, 2026 and 2036
- Figure 67: Latin America Market Y-o-Y Growth Comparison by Integration Layer, 2026-2036
- Figure 68: Latin America Market Attractiveness Analysis by Integration Layer
- Figure 69: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 70: Western Europe Market Value Share and BPS Analysis by Solution Type , 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Solution Type , 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Solution Type
- Figure 73: Western Europe Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Deployment Model
- Figure 76: Western Europe Market Value Share and BPS Analysis by Buyer Type, 2026 and 2036
- Figure 77: Western Europe Market Y-o-Y Growth Comparison by Buyer Type, 2026-2036
- Figure 78: Western Europe Market Attractiveness Analysis by Buyer Type
- Figure 79: Western Europe Market Value Share and BPS Analysis by Clinical Scope, 2026 and 2036
- Figure 80: Western Europe Market Y-o-Y Growth Comparison by Clinical Scope, 2026-2036
- Figure 81: Western Europe Market Attractiveness Analysis by Clinical Scope
- Figure 82: Western Europe Market Value Share and BPS Analysis by Core Function, 2026 and 2036
- Figure 83: Western Europe Market Y-o-Y Growth Comparison by Core Function, 2026-2036
- Figure 84: Western Europe Market Attractiveness Analysis by Core Function
- Figure 85: Western Europe Market Value Share and BPS Analysis by Integration Layer, 2026 and 2036
- Figure 86: Western Europe Market Y-o-Y Growth Comparison by Integration Layer, 2026-2036
- Figure 87: Western Europe Market Attractiveness Analysis by Integration Layer
- Figure 88: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Solution Type , 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Solution Type , 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Solution Type
- Figure 92: Eastern Europe Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 93: Eastern Europe Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 94: Eastern Europe Market Attractiveness Analysis by Deployment Model
- Figure 95: Eastern Europe Market Value Share and BPS Analysis by Buyer Type, 2026 and 2036
- Figure 96: Eastern Europe Market Y-o-Y Growth Comparison by Buyer Type, 2026-2036
- Figure 97: Eastern Europe Market Attractiveness Analysis by Buyer Type
- Figure 98: Eastern Europe Market Value Share and BPS Analysis by Clinical Scope, 2026 and 2036
- Figure 99: Eastern Europe Market Y-o-Y Growth Comparison by Clinical Scope, 2026-2036
- Figure 100: Eastern Europe Market Attractiveness Analysis by Clinical Scope
- Figure 101: Eastern Europe Market Value Share and BPS Analysis by Core Function, 2026 and 2036
- Figure 102: Eastern Europe Market Y-o-Y Growth Comparison by Core Function, 2026-2036
- Figure 103: Eastern Europe Market Attractiveness Analysis by Core Function
- Figure 104: Eastern Europe Market Value Share and BPS Analysis by Integration Layer, 2026 and 2036
- Figure 105: Eastern Europe Market Y-o-Y Growth Comparison by Integration Layer, 2026-2036
- Figure 106: Eastern Europe Market Attractiveness Analysis by Integration Layer
- Figure 107: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 108: East Asia Market Value Share and BPS Analysis by Solution Type , 2026 and 2036
- Figure 109: East Asia Market Y-o-Y Growth Comparison by Solution Type , 2026-2036
- Figure 110: East Asia Market Attractiveness Analysis by Solution Type
- Figure 111: East Asia Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 112: East Asia Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 113: East Asia Market Attractiveness Analysis by Deployment Model
- Figure 114: East Asia Market Value Share and BPS Analysis by Buyer Type, 2026 and 2036
- Figure 115: East Asia Market Y-o-Y Growth Comparison by Buyer Type, 2026-2036
- Figure 116: East Asia Market Attractiveness Analysis by Buyer Type
- Figure 117: East Asia Market Value Share and BPS Analysis by Clinical Scope, 2026 and 2036
- Figure 118: East Asia Market Y-o-Y Growth Comparison by Clinical Scope, 2026-2036
- Figure 119: East Asia Market Attractiveness Analysis by Clinical Scope
- Figure 120: East Asia Market Value Share and BPS Analysis by Core Function, 2026 and 2036
- Figure 121: East Asia Market Y-o-Y Growth Comparison by Core Function, 2026-2036
- Figure 122: East Asia Market Attractiveness Analysis by Core Function
- Figure 123: East Asia Market Value Share and BPS Analysis by Integration Layer, 2026 and 2036
- Figure 124: East Asia Market Y-o-Y Growth Comparison by Integration Layer, 2026-2036
- Figure 125: East Asia Market Attractiveness Analysis by Integration Layer
- Figure 126: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 127: South Asia and Pacific Market Value Share and BPS Analysis by Solution Type , 2026 and 2036
- Figure 128: South Asia and Pacific Market Y-o-Y Growth Comparison by Solution Type , 2026-2036
- Figure 129: South Asia and Pacific Market Attractiveness Analysis by Solution Type
- Figure 130: South Asia and Pacific Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 131: South Asia and Pacific Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 132: South Asia and Pacific Market Attractiveness Analysis by Deployment Model
- Figure 133: South Asia and Pacific Market Value Share and BPS Analysis by Buyer Type, 2026 and 2036
- Figure 134: South Asia and Pacific Market Y-o-Y Growth Comparison by Buyer Type, 2026-2036
- Figure 135: South Asia and Pacific Market Attractiveness Analysis by Buyer Type
- Figure 136: South Asia and Pacific Market Value Share and BPS Analysis by Clinical Scope, 2026 and 2036
- Figure 137: South Asia and Pacific Market Y-o-Y Growth Comparison by Clinical Scope, 2026-2036
- Figure 138: South Asia and Pacific Market Attractiveness Analysis by Clinical Scope
- Figure 139: South Asia and Pacific Market Value Share and BPS Analysis by Core Function, 2026 and 2036
- Figure 140: South Asia and Pacific Market Y-o-Y Growth Comparison by Core Function, 2026-2036
- Figure 141: South Asia and Pacific Market Attractiveness Analysis by Core Function
- Figure 142: South Asia and Pacific Market Value Share and BPS Analysis by Integration Layer, 2026 and 2036
- Figure 143: South Asia and Pacific Market Y-o-Y Growth Comparison by Integration Layer, 2026-2036
- Figure 144: South Asia and Pacific Market Attractiveness Analysis by Integration Layer
- Figure 145: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 146: Middle East & Africa Market Value Share and BPS Analysis by Solution Type , 2026 and 2036
- Figure 147: Middle East & Africa Market Y-o-Y Growth Comparison by Solution Type , 2026-2036
- Figure 148: Middle East & Africa Market Attractiveness Analysis by Solution Type
- Figure 149: Middle East & Africa Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 150: Middle East & Africa Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 151: Middle East & Africa Market Attractiveness Analysis by Deployment Model
- Figure 152: Middle East & Africa Market Value Share and BPS Analysis by Buyer Type, 2026 and 2036
- Figure 153: Middle East & Africa Market Y-o-Y Growth Comparison by Buyer Type, 2026-2036
- Figure 154: Middle East & Africa Market Attractiveness Analysis by Buyer Type
- Figure 155: Middle East & Africa Market Value Share and BPS Analysis by Clinical Scope, 2026 and 2036
- Figure 156: Middle East & Africa Market Y-o-Y Growth Comparison by Clinical Scope, 2026-2036
- Figure 157: Middle East & Africa Market Attractiveness Analysis by Clinical Scope
- Figure 158: Middle East & Africa Market Value Share and BPS Analysis by Core Function, 2026 and 2036
- Figure 159: Middle East & Africa Market Y-o-Y Growth Comparison by Core Function, 2026-2036
- Figure 160: Middle East & Africa Market Attractiveness Analysis by Core Function
- Figure 161: Middle East & Africa Market Value Share and BPS Analysis by Integration Layer, 2026 and 2036
- Figure 162: Middle East & Africa Market Y-o-Y Growth Comparison by Integration Layer, 2026-2036
- Figure 163: Middle East & Africa Market Attractiveness Analysis by Integration Layer
- Figure 164: Global Market - Tier Structure Analysis
- Figure 165: Global Market - Company Share Analysis

