Helicobacter Pylori Non-Invasive Testing Market : Global Industry Analysis 2015 - 2024 and Opportunity Assessment 2025 - 2035
Helicobacter Pylori Non-Invasive Testing Market Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
Helicobacter Pylori Non-Invasive Testing Market Analysis Size and Share Forecast Outlook 2025 to 2035
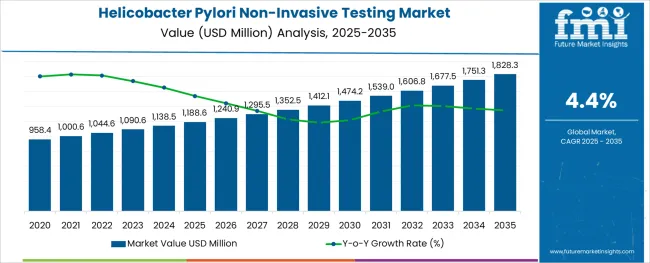
The Helicobacter Pylori Non-Invasive Testing Market is estimated to be valued at USD 1188.6 million in 2025 and is projected to reach USD 1828.3 million by 2035, registering a compound annual growth rate (CAGR) of 4.4% over the forecast period.
Quick Stats for Helicobacter Pylori Non-Invasive Testing Market
- Helicobacter Pylori Non-Invasive Testing Market Industry Value (2025): USD 1188.6 million
- Helicobacter Pylori Non-Invasive Testing Market Forecast Value (2035): USD 1828.3 million
- Helicobacter Pylori Non-Invasive Testing Market Forecast CAGR: 4.4%
- Leading Segment in Helicobacter Pylori Non-Invasive Testing Market in 2025: Serology Test (41.7%)
- Key Growth Region in Helicobacter Pylori Non-Invasive Testing Market: North America, Asia-Pacific, Europe
- Top Key Players in Helicobacter Pylori Non-Invasive Testing Market: DiaSorin S.p.A., Meridian Bioscience, Inc., Exalenz Bioscience Ltd., Alere, Thermo Fisher Scientific, Biomerica, Inc., Certest Biotec S.L., Sekisui Diagnostics, Coris Bioconcept SPRL, Shenzhen Zhonghe Headway Bio-Sci & Tech Co., Ltd
Rationale for Segmental Growth in the Alginic Acid Market
Market Overview
The alginic acid market is undergoing consistent growth, propelled by rising demand from food processing, pharmaceutical formulations, and cosmetic applications. A growing focus on natural and sustainable ingredients in manufacturing processes has positioned alginic acid as a preferred biopolymer across industries.
The market is further supported by its diverse functional benefits, including water retention, gelling, and stabilizing properties, which make it indispensable in high-performance formulations. Increased regulatory acceptance of alginates as safe additives and the push towards cleaner labeling in food and personal care products have reinforced their adoption.
Ongoing innovation in extraction technologies and the utilization of algae as a renewable resource are paving the way for cost-effective production and expanded applications, ensuring sustained market expansion in the coming years.
Segmental Analysis
The market is segmented by Non-invasive Test Type, Test Type, and End-user and region. By Non-invasive Test Type, the market is divided into Serology Test, Stool Antigen Test, and Urea Breath Test. In terms of Test Type, the market is classified into Laboratory-based Tests and Point-of-Care Tests. Based on End-user, the market is segmented into Hospitals, Diagnostic Labs, and Clinics. Regionally, the market is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the Sodium Alginate Salts Segment
When segmented by salts, sodium alginate is expected to command 27.5 % of the market revenue in 2025, marking it as the leading subsegment in this category. This leadership has been driven by its superior solubility, ease of incorporation into formulations, and versatility in a wide range of applications.
Its ability to form stable gels and maintain viscosity under varying conditions has made it highly sought after in both food and pharmaceutical sectors. Manufacturers have prioritized sodium alginate due to its consistent performance, cost-effectiveness, and regulatory acceptance, which have collectively strengthened its position.
The segment’s prominence has also been enhanced by its adaptability to evolving consumer preferences for plant-derived and sustainable ingredients, reinforcing its market share.
Insights into the Food Industry End User Industry Segment
Segmented by end user industry, the food industry is projected to hold 33.0 % of the market revenue in 2025, positioning it as the most prominent sector. This dominance has been shaped by the industry’s increasing reliance on alginic acid and its derivatives to deliver desirable textures, stabilize emulsions, and improve shelf life of processed foods.
As consumer demand for clean label and natural additives has intensified, the food sector has responded by integrating alginates into bakery, dairy, and confectionery products. Enhanced production efficiency and compliance with food safety standards have further encouraged widespread use.
The segment’s leadership has also been supported by product differentiation strategies where alginates contribute to premium quality and innovation, securing their role in modern food formulations.
Insights into the Thickening Agent Functionality Segment
When analyzed by functionality, thickening agents are forecast to account for 29.0 % of the market revenue in 2025, establishing themselves as the dominant functional category. This preeminence has been underpinned by the growing need for consistent texture and viscosity in a wide array of end products.
Alginic acid’s natural origin and high efficiency in creating uniform, stable thickness without altering taste or color have solidified its appeal. The demand for thickeners in both edible and topical applications has expanded, with manufacturers leveraging its rheological properties to meet performance and regulatory requirements.
The functionality’s leading share has also been reinforced by the ability to deliver cost savings through lower dosages and its compatibility with other ingredients, securing its position as an indispensable component in formulation strategies.
Key Success Factors Driving the Helicobacter Pylori Non-Invasive Testing Industry
Immunoassays are biochemical tests that determine how much of an analyte is present. It doesn't matter what size the analyte is; it might be a huge protein, an antibody the body has created in response to an illness or a little molecule. The accuracy and reliability of immunoassays are unparalleled. They are very specific since antibodies and purified antigens are used as reagents. Diseases can be diagnosed more precisely using this assay since it measures antigen presence. It is a highly selective bioanalytical approach that uses an antibody or antigen as a biorecognition agent to quantify the presence or quantity of analytes ranging from tiny molecules to macromolecules in solution.
Due to the presence of several advanced technologies, such as the enzyme immunoassays, which are used for the detection of antibodies, the immunoassay segment currently holds a significant market share in the helicobacter pylori non-invasive testing market and is expected to show a similar trend over the forecast period. Key factors propelling the immunoassay market are the growing incidence of gastritis and infections caused by Helicobacter pylori, as well as technical developments.
For instance, in 2024, the worldwide prevalence of gastritis was estimated to be between 50.0% and 56.0%, according to a study titled "Diagnosis of Helicobacter pylori Infection and Recent Advances," with the prevalence of chronic atrophic gastritis falling within that range between 20% and 30%. Helicobacter pylori infection was shown to be proportional to gastritis prevalence, and cases of Helicobacter pylori-positive gastritis were found to account for more than 80.0% of all cases. Since such illnesses are typically detected by immunoassays, rising instances of Helicobacter pylori infection and gastritis are expected to drive the segment growth throughout the forecast period.
Major Trends, Drivers, and Challenges Impacting the Helicobacter Pylori Non-Invasive Testing Market
Increased Patient Requirement for POC Increases the Demand for H. pylori Tests Drives the Development of the Industry as a Whole
The National Institutes of Health (NIH) research states that IgG serology, the C-Urea Breath Test (UBT), and the monoclonal stool antigen test are all non-invasive, Point-of-care (POC) diagnostic options for primary care settings that can detect H. pylori. UBT is the only one of these that reliably verifies whether an infection is present or has been eradicated. According to the research published in Gut and Liver, UBT is an accurate, non-invasive option that is safe for use on both children and pregnant women.
In addition, H. pylori laboratory test results can now be obtained in a shorter amount of time owing to Point-of-care Testing (POCT), a tried-and-true method. Popularity, the number of people getting tested, and the variety of therapeutic uses for POCTs have all increased as leading companies keep releasing new models. In addition, Point-of-care Testing (POCT) is now widely used in all hospitals and is increasingly being adopted as the norm in other types of healthcare facilities. The global helicobacter pylori non-invasive testing market is expected to expand because of the aforementioned factors throughout the forecast period. This means that the increasing number of H. pylori sufferers is a key factor propelling the helicobacter pylori non-invasive testing market forward.
The industry's growth may be hampered by a lack of educated doctors to perform intrusive tests.
To accurately diagnose Helicobacter pylori infection and determine the best course of therapy, a surgeon or medical professional must perform an invasive procedure that often includes a biopsy and endoscopy. Market growth could be stymied, however, if countries with fewer healthcare resources and a corresponding shortage of trained professionals saw a decline in the number of people getting their diseases checked out. Unfortunately, many nations with low or middle income lack the hospital and medical training infrastructure necessary to produce highly qualified physicians. However, there are additional difficulties for doctors when treating Helicobacter pylori, as no first-line therapy is effective for everyone.
The helicobacter pylori non-invasive testing market is expected to expand at a modest rate worldwide. Although testing has become more common, diagnosis has been hindered by the fact that HIV infection often has no outward symptoms. This is a major factor in the market's slow expansion. It is also expected that h. pylori non-invasive testing is expected to decrease as attention is redirected to ending the current coronavirus pandemic.
Helicobacter Pylori Non-Invasive Testing Industry Analysis by Type, Method, and End Use
From 2025 to 2035, the clinic market is expected to grow at a CAGR of over 6%, as more people choose clinics over hospitals because of their lower costs and higher quality of care. The reason for this is that people are more likely to seek help from clinics since they offer high-quality care and respond quickly to patient needs, resulting in effective treatment at an affordable price. The premium services provided by clinics as a result of efficient disease management tactics are also expected to fuel market growth.
Due to its non-invasive nature and its ability to offer speedy findings, the POC testing segment accounted for more than 35% of the business. This is due, in large part, to rising Research and Development spending and joint research activities amongst industry heavyweights that are fostering innovation and new product development.
The helicobacter pylori non-invasive testing market may be broken down into two distinct subsegments: invasive helicobacter pylori tests and non-invasive helicobacter pylori tests. Since quick urease tests are both effective and inexpensive, this market subsegment is expected to grow at a CAGR of 6.5% between 2025 and 2035. Patients who do not respond well to antibiotic medication, who have symptoms of severe gastrointestinal issues, and who require gastric endoscopy have a significant demand for invasive diagnostic methods.
Top Regions Deploying Helicobacter Pylori Non-Invasive Testing Solutions
How is the USA Helicobacter Pylori Non-Invasive Testing Market Developing?
Globally, North America accounts for the largest helicobacter pylori non-invasive testing market. The expansion of healthcare infrastructure in addition to the rising incidence of stomach cancer, duodenal ulcers, and gastric ulcers in North America are driving forces behind the helicobacter pylori non-invasive testing market's expansion.
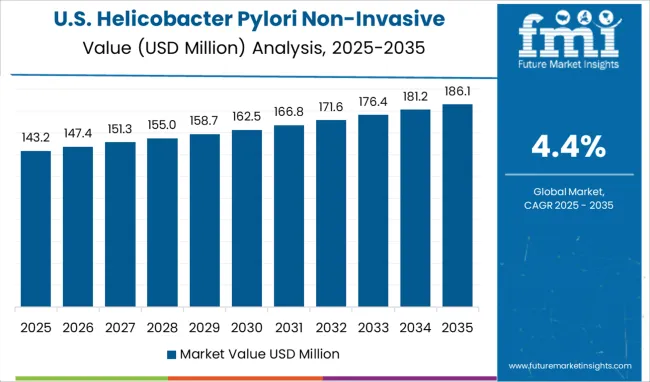
The NIH found that 5 percent of USA youngsters under the age of 10 have H. pylori in their systems. In addition, 30% to 40% of Americans have H. pylori infection, according to research from the USA Department of Health and Human Services. Furthermore, socioeconomic variables such as poor income, less education, home crowding, and immigration all contribute to a higher prevalence of H. pylori infection in the USA's Hispanic and Black populations. In order to treat H. pylori, the FDA recommends either Proton Pump Inhibitors (PPI) and two antibiotics or bismuth subsalicylate, acid suppressors, and two antibiotics, as detailed in a paper available from the ClinMed International Library.
There is resistance to effectiveness with the currently available first-line medication regimens. Due to resistance in H. pylori to common antibiotics, the USA, for example, observed 75% eradication rates utilizing these regimens. The largest rates of resistance are shown with Clarithromycin and Metronidazole and characteristics related to resistance include race, age, location, and the presence or absence of ulcer disease.
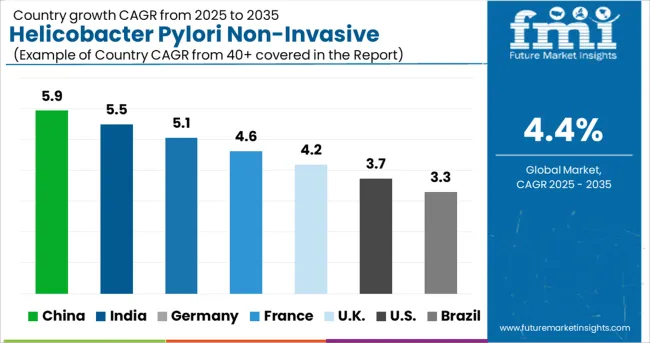
What are the driving factors for the Chinese Helicobacter Pylori Non-Invasive Testing Market?
The market for non-invasive testing for Helicobacter Pylori (H. pylori) in Asia and the Pacific is dominated by China. When it comes to healthcare and pharmaceuticals, China is a developing powerhouse in the Asia-Pacific region. Many international manufacturers of medical, pharmaceutical, and biotechnology products are based in China. High rates of H. pylori infection and gastric (stomach) cancer cases have spurred the development of diagnostic testing kits and technologies for H. pylori detection by pharmaceutical and biotechnology firms.
What are the defining characteristics of the Japanese Helicobacter Pylori Non-Invasive Testing Market?
The Japanese medical industry is among the world's most advanced markets. Due to widespread infection with H. pylori and a penchant for consuming smoked and salted foods, Roswell Park Comprehensive Cancer Center reports that Japan has an alarmingly high incidence of stomach cancer. In Japan, 50,000 individuals each year are killed by stomach cancer, making it the third deadliest form of the disease. In addition, 98% of gastric cancer in Japan can be attributed to H. pylori, according to a study published in 2024 by Wiley Online Library.
Recent investigations have indicated that H. pylori infection is the primary cause of stomach cancer, while it is one of many factors. The expansion of the helicobacter pylori non-invasive testing market in Japan is anticipated to benefit from the aforementioned factors over the forecast period.
Leading Suppliers of Helicobacter Pylori Non-Invasive Testing Kits
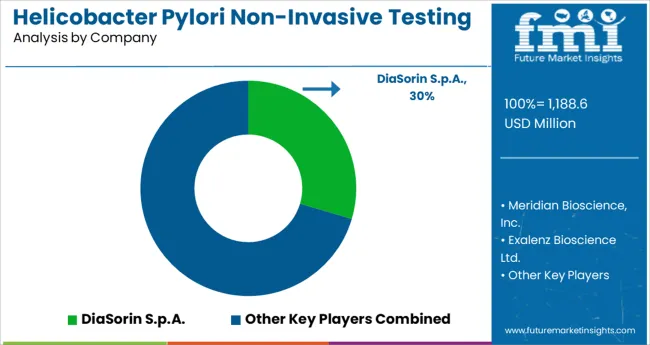
Companies like DiaSorin S.p.A., Meridian Bioscience, Inc., Exalenz Bioscience Ltd., Alere, Thermo Fisher Scientific, Biomerica, Inc., Certest Biotec S.L., Sekisui Diagnostics, CorisBioconcept SPRL, and Shenzhen Zhonghe Headway Bio-Sci & Tech Co., Ltd. are all major players in the global helicobacter pylori market. These leaders are anticipated to retain their positions of prominence during the prediction horizon.
Some recent developments in the helicobacter pylori non-invasive testing market include:
- Meridian Bioscience, Inc. announced in March 2024 that its novel Curian and Curian HpSA assay had been approved by the Food and Drug Administration. They hope that their efforts to preserve their position as the industry leader in testing for gastrointestinal diseases would be aided by the introduction of this analyzer and its initial assay.
- Thermo Fischer Scientific announced in December 2024 that it would be purchasing PPD, Inc. for USD17.4 billion. PPD is a top global provider of clinical research services to the biopharma & biotech industry.
Top Investment Segments in the Helicobacter Pylori Non-Invasive Testing Market
By Non-invasive Test Type:
- Serology Test
- Stool Antigen Test
- Urea Breath Test
By Test Type:
- Laboratory-based Tests
- Point-of-Care Tests
By End-user:
- Hospitals
- Diagnostic Labs
- Clinics
By Region:
- North America
- Latin America
- Asia Pacific
- MEA (Middle East and Africa)
- Europe
Frequently Asked Questions
How big is the helicobacter pylori non-invasive testing market in 2025?
The global helicobacter pylori non-invasive testing market is estimated to be valued at USD 1,188.6 million in 2025.
What will be the market size for the helicobacter pylori non-invasive testing market in 2035?
It is projected to reach USD 1,828.3 million by 2035.
How much will be the CAGR of the helicobacter pylori non-invasive testing market between 2025 and 2035?
The market is expected to grow at a 4.4% CAGR between 2025 and 2035.
What are the key product types in the helicobacter pylori non-invasive testing market?
The key product types are serology test, stool antigen test and urea breath test.
Which laboratory-based tests segment is expected to dominate the helicobacter pylori non-invasive testing market in 2025?
laboratory-based tests segment is expected to dominate with a 54.2% industry share in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Non-invasive Test Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Non-invasive Test Type , 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Non-invasive Test Type , 2025 to 2035
- Serology Test
- Stool Antigen Test
- Urea Breath Test
- Y-o-Y Growth Trend Analysis By Non-invasive Test Type , 2020 to 2024
- Absolute $ Opportunity Analysis By Non-invasive Test Type , 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Test Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Test Type, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Test Type, 2025 to 2035
- Laboratory-based Tests
- Point-of-Care Tests
- Y-o-Y Growth Trend Analysis By Test Type, 2020 to 2024
- Absolute $ Opportunity Analysis By Test Type, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By End-user
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End-user, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End-user, 2025 to 2035
- Hospitals
- Diagnostic Labs
- Clinics
- Y-o-Y Growth Trend Analysis By End-user, 2020 to 2024
- Absolute $ Opportunity Analysis By End-user, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2020 to 2024
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- USA
- Canada
- Mexico
- By Non-invasive Test Type
- By Test Type
- By End-user
- By Country
- Market Attractiveness Analysis
- By Country
- By Non-invasive Test Type
- By Test Type
- By End-user
- Key Takeaways
- Latin America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Non-invasive Test Type
- By Test Type
- By End-user
- By Country
- Market Attractiveness Analysis
- By Country
- By Non-invasive Test Type
- By Test Type
- By End-user
- Key Takeaways
- Western Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Non-invasive Test Type
- By Test Type
- By End-user
- By Country
- Market Attractiveness Analysis
- By Country
- By Non-invasive Test Type
- By Test Type
- By End-user
- Key Takeaways
- Eastern Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Non-invasive Test Type
- By Test Type
- By End-user
- By Country
- Market Attractiveness Analysis
- By Country
- By Non-invasive Test Type
- By Test Type
- By End-user
- Key Takeaways
- East Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By Non-invasive Test Type
- By Test Type
- By End-user
- By Country
- Market Attractiveness Analysis
- By Country
- By Non-invasive Test Type
- By Test Type
- By End-user
- Key Takeaways
- South Asia and Pacific Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Non-invasive Test Type
- By Test Type
- By End-user
- By Country
- Market Attractiveness Analysis
- By Country
- By Non-invasive Test Type
- By Test Type
- By End-user
- Key Takeaways
- Middle East & Africa Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Non-invasive Test Type
- By Test Type
- By End-user
- By Country
- Market Attractiveness Analysis
- By Country
- By Non-invasive Test Type
- By Test Type
- By End-user
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By Non-invasive Test Type
- By Test Type
- By End-user
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Non-invasive Test Type
- By Test Type
- By End-user
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Non-invasive Test Type
- By Test Type
- By End-user
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Non-invasive Test Type
- By Test Type
- By End-user
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Non-invasive Test Type
- By Test Type
- By End-user
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Non-invasive Test Type
- By Test Type
- By End-user
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By Non-invasive Test Type
- By Test Type
- By End-user
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Non-invasive Test Type
- By Test Type
- By End-user
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Non-invasive Test Type
- By Test Type
- By End-user
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Non-invasive Test Type
- By Test Type
- By End-user
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Non-invasive Test Type
- By Test Type
- By End-user
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Non-invasive Test Type
- By Test Type
- By End-user
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Non-invasive Test Type
- By Test Type
- By End-user
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Non-invasive Test Type
- By Test Type
- By End-user
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Non-invasive Test Type
- By Test Type
- By End-user
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Non-invasive Test Type
- By Test Type
- By End-user
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Non-invasive Test Type
- By Test Type
- By End-user
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Non-invasive Test Type
- By Test Type
- By End-user
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Non-invasive Test Type
- By Test Type
- By End-user
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Non-invasive Test Type
- By Test Type
- By End-user
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Non-invasive Test Type
- By Test Type
- By End-user
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Non-invasive Test Type
- By Test Type
- By End-user
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Non-invasive Test Type
- By Test Type
- By End-user
- Competition Analysis
- Competition Deep Dive
- DiaSorin S.p.A.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Meridian Bioscience, Inc.
- Exalenz Bioscience Ltd.
- Alere
- Thermo Fisher Scientific
- Biomerica, Inc.
- Certest Biotec S.L.
- Sekisui Diagnostics
- Coris Bioconcept SPRL
- Shenzhen Zhonghe Headway Bio-Sci & Tech Co., Ltd
- DiaSorin S.p.A.
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2020-2035
- Table 2: North America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 3: Latin America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 4: Western Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 5: Eastern Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 6: East Asia Market Value (USD Million) Forecast by Country, 2020-2035
- Table 7: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020-2035
- Table 8: Middle East & Africa Market Value (USD Million) Forecast by Country, 2020-2035
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020-2035
- Figure 3: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 4: Global Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 5: Global Market Attractiveness Analysis by Region
- Figure 6: North America Market Incremental $ Opportunity, 2025-2035
- Figure 7: Latin America Market Incremental $ Opportunity, 2025-2035
- Figure 8: Western Europe Market Incremental $ Opportunity, 2025-2035
- Figure 9: Eastern Europe Market Incremental $ Opportunity, 2025-2035
- Figure 10: East Asia Market Incremental $ Opportunity, 2025-2035
- Figure 11: South Asia and Pacific Market Incremental $ Opportunity, 2025-2035
- Figure 12: Middle East & Africa Market Incremental $ Opportunity, 2025-2035
- Figure 13: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 14: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 15: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 16: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 17: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 18: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 19: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 20: Global Market - Tier Structure Analysis
- Figure 21: Global Market - Company Share Analysis

