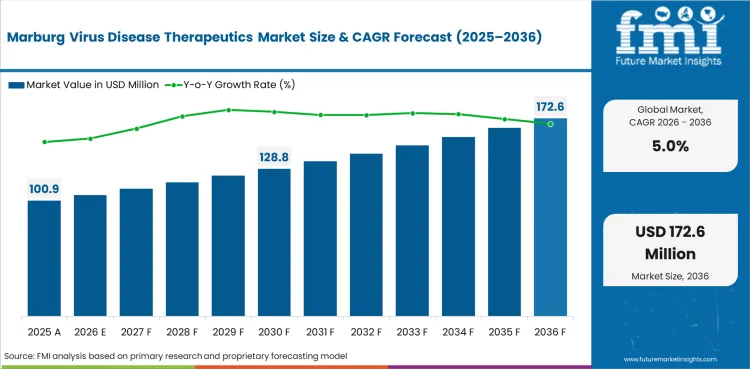
The Marburg virus disease therapeutics market covers therapeutic products and supportive care solutions used in the management of Marburg virus infection, including antibiotics for secondary infection management, antiemetic agents, nitro-glycerine formulations, loperamide, and other supportive care pharmaceuticals distributed through hospital, retail, and online pharmacy channels.
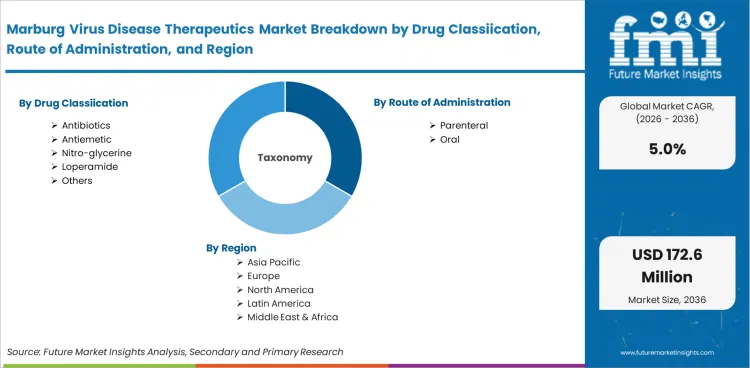
Market scope covers all commercially available Marburg virus disease therapeutic products categorized by drug classification (antibiotics, antiemetic, nitro-glycerine, loperamide, others), route of administration (parenteral, oral), end use (hospitals, specialty clinics, ambulatory surgical centres, others), and distribution channel (hospital pharmacy, retail pharmacy, online pharmacy). Revenue coverage spans 2026 to 2036.
The scope does not include experimental vaccines in preclinical development stages, diagnostic testing kits for Marburg virus detection, or personal protective equipment used in outbreak containment.
Demand for Marburg virus disease therapeutics reflects the intersection of outbreak preparedness spending and ongoing pharmaceutical research into filovirus treatments. The sporadic but severe nature of Marburg virus outbreaks creates a procurement pattern driven by government and institutional stockpile maintenance rather than continuous clinical consumption. National health agencies in the USA, Europe, and parts of Asia maintain therapeutic reserves as part of broader biodefense programs.
The majority of treatment delivery is done through parenteral administration routes. This is because Marburg virus infection is very serious and needs to be treated quickly in hospitals. Current treatment plans are based on intravenous antibiotics, fluid replacement, and supportive care drugs. Because there are no approved antiviral-specific treatments, management has to use broad-spectrum supportive care methods.
The limited supply of Marburg virus therapeutics is due to the fact that research and testing must be done in biosafety level 4 facilities. The small number of patients who can be reached outside of outbreaks makes commercial investment less appealing. This means that government funding and partnerships with international health organizations are essential for keeping the development pipeline going.
The marburg virus disease therapeutics market is segmented by drug classification, route of administration, end use, and distribution channel. Each segment reflects distinct demand dynamics shaped by end-user requirements, regulatory conditions, and procurement preferences.
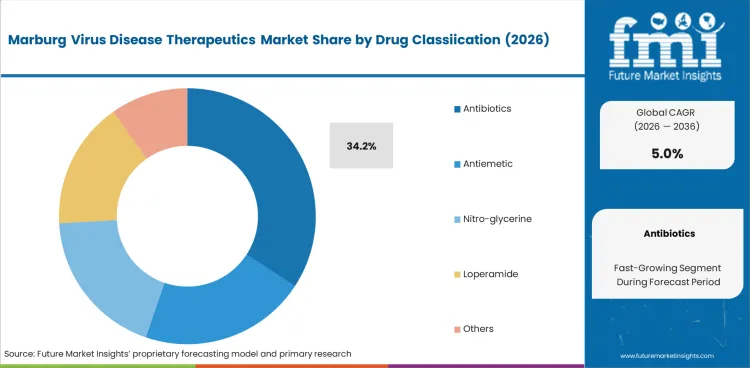
In 2026, antibiotics is expected to account for 34.2% of the drug classification segment. Antibiotics maintain their leading position due to the high incidence of secondary bacterial infections in patients with severe viral hemorrhagic fevers. Broad-spectrum antibiotics are essential components of Marburg virus disease supportive care protocols, used to manage complications that arise from compromised immune function during acute infection.
Parenteral is expected to represent 61.5% of the route of administration segment in 2026. Parenteral administration dominates due to the clinical severity of Marburg virus infection, which typically requires hospital-based intravenous therapy for fluid replacement, antibiotic delivery, and supportive care management. The rapid progression of the disease necessitates injectable formulations for immediate therapeutic effect.
The marburg virus disease therapeutics market is shaped by regulatory requirements, technology evolution, and shifting procurement structures across healthcare and institutional settings. Growth reflects both mandated adoption in established markets and expanding access in emerging regions.
Demand is shaped by national biodefense budgets that maintain therapeutic stockpiles for hemorrhagic fever outbreak response. The USA, EU member states, and selected East Asian governments allocate recurring procurement budgets for Marburg virus supportive care pharmaceuticals as part of broader epidemic preparedness frameworks.
Growth reflects the advancing clinical pipeline for Marburg virus-specific therapeutics, including monoclonal antibody candidates being evaluated for cross-protective activity against filoviruses and mRNA vaccine platforms adapted from COVID-19 development experience.
The sporadic nature of outbreaks and small addressable patient population outside epidemic events create limited commercial return potential, constraining private sector investment. Orphan drug designations and government-funded development programs partially offset this barrier.
International coordination on outbreak response protocols creates standardized procurement requirements for supportive care pharmaceuticals, providing consistency in therapeutic demand across geographically dispersed outbreak events.
.webp)
| Country | CAGR |
|---|---|
| USA | 5.1% |
| South Korea | 5.1% |
| EU | 5% |
| UK | 4.9% |
| Japan | 4.9% |
The global marburg virus disease therapeutics market is expected to grow at a rate of 5% per year from 2026 to 2036. The study covers more than 30 countries, and the main markets are listed below.
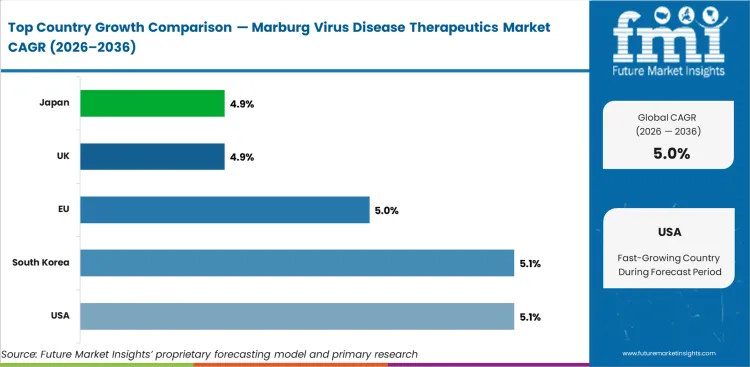
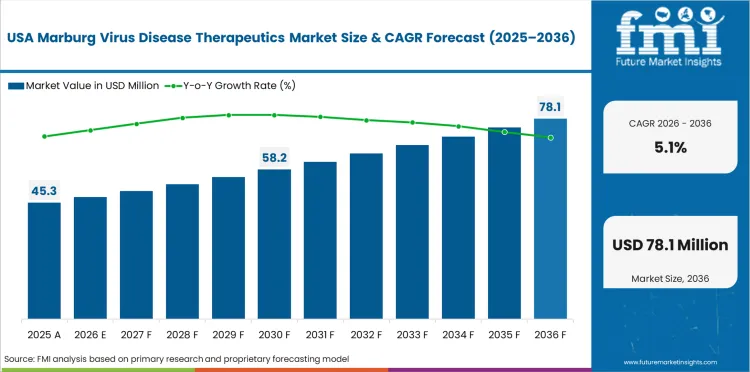
USA is expected to grow at 5.1% through 2036, supported by established healthcare infrastructure, regulatory compliance requirements, and high investment in technology adoption.
South Korea is expected to grow at 5.1% through 2036, supported by healthcare modernization programs, expanding diagnostic capabilities, and government-backed technology investment.
Opportunity Analysis of Marburg Virus Disease Therapeutics Market in the European Union
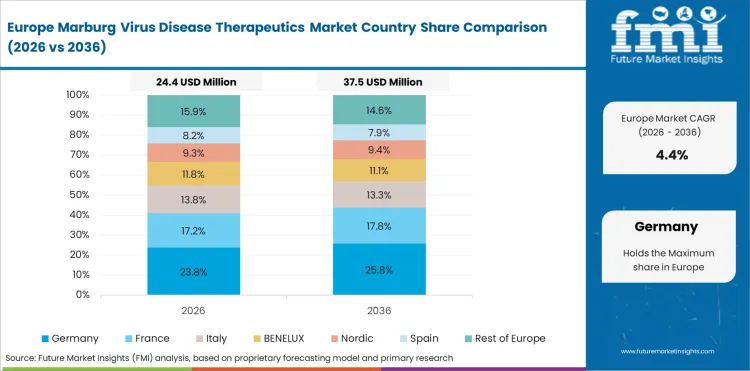
The European Union is expected to grow at 5% through 2036, supported by harmonized regulatory frameworks, cross-border procurement standards, and sustainability-driven investment programs.
In-depth Analysis of Marburg Virus Disease Therapeutics Market in the United Kingdom
The UK is expected to grow at 4.9% through 2036, supported by established healthcare system standards, government modernization programs, and expanding clinical coverage.
Japan is expected to grow at 4.9% through 2036, supported by aging population demographics, advanced manufacturing infrastructure, and high-quality standard requirements.
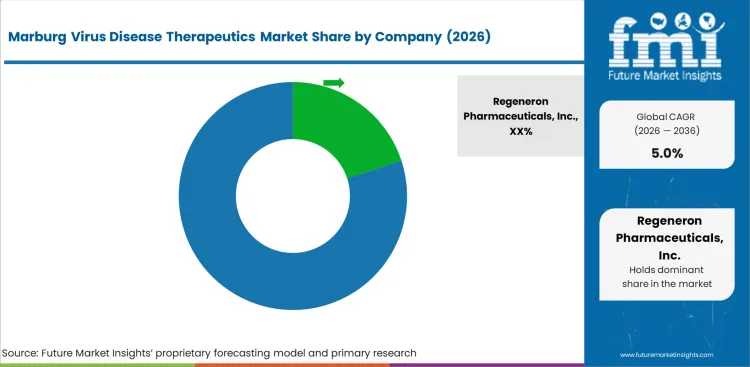
Regeneron Pharmaceuticals, Inc. leads the market through its monoclonal antibody development platform, which has demonstrated cross-protective potential against multiple filoviruses including Marburg virus. The company benefits from established government contract relationships for biodefense therapeutic procurement.
Emergent BioSolutions Inc. maintains a position through its focus on biodefense and public health emergency response therapeutics, providing established manufacturing and distribution infrastructure for government stockpile procurement programs.
Moderna, Inc. is advancing mRNA-based vaccine candidates for Marburg virus, leveraging its platform technology validated through COVID-19 vaccine development. This positions the company for future procurement contracts as vaccine candidates progress through clinical evaluation.
Commercial barriers include the high cost of BSL-4 research facilities, limited patient populations for clinical trial enrollment, and the dependence on government funding for both development and procurement. Companies with existing biodefense contract relationships hold structural advantages.
Key Companies in the Marburg Virus Disease Therapeutics Market
Key global companies leading the marburg virus disease therapeutics market include:
| Company | Therapeutic Pipeline | Government Contract Access | Manufacturing Capability | Geographic Reach |
|---|---|---|---|---|
| Regeneron Pharmaceuticals, Inc. | High | Strong | High | Global |
| Emergent BioSolutions Inc. | Medium | Strong | High | N. America, Global |
| Moderna, Inc. | High | Moderate | High | Global |
| Janssen Pharmaceuticals | Medium | Strong | High | Global |
| Gilead Sciences, Inc. | Medium | Moderate | High | Global |
| BioCryst Pharmaceuticals | Medium | Moderate | Low | N. America |
| Vaxart, Inc. | Low | Low | Low | N. America |
| Tonix Pharmaceuticals | Low | Low | Low | N. America |
| Inovio Pharmaceuticals | Medium | Moderate | Low | N. America |
| CEPI | High | Strong | Medium | Global |
Source: Future Market Insights competitive analysis, 2026.
Key Developments in Marburg Virus Disease Therapeutics Market
Major Global Players
Emerging Players/Startups
BioCryst Pharmaceuticals
| Parameter | Details |
|---|---|
| Quantitative Units | USD 105.95 million to USD 172.57 million, at a CAGR of 5% |
| Market Definition | The Marburg virus disease therapeutics market covers therapeutic products and supportive care solutions used in the management of Marburg virus infection, including antibiotics for secondary infection management, antiemetic agents, nitro-glycerine formulations, loperamide, and other supportive care pharmaceuticals distributed through hospital, retail, and online pharmacy channels. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | USA, UK, France, Germany, Italy, South Korea, Japan, China, India, 30 plus countries |
| Key Companies Profiled | Regeneron Pharmaceuticals, Inc., Emergent BioSolutions Inc., Moderna, Inc., Janssen Pharmaceuticals (Johnson & Johnson), Gilead Sciences, Inc., BioCryst Pharmaceuticals, Vaxart, Inc., Tonix Pharmaceuticals, Inovio Pharmaceuticals, CEPI (Coalition for Epidemic Preparedness Innovations) |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down methodology starting with verified transaction data, projecting adoption velocity across segments and regions. |
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with publication dates, URLs, and supporting data for all cited works.
What is the global market demand for Marburg Virus Disease Therapeutics in 2026?
In 2026, the global marburg virus disease therapeutics market is expected to be worth USD 105.95 million.
How big will the Marburg Virus Disease Therapeutics Market be in 2036?
By 2036, the marburg virus disease therapeutics market is expected to be worth USD 172.57 million.
How much is demand expected to grow between 2026 and 2036?
Between 2026 and 2036, demand is expected to grow at a CAGR of 5%.
Which Drug Classification segment is expected to lead in 2026?
Antibiotics is expected to account for 34.2% of the drug classification segment in 2026.
What is causing demand to rise in USA?
USA is expected to grow at 5.1% through 2036, supported by government biodefense investment, advanced pharmaceutical research infrastructure, and leadership in clinical trial activity for filovirus therapeutics.
What is causing demand to rise in South Korea?
South Korea is expected to grow at 5.1% through 2036, supported by investment in epidemic preparedness, participation in global health security programs, and expansion of biodefense pharmaceutical stockpile procurement.
What does this report mean by Marburg Virus Disease Therapeutics Market definition?
The Marburg virus disease therapeutics market covers therapeutic products and supportive care solutions used in the management of Marburg virus infection, including antibiotics for secondary infection.
How does FMI make the forecast?
Forecasting uses a hybrid bottom-up and top-down methodology starting with verified transaction data, projecting adoption velocity across segments and regions.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.