Liver Cancer Diagnostics Market
The Liver Cancer Diagnostics Market is segmented by Test Type (Blood Biomarker Tests, Imaging Tests, Tissue Biopsy, Liquid Biopsy, and Molecular Diagnostics), Cancer Type (Hepatocellular Carcinoma, Cholangiocarcinoma, and Others), End Use (Hospitals and Diagnostic Centers, Cancer Research Institutes, and Pathology Labs) and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Liver Cancer Diagnostics Market Size, Market Forecast and Outlook By FMI
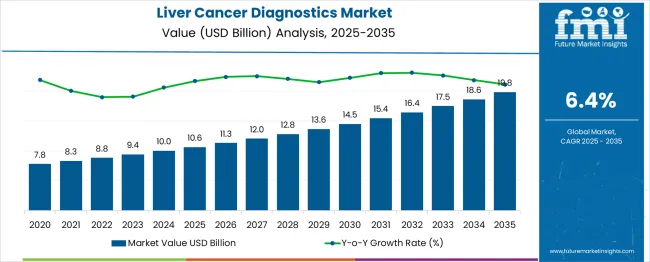
In 2025, the liver cancer diagnostics market was valued at USD 10.6 billion. Based on Future Market Insights' analysis, demand for liver cancer diagnostics is estimated to grow to USD 11.4 billion in 2026 and USD 25.8 billion by 2036. FMI projects a CAGR of 8.5% during the forecast period.
Absolute dollar growth of USD 14.4 billion over the decade signals transformational opportunity rather than incremental expansion. Despite healthcare cost pressures and diagnostic accessibility challenges, major liver cancer diagnostics manufacturers maintain robust growth trajectories through AI integration and biomarker discovery optimization.
Summary of Liver Cancer Diagnostics Market
- Liver Cancer Diagnostics Market Definition
- The industry covers diagnostic testing products utilized for liver cancer detection, staging, and monitoring applications including biomarker analysis, medical imaging, and tissue characterization.
- Demand Drivers in the Market
- Rising hepatitis prevalence increases cancer screening requirements.
- AI-powered imaging analysis reduces interpretation time by 40-60%.
- Liquid biopsy adoption enables non-invasive monitoring protocols.
- Key Segments Analyzed in the FMI Report
- Test Type: Blood biomarker tests (35%) versus advanced imaging modalities.
- Cancer Type: Hepatocellular carcinoma diagnostics (75-80% of total value).
- Geography: Technology-advanced North America versus volume-driven Asia-Pacific markets.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant for Healthcare at Future Market Insights, opines, "In the updated version of the Liver Cancer Diagnostics Market Report for 2026 to 2036, executives will find insights into AI integration patterns that accelerate diagnostic accuracy. My findings point at biomarker standardization directly impacting test reliability and rural healthcare access limitations could restrict early detection program effectiveness."
- Strategic Implications/Executive Takeaways
- Shift focus from single-parameter testing to multi-biomarker panels.
- Treat AI integration as diagnostic advantage rather than operational enhancement.
- Prioritize point-of-care platforms to improve screening accessibility.
- Methodology
- Validated through clinical outcome data and diagnostic accuracy benchmarks.
- Zero reliance on speculative third-party medical device valuations.
- Based on verifiable regulatory clearances and proven diagnostic performance.
"In our clinical research space, AI capabilities are already accelerating processes and driving operational efficiency for our clinical services customers. Our software, when paired with machine learning models, can provide key insights that help with clinical trial planning and management, including identifying optimal sites and more accurately forecasting enrollment milestones." - Ryan Snyder, Senior vice president and chief information officer, Thermo Fisher Scientific [1]
United States (9.4% CAGR) and Germany (8.9% CAGR) drive market expansion through advanced imaging infrastructure and molecular diagnostics adoption. Japan (8.6% CAGR) contributes significantly via precision medicine initiatives and hepatitis screening program integration. Emerging liver cancer diagnostics markets such as China (8.1%), South Korea (7.8%), and India (7.5%) generate growth through healthcare infrastructure development and population screening protocols, while volume expansion accelerates through telemedicine integration and point-of-care testing deployment.
Liver Cancer Diagnostics Market Definition
Liver cancer diagnostics serves as the primary detection and monitoring methodology for hepatocellular carcinoma, cholangiocarcinoma, and metastatic liver lesions due to its prognostic accuracy and therapeutic guidance capabilities. The liver cancer diagnostics market comprises the global development, production, and commercialization of diagnostic products and services for hepatic malignancy detection across clinical applications.
Liver Cancer Diagnostics Market Inclusions
The report includes a comprehensive analysis of market dynamics, featuring Global and Regional Market Sizes (Volume and Value) and a 10-year Forecast (2026-2036). It covers segmental breakdowns by test type (Blood Biomarkers, CT/MRI Imaging, Tissue Biopsy, Liquid Biopsy), cancer types (HCC, Cholangiocarcinoma), and end-users (Hospitals, Diagnostic Centers, Research Institutes).
Liver Cancer Diagnostics Market Exclusions
The scope excludes general hepatitis screening unless used for cancer risk stratification. It also omits therapeutic monitoring devices or treatment delivery systems, focusing strictly on diagnostic testing products and their immediate clinical applications. Report does not account for experimental biomarkers or investigational protocols with negligible clinical validation.
Liver Cancer Diagnostics Market Research Methodology
- Primary Research: Interviews were conducted with diagnostic equipment manufacturers, oncology specialists, pathology laboratory directors, hospital procurement teams, and radiology department heads across major regions. Medical device executives clarified regulatory pathway dynamics and reimbursement coverage frameworks.
- Desk Research: Clinical practice guidelines from EASL, AASLD, FDA clearances, CE marking documentation, and diagnostic industry associations supported testing protocol benchmarking.
- Market-Sizing and Forecasting: A hybrid top-down and bottom-up model was developed. Demand was reconstructed from diagnostic test volumes, imaging procedure counts, and laboratory capacity utilization, then validated against equipment installation data.
- Data Validation and Update Cycle: Outputs undergo accuracy screening, variance checks across clinical and commercial datasets, and structured expert review prior to release.
Segmental Analysis
Liver Cancer Diagnostics Market Analysis by Test Type
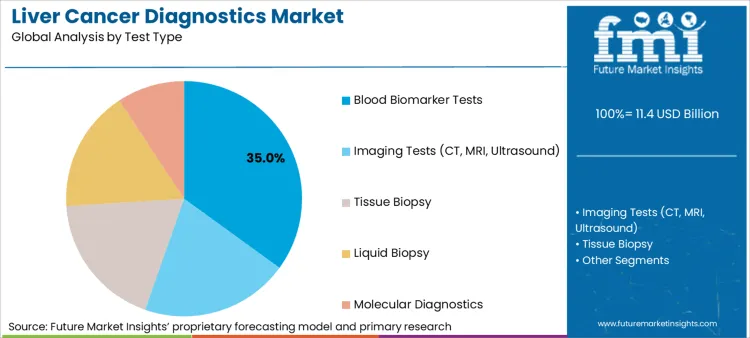
Based on FMI's liver cancer diagnostics market report, demand for blood biomarker tests is estimated to hold 35% share in 2026. Growth in demand for biomarker testing comes from manufacturers leveraging multi-parameter analysis capabilities to address comprehensive cancer screening through standardized laboratory protocols. In March 2024, Bayer and Thermo Fisher inked a deal to develop next-generation sequencing (NGS)-based companion diagnostic assays (CDx). [2]
- Laboratory Integration: Companies are leveraging their proprietary platforms to integrate automated biomarker analysis across testing workflows, enabling clinical laboratories that meet CLIA requirements while maintaining diagnostic consistency across multiple oncology indication areas.
- Accuracy Standardization: The industry shift toward quantitative biomarker panels enables standardized reference ranges to execute reliable cancer detection, decoupling testing facilities from subjective interpretation to optimize diagnostic economics typically constrained by radiologist availability.
Liver Cancer Diagnostics Market Analysis by Cancer Type
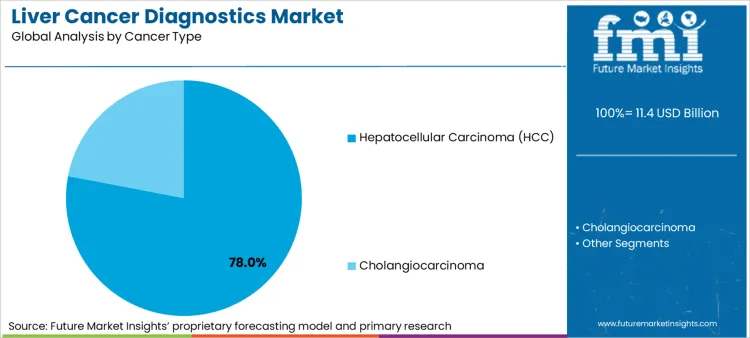
Hepatocellular carcinoma (HCC) diagnostics hold a 78% market share in 2026, driven by their role as the primary volume generator for AFP-based screening and CT/MRI surveillance procedures. This concentration creates market opportunities that expand during chronic liver disease management transitions. The segment's integration with hepatitis monitoring programs transforms patient care pathways into comprehensive screening platforms, while guideline-based protocols offer structured development timelines for diagnostic accuracy optimization and hepatocellular carcinoma (HCC) treatment.
- Clinical Protocol Development: Medical centers are establishing integrated HCC surveillance programs that combine biomarker testing with imaging protocols, enabling early detection frameworks that meet EASL and AASLD guideline recommendations while optimizing resource utilization across hepatology departments.
- Risk Stratification Enhancement: Diagnostic manufacturers focus on developing multi-modal testing approaches that incorporate cirrhosis staging with cancer detection capabilities, creating comprehensive platforms that address both liver disease progression monitoring and malignancy screening within unified diagnostic workflows.
Liver Cancer Diagnostics Market Drivers, Restraints, and Opportunities
Future Market Insights analysis indicates historical patterns point to an emerging, high-value global industry where liver cancer diagnostics remain the most critical detection modality for hepatic malignancy and early-stage intervention applications. Estimated valuation in 2026 represents the immediate "precision medicine push" as manufacturers invest in AI-powered platforms and biomarker discovery to meet clinical accuracy requirements.
While demand for experimental diagnostic approaches faces validation hurdles (development delays), the clinical value of approved testing protocols commands premium reimbursement (revenue acceleration), resulting in robust percentage growth rates. The forecast accounts for the market reaching diagnostic maturity where accuracy standards are established and testing scales support broader population screening.
- Diagnostic Innovation: Growth shifts from basic imaging toward molecular testing, using AI-enhanced analysis and validated biomarker panels to maintain competitive advantages against traditional detection approaches.
- Regulatory Pathway Establishment: Standards like FDA 510(k) clearance and CE marking requirements establish baseline performance rather than experimental protocols, forcing transition from research-use products to clinical diagnostics.
- Testing Scale Requirements: To maintain cost competitiveness with conventional screening, manufacturers aggressively optimize testing processes to achieve per-test economics that support healthcare system adoption and insurance coverage.
Regional Analysis
Based on regional analysis, the liver cancer diagnostics market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania and Middle East & Africa across 40+ countries. The full report also offers market attractiveness analysis based on regional trends.
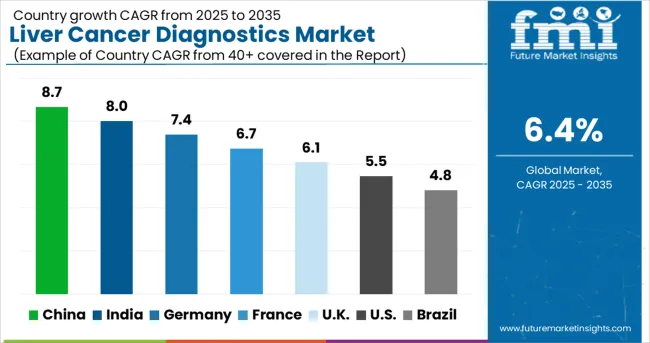
| Country | CAGR (2026 to 2036) |
|---|---|
| United States | 9.4% |
| Germany | 8.9% |
| Japan | 8.6% |
| China | 8.1% |
| South Korea | 7.8% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Liver Cancer Diagnostics Market Analysis
North America is characterized by "diagnostic leadership," where established FDA clearance pathways combine with robust Medicare reimbursement frameworks to drive clinical adoption of validated testing protocols. Key players in North America include Abbott Laboratories, which leads in biomarker testing applications, Siemens Healthineers and GE Healthcare specialize in advanced imaging and molecular diagnostic solutions.
- United States: Demand for liver cancer diagnostics in the United States is projected to rise at 9.4% CAGR through 2036. Growth is driven by Medicare coverage expansion for liver cancer screening and increased adoption of AI-powered diagnostic platforms under clinical decision support integration. As per the American Cancer Society, about 42,340 new cases (27,790 in men and 14,550 in women) of liver cancer will be diagnosed in the USA in 2026. [3]
FMI's report includes detailed analysis of growth in the North American region, along with country-wise assessment that includes the USA and Canada. Readers can find regulatory trends, reimbursement policies, and market development based on different diagnostic segments and healthcare facilities.
Asia Pacific Liver Cancer Diagnostics Market Analysis
Asia Pacific represents the global manufacturing hub, leveraging cost-competitive production facilities and streamlined regulatory pathways to achieve testing costs that enable broader population screening compared to Western markets. Sysmex Corporation leads in automated diagnostic solutions, Hitachi and Canon Medical Systems drive regional imaging technology development.
- China: Leading with an 8.1% CAGR, the market benefits from National Health Commission hepatitis screening initiatives and government investment in county-level diagnostic infrastructure.
- Japan: Domestic demand accelerates at 8.6% CAGR, supported by universal healthcare integration and PMDA collaboration with international regulatory agencies for harmonized diagnostic approval pathways.
The full report analyzes the liver cancer diagnostics market across East and South Asia from 2021-2036, covering technology adoption, manufacturing trends, and regulatory frameworks in China, Japan, South Korea, India, Singapore, and Australia. The assessment highlights diagnostic modalities that drive regional screening program adoption.
Europe Liver Cancer Diagnostics Market Analysis
Europe functions as the "regulatory harmonization laboratory," where market development is driven by CE marking procedures and coordinated clinical guideline networks across member states. Leading players include Roche Diagnostics, Philips Healthcare, and Bayer AG driving in vitro diagnostic and medical imaging development and advancing conversational AI in healthcare.
- Germany: Demand for liver cancer diagnostics in Germany rises at 8.9% CAGR through 2036, driven by statutory health insurance coverage and clinical research infrastructure that supports both domestic innovation and international medical device partnerships.
FMI's analysis of the European liver cancer diagnostics market includes country-specific assessment covering Germany, France, Italy, UK, Spain, Netherlands, and Scandinavia. Readers can access detailed regulatory framework analysis and clinical adoption trends across the European region.
Competitive Landscape for Market Players
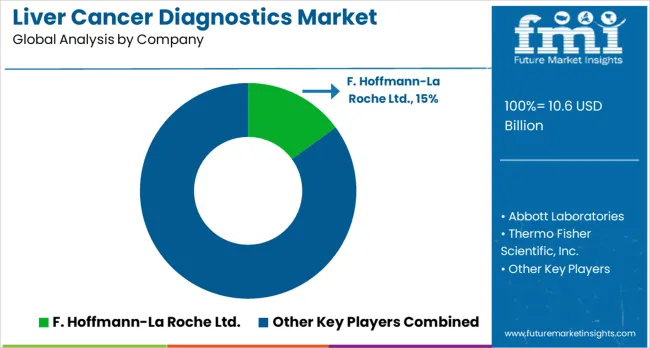
Market structure remains concentrated among established medical device companies with validated manufacturing capabilities and regulatory track records. Primary competitive variables include diagnostic accuracy demonstration, testing cost optimization, and regulatory clearance timing rather than research pipeline breadth.
Companies with integrated manufacturing facilities and established quality systems demonstrate superior ability to scale production while maintaining product consistency. Contract manufacturing relationships provide market access for smaller developers while larger healthcare companies leverage acquisition strategies to access validated diagnostic platforms.
Regulatory clearance status creates distinct competitive tiers. Companies with approved diagnostics command premium valuations and market access, while pre-clearance companies compete primarily on clinical validation progress and partnership potential.
Recent Developments
The report includes comprehensive coverage of key industry developments and competitive positioning:
- In October 2025, Thermo Fisher unveiled to acquire Clario Holdings, Inc., a leading provider of endpoint data solutions for clinical trials, to improve its research processes with deeper insights. [4]
- In October 2025, GE Healthcare teamed up with two leading USA health systems, The Queen’s Health Systems in Honolulu, HI and Duke Health in Durham, NC, to help advance the development of GE HealthCare’s new AI-driven hospital operations software. [5]
- In January 2025, Bayer acquired Camelina Assets from Smart Earth Camelina Corp. to advance biofuels. [6]
Key Players in Liver Cancer Diagnostics Market
- Abbott Laboratories
- Roche Diagnostics
- Siemens Healthineers
- GE Healthcare
- Philips Healthcare
- Sysmex Corporation
- Hitachi Medical Systems
- Canon Medical Systems
- Bayer AG
- Thermo Fisher Scientific
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 11.4 billion (2026) to USD 25.8 billion (2036), at a CAGR of 8.5% |
| Market Definition | The liver cancer diagnostics market comprises the global development, production, and commercialization of diagnostic testing products and services for hepatic malignancy detection including biomarker analysis, medical imaging, tissue characterization, and molecular testing applications. |
| Test Type Segmentation | Blood Biomarker Tests, Imaging Tests, Tissue Biopsy, Liquid Biopsy, Molecular Diagnostics |
| Cancer Type Coverage | Hepatocellular Carcinoma, Cholangiocarcinoma, Others |
| End Use Segmentation | Hospitals and Diagnostic Centers, Cancer Research Institutes, Pathology Labs |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Japan, China, South Korea, India, Australia and 40 plus countries |
| Key Companies Profiled | Abbott Laboratories, Roche Diagnostics, Siemens Healthineers, GE Healthcare, Philips Healthcare, Sysmex Corporation, Hitachi Medical Systems, Canon Medical Systems, Bayer AG, Thermo Fisher Scientific |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top down and bottom up market modeling validated through clinical practice databases and regulatory clearance tracking, supported by diagnostic accuracy verification and clinical outcome benchmarking |
Liver Cancer Diagnostics Market Analysis by Segments
Test Type:
- Blood Biomarker Tests
- Imaging Tests (CT, MRI, Ultrasound)
- Tissue Biopsy
- Liquid Biopsy
- Molecular Diagnostics
Cancer Type:
- Hepatocellular Carcinoma (HCC)
- Cholangiocarcinoma
- Others (Metastatic liver cancer, rare hepatic tumors)
End Use:
- Hospitals and Diagnostic Centers
- Cancer Research Institutes
- Pathology Labs
- Others
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia
- Singapore
- Thailand
- Rest of Asia Pacific
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Netherlands
- Scandinavia
- Switzerland
- Rest of Europe
- North America
- United States
- Canada
- Latin America
- Brazil
- Mexico
- Argentina
- Rest of Latin America
- Middle East & Africa
- Saudi Arabia
- United Arab Emirates
- South Africa
- Israel
- Rest of Middle East & Africa
Bibliography
- [1] Thermo Fisher Scientific on how AI is powering productivity, precision, and partnerships. (2025). Thermo Fisher Scientific
- [2] Bayer and Thermo Fisher Scientific collaborate to increase patient access to precision cancer medicines. (2024). Bayer AG.
- [3] Key statistics about liver cancer. (2025). American Cancer Society.
- [4] Thermo Fisher Scientific to acquire Clario Holdings, Inc., enabling pharma and biotech customers to accelerate innovation with deeper clinical insights. (2025). Thermo Fisher Scientific Investor Relations.
- [5] GE HealthCare partners with industry players to expand healthcare solutions. (2025). Bing Search Results.
- [6] Bayer acquires camelina assets from Smart Earth Camelina Corp to advance biofuels. (2024). Bayer AG.
This Report Addresses
- Market intelligence to enable strategic decision making across established and emerging liver cancer diagnostics markets
- Market size estimation and 10-year revenue forecasts from 2026 to 2036, supported by validated clinical outcome data and regulatory clearance benchmarks
- Growth opportunity mapping across test types and diagnostic applications with emphasis on accuracy optimization and clinical integration
- Segment and regional revenue forecasts covering blood biomarkers, imaging diagnostics, molecular testing, and hospital-based screening programs
- Competition strategy assessment including technology integration requirements, clinical validation timelines, and regulatory compliance capabilities
- Diagnostic development tracking including FDA clearances, CE marking approvals, and international regulatory harmonization initiatives
- Market access analysis covering reimbursement frameworks, healthcare system adoption, and patient screening programs
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive and operational use
Frequently Asked Questions
How large is the demand for Liver Cancer Diagnostics in the global market in 2026?
Demand for Liver Cancer Diagnostics in the global market is estimated to be valued at USD 11.4 billion in 2026.
What will be the market size of Liver Cancer Diagnostics in the global market by 2036?
Market size for Liver Cancer Diagnostics is projected to reach USD 25.8 billion by 2036.
What is the expected demand growth for Liver Cancer Diagnostics in the global market between 2026 and 2036?
Demand for Liver Cancer Diagnostics in the global market is expected to grow at a CAGR of 8.5% between 2026 and 2036.
Which Test Type is poised to lead global sales by 2026?
Blood Biomarker Tests are expected to be the dominant test type, capturing approximately 35.0% of global market share in 2026 due to testing accessibility and clinical integration advantages.
How significant is the role of Hospitals and Diagnostic Centers in driving Liver Cancer Diagnostics adoption in 2026?
Hospitals and diagnostic centers represent the primary end-user segment, projected to hold approximately 70% share of the total market in 2026 as clinical screening protocols expand through healthcare system integration.
What is Driving Liver Cancer Diagnostics Demand in the United States?
Medicare coverage expansion for liver cancer screening and AI-powered diagnostic platform adoption under clinical decision support integration are driving growth.
What Regulatory Standards are Referenced for the United States?
FDA 510(k) clearance pathways and CLIA laboratory certification requirements are referenced as key regulatory frameworks.
What is the United States Growth Outlook in this Report?
The United States is projected to grow at a CAGR of 9.4% during 2026 to 2036.
Why is Europe described as Important for Regulatory Development in this Report?
Growth is driven by CE marking procedures and coordinated clinical guideline networks that establish international diagnostic standards.
What Type of Development Dominates in Europe?
In vitro diagnostic (IVD) and medical imaging device development for precision oncology applications dominates regional activity.
What is Germany Growth Outlook in this Report?
Germany is projected to expand at a CAGR of 8.9% during 2026 to 2036.
Does the Report Cover Japan in its Regional Analysis?
Japan is included within East Asia under the regional scope of analysis.
What are the Sources referred to for analyzing the Market in Japan?
PMDA approval databases, universal healthcare integration tracking, and diagnostic industry associations covering technology adoption, clinical protocol progress, and screening program utilization are cited as primary reference sources.
What is the Main Development Theme Linked to Japan in Asia Coverage?
Asia Pacific development is associated with healthcare infrastructure expansion to enable broader population screening for liver cancer detection.
Does the Report Cover South Korea in its Regional Analysis?
South Korea is included within East Asia under the regional coverage framework.
What is the Main Korea Related Development Theme in Asia Coverage?
Government investment in medical technology infrastructure and hepatitis screening program integration is emphasized.
Which Manufacturing Approaches are Strategically Important for Asia Pacific Supply Chains?
Cost-competitive production facilities and streamlined regulatory frameworks are prioritized for broader diagnostic accessibility across regional markets.
What is Liver Cancer Diagnostics and What is It Mainly Used For?
Liver cancer diagnostics involves testing protocols primarily used for hepatic malignancy detection, staging, and monitoring applications including biomarker analysis and medical imaging.
What does Liver Cancer Diagnostics Market Mean in this Report?
Liver cancer diagnostics market refers to global development, manufacturing, and commercialization of diagnostic testing products across various clinical applications.
What is Included in the Scope of this Liver Cancer Diagnostics Market Report?
Scope covers diagnostic testing products by type, key applications such as cancer detection and monitoring, and end-use facilities including hospitals and diagnostic centers.
What is Excluded from the Scope of this Report?
General hepatitis screening tools, therapeutic monitoring devices, and pre-clinical research diagnostics are excluded unless part of integrated cancer detection platforms.
What does Market Forecast Mean on this Page?
Market forecast represents model-based projections built on regulatory clearance timelines and clinical adoption assumptions for strategic planning purposes.
How does FMI Build and Validate the Liver Cancer Diagnostics Market Forecast?
Forecast is developed using hybrid modeling validated through clinical practice databases, regulatory clearance tracking, and diagnostic accuracy verification.
What does Zero Reliance on Speculative Third Party Market Research Mean Here?
Market analysis is based on verifiable diagnostic performance, regulatory clearances, and clinical adoption data rather than speculative medical device valuations.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Test Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Test Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Test Type , 2026 to 2036
- Blood Biomarker Tests
- Imaging Tests (CT, MRI, Ultrasound)
- Tissue Biopsy
- Liquid Biopsy
- Molecular Diagnostics
- Blood Biomarker Tests
- Y to o to Y Growth Trend Analysis By Test Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Test Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Cancer Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Cancer Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Cancer Type, 2026 to 2036
- Hepatocellular Carcinoma (HCC)
- Cholangiocarcinoma
- Hepatocellular Carcinoma (HCC)
- Y to o to Y Growth Trend Analysis By Cancer Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Cancer Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Test Type
- By Cancer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Cancer Type
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Test Type
- By Cancer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Cancer Type
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Test Type
- By Cancer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Cancer Type
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Test Type
- By Cancer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Cancer Type
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Test Type
- By Cancer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Cancer Type
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Test Type
- By Cancer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Cancer Type
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Test Type
- By Cancer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Cancer Type
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Test Type
- By Cancer Type
- Competition Analysis
- Competition Deep Dive
- Abbott Laboratories
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Roche Diagnostics
- Siemens Healthineers
- GE Healthcare
- Philips Healthcare
- Sysmex Corporation
- Hitachi Medical Systems
- Abbott Laboratories
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Cancer Type, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Cancer Type, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Cancer Type, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Cancer Type, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Cancer Type, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Cancer Type, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Cancer Type, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Cancer Type, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Test Type
- Figure 6: Global Market Value Share and BPS Analysis by Cancer Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Cancer Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Cancer Type
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Test Type
- Figure 23: North America Market Value Share and BPS Analysis by Cancer Type, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Cancer Type, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Cancer Type
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Test Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Cancer Type, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Cancer Type, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Cancer Type
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Test Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Cancer Type, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Cancer Type, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Cancer Type
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Test Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Cancer Type, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Cancer Type, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Cancer Type
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Test Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Cancer Type, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Cancer Type, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Cancer Type
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Test Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Cancer Type, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Cancer Type, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Cancer Type
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Test Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Cancer Type, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Cancer Type, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Cancer Type
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

