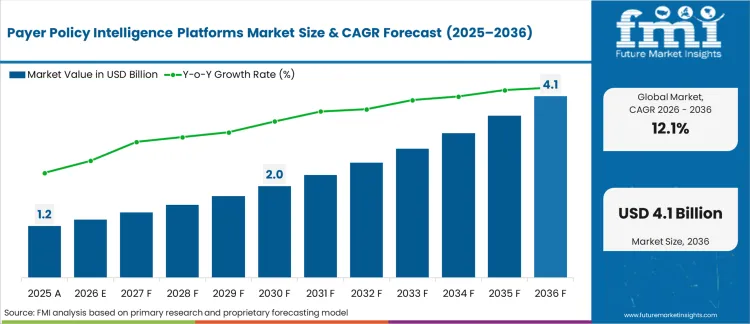
The payer policy intelligence platforms market was valued at USD 1.2 billion in 2025 and is projected to reach USD 1.3 billion in 2026, reflecting a CAGR of 12.06%. Industry is expected to grow to USD 4.1 billion by 2036, driven by the adoption of NLP‑enabled systems that streamline the interpretation of complex commercial and Medicare policies and reduce delays in market access processes.
Transitioning from manual PDF scraping to algorithmic policy tracking forces Pharmaceutical Market Access Directors to upgrade their commercialization infrastructure. Relying on static policy reviews creates massive blind spots when daily medical policy updates exceed manual review capacity. Market access teams failing to track these changes lose peak product revenue as favorable coverage criteria revert or tighten without their knowledge. The critical failure point is mapping ambiguous clinical criteria rather than just extracting raw policy text. Commercial leaders integrating these engines with healthcare data integration architectures bypass traditional operational silos.
Algorithmic tracking achieves self-sustaining adoption once market access teams must map concurrent real-time criteria changes across 500+ commercial plans simultaneously to maintain Tier 2 formulary status. Pharma launch coordinators must link their patient assistance programs to dynamic policy feeds before post-launch commercialization windows close. This forces an immediate migration away from ad-hoc consultant reports toward continuous API-fed intelligence platforms.
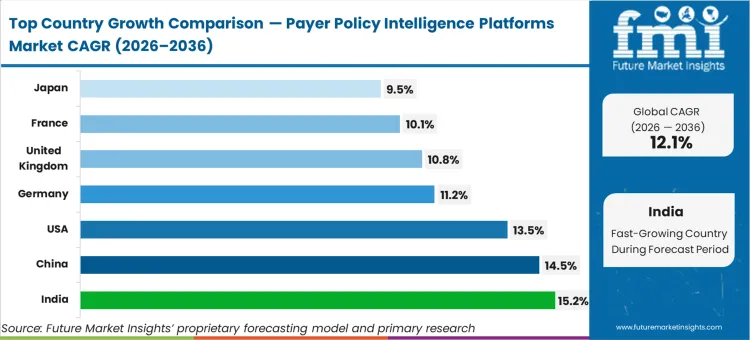
India tracks 15.2% compound growth, driven by Ayushman Bharat Digital Mission mandates requiring unstructured coverage data mapping for national registries. China advances at 14.5% as provincial reimbursement criteria consolidate under centralised negotiation frameworks. The United States follows at 13.5% through 2036. Germany records an 11.2% rate, while the United Kingdom expands at 10.8%. France and Japan post growth of 10.1% and 9.5% respectively. This structural spread exists because single-payer systems require localized policy linkage, whereas multi-payer commercial markets force exponentially higher ingestion volume to maintain baseline revenue cycle visibility.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 1.3 billion |
| Industry Value (2036) | USD 4.1 billion |
| CAGR (2026-2036) | 12.06% |
The Payer Policy Intelligence Platforms Market encompasses software systems and managed services that autonomously ingest, interpret, and track medical policies, formulary updates, and reimbursement criteria from public and commercial health insurers. These platforms convert unstructured payer documents into structured, queryable databases. The market is strictly separated from general clinical trial management tools by its exclusive focus on post-approval coverage rules, billing codes, and health technology assessment tracking.
Scope includes AI-driven policy document extraction tools, real-time formulary status dashboards, prior authorization criteria monitors, and Medicare Local Coverage Determination (LCD) tracking modules. Natural language processing engines built specifically for medical necessity interpretation fall within this boundary. Systems that map payer requirements directly to specialized medical billing software frameworks are included, alongside subscription-based policy alert services designed for medical device commercialization teams.
General electronic health record (EHR) systems and patient-facing claim dispute portals are explicitly excluded. Contract lifecycle management systems that handle payer-provider rate negotiations but do not interpret clinical coverage policies are outside scope. Clinical decision support systems used purely for diagnostic assistance, without integrating insurer-specific reimbursement rules, do not qualify for this market analysis.
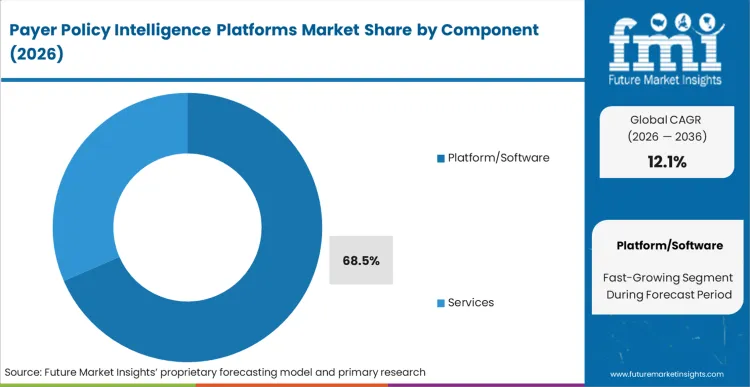
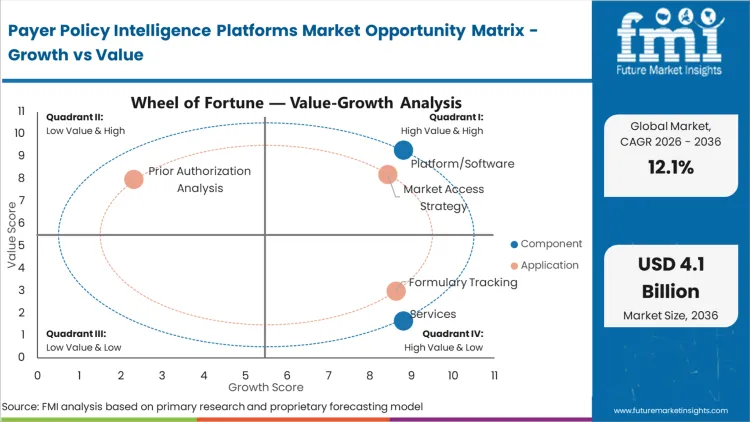
The replacement of slow, manual consultant-led policy audits with persistent NLP tracking systems accelerates adoption across the life sciences sector. Platform/Software holds 68.5% share in 2026 due to the sheer volume of daily policy bulletins, which far exceeds human processing capacity. According to FMI's estimates, vendors embedding clinical natural language processing directly into market access dashboards create an indispensable technical advantage. Market access directors must transition from project-based service contracts to continuous software subscriptions to avoid fatal launch delays caused by outdated coverage assumptions.
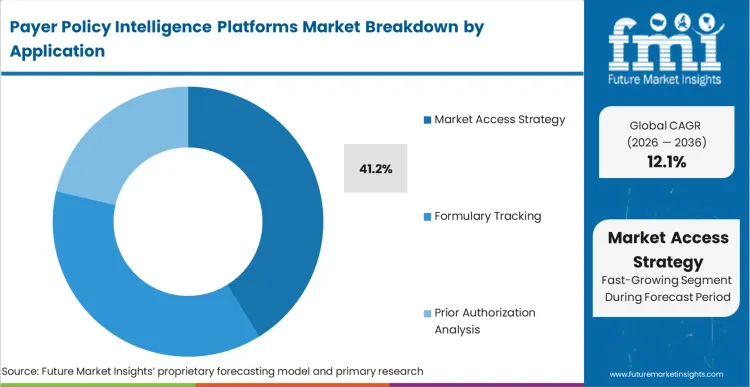
The operational gap between securing regulatory approval and actually navigating complex reimbursement pathways drives demand for specialised modelling. Market Access Strategy leads with 41.2% share in 2026, as drug manufacturers rely on these tools to secure and defend Tier 2 or Tier 3 formulary placements.
As per FMI's projection, the ability to simulate the financial impact of a competitor's favorable policy change forces companies to invest heavily in strategic modelling platforms. Incorporating these insights into broader revenue cycle management strategies allows organizations to anticipate cash flow disruptions. Commercial launch leaders must validate payer criteria constraints prior to deploying expensive field sales forces or face devastating budget overruns.
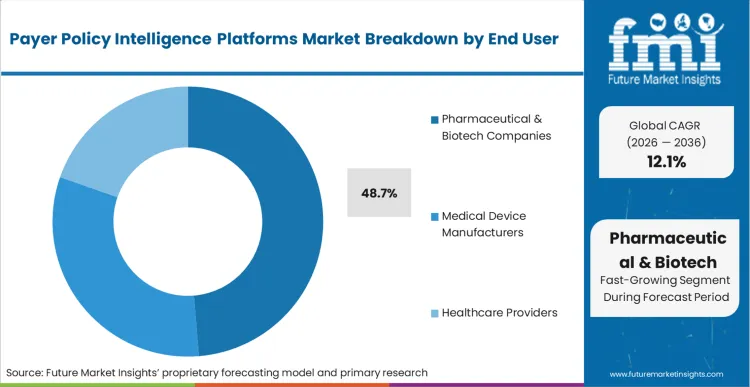
The rigid compliance threshold surrounding specialty drug reimbursement dictates technology procurement for major drug developers. Pharmaceutical & Biotech Companies capture 48.7% share in 2026 because their high-margin portfolios are disproportionately targeted by aggressive payer cost-containment measures. FMI analysts opine that integrating these intelligence feeds with emerging digital therapeutics commercialization efforts will become a standard industry requirement. Pharma launch executives must deploy comprehensive policy surveillance to protect peak product revenues from sudden, unannounced criteria restrictions.
The CMS Interoperability and Prior Authorization Final Rule (CMS-0057-F) forces hospital revenue cycle teams to implement automated policy tracking architectures. This regulatory mandate creates an urgent operational deadline, as manual denial management becomes mathematically unsustainable under the new data-sharing requirements. Health systems that adopt NLP-driven intelligence platforms drastically reduce their write-off ratios by aligning physician orders with precise, real-time payer criteria. Operations vice presidents who ignore this technical shift face catastrophic claim denial rates and subsequent cash flow paralyzation.
Heavy fragmentation in regional payer terminology creates severe algorithmic friction. Small health plans frequently use non-standardized language for medical necessity, which confuses baseline NLP models trained strictly on Medicare standards. To circumvent this, vendors deploy human-in-the-loop validation teams, significantly increasing the cost of goods sold and limiting the scalability of pure-play software models in highly localized markets.
Based on the regional analysis, the Payer Policy Intelligence Platforms market is segmented into North America, Latin America, Europe, East Asia, South Asia & Pacific, and Middle East & Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 15.2% |
| China | 14.5% |
| United States | 13.5% |
| Germany | 11.2% |
| United Kingdom | 10.8% |
| France | 10.1% |
| Japan | 9.5% |
Specific policy mandates driven by national digital health infrastructure overhauls dictate this region's aggressive platform adoption. Integrating fragmented private insurer data with emerging centralized registries forces local technology vendors to build sophisticated data ingestion engines. Based on FMI's assessment, the push for standardized health records requires deep linkage between policy text and population health management frameworks to ensure broad clinical accessibility.
FMI's report includes comprehensive evaluation of Australia, Indonesia, and Malaysia. The rapid proliferation of private micro-insurance models across these nations accelerates the need for hyper-localized policy tracking algorithms.
Centralized procurement practices and aggressive state-negotiated reimbursement criteria characterize the East Asian market structure. The sheer volume of population-level formulary adjustments requires life sciences companies to utilize real-time surveillance to protect their contracted margins. In FMI's view, pharmaceutical manufacturers operating here cannot rely on legacy consulting models due to the rapid execution timelines mandated by provincial health authorities.
FMI's report includes detailed analysis of South Korea and Taiwan. Both countries utilize strict health technology assessment (HTA) gateways, requiring advanced policy tracking for successful market entry.
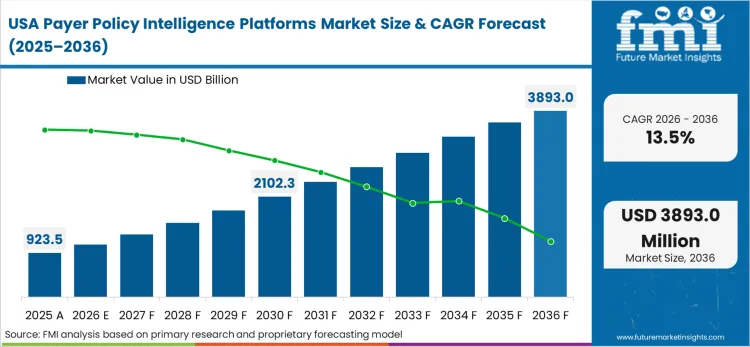
Complex multi-payer economics and erratic commercial coverage variations create the highest baseline demand for policy tracking globally. The structural necessity to monitor thousands of disparate health plan bulletins simultaneously makes manual oversight impossible for organizations operating at scale. As per FMI's projection, the volume of data generated by the American healthcare billing apparatus drives unmatched investment in natural language processing architectures.
FMI's report includes coverage of Canada. The transition toward harmonized provincial drug review systems drives parallel demand for specialized policy mapping software north of the border.
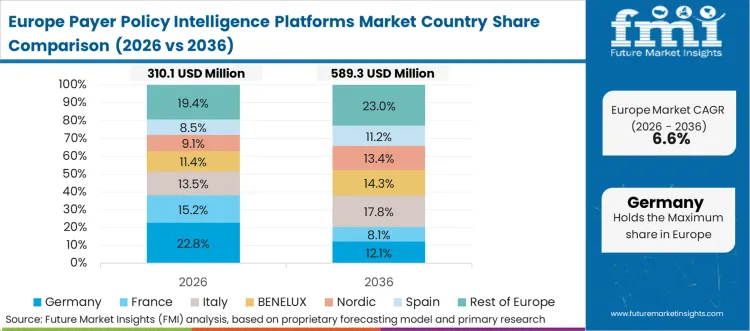
Strict health technology assessment protocols and unified data privacy directives heavily influence platform design in this region. The need to prove clinical and economic value against established standard-of-care policies forces drug developers to maintain massive repositories of historical payer decisions. FMI analysts opine that navigating these unified yet complex national health systems requires platforms built explicitly for European value-based pricing models.
FMI's report includes comprehensive evaluation of Italy, Spain, and the Nordics. The expansion of regional value-based procurement contracts requires deep integration with local hospital formulary data.
The payer policy intelligence market exhibits a moderately consolidated structure, anchored by high barriers to entry related to historical data accumulation and payer network integration. Challengers struggle to replicate the extensive archives of commercial medical policies maintained by dominant firms like Clarivate (MMIT) and Policy Reporter. Buyers distinguish qualified vendors primarily through the latency of their data ingestion—specifically, whether a platform alerts them to a policy change within 24 hours versus a week. This behavior shift forces incumbent platforms to abandon batch-processing methods in favor of real-time API connectivity.
Firms like Panalgo and Komodo Health hold specific structural advantages by linking raw policy text directly to massive real-world patient data repositories. A challenger entering this space must build proprietary cross-referencing capabilities that match specific billing codes to exact diagnostic criteria, rather than simply presenting unstructured PDFs. Vendors embedding these capabilities into comprehensive healthcare cloud computing infrastructures capture the most lucrative life sciences contracts. Challengers providing only surface-level scraping tools face rapid displacement.
Large pharmaceutical buyers actively prevent vendor lock-in by utilizing multi-vendor strategies, often pairing a dominant formulary tracking platform with a specialized predictive analytics tool. This structural tension prevents dominant intelligence providers from exerting absolute pricing power over enterprise contracts, as sophisticated market access teams can insource basic data aggregation if software licensing fees become punitive. The competitive trajectory to 2036 indicates further consolidation as leading platforms acquire specialized NLP startups to enhance their ability to parse highly ambiguous oncology and rare disease criteria.
| Metric | Value |
|---|---|
| Quantitative Units | USD 1.3 billion to USD 4.1 billion, at a CAGR of 12.06% |
| Market Definition | Software and services that autonomously ingest, interpret, and track medical policies, formulary updates, and reimbursement criteria from public and commercial health insurers. |
| Component Segmentation | Platform/Software, Services |
| Application Segmentation | Market Access Strategy, Formulary Tracking, Prior Authorization Analysis, Coding & Billing Compliance |
| End User Segmentation | Pharmaceutical & Biotech Companies, Medical Device Manufacturers, Healthcare Providers |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia & Pacific, Middle East & Africa |
| Countries Covered | India, China, United States, Germany, United Kingdom, France, Japan, and 40 plus countries |
| Key Companies Profiled | Policy Reporter (Mercalis), Clarivate (MMIT), Panalgo, Model N, Komodo Health, Definitive Healthcare, Veeva Systems, Zitter Insights |
| Forecast Period | 2026 to 2036 |
| Approach | Primary interviews with Pharma Market Access Directors and Health System Revenue Cycle VPs. Market sizing validated against enterprise software licensing contracts and API transaction volumes. |
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the Payer Policy Intelligence Platforms Market in 2026?
The market is projected to reach USD 1.3 billion in 2026, driven by life sciences firms migrating away from manual PDF reviews to continuous API-fed intelligence subscriptions.
What will it be valued at by 2036?
The valuation reaches USD 4.1 billion through 2036 as NLP-driven coverage ingestion automates the interpretation of scattered commercial and Medicare policies, eliminating fatal delays in market access.
What CAGR is projected?
A 12.06% compound annual growth rate is structurally supported by the operational necessity to map ambiguous clinical criteria to specific patient cohorts continuously.
Which Component segment leads?
Platform/Software holds 68.5% share in 2026 because the sheer daily volume of policy bulletins far exceeds human consultant processing capacity.
Which Application segment leads?
Market Access Strategy commands 41.2% share, dictated by the urgent commercial need to simulate projected revenue impacts before new clinical criteria edits take effect.
Which End User segment leads?
Pharmaceutical & Biotech Companies capture 48.7% share, as their high-margin specialty drug portfolios are disproportionately targeted by aggressive payer cost-containment measures.
What drives rapid growth?
The CMS Interoperability and Prior Authorization Final Rule forces health systems to adopt automated transparency tools, while compressed product launch windows compel pharma directors to secure peak sales velocity.
What is the primary restraint?
Heavy fragmentation in regional payer terminology creates severe algorithmic friction, requiring expensive human-in-the-loop validation teams to interpret non-standardized medical necessity language.
Which country grows fastest?
India advances at 15.2% annually, accelerated by the Ayushman Bharat Digital Mission requiring unstructured coverage data mapping for national registry inclusion.
How does CMS-0057-F impact this sector?
This mandate forces healthcare system revenue cycle teams to adopt API-driven policy transparency tools to manage skyrocketing denial rates.
What limits manual policy extraction?
Static policy scraping causes massive operational blind spots when daily medical policy updates exceed a market access team's manual review threshold of approximately 50 changes per week.
How do vendors compete on latency?
Buyers distinguish qualified vendors primarily by whether their platform alerts them to a localized policy change within 24 hours versus a multi-day processing lag.
Why is China's market expanding rapidly?
The centralized National Reimbursement Drug List negotiations compel domestic and foreign pharmaceutical firms to simulate precise pricing and coverage trade-offs prior to bidding.
What accelerates adoption in the United States?
Tier-1 pharmaceutical distributors face continuous policy volatility from hundreds of independent commercial payers altering step-therapy and prior authorization rules daily.
How does Germany's AMNOG framework drive procurement?
The regulation requires pharmaceutical companies to continuously track comparator drug policies and reference pricing shifts against evolving Federal Joint Committee criteria.
Why is MedTech adoption accelerating?
Erratic Medicare Local Coverage Determinations compel medtech commercialization leads to deploy dynamic tracking to prevent widespread reimbursement eligibility loss.
What happens if market access teams delay adoption?
Pharma teams failing to track these changes lose peak product revenue as favorable coverage criteria revert or tighten without their immediate knowledge.
Why are predictive algorithms becoming necessary?
Predictive engines calculate potential revenue loss from impending prior authorization rollouts, allowing financial analysts to adjust quarterly revenue guidance before the policy officially activates.
How do buyers avoid vendor lock-in?
Large pharmaceutical buyers utilize multi-vendor strategies, often pairing a dominant formulary tracking platform with a specialized predictive analytics tool.
What defines the crossover point for technology adoption?
The tipping point occurs when market access teams must map concurrent real-time criteria changes across 500+ commercial plans simultaneously to defend Tier 2 formulary status.
How does historical linkage provide an advantage?
Preserving archived policy versions allows analysts to identify payer criteria tightening trends over time, enabling accurate modeling of future coverage erosion for specialty therapies.
Why is genomic testing policy tracking an opportunity?
Specialized modules track the rapidly evolving reimbursement rules for companion diagnostics, accelerating precision medicine protocols across cautious payer networks.
How does API connectivity change the workflow?
Embedding policy alerts directly into the physician's electronic prescribing interface stops unapproved scripts before they reach the pharmacy, drastically reducing administrative burden.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.