The global genital herpes treatment market is on track to achieve a valuation of USD 3,957.6 Million by 2036, accelerating from USD 2,448.9 Million in 2026 at a CAGR of 4.9%. As per Future Market Insights, expansion is structurally underpinned by a maturing antiviral pipeline that is transitioning from legacy nucleoside analogues to novel helicase-primase inhibitors and mRNA-based therapeutic vaccines. The World Health Organization estimated in 2024 that approximately 491 million people aged 15 to 49 worldwide are living with HSV-2 infection, confirming the scale of unmet therapeutic need. This compels manufacturers to invest in scalable production and distribution infrastructure that can meet accelerating demand. Simultaneously the regulatory and competitive landscape is shifting from reliance on generic acyclovir regimens to a pipeline-driven competitive dynamic where novel mechanisms command premium pricing and attract multi-hundred-million-dollar licensing deals.
Jared Baeten, MD, PhD, SVP Clinical Development, Gilead, stated: 'Our research partnership with Assembly Bio has been highly fruitful, and we are excited to continue the clinical development of our herpes simplex virus candidates.' This statement was made in the context of regarding the licensing of abi-5366 and abi-1179. FMI opines that this reflects a broader structural shift in the industry where established players are accelerating investment to secure competitive positioning in a market defined by rapid evolution.
The operational reality for genital herpes treatment developers is defined by a rapidly maturing pipeline that is shifting competitive dynamics away from generic nucleoside analogues toward novel mechanisms with curative potential. Gilead Sciences exercised its option to license two helicase-primase inhibitors, ABI-5366 and ABI-1179, from Assembly Biosciences in December 2025, triggering a USD 35 million payment that confirms the commercial value assigned to next-generation antiviral targets. Alfasigma signed a EUR 125 million deal with Innovative Molecules in January 2026 for the rights to an injectable form of adibelivir, directing capital toward the acceleration of Phase 2 trials for a compound that could redefine suppressive therapy. Moderna continued to advance its Phase 1/2 clinical trial for mRNA-1608, a therapeutic vaccine candidate for recurrent HSV-2, with study updates posted in May 2025 confirming ongoing dose-ranging evaluation. As per FMI, the scale of licensing valuations and the diversity of modalities entering mid-stage development confirm that the treatment paradigm is shifting from symptom management to viral load reduction and potential functional cure. BioNTech continued its Phase 1 trial for BNT163 through October 2025, adding a second mRNA platform to the competitive pipeline.
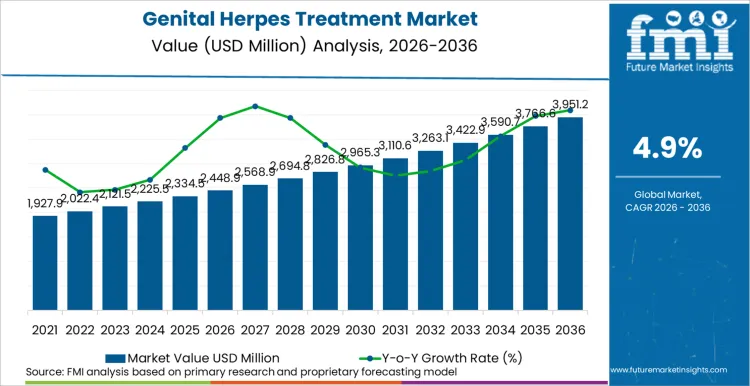
Future Market Insights projects the genital herpes treatment market to expand at a CAGR of 4.9% from 2026 to 2036, increasing from USD 2,448.9 Million in 2026 to USD 3,957.6 Million by 2036.
FMI Research Approach: FMI proprietary forecasting model based on demand-supply correlation and end-use consumption analysis.
FMI analysts perceive the market evolving toward a pipeline-driven competitive dynamic where novel mechanisms such as helicase-primase inhibitors and mRNA therapeutics command premium pricing and reshape treatment paradigms beyond generic nucleoside analogues.
FMI Research Approach: Regulatory convergence analysis and competitive landscape mapping.
The United States holds a significant share of the global genital herpes treatment market by value, supported by the largest pharmaceutical R&D expenditure globally and a concentration of pipeline-stage assets from Gilead, Moderna, and Assembly Biosciences.
FMI Research Approach: FMI country-level revenue modelling by end-use consumption and trade data.
The global genital herpes treatment market is projected to reach USD 3,957.6 Million by 2036.
FMI Research Approach: FMI long-term revenue forecast derived from end-use demand projections and macroeconomic indicators.
The genital herpes treatment market includes revenue generated from therapeutic products for the management and treatment of genital herpes simplex virus (HSV-2) infections, covering antiviral medications, topical treatments, and pipeline-stage vaccines and novel inhibitors.
FMI Research Approach: FMI market taxonomy and inclusion-exclusion framework.
Globally unique trends include the advancement of mRNA therapeutic vaccine platforms into mid-stage clinical trials, the licensing of novel helicase-primase inhibitors at multi-hundred-million-dollar valuations, and the development of topical alternatives to systemic antiviral regimens.
FMI Research Approach: Competitive intelligence and regulatory directive tracking.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 2,448.9 Million |
| Industry Value (2036) | USD 3,957.6 Million |
| CAGR (2026 to 2036) | 4.9% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
High HSV infection rates, the excellent presence of leading pharmaceutical companies, and increasing use of suppressive antiviral therapies are responsible for the growth in the North American genital herpes treatment market. The USA leads the region with large STI screening programs, access to high-tech treatment, and growing public health efforts to prevent HSV transmission.
Barriers to market expansion include a relatively low degree of knowledge about asymptomatic infections, high fees for long-term antiviral treatment, and significant social stigma impacting treatment-seeking behavior. Growing interest in HSV vaccine development, increasing direct-to-consumer diagnostic testing, and growing investment in new antiviral drug formulations will all be additional drivers that will fuel the anticipated market growth in North America.
Europe is a major market for genital herpes treatment, led by growing STI awareness initiatives, growing investment in HSV therapeutic research, and robust healthcare policies favoring STI management. Germany, France, and the UK are the major markets, with well-developed sexual health clinics, growing use of suppressive therapy, and growth of national screening programs for herpes simplex virus.
Yet, difficulties like rigorous regulatory approval procedures for new antiviral medicines, public healthcare system pricing pressures, and inequalities in STI treatment availability between countries could affect market growth.
The rising development of immune-modulating treatments, widening combination antiviral regimens, and increased collaboration between biotech companies and research institutions for HSV vaccine trials are defining the European market scenario. Also, increased demand for herbal and natural HSV treatments is shaping consumer behavior.
The Asia-Pacific region is seeing fast growth in the genital herpes treatment market based on rising rates of STIs, enhanced access to diagnostic and treatment services, and enhanced sexual health awareness. China, Japan, and India are among major markets, driven by growing pharmaceutical manufacturing capacity, rising government STI prevention campaigns, and emerging demand for lower-cost antiviral drugs.
Yet, problems like cultural STI taboos, limited standardized treatment regimen, and rarity of specialized clinics for sexual health may slow market penetration. Expansion of global antiviral pharmaceutical companies, the growth of public-private partnerships to educate on STIs, and the integration of telemedicine networks for HSV therapy consultation are accelerating market growth.
In addition, progress in CRISPR-based HSV gene editing and rising investment in therapeutic vaccines are enhancing the long-term treatment opportunities in the region.
From high costs to transplant rejection: Key Challenges Hindering Genital Herpes Treatment Advancements
The deficiencies faced by the market of genital herpes treatment are many, ranging from lack of proper antiviral medications capable of inflammation elimination, increasing resistance to acyclovir-based treatments, and the emotional Strain an HSV diagnosis places on suffering patients. Preventive measures are hindered by the lack of an HSV vaccine while long-term antiviral suppression becomes the only suitable option to manage the condition.
Moreover, the social stigma associated with genital herpes leads to late diagnosis and underdiagnosing of infections, posing a threat to effective disease management. Further, regulatory constraints on new antiviral drugs and scarce funding for STI public health reimbursement add even more delays to the progress of the market.
Advancements and Innovations Driving Growth in the Genital Herpes Treatment Market
There is ample room for growth in the treatment of genital herpes due to the rapid development and adoption of immune based herpes simplex virus therapies, the proliferation of non prescriptions and over the counter antiviral drugs, and the increasing funding of therapeutic vaccines creating opportunities.
The use of Artificial Intelligence driven drug designing and expanding rapid point of care diagnostic tests, along with public health campaigns aimed at improving awareness of STI are fueling the growth in this market. Along these lines, direct public health initiatives are increasingly popular to increase awareness and diagnosis of STIs, which are expanding the range of such diagnostic tests.
These factors are so important in driving market growth along with increasing focus on personalized medicine interventions for the management of herpes simplex virus.
Emerging Trends
The continued development of extended-release antiviral drugs and topical microbicides is improving the treatment of HSV symptoms and reducing outbreak frequency. The increased focus on HSV vaccine and immune-based therapy development is expanding the repertoire of long-term prevention and treatment for genital herpes. Greater investment in CRISPR-based HSV eradication and combination antiviral therapies is driving innovation in the long-term suppression of the herpes virus and cure.
Between 2021 and 2025, the global genital herpes treatment market is projected to continue expanding, supported by rising incidence of herpes simplex virus (HSV) infections, growing awareness of sexually transmitted diseases, and improved antiviral treatment.
Widespread use of oral and topical antiviral medications including acyclovir, valacyclovir and famciclovir continued as the standard of care, relieving symptoms and decreasing viral shedding. Market dimensions were also expanded through initiatives for early detection and patient awareness. But there were challenges: there was no curative treatment, and drug resistance was a concern.
Development of therapeutic vaccines, gene-editing approaches, and new long-acting antivirals is likely to follow through into the 2026 to 2036 market. Potential long-term suppression or even eradication of HSV with novel CRISPR-based therapies and immunotherapies contributing to the growth.
Moreover, telemedicine and digital health platforms are expected to increase access to diagnosis and treatment. As such, the market sees drastic changes both in treatment options and patient care models with greater investment into research and regulatory approval of novel therapies.
| Category | 2021 to 2025 Trends |
|---|---|
| Regulatory Landscape | Approval of existing antiviral drugs ensuring safety and efficacy. |
| Technological Advancements | Adoption of oral and topical antivirals to manage outbreaks. |
| Consumer Demand | Increased awareness leading to higher demand for fast-acting, symptom-relieving treatments. |
| Market Growth Drivers | Rising incidence of HSV infections and improved access to diagnostic testing. |
| Sustainability | Initial adoption of eco-friendly pharmaceutical manufacturing. |
| Supply Chain Dynamics | Reliance on traditional pharmacy-based distribution networks. |
| Category | 2026 to 2036 Projections |
|---|---|
| Regulatory Landscape | Expected regulatory approvals for HSV vaccines and gene-editing therapies, streamlining access to innovation. |
| Technological Advancements | Introduction of therapeutic vaccines, CRISPR-based therapies, and long-acting antivirals for prolonged efficacy. |
| Consumer Demand | Rising preference for preventive therapies and long-term HSV suppression strategies. |
| Market Growth Drivers | Expansion of personalized medicine, increased research funding, and broader treatment accessibility. |
| Sustainability | Greater focus on biodegradable packaging, green chemistry in drug production, and sustainable healthcare initiatives. |
| Supply Chain Dynamics | Expansion of telehealth platform |
The increasing incidence of herpes simplex virus and the rise in awareness of sexually transmitted infections (STIs) along with antiviral therapy development is sustaining the growth of the genital herpes treatment market in the United States.
The over-the-counter (OTC) and prescription antiviral drugs acyclovir, valacyclovir, and famciclovir play key roles in symptom management and outbreak reduction as major components of care. Therapeutic vaccines in conjunction with long-acting antiviral drugs are a key enabling factor in projected market growth.
Market Growth Factors
Market Forecast
| Country | CAGR (2026 to 2036) |
|---|---|
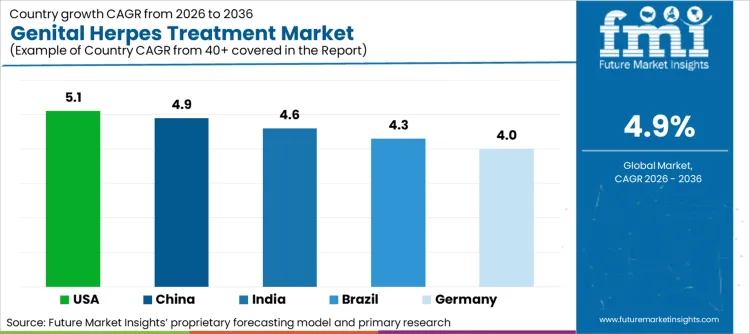
| United States | 5.1% |
Germany’s genital herpes treatment market is supported by a well-established healthcare system, insurance coverage for antiviral drugs, and significant investment in herpes research. Patients have access to advanced diagnostic tools and prescription medications, while pharmaceutical companies are actively engaged in developing novel therapies and vaccines.
Market Growth Factors
Market Forecast
| Country | CAGR (2026 to 2036) |
|---|---|
| Germany | 4.0% |
Genital herpes treatment market in India witnessed strong growth due to the rising burden of STIs, growing access to healthcare and rising penetration of digital health platforms. Due to generic drug making at a large scale, antiviral treatments remain widely available and inexpensive. STI prevention campaigns are implemented by both government and private organizations, thus increasing the corresponding diagnosis and treatment rates as well.
Market Growth Factors
Market Forecast
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 4.6% |
The genital herpes treatment market in China is booming with its huge population base, ever-increasing STI incidences and widening access to healthcare. These aspects play a significant role in the boosting of the market in China with the Chinese government promoting for STI awareness along with diagnostic services. The rising availability of cheap generic antiviral agents and rising R&D activities for the development of a herpes vaccine also accelerate the market.
Market Growth Drivers
Market Forecast
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 4.9% |
Brazil’s genital herpes treatment market is growing due to increased STI prevalence, government-led sexual health programs, and expanded access to prescription drugs. The availability of both branded and generic antiviral drugs makes treatment more accessible, while growing digital health adoption supports online consultations and prescription services.
Market Growth Drivers
Market Forecast
| Country | CAGR (2026 to 2036) |
|---|---|
| Brazil | 4.3% |
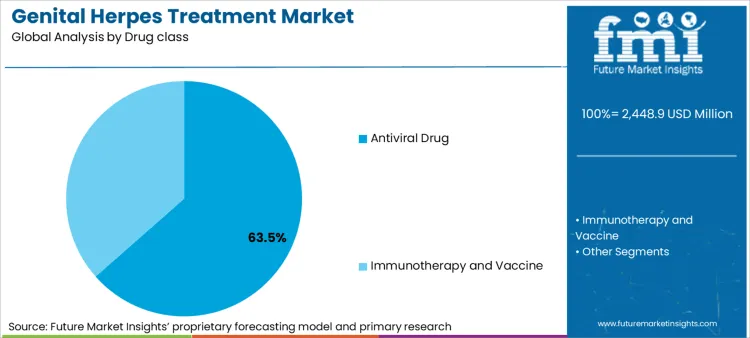
Antiviral Drugs (Nucleoside Analogues: Acyclovir, Valacyclovir, and Famciclovir)
Antiviral drugs are the first-line treatment for genital herpes, helping to reduce outbreak severity, decrease viral shedding, and lower transmission risk. Drugs such as acyclovir, valacyclovir, and famciclovir work by inhibiting the replication of the herpes simplex virus (HSV-1 and HSV-2). The rising prevalence of genital herpes worldwide, increasing awareness of suppressive therapy to prevent recurrent outbreaks, and growing use of antivirals for asymptomatic viral shedding reduction are driving market demand.
North America and Europe lead in antiviral drug adoption, while Asia-Pacific is witnessing growth due to increasing access to sexual health treatments and awareness programs. Future innovations include long-acting antiviral formulations, AI-driven outbreak prediction models, and novel antiviral combination therapies for improved efficacy.
Immunotherapy and Vaccine Development: The Growing Need to Prevent and Treat Herpes Virus
Immunotherapy is an emerging treatment approach for genital herpes, aiming to enhance the body's immune response to suppress the virus and reduce recurrences.Several therapeutic vaccines and monoclonal antibody-based therapies are currently in clinical trials, focusing on long-term viral suppression and potential functional cures. The rising investment in herpes vaccine research, increasing demand for alternative treatment options beyond antiviral drugs, and advancements in mRNA-based vaccine technologies are fueling market expansion.
North America and Europe dominate in herpes immunotherapy research, while Asia-Pacific is witnessing rising interest due to increasing sexually transmitted infection (STI) awareness programs. Future trends include CRISPR-based gene-editing therapies for viral eradication, AI-driven vaccine design, and combination immunotherapies with antiviral drugs for sustained viral suppression.
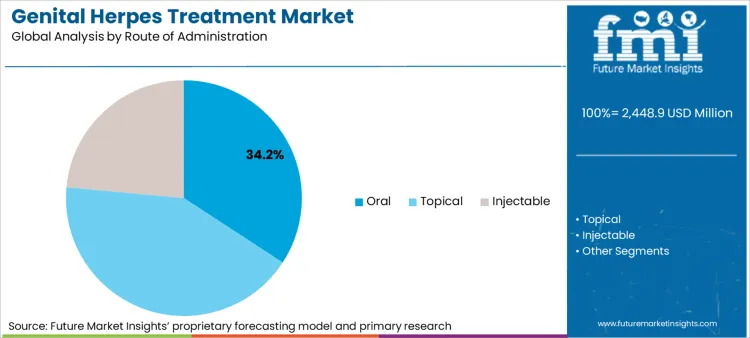
Oral Route: The preferred once due to convenience
Oral antiviral therapy is the mainstay of treatment for genital herpes, as this route of administration provides appropriate long-term, convenient, effective control of recurrent outbreaks. Antivirals such as valacyclovir and famciclovir are commonly prescribed not only for episodic treatment, but for suppressive therapy to prevent recurrent episodes.
Demand is driven by a recent shift towards oral antiviral therapy over topical therapy, increasing availability of generic oral formulations and increasing use of long-term suppressive therapy as a method to reduce risk of transmission. North America and Europe are almost completely cornering the market for sales on these increasingly scarce oral antivirals.
The region of Asia-Pacific is witnessing increased adoption as a result of development of STI healthcare infrastructure and awareness building programs. Further innovations coming down the pipeline include extended-release oral formulations for improved adherence to therapy, AI-powered bespoke therapy recommendations, and next generation prodrug antiviral compounds with enhanced bioavailability.
Topical Therapy- Allowing for convenient self-care and early symptom management
Topical antivirals creams and ointments like acyclovir and penciclovir formulations helps for symptomatic relief of short term (pain relief of lesion), speed up healing of herpes infection. These treatments are usually used in conjunction with oral antivirals for greater effectiveness. For example, the growing preference for over-the-counter (OTC) topical treatments, increasing demand for consumer self-care solutions, and the growing awareness of early symptom management are some of the factors driving the growth of the segment.
Topical herpes treatment value is mainly dependent on North America and Europe while Asia-Pacific has high opportunities with growth in pharmacy access to OTC antiviral creams. There may be new AI lesion monitoring apps for facilitating early treatment interventions, nanoparticle-based future topical antivirals, and perhaps combination topical therapies of topical pain relief with topical antivirals.
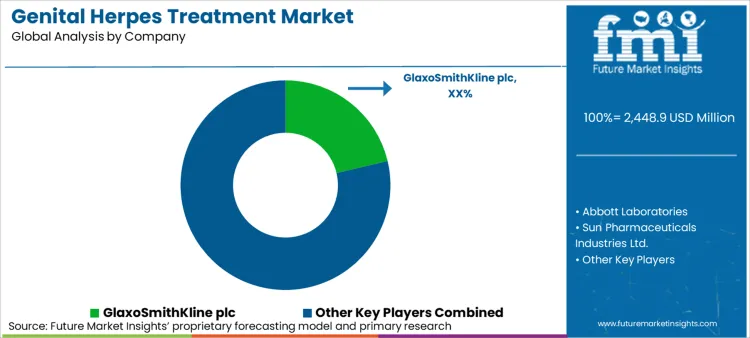
The genital herpes treatment market is highly competitive, driven by the increasing prevalence of herpes simplex virus (HSV) infections, growing awareness of antiviral therapies, and advancements in vaccine research.
Companies are investing in next-generation antiviral drugs, suppressive therapy options, and immunotherapy-based treatments to maintain a competitive edge. The market is shaped by well-established pharmaceutical firms, biotechnology innovators, and emerging antiviral drug developers, each contributing to the evolving landscape of genital herpes management.
Market Share Analysis by Company
| Company Name | Estimated Market Share (%) |
|---|---|
| GlaxoSmithKline (GSK) | 22-26% |
| Merck & Co., Inc. | 18-22% |
| Novartis AG | 10-14% |
| Abbott Laboratories | 8-12% |
| Others | 30-40% |
GlaxoSmithKline (GSK) (22-26%)
A dominant player in the genital herpes treatment market, GSK leads with its antiviral portfolio, including Valtrex, a widely used suppressive therapy.
Merck & Co., Inc. (18-22%)
A leader in antiviral drug development, Merck continues to innovate in herpes therapeutics and vaccine research.
Novartis AG (10-14%)
A key innovator in immune response modulation, Novartis is actively developing treatments for recurrent HSV infections.
Abbott Laboratories (8-12%)
A major player in diagnostics, Abbott supports early herpes detection and management through advanced testing solutions.
Other Key Players (25-35% Combined)
Beyond the leading companies, several other manufacturers contribute significantly to the market, enhancing product diversity and technological advancements. These include:
These companies focus on expanding the reach of genital herpes treatment solutions, offering competitive pricing and cutting-edge innovations to meet diverse patient and healthcare provider needs.
The genital herpes treatment market represents revenue generated from therapeutic products for the management and treatment of genital herpes simplex virus (HSV-2) infections. The market measures the value of antiviral medications, topical treatments, and pipeline-stage vaccines and novel inhibitors sold through prescription and hospital channels.
Inclusions cover oral antivirals (acyclovir, valacyclovir, famciclovir), topical treatments, novel helicase-primase inhibitors, mRNA therapeutic vaccines in clinical development, and combination therapy regimens. It includes both branded and generic formulations distributed through retail pharmacies, hospital pharmacies, and specialty channels.
Exclusions include diagnostic testing kits and laboratory services for HSV detection, over-the-counter cold sore treatments for oral HSV-1 that are not indicated for genital herpes, and preventive (prophylactic) vaccines not yet in clinical trials. General antiviral drugs not specifically indicated for HSV-2 are outside the scope.
Antiviral Drug, and Immunotherapy and Vaccine Development
Oral, Topical and Injectable
HSV-1 and HSV-2
Hospital Pharmacies, Retail Pharmacies and Mail Order Pharmacies
North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, Middle East & Africa
What is the current global market size for the Genital Herpes Treatment Market?
The market is valued at USD 2,448.9 Million in 2026, driven by a maturing clinical pipeline of novel antiviral and mRNA therapeutic vaccine candidates.
What is the projected Compound Annual Growth Rate (CAGR) for the market over the next 10 years?
The market is projected to grow at a CAGR of 4.9% from 2026 to 2036.
Which regions are experiencing the fastest expansion?
Asia Pacific leads with the highest growth rate, driven by rising healthcare and consumer spending, followed by North America with its large installed base and innovation pipeline.
What are the primary market drivers?
Strategic investment from players such as Gilead Sciences / Assembly Bio and Alfasigma / Innovative Molecules, combined with regulatory mandates and evolving end-user demand, are the primary drivers of adoption.
Who are the leading companies in the industry?
Gilead Sciences / Assembly Bio, Alfasigma / Innovative Molecules, Moderna, BioNTech SE are key players, differentiating through strategic investment, product innovation, and regional expansion.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.