AI Radiology Worklist Orchestration Market
The AI Radiology Worklist Orchestration Market Is Segmented By Component (Software Platforms, Implementation Services, Monitoring Services, Analytics Modules), Deployment (Cloud, Hybrid, On-Premise), Modality Focus (CT, X-Ray, MRI, Multi-Modality), Workflow Use Case (Triage Prioritization, Study Routing, Follow-Up Tracking, Escalation Alerts), End Use (Hospitals, Imaging Centers, Teleradiology Groups, Integrated Networks), And Region. Forecast For 2026 To 2036.
AI Radiology Worklist Orchestration Market Size, Market Forecast and Outlook By FMI
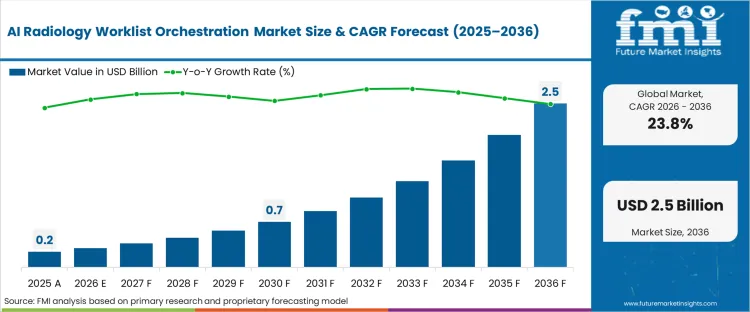
The AI radiology worklist orchestration market surpassed the estimation of USD 0.2 billion in 2025, continuing to escalate the industry revenue by reaching the assessed USD 0.3 billion in 2026 at a CAGR of 23.8% during the decade. Consistent market growth further influences the significant scale of upto USD 1.8 billion as reading backlogs exceed human diagnostic capacity, forcing hospital networks to automate study prioritization over simple chronologic queuing.
Radiology department heads face daily triage failures where critical intracranial hemorrhages wait behind routine outpatient knee scans purely based on chronological arrival. Teleradiology directors cannot simply hire more readers to clear weekend backlogs, because raw cross-sectional imaging volumes outpace graduating radiologist cohorts globally. Delays identifying positive acute findings translate directly into higher length-of-stay metrics and missed intervention windows, prompting clinical directors to prioritize solutions that reduce radiology backlog with AI. Purchasing committees evaluate radiology structured reporting automation not just for transcription speed, but to standardize how algorithmic flags populate preliminary text. What vendor pitches ignore is integrating an imaging AI orchestration layer often breaks existing hanging protocols, creating silent resistance from attending physicians who prioritize reading rhythm over algorithmic suggestions.
Once emergency departments mandate sub-hour turnaround times for all trauma scans, AI-powered radiology case prioritization shifts from an innovation budget line item to core operational requirement. Passing this threshold requires interoperability with legacy archiving environments, allowing positive study alerts to bypass standard lists entirely. Volume thresholds dictate adoption timing, where facilities processing over fifty thousand annual scans face catastrophic bottlenecking without a functional PACS RIS AI orchestration platform.
Summary of AI Radiology Worklist Orchestration Market
- AI Radiology Worklist Orchestration Market Definition
- Algorithmic study routing sits between scanners and radiologists, reordering reading queues based on automated pixel analysis rather than chronological arrival.
- Demand Drivers in the Market
- Shrinking reimbursement rates per scan compel imaging center directors to deploy radiology turnaround time reduction software to demand higher daily throughput from reading staff.
- Emergency department turnaround mandates force clinical informatics teams to bypass chronologic queuing for trauma cases.
- Sub-specialty reading requirements push teleradiology coordinators across this radiologist shortage workflow automation market to route complex scans automatically to available neuro-radiologists.
- Key Segments Analyzed in the FMI Report
- Software Platforms: 68.0% share in 2026, driven by continuous subscription revenue models overlaying legacy hospital IT infrastructure.
- Cloud: 47.0% share in 2026, reflecting off-site computational requirements for rapid deep learning inference.
- CT: 34.0% share in 2026, as high-slice-count trauma scans generate unsustainable manual review burdens.
- Triage Prioritization: 39.0% share in 2026, addressing immediate critical care bottlenecks in stroke and pulmonary embolism pathways.
- Hospitals: 52.0% share in 2026, possessing necessary capital budgets and scale to justify enterprise-wide algorithmic deployment.
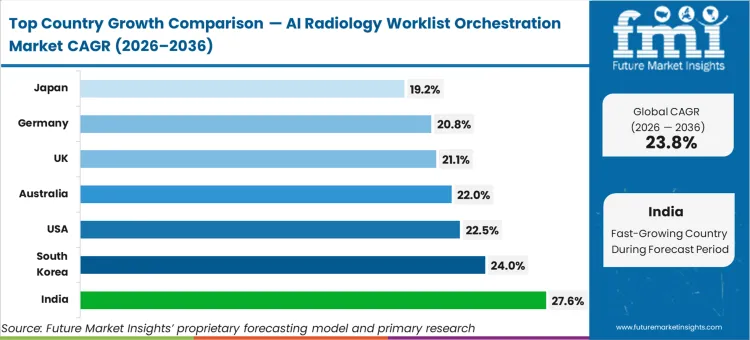
- India: 27.6% CAGR, reflecting rapid corporate hospital expansion seeking to centralize limited sub-specialist reading capacity.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Analyst, Healthcare, at FMI, observes that, "Hospital administrators assume funding algorithmic triage solves their turnaround time crisis. In reality, purchasing software creates severe bottlenecks downstream. Algorithm accuracy represents an easy metric to validate during procurement evaluations. Rewiring how fifty sub-specialized attending physicians actually accept, verify, and document those algorithmic flags is where deployments stall. Teleradiology networks buy these systems to protect margins, but individual readers often disable orchestration alerts because constant interruption breaks concentration on complex cases."
- Strategic Implications / Executive Takeaways
- PACS administrators must evaluate vendor APIs for bidirectional communication to ensure algorithmic flags actually alter native hanging protocols to capture true AI worklist prioritization ROI radiology.
- Teleradiology directors face immediate pressure to standardize orchestration layers across dispersed remote reading stations to maintain service level agreements.
- Corporate procurement teams risk massive sunk costs if purchased algorithms fail to integrate cleanly with incumbent dictation software platforms.
- Methodology
- PACS administrators, radiology department chairs, and clinical informatics directors.
- FDA 510(k) clearance databases, Integrating Healthcare Enterprise interoperability profiles, and software medical device registries.
- Annual cross-sectional imaging volumes requiring radiologist interpretation across defined healthcare networks.
- Vendor integration announcements with major enterprise imaging platforms cross-referenced against hospital procurement disclosures.
India tracks at 27.6% as acute specialist shortages outside tier-one urban centers force corporate hospital chains to centralize diagnostic functions. South Korea expands at 24.0% through heavy government subsidization targeting hospital digitalization initiatives, while United States facilities follow at 22.5% driven by aggressive private equity consolidation of outpatient imaging groups expanding North America AI radiology worklist orchestration market footprints. Australia advances at 22.0%, outpacing United Kingdom clinics at 21.1% and Germany at 20.8%, reflecting vast geographic disparities making algorithmic triage essential for rural health outposts. Japan grows at 19.2%, representing mature adoption curves where aging demographics fundamentally alter scan utilization rates across geriatric care networks.
AI Radiology Worklist Orchestration Market Definition
Algorithmic study routing sits between scanners and radiologists, reordering reading queues based on automated pixel analysis rather than chronological arrival. Buyers researching what is AI radiology worklist orchestration discover software categories analyzing incoming pixel data in background processes, detecting suspected critical abnormalities, and instantly pushing those specific studies to priority viewing status. Operational execution requires deep integration with existing picture archiving systems and radiology information system architectures.
AI Radiology Worklist Orchestration Market Inclusions
Core components cover triage prioritization algorithms, automated routing engines, and escalation alert modules embedded within diagnostic workflows. Integration encompasses cloud-hosted processing platforms, on-premise edge computing nodes, and hybrid deployment models managing DICOM data flow. Scope also contains specific AI radiology tool interfaces displaying confidence intervals and algorithmic bounding boxes directly to interpreting physicians accessing any radiology study routing platform.
AI Radiology Worklist Orchestration Market Exclusions
Diagnostic algorithms generating standalone clinical diagnoses without physician review fall outside scope, as they represent autonomous medical practice rather than orchestration. Enterprise resource planning systems managing billing or patient scheduling remain excluded because they lack pixel-level analysis capabilities. Hardware modalities, standalone dictation software, and basic chronologic queuing systems are omitted due to their inability to perform content-aware algorithmic triage.
AI Radiology Worklist Orchestration Market Research Methodology
- Primary Research: PACS administrators, radiology department chairs, and clinical informatics directors.
- Desk Research: FDA 510(k) clearance databases, Integrating Healthcare Enterprise interoperability profiles, and software medical device registries.
- Market-Sizing and Forecasting: Annual cross-sectional imaging volumes requiring radiologist interpretation across defined healthcare networks determining radiology worklist orchestration market size.
- Data Validation and Update Cycle: Vendor integration announcements with major enterprise imaging platforms cross-referenced against hospital procurement disclosures.
Segmental Analysis
AI Radiology Worklist Orchestration Market Analysis By Component
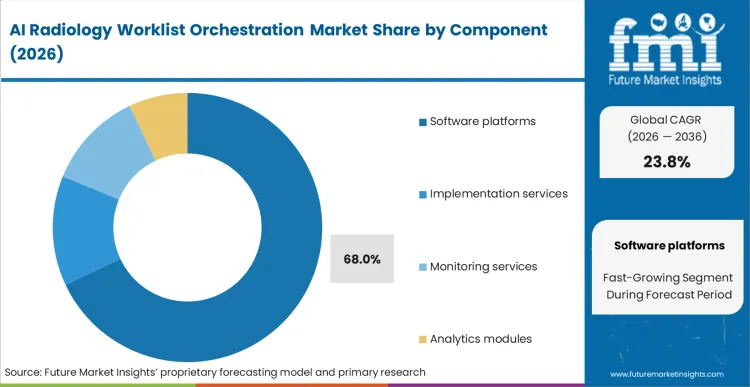
Hospital procurement teams evaluating orchestration investments weigh immediate deployment speed against long-term maintenance burdens. FMI's analysis indicates Software platforms command 68.0% share, reflecting preference for comprehensive digital overlays rather than piecemeal consulting engagements. PACS administrators install these central engines to intercept DICOM traffic before it reaches interpreting physicians, establishing single control points for seamless radiology AI workflow orchestration. Software interfaces must integrate cleanly with clinical workflow solutions to prevent duplicate data entry tasks. What vendor implementation specialists rarely document is that algorithm performance degrades significantly when installed across heterogenous scanner fleets producing variable image quality. This calibration gap explains why analytics modules struggle to gain traction independently; hospitals refuse to pay separately for performance dashboards that should be native to core software platforms. Department chairs who underinvest in continuous monitoring services face rapid algorithmic drift, resulting in alert fatigue that causes attending physicians to ignore automated prioritization entirely.
- False-Positive Fatigue: Uncalibrated software engines flag benign anomalies constantly. Attending physicians actively disable priority alerts, neutralizing expected efficiency gains completely.
- Workflow Fragmentation: Standalone triage screens force readers to toggle between applications. PACS administrators must write custom scripts to unify viewing experiences.
- Continuous Recalibration: Scanner protocol updates alter pixel distributions unpredictably. Clinical informatics teams must establish ongoing validation routines to maintain algorithmic sensitivity.
AI Radiology Worklist Orchestration Market Analysis By Deployment
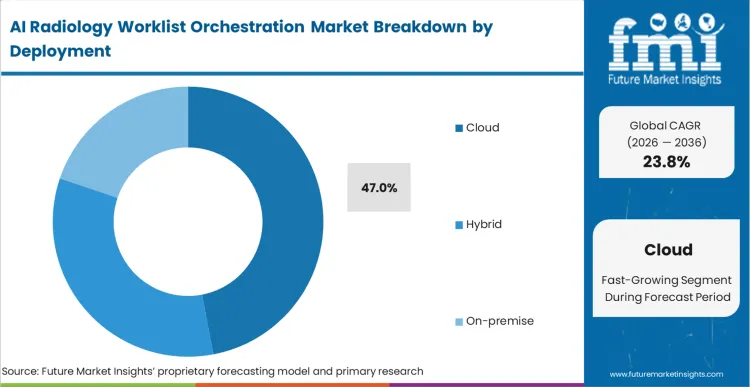
Capital expenditure constraints clash directly with data security mandates regarding patient health information. Cloud configurations capture 47.0% share, driven by massive computational demands inherent in deep learning inference models. Chief information officers at mid-sized networks transition away from on-premise server maintenance, preferring scalable remote processing that dynamically adjusts to weekend trauma spikes. Based on FMI's assessment, cloud architecture simplifies delivering crucial updates to healthcare AI computer vision algorithms without requiring physical site visits. What cloud adoption metrics obscure is severe latency issues plaguing facilities with subpar broadband infrastructure, making remote processing dangerous for stroke triage protocols demanding minute-level turnaround across any multi-vendor radiology AI platform market. Imaging center directors operating in rural territories face no choice but to absorb expensive hybrid deployment models, keeping critical inference engines on-site while offloading long-term data storage to remote servers.
- Infrastructure Overhead: Maintaining physical servers requires specialized IT personnel. Chief information officers eliminate these labor costs by migrating orchestration layers off-site.
- Latency Penalties: Substandard internet connectivity delays algorithmic inference returns. Stroke team coordinators experience missed intervention windows when cloud communication stalls.
- Data Egress Fees: Frequent retrieval of massive imaging files generates unpredictable operational costs. Financial directors demand capped-fee structures from cloud vendors before signing multi-year contracts.
AI Radiology Worklist Orchestration Market Analysis By Modality Focus
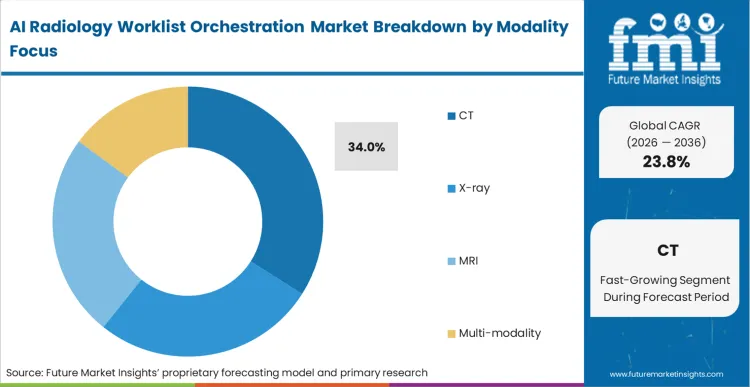
High-density pixel volumes generated during acute trauma evaluations create unsustainable manual review burdens. CT modalities hold 34.0% share because these massive datasets contain subtle indicators of catastrophic bleeds or embolisms easily missed during rushed initial passes. Emergency department coordinators rely on algorithmic pre-reading to bump positive CT findings above dozens of pending outpatient examinations. FMI observes that successful AI enabled medical devices focus overwhelmingly on cross-sectional imaging rather than flat planar x-rays due to higher reimbursement rates justifying software costs. What modality share figures mask is structural difficulty plaguing any multi-modality radiology AI workflow platform; algorithms trained on specific CT slice thicknesses fail completely when applied to variable MRI sequences. Radiology directors attempting to unify triage rules across all scanner types invariably experience catastrophic routing errors, forcing them to silo their orchestration tools by specific imaging hardware categories.
- Acute Triage Initiation: Stroke protocols require immediate intracranial evaluation. Emergency physicians rely on orchestration algorithms to bypass routine outpatient scan queues.
- Slice-Count Validation: Modern scanners produce thousands of images per study. Radiologists demand automated tools to highlight specific slices containing suspected pathologies.
- Subscription Expansion: Initial deployments target limited trauma pathways. Department chairs justify budget increases by expanding orchestration capabilities across routine oncology screening workflows.
AI Radiology Worklist Orchestration Market Analysis By Workflow Use Case
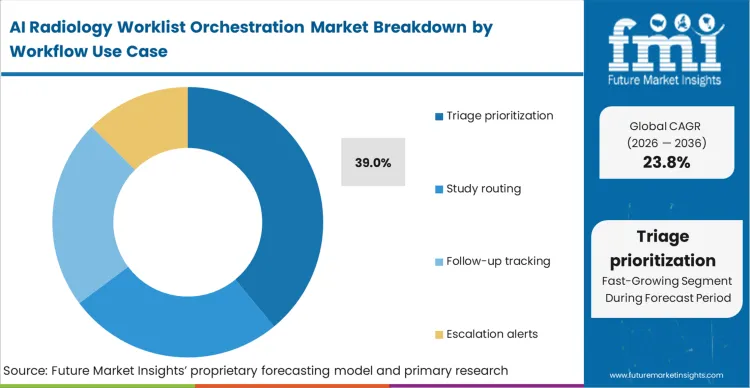
Legacy queuing models built on chronological arrival times fail catastrophically during mass casualty events or severe weekend staffing shortages. According to FMI's estimates, integrating clinical decision support logic directly into routing engines prevents misallocation of scarce sub-specialist time. The aspect that utilization metrics fail to reveal is that algorithmic prioritization completely destroys traditional productivity measuring systems; readers assigned exclusively to complex, AI-flagged cases appear significantly slower on paper than colleagues reading routine normal scans, complicating incidental findings follow-up orchestration radiology workflows. Practice managers who evaluate radiologist performance using legacy volume metrics penalize their most valuable sub-specialists, driving physician burnout across highly optimized triage environments. Triage prioritization captures 39.0% share by fundamentally rewriting how work flows to available physicians. Teleradiology coordinators utilize these tools to route suspected positive cases directly to specialized readers, ensuring critical findings receive immediate expert attention.
- Baseline Disruption: Chronological lists ignore clinical severity entirely. Teleradiology coordinators deploy algorithmic triage to identify life-threatening anomalies buried deep inside routine queues.
- Edge-Case Failure: Rare pathologies lacking sufficient training data bypass prioritization triggers. Attending physicians must maintain manual oversight to catch unusual presentations algorithms miss.
- Protocol Standardization: Unpredictable study routing creates liability risks. Practice managers must establish strict rules defining when physicians can override automated priority assignments.
AI Radiology Worklist Orchestration Market Analysis By End Use
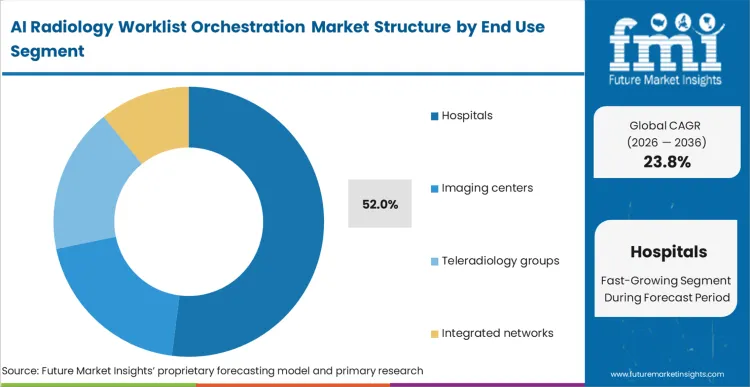
Massive daily study volumes combined with severe sub-specialist shortages make manual assignment processes functionally impossible. As per FMI's projection, digital healthcare initiatives mandate seamless integration between orchestration layers and enterprise electronic health records. The fact that hospital procurement data hides is extreme friction between central IT departments and decentralized reading groups; purchased software often sits dormant because private radiology practices contracted by hospitals refuse to alter their proprietary viewing systems. Integrated networks that fail to enforce standardized orchestration protocols across all affiliated clinics suffer massive data fragmentation, neutralizing expected efficiency gains entirely. Hospitals dominate with 52.0% share because their diverse clinical service lines demand sophisticated routing logic capable of distinguishing between trauma bay criticals and routine inpatient follow-ups. Chief medical officers approve these massive software expenditures to secure the best radiology AI workflow software for hospitals to reduce length-of-stay metrics tied directly to delayed diagnostic reports.
- Academic Pioneers: Research institutions possess necessary technical talent. Clinical informatics directors build custom integrations linking orchestration algorithms directly to proprietary viewer applications.
- Corporate Consolidation: Aggressive acquisition strategies create heterogenous IT environments. Enterprise imaging directors deploy unified orchestration layers to standardize routing across dozens of newly acquired clinics.
- Rural Capitulation: Isolated facilities lack specialized reading staff. Outpatient clinic managers adopt algorithmic triage out of desperation, relying heavily on remote teleradiology connections.
AI Radiology Worklist Orchestration Market Drivers, Restraints, and Opportunities
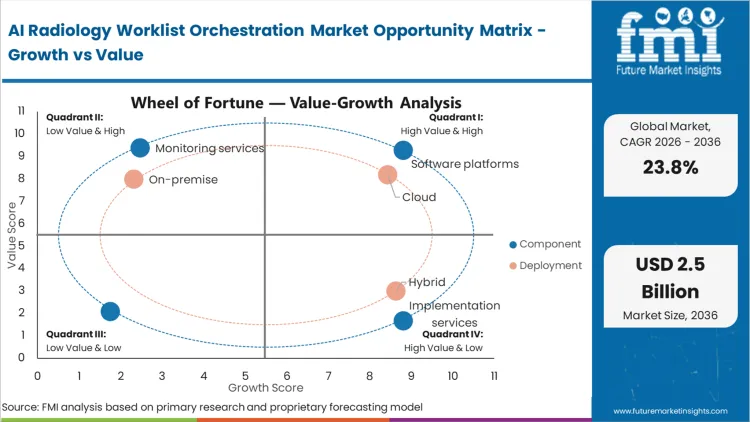
Shrinking reimbursement rates per scan compel imaging center directors to demand higher daily throughput from reading staff. This intense commercial pressure forces practice managers to eliminate every second wasted manually sorting through study lists or searching for prior comparative examinations, turning to worklist prioritization software for radiology. Teleradiology network executives cannot maintain profitability unless their readers operate at maximum efficiency, driving rapid adoption of teleradiology services integrated with intelligent routing engines capable of executing teleradiology workflow orchestration AI. Delays in deployment mean losing lucrative hospital contracts to competitors who guarantee sub-hour turnaround times for critical findings, making this technology mandatory for basic commercial survival.
Disparate legacy IT infrastructure prevents uniform algorithmic deployment across merged hospital networks. Enterprise imaging directors struggle constantly with outdated picture archiving systems lacking necessary application programming interfaces to accept external routing commands. This structural friction forces clinical informatics teams to build custom middleware for each individual hospital site, transforming rapid software deployments into multi-year integration nightmares. Interoperability protocols remain fragmented, capping adoption velocity even when capital budgets exist to fund current radiology orchestration software pricing models.
Opportunities in the AI Radiology Worklist Orchestration Market
- Middleware Bridge Deployment: Legacy archive systems lack modern communication standards. Enterprise imaging directors require imaging interoperability middleware to translate algorithmic routing commands into usable workflow alterations.
- Regulatory-Backed Acceleration: Purchasing committees demand proven safety profiles before authorizing massive deployments. Developers securing FDA-cleared radiology triage software market approvals instantly bypass prolonged security audits.
- Edge Computing Localization: Rural clinics face severe bandwidth limitations. Outpatient managers demand localized inference engines capable of prioritizing critical studies without requiring massive cloud data transfers.
Regional Analysis
.webp)
Based on regional analysis, AI Radiology Worklist Orchestration is segmented into North America, Europe, Asia Pacific, and others across 40 plus countries. Geographic constraints and specific localized funding mechanisms fundamentally dictate how algorithmic routing integrates across global health networks. Procurement patterns reveal a sharp divide between regions optimizing for sub-specialist scarcity versus those managing absolute imaging volumes.
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 27.6% |
| South Korea | 24.0% |
| United States | 22.5% |
| Australia | 22.0% |
| United Kingdom | 21.1% |
| Germany | 20.8% |
| Japan | 19.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America AI Radiology Worklist Orchestration Market Analysis
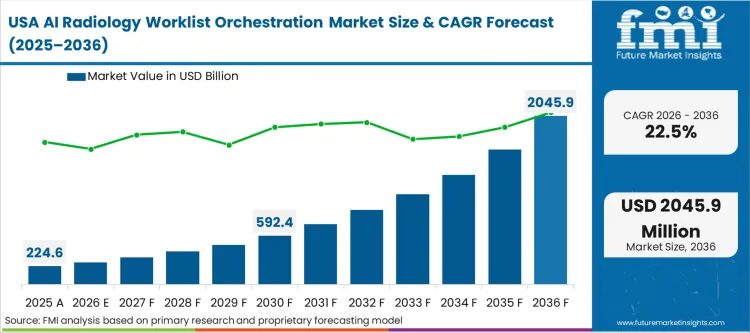
Corporate directors currently mandate unified teleradiology workflows across dozens of acquired clinics, demanding centralized orchestration engines to balance reading loads efficiently. Rigid compliance requirements surrounding patient data security push procurement teams toward highly secure, enterprise-grade routing platforms over fragmented startup solutions, shaping the broader US radiology AI orchestration market. Aggressive private equity consolidation of outpatient imaging centers ultimately dictates this technological adoption across North America.
- United States: Expanding at a 22.5% CAGR, massive conglomerates utilize algorithmic triage to maintain strict service level agreements across acquired facilities. Private equity buyouts force disparate clinics onto unified IT networks, prompting enterprise imaging directors to deploy orchestration software that routes scans efficiently to centralized reading hubs. Successful deployment allows corporate entities to maximize radiologist utilization, directly improving bottom-line profitability.
FMI's report includes Canada. Complex cross-border data residency regulations force vendors to establish localized cloud instances before securing major hospital contracts.
Europe AI Radiology Worklist Orchestration Market Analysis
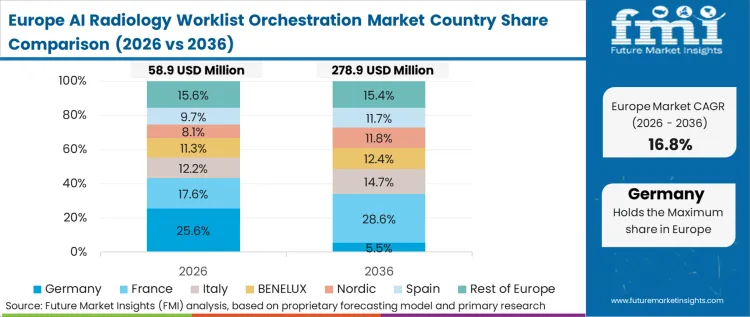
Clinical informatics directors operating within constrained public budgets prioritize orchestration tools demonstrating immediate reductions in patient length-of-stay metrics, accelerating the Europe radiology workflow AI market. Stringent data privacy frameworks heavily penalize compliance failures, forcing vendors to prove absolute algorithmic transparency during public tenders. Nationalized health systems grappling with severe radiologist shortages implement algorithmic triage specifically to manage these expanding imaging backlogs.
- United Kingdom: Trust administrators adopt intelligent routing to ensure suspected cancer cases bypass massive chronological queues, expanding UK radiology workflow automation AI footprints. Regional adoption depends heavily on proving orchestration tools meet strict clinical safety standards, fueling growth at 21.1% over the forecast period. European competitors must secure specific regional certifications to access these lucrative public procurement channels.
- Germany: Department chairs implement cross-network orchestration routing to balance diagnostic workloads when decentralized regional hospital networks struggle to share sub-specialist reading capacity. Transitioning away from legacy systems introduces significant workflow disruptions attending physicians actively resist, thereby, supporting the expansion of CAGR at 20.8%, requiring deep integration with existing medical imaging software deeply entrenched within regional clinics.
FMI's report includes France and Italy. Heavy unionization among public healthcare workers necessitates extensive change management consulting before orchestration algorithms can legally alter established work routines.
Asia Pacific AI Radiology Worklist Orchestration Market Analysis
Corporate hospital chains actively deploy orchestration engines to route complex scans from remote outposts directly to centralized expert teams. Successful vendors must navigate highly variable broadband infrastructure, requiring robust oncology imaging software connections capable of functioning during frequent network interruptions. Vast geographic disparities separating hyper-modern urban medical centers from severely under-resourced rural clinics define this territory, prompting analysts to monitor which countries are adopting radiology workflow AI fastest.
- India: Tracking a 27.6% of CAGR, rapid adoption allows urban specialists to service dozens of rural spoke clinics simultaneously. Network executives deploy algorithmic triage to funnel critical neuro-imaging studies toward limited expert reading pools because corporate hospital expansion outpaces sub-specialist training rates drastically. Securing major corporate contracts grants vendors massive data access crucial for training region-specific deep learning models and fueling the India radiology AI workflow market.
- South Korea: Chief information officers replace outdated chronological lists with sophisticated orchestration engines following government initiatives subsidizing digital healthcare transformation. Aggressive digitalization completely alters how physicians interact with diagnostic imaging platforms daily, pushing demand forward at 24.0% annually. Massive capital injections for hospital IT upgrades enable this rapid architectural shift.
- Australia: Teleradiology coordinators utilize algorithmic prioritization to manage massive overnight trauma volumes from remote mining towns. Massive physical distances separating patients from diagnostic specialists make intelligent routing mandatory rather than optional. Sustaining a CAGR of 22.0%, successful orchestration relies heavily on seamless integration with national electronic health record systems.
- Japan: Department chairs deploy orchestration tools to filter massive volumes of normal scans away from highly paid sub-specialists as super-aging demographics generate immense quantities of routine geriatric imaging. Mature infrastructure allows rapid deployment of software upgrades across established hospital networks, registering a 19.2% CAGR trajectory. Mature adoption curves indicate a structural shift toward highly specialized algorithms targeting specific disease pathways.
FMI's report includes China and Southeast Asia. Strict data localization laws prevent external vendors from processing pixel data on international cloud servers, demanding dedicated localized infrastructure.
Competitive Aligners for Market Players
Algorithmic accuracy no longer guarantees commercial success; seamless integration into legacy hospital IT architecture dictates vendor survival. AIdoc and Viz.AI dominate procurement discussions by demonstrating functional bidirectional communication with existing viewing platforms, eliminating disruptive workflow toggling. Purchasing committees dismiss standalone point solutions lacking comprehensive vendor neutral archive compatibility, refusing to manage dozens of disparate software interfaces when they compare radiology triage AI platforms for hospitals. This dynamic force radiology AI vendors to prioritize interoperability over pure diagnostic sensitivity, especially when evaluating AIdoc vs viz AI radiology capabilities.
Incumbents possess massive libraries of real-world workflow data, allowing them to anticipate and solve hanging protocol conflicts before implementation begins, answering who are the leading companies in AI radiology worklist orchestration. Qure.AI and Harrison.AI leverage deep partnerships with dominant archiving vendors, ensuring their routing commands actually execute within native physician viewing screens. Challengers lacking these structural partnerships face immense resistance from hospital IT departments unwilling to build custom integration bridges for unproven algorithms within the radiology triage software market.
Large teleradiology networks resist vendor lock-in by demanding modular orchestration architectures supporting third-party algorithms. Gleamer and deepc secure contracts by offering open platform designs, allowing corporate directors to swap out underperforming triage models without replacing entire routing engines. Structural tension between closed proprietary ecosystems and open interoperability standards shapes procurement strategy, as massive imaging conglomerates evaluate radiology workflow automation vendors to avoid surrendering workflow control to single algorithm developers.
Key Players in AI Radiology Worklist Orchestration Market
- Aidoc
- Viz.AI
- Qure.AI
- Harrison.AI
- Gleamer
- deepc
- Blackford
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 0.3 billion to USD 1.8 billion, at a CAGR of 23.8% |
| Market Definition | Algorithmic study routing sits between scanners and radiologists, reordering reading queues based on automated pixel analysis rather than chronological arrival. |
| Segmentation | Component, Deployment, Modality focus, Workflow use case, End user, and Region |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | United States, Canada, Brazil, Mexico, Germany, United Kingdom, France, Italy, Spain, China, Japan, South Korea, India, ASEAN, Australia, New Zealand, GCC, South Africa |
| Key Companies Profiled | Aidoc, Viz.AI, Qure.AI, Harrison.AI, Gleamer, deepc, Blackford |
| Forecast Period | 2026 to 2036 |
| Approach | Annual cross-sectional imaging volumes requiring radiologist interpretation across defined healthcare networks |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Key Segments:
Component
- Software platforms
- Implementation services
- Monitoring services
- Analytics modules
Deployment
- Cloud
- Hybrid
- On-premise
Modality Focus
- CT
- X-ray
- MRI
- Multi-modality
Workflow Use Case
- Triage prioritization
- Study routing
- Follow-up tracking
- Escalation alerts
End Use
- Hospitals
- Imaging centers
- Teleradiology groups
- Integrated networks
Regions:
- North America & Latin America
- United States
- Canada
- Mexico
- Brazil
- Argentina
- Chile
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Asia-Pacific
- China
- Japan
- South Korea
- India
- ASEAN
- ANZ
- Middle East & Africa
- UAE
- Saudi Arabia
- South Africa
Bibliography
- Imaging Technology News. (2025, May 6). NewVue.ai, MD.AI announce integration partnership for cloud-native radiology workflow orchestration.
- Tejani, A. S., Cook, T. S., Allen, B., Chokshi, F. H., Dreyer, K. J., Lee, C. I., Nicolson, N. G., & Rosenthal, D. I. (2024). Integrating and adopting AI in the radiology workflow: A primer for standards and Integrating the Healthcare Enterprise (IHE) profiles. Radiology, 312(1), e232653.
- Gu, Z., Dogra, S., Siriruchatanon, M., Tay, K., Bhargavan-Chatfield, M., Cook, T. S., & Tejani, A. S. (2026). Radiology workflow assistance with artificial intelligence: Establishing the link to outcomes. Journal of the American College of Radiology, 23(3), 389-398.
- Windecker, D., Baj, G., Shiri, I., Khera, R., Trivedi, H., Kohli, M. D., Liu, Y., & Topol, E. J. (2025). Generalizability of FDA-approved AI-enabled medical devices for clinical use. JAMA Network Open, 8(4), e258052.
- Dean, G., Park, S. H., & Geis, J. R. (2025). Real-world monitoring of artificial intelligence in radiology. Korean Journal of Radiology, 26(10).
- Christensen, E. W., Canelo-Aybar, C., Abbott, J. G., & Hughes, D. R. (2025). Projected US radiologist supply, 2025 to 2055. Journal of the American College of Radiology, 22(4), 592-600.
- Zhang, K., Allen, B., Dreyer, K. J., & Wu, C. C. (2024). FDA review of radiologic AI algorithms: Process and policy. Radiology, 311(2), e230242.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Emergency department turnaround mandates forcing clinical informatics teams to bypass chronologic queuing for trauma cases.
- Legacy IT infrastructure preventing uniform algorithmic deployment across merged hospital networks.
- Algorithmic triage shifting from an innovation budget line item to core operational requirement.
- False-positive fatigue causing attending physicians to actively disable priority alerts.
- Data egress fees generating unpredictable operational costs for mid-sized networks.
- Multi-modality orchestration structural difficulties creating catastrophic routing errors.
- Teleradiology coordinators utilizing tools to route suspected positive cases directly to specialized readers.
- Massive physical distances separating patients from diagnostic specialists making intelligent routing mandatory.
Frequently Asked Questions
What is AI radiology worklist orchestration?
Algorithmic study routing sits between scanners and radiologists, reordering reading queues based on automated pixel analysis rather than chronological arrival to push specific studies to priority viewing status.
How does radiology worklist prioritization software work?
Software engines intercept DICOM traffic before it reaches interpreting physicians, analyzing incoming pixel data in background processes to detect critical abnormalities and instantly push those flagged studies to the top of viewing lists.
Why are hospitals adopting radiology workflow AI tools?
Chief medical officers approve these massive software expenditures to alleviate severe specialist burnout and reduce length-of-stay metrics tied directly to delayed diagnostic reports amid shrinking reimbursement rates and exploding scan volumes.
Which vendors lead the AI radiology worklist orchestration market?
Aidoc, Viz.AI, Qure.AI, and Harrison.AI dominate current procurement discussions by demonstrating functional bidirectional communication with existing viewing platforms, while challengers like Gleamer and deep secure contracts by offering open platform designs.
How is FDA clearance shaping this market?
Developers securing regulatory approvals instantly bypass prolonged security audits, while those lacking clearance face impossible barriers competing for lucrative hospital contracts because purchasing committees demand proven safety profiles before authorizing massive deployments.
Which imaging modalities use worklist AI most?
High-density CT modalities hold the largest share because emergency department coordinators rely on algorithmic pre-reading to rapidly bump positive findings containing subtle indicators of catastrophic bleeds easily missed during rushed initial passes.
What is the difference between triage AI and reporting AI in radiology?
Triage algorithms reorder viewing queues based on suspected abnormalities to accelerate expert review, whereas reporting AI autonomously generates standalone clinical diagnoses or populates preliminary text fields.
How much does radiology orchestration software typically cost?
Facilities pay ongoing subscription fees layered upon initial integration expenses required to build custom middleware bridges, with pricing models varying wildly depending on institutional scale and modality coverage.
Which countries are growing fastest in radiology workflow AI?
India tracks at a 27.6% CAGR as corporate hospital chains centralize diagnostic functions, while South Korea expands at 24.0% through heavy government subsidization targeting hospital digitalization initiatives.
What ROI do hospitals seek from AI worklist orchestration?
Administrators demand immediate reductions in emergency department turnaround times to maximize sub-specialist reading efficiency, directly improving bottom-line profitability while minimizing expensive malpractice liabilities associated with missed critical findings.
What limits widespread adoption of cloud-based algorithmic routing?
Severe latency issues plague facilities with subpar broadband infrastructure, delaying algorithmic inference returns and causing stroke team coordinators to experience missed intervention windows when cloud communication stalls.
Why do analytics modules struggle to gain independent traction?
Department chairs demand integrated analytics within base subscriptions, treating standalone tracking modules as unnecessary administrative overhead rather than paying separately for performance dashboards that should be native to core software platforms.
How does algorithmic prioritization disrupt traditional performance metrics?
Practice managers evaluating radiologist performance using legacy volume metrics penalize their most valuable sub-specialists because readers assigned exclusively to complex, AI-flagged cases appear significantly slower on paper than colleagues reading routine normal scans.
Why do multi-modality orchestration tools face structural resistance?
Radiology directors attempting to unify triage rules across all scanner types invariably experience catastrophic routing errors because algorithms trained on specific CT slice thicknesses fail completely when applied to variable MRI sequences.
What specific operational consequence do hospitals face from false-positive fatigue?
Uncalibrated software engines flag benign anomalies constantly, causing attending physicians to actively disable priority alerts, which completely neutralizes expected efficiency gains and wastes expensive software subscriptions.
What role do teleradiology networks play in algorithmic deployment?
Teleradiology coordinators utilize algorithmic prioritization to manage massive overnight trauma volumes from remote clinics by routing suspected positive cases directly to specialized readers to ensure critical findings receive immediate expert attention.
Why do mid-sized networks prefer cloud deployment configurations?
Chief information officers eliminate specialized IT personnel costs by migrating orchestration layers off-site, leveraging cloud architecture to simplify delivering crucial algorithm updates without requiring physical site visits.
What operational risk do standalone triage screens introduce?
Fragmented workflows force readers to toggle between applications, creating dangerous distractions during complex case reviews that force PACS administrators to write custom scripts to unify viewing experiences.
How do slice-count volumes impact software requirements?
Because modern scanners produce thousands of images per study that overwhelm manual review capacity, radiologists demand automated tools highlighting specific slices containing suspected pathologies to accelerate reporting.
Why do enterprise imaging directors require interoperability middleware?
Middleware translates algorithmic routing commands into usable workflow alterations without requiring expensive wholesale PACS replacements when integrating with legacy archive systems that lack modern communication standards.
What specific workflow changes when orchestration algorithms identify critical findings?
Software instantly pushes suspected positive studies to priority viewing status, bypassing chronological arrival lists to deliver immediate alerts to radiologists within their native viewing applications.
Why do isolated rural clinics adopt algorithmic triage out of desperation?
Outpatient clinic managers completely lacking specialized reading staff rely heavily on remote teleradiology connections augmented by intelligent routing to safely manage acute trauma cases.
What structural friction prevents uniform deployment across merged hospital networks?
Disparate legacy IT infrastructure prevents standard communication protocols, forcing clinical informatics teams to build custom middleware for each individual hospital site and transforming rapid software deployments into multi-year integration nightmares.
How do corporate consolidation strategies alter IT requirements?
Because aggressive acquisitions create heterogenous IT environments, enterprise imaging directors must deploy unified orchestration layers to standardize routing across dozens of newly acquired clinics.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Component
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Component , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Component , 2026 to 2036
- Software platforms
- Implementation services
- Monitoring services
- Analytics modules
- Software platforms
- Y to o to Y Growth Trend Analysis By Component , 2021 to 2025
- Absolute $ Opportunity Analysis By Component , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Deployment
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Deployment, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Deployment, 2026 to 2036
- Cloud
- Hybrid
- On-premise
- Cloud
- Y to o to Y Growth Trend Analysis By Deployment, 2021 to 2025
- Absolute $ Opportunity Analysis By Deployment, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Modality Focus
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Modality Focus, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Modality Focus, 2026 to 2036
- CT
- X-ray
- MRI
- Multi-modality
- CT
- Y to o to Y Growth Trend Analysis By Modality Focus, 2021 to 2025
- Absolute $ Opportunity Analysis By Modality Focus, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Workflow Use Case
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Workflow Use Case, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Workflow Use Case, 2026 to 2036
- Triage prioritization
- Study routing
- Follow-up tracking
- Escalation alerts
- Triage prioritization
- Y to o to Y Growth Trend Analysis By Workflow Use Case, 2021 to 2025
- Absolute $ Opportunity Analysis By Workflow Use Case, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Hospitals
- Imaging centers
- Teleradiology groups
- Integrated networks
- Hospitals
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Component
- By Deployment
- By Modality Focus
- By Workflow Use Case
- By End Use
- Competition Analysis
- Competition Deep Dive
- Aidoc
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Viz.AI
- Qure.AI
- Harrison.AI
- Gleamer
- deepc
- Blackford
- Aidoc
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Modality Focus, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Workflow Use Case, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Modality Focus, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Workflow Use Case, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Modality Focus, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Workflow Use Case, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Modality Focus, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Workflow Use Case, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Modality Focus, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Workflow Use Case, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Modality Focus, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Workflow Use Case, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Modality Focus, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Workflow Use Case, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Modality Focus, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Workflow Use Case, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Component
- Figure 6: Global Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Deployment
- Figure 9: Global Market Value Share and BPS Analysis by Modality Focus, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Modality Focus, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Modality Focus
- Figure 12: Global Market Value Share and BPS Analysis by Workflow Use Case, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Workflow Use Case, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Workflow Use Case
- Figure 15: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by End Use
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Component
- Figure 32: North America Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Deployment
- Figure 35: North America Market Value Share and BPS Analysis by Modality Focus, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Modality Focus, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Modality Focus
- Figure 38: North America Market Value Share and BPS Analysis by Workflow Use Case, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Workflow Use Case, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Workflow Use Case
- Figure 41: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by End Use
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Component
- Figure 48: Latin America Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Deployment
- Figure 51: Latin America Market Value Share and BPS Analysis by Modality Focus, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Modality Focus, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Modality Focus
- Figure 54: Latin America Market Value Share and BPS Analysis by Workflow Use Case, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Workflow Use Case, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Workflow Use Case
- Figure 57: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by End Use
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Component
- Figure 64: Western Europe Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Deployment
- Figure 67: Western Europe Market Value Share and BPS Analysis by Modality Focus, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Modality Focus, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Modality Focus
- Figure 70: Western Europe Market Value Share and BPS Analysis by Workflow Use Case, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Workflow Use Case, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Workflow Use Case
- Figure 73: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by End Use
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Component
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Deployment
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Modality Focus, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Modality Focus, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Modality Focus
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Workflow Use Case, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Workflow Use Case, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Workflow Use Case
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Component
- Figure 96: East Asia Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Deployment
- Figure 99: East Asia Market Value Share and BPS Analysis by Modality Focus, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Modality Focus, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Modality Focus
- Figure 102: East Asia Market Value Share and BPS Analysis by Workflow Use Case, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Workflow Use Case, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Workflow Use Case
- Figure 105: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by End Use
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Component
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Deployment
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Modality Focus, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Modality Focus, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Modality Focus
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Workflow Use Case, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Workflow Use Case, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Workflow Use Case
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Component
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Deployment
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Modality Focus, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Modality Focus, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Modality Focus
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Workflow Use Case, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Workflow Use Case, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Workflow Use Case
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

