Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments Market Size, Market Forecast and Outlook By FMI
Summary of the Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments
- Product and Segment View
- Pharma companies segment is expected to account for 37.0% share in 2026 because internal formulation development and process transfer work require regulated analytical review.
- CDMOs and CROs and academic institutes create demand for the instruments. Sponsor-led programs bring in higher-value revenue because instrument use extends from early formulation to later analytical comparison.
- Demand and Growth Drivers
- FDA guidance for micro liposome delivery products and nanomaterial-containing drug products supports continued use of particle size and zeta potential characterization in formal product development.
- ISO 13099-2:2025 provides an updated optical-method framework for zeta-potential determination in colloidal systems.
- Replacement demand is supported by software upgrades ans broader workflow requirements with updated instrument additions in LNP and advanced drug delivery laboratories.
- Geography and Competitive Outlook
- China and India record faster growth because local biopharma investment is rising and fresh analytical laboratory capacity is coming online.
- The United States and Europe possess a larger installed base because advanced therapy activity remains high and regulated development work is established across particle-analysis fleets.
- Supplier rank depends on particle-sizing accuracy and zeta-potential workflow fit. Supplier rank depends on low sample consumption and software readiness for regulated use.
- Analyst Opinion
- Nikhil Kaitwade, Principal Analyst at FMI states that, “Industry is aligning toward multi‑method platforms and closer integration with LNP development programs. Growth potential is concentrated in CDMO capacity expansion, and replacement demand driven by upgrades of legacy particle characterization systems. Investment in advanced formulation laboratories is increasing as firms expand early-stage development capacity and shorten iteration cycles between formulation design and analytical validation.”
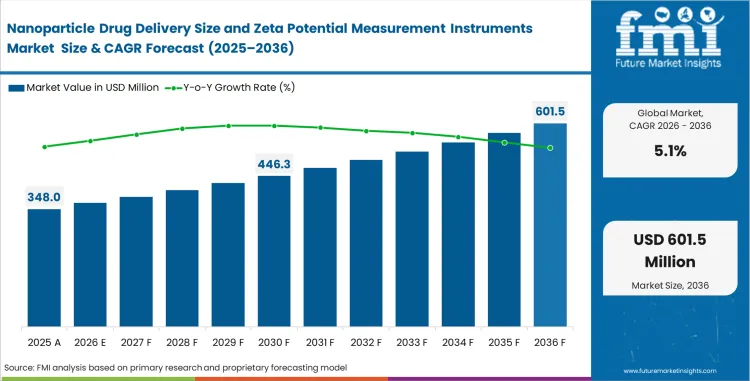
- Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments Market Value Analysis
- Industry growth from USD 348.00 million in 2026 to USD 573.00 million in 2036 reflects an analytical-instrument niche within drug delivery development.
- Revenue contribution is higher from regulated pharma programs and large CDMO accounts and from formulation laboratories.
- Long-cycle expansion in RNA delivery and nanomedicine work supports a steady demand base. FDA guidance treats particle size distribution and surface charge as important analytical parameters in nanomaterial-based drug product evaluation.
Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments Market Definition
Nanoparticle drug delivery size and zeta potential measurement instruments are analytical systems used to measure particle size and its distribution, and zeta potential in drug delivery nanoparticles. These instruments support evaluation of colloidal behavior and formulation stability along with particle-profile consistency. The market includes DLS systems and zeta-potential analyzers. It also includes nanoparticle tracking analysis systems and related multi-mode particle characterization platforms that are used across regulated and research drug delivery workflows.
Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments Market Inclusions
Market scope includes DLS systems and electrophoretic light scattering systems. It also includes zeta-potential instruments and nanoparticle tracking analysis systems, as well as integrated particle characterization platforms. Related software and instrument-linked workflows used in size measurement and formulation stability studies are also included. Scope also includes pharma and CDMO. CRO and public laboratory demand are related to nanoparticle drug delivery technology analysis.
Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments Market Exclusions
Scope of the market does not include general microscopy systems and full chromatography platforms. It also excludes contract testing revenue and general materials science instruments along with routine laboratory consumables not tied to installed particle characterization systems.
Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments Market Research Methodology
- Primary Research: Official company product pages and technical capability disclosures were reviewed. Advanced drug delivery workflow material was reviewed.
- Desk Research: FDA guidance and ISO standards were studied in depth. Clinical activity and public biopharma workflow material were reviewed.
- Market sizing and forecasting: Bottom-up narrowing was applied to advanced drug delivery analytical demand inside pharma and CDMO and CRO settings.
- Data validation: Segment shares and country growth rates were cross-checked against public supplier disclosures and standards and regulatory guidance.
Why is the Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments Market Growing?
- FDA guidance for liposome and nanomaterial-containing drug products keeps particle size and zeta potential inside formal characterization work.
- LNP and viral vector programs and formulation development programs are expected to create future revenue opportunities.
- Replacement demand is expanding with installed-base maturity and software upgrades.
Nanoparticle drug delivery solutions face aggregation risk and formulation instability during development and scale-up. A weak particle profile can reduce process control and product quality in regulated settings. Particle size and zeta potential measurements serve a direct analytical function rather than a broad exploratory role. FDA guidance for liposome drug products lists net charge, typically measured as zeta potential, among useful physicochemical properties, while FDA nanomaterial guidance requires reporting of average particle size and particle size distribution including stability.
Advanced particle characterization systems gain preference because these support low sample volumes and repeated analysis. It also supports broader workflow use inside one platform. Service and workflow expansion are other factors driving market growth. Instrument suppliers are increasing commercial focus on biopharma and advanced therapeutics.
Market Segmentation Analysis
- The DLS systems segment is estimated to account for 39.0% share in 2026, supported by established use in routine particle size measurement and wider fit across formulation workflows.
- The size measurement segment is estimated to account for 35.0% share in 2026, because particle sizing is the first analytical screen in most nanoparticle development programs.
- The process development segment is projected to hold 30.0% market share in 2026, driven by repeated optimization and scale-up work.
- The lipid nanoparticles segment is expected to garner 31.0% market share in 2026, due to expanding RNA delivery and LNP formulation activity.
- In 2026, the pharma companies segment is likely to secure 37.0% market share, as internal development and stability studies are handled at the sponsor stage.
The market is divided into instrument type, application, workflow stage, particle class, end user, and region. Instrument type includes DLS systems, zeta potential systems, NTA systems, multi-mode systems, and particle characterization platforms. Application includes size measurement, zeta potential measurement, concentration analysis, stability screening, and formulation comparison. Workflow stage includes discovery, preclinical development, process development, QC testing, and stability studies. Particle class includes lipid nanoparticles, liposomes, polymeric nanoparticles, nanocrystals, and micelles. End user includes pharma companies, CDMOs, CROs, academic institutes, and public laboratories.
Insights into the DLS Systems Segment
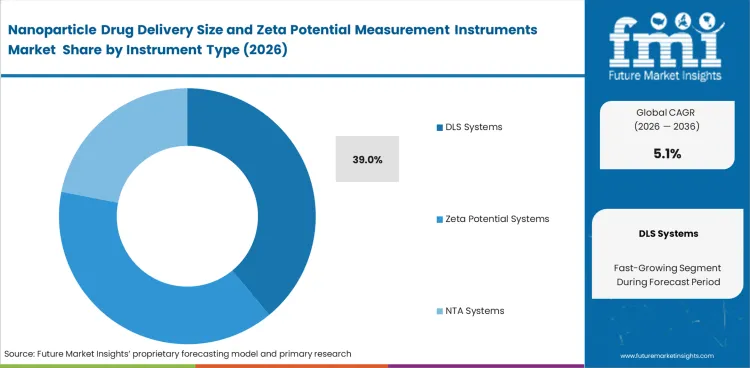
- In 2026 DLS systems are expected to contribute 39.0% of total market share because nanoparticle sizing remains a routine analytical step across development workflows.
- DLS holds one of the largest installed base in routine particle size work as these systems support dynamic light scattering and particle concentration in one platform family.
Insights into the Size Measurement Segment
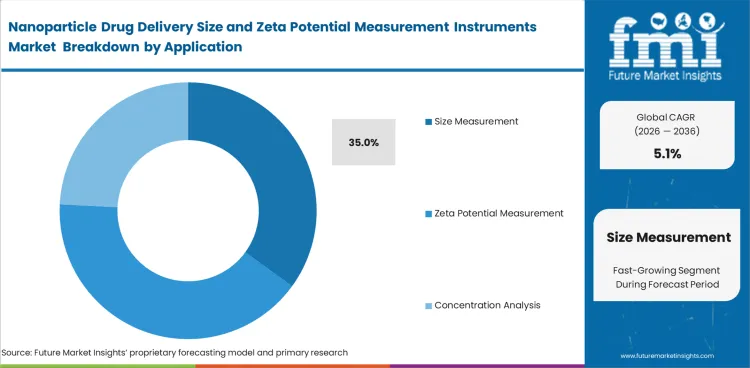
- Size measurement is expected to hold 35.0% share in 2026 because particle size is one of the first controlled attributes in nanoparticle drug product evaluation.
- The scope includes zeta potential measurement and concentration analysis. Formulation comparison is part of the market scope as well. FDA nanomaterial guidance calls for reporting average particle size and particle size distribution in premarket applications.
Insights into the Process Development Segment
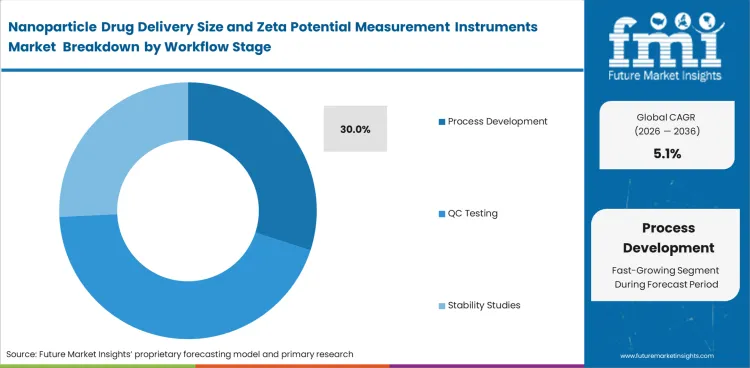
- Process development is projected to capture 30.0% of the market in 2026 because repeated formulation adjustment and scale-up studies require frequent particle checks. Process comparability work adds further demand across programs that move through repeated adjustment cycles.
- Discovery work and preclinical work including QC testing promote the demand across programs. Process development holds a wider instrument-use base as one program can require repeated particle checks across multiple batches.
Insights into the Lipid Nanoparticles Segment
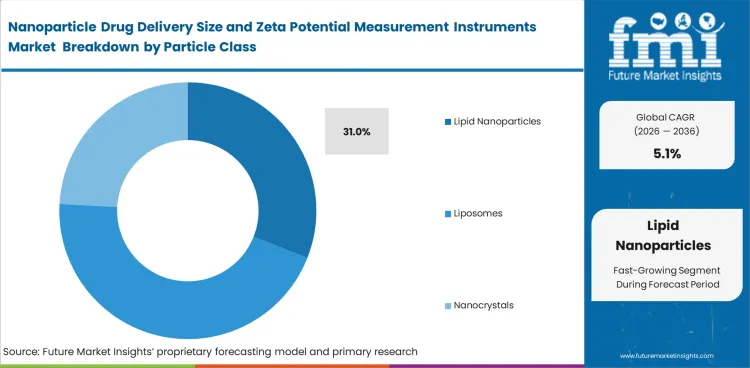
- Lipid nanoparticles are expected to represent 31.0% of market share in 2026 because RNA delivery and related therapeutics continue to expand.
- Liposomes and polymeric nanoparticles remain in high demand. Manufacturers position LNP-related workflows as active areas for particle analysis across current development programs.
Insights into the Pharma Companies Segment
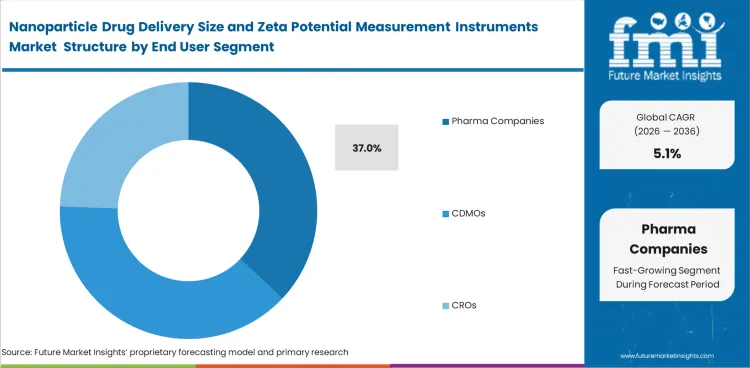
- Pharma companies segment is poised to garner 37.0% share in 2026, owing to internal formulation development and process transfer work including regulated analytical review.
- CDMOs and CROs as well as academic institutes are creating high create demand for the instruments. Sponsor-led programs bring in more valuable revenue because instrument use extends from early formulation to later analytical comparison.
Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments Market Drivers, Restraints, and Opportunities
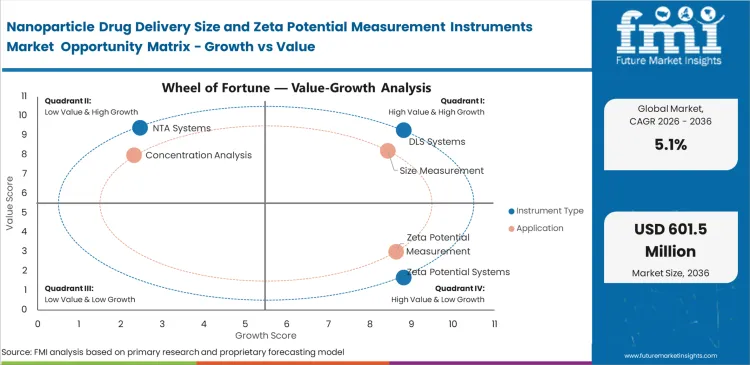
- Growth is supported by formal characterization needs and LNP development and wider use of integrated particle analysis systems.
- Adoption is limited by high instrument cost and validation time in specialized laboratories.
- Opportunity is expanding in low-sample-volume systems and multi-mode platforms. CDMO-linked analytical build-out adds another route to demand.
Demand for these instruments is shaped by regulated biopharma development activity rather than by general laboratory purchasing trends. Buyer demand is driven by the need for consistent measurement repeatability and dependable data integrity across formulation and quality workflows. Pharma companies and CDMOs prioritize systems that integrate smoothly into existing development and quality frameworks, as instruments that increase method‑transfer effort or introduce analytical risk tend to face lower adoption. Players in the industry are concentrating on platforms that preserve workflow continuity and regulatory confidence, rather than on systems differentiated only by expanded feature sets.
Qualification and Method Transfer Constraints
FDA nanomaterial guidance states that critical quality attributes should capture nanomaterial-specific properties such as particle size distribution and physical stability, while also pointing to changes in surface charge such as zeta potential among stability concerns. Instrument switching is slower in that setting because methods must keep analytical continuity across development stages.
Multi-Measurement Platform Opportunity
Demand is increasingly shifting toward multi‑measurement platforms as laboratories look to expand nanoparticle characterization without proportionally increasing instrument footprints. ISO 13099‑2:2025 strengthens this demand by providing an updated optical‑method framework for zeta‑potential determination, reinforcing the need for standardized, regulator‑aligned measurements within integrated workflows. Multi‑measurement platforms gain preference because they allow laboratories to meet broader analytical requirements and optimize capital and bench space by consolidating multiple functions into one validated system
Analysis of Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments Market By Key Countries
.webp)
| Country |
CAGR |
| India |
6.5% |
| China |
6.3% |
| South Korea |
5.6% |
| Germany |
4.9% |
| United States |
4.8% |
| United Kingdom |
4.7% |
| Japan |
4.5% |
Source: FMI analysis based on primary research and proprietary forecasting model
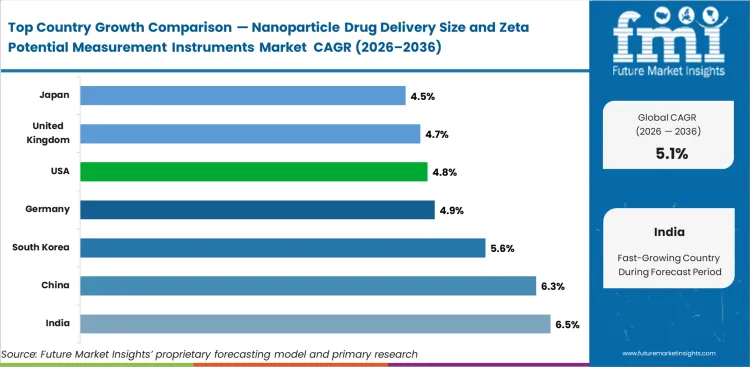
Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments Market Cagr Analysis By Country
- Demand for nanoparticle drug delivery size and zeta potential measurement instruments in India is anticipated to rise at a CAGR of 6.5% through 2036, supported by CDMO expansion and fresh analytical‑laboratory additions.
- Sales of instruments in China are expected to increase at a CAGR of 6.3% during the forecast period, driven by local biopharma scale‑up and advanced therapy activity.
- The market in South Korea is poised to expand at a CAGR of 5.6% through 2036, due to biologics and process‑development growth.
- Germany is set to record a CAGR of 4.9% during the assessment period, supported by precision analytical demand and established pharma development capacity.
- The market in the United States is likely to advance at a CAGR of 4.8% by 2036, owing to a large installed base and sustained sponsor activity.
- The United Kingdom is projected to witness a 4.7% CAGR in the nanoparticle drug delivery size and zeta potential measurement instruments market through 2036.
- Japan is likely to post a CAGR of 4.5% in the market by 2036, reflecting mature installed bases and steady regulated development demand.
The country outlook reflects a balanced demand profile across mature biopharma hubs and faster‑growing analytical markets.
Demand Outlook for Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments in India
India records one of the fastest growth rates in the market because CDMO expansion and analytical laboratory additions are increasing. Demand for nanoparticle drug delivery size and zeta potential measurement instruments in India is projected to rise at a CAGR of 6.5% through 2036 because biosimilar development and fresh instrument purchases are increasing. Supplier access improves in programs requiring lower sample use and faster workflow deployment.
- CDMO expansion supports new instrument demand because new development lines require particle sizing across repeated formulation and stability workflows.
- Fresh lab additions widen future sourcing scope because newly equipped analytical centers need installed measurement capacity from the start.
- Outsourced development supports repeated workflow use because sponsor programs rely on external laboratories through multiple formulation cycles.
Future Outlook for Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments in China
China records high growth in the market because local biopharma investment and formulation work are increasing. Sales of nanoparticle drug delivery size and zeta potential measurement instruments in China are expected to increase at a CAGR of 6.3% during the forecast period because fresh laboratory capacity and wider LNP development activity are expanding. Approved suppliers can gain ground across research and regulated development demand.
- Local biopharma growth expands particle-analysis demand because more formulation programs require routine size and charge measurement.
- New laboratory capacity supports first-system purchases because new facilities need installed analytical capability before program intake rises.
- LNP work improves instrument intensity because developers repeat particle checks through formulation adjustment and stability review.
Opportunity Analysis of Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments in South Korea
The South Korea market growth is supported by biologics activity and process-development work and therapy expansion. The nanoparticle drug delivery size and zeta potential measurement instruments market in South Korea is projected to expand at a CAGR of 5.6% through 2036 because analytical intensity is rising in newer laboratories. Low sample use and wider software support improve supplier position in this market.
- Biologics work broadens demand mix because particle characterization is used across repeated formulation and development checks.
- Newer laboratories support multi-mode system demand because buyers prefer wider workflow coverage from each installed platform.
- Process studies increase repeated instrument use because scale-up work needs frequent particle verification across multiple batches.
In-depth Analysis of Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments in Germany
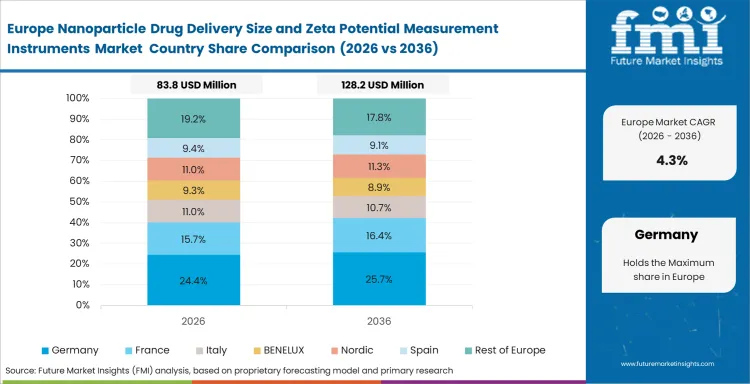
Germany's growth is driven by regulated pharma development and high analytical discipline. Germany is projected to record a CAGR of 4.9% in nanoparticle drug delivery size and zeta potential measurement instruments during the assessment period because method quality and laboratory process control remain established. High-spec development programs support premium product mix.
- Analytical rigor supports repeat instrument use because validated workflows depend on consistent particle characterization results.
- Process control favors proven suppliers because laboratories prioritize repeatability and documented method performance.
- Premium workflows keep value density high because specialized development work requires higher-spec analytical systems.
Sales Analysis of Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments in the United States
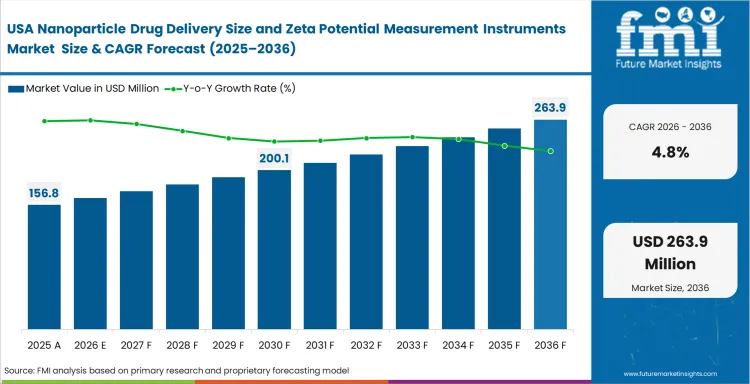
The United States benefits from high sponsor concentration and a large active installed base. The nanoparticle drug delivery size and zeta potential measurement instruments market in the United States is projected to advance at a CAGR of 4.8% by 2036 because regulated development activity and replacement demand remain active.
- Large installed bases support recurring upgrades because mature laboratories replace systems as workflow expectations change.
- Sponsor activity broadens demand mix because internal development programs require repeated analytical instrument use.
- Regulated development sustains premium system use because validated workflows require dependable particle characterization data.
Demand Outlook for Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments in the United Kingdom
Product demand in the United Kingdom is rising because biotech research depth and formulation expertise remain established. The United Kingdom is projected to witness 4.7% CAGR in the nanoparticle drug delivery size and zeta potential measurement instruments market through 2036 because particle-analysis systems hold a direct role in drug delivery programs and translational research.
- Biotech research supports particle-analysis work because formulation programs need repeated size and charge measurement.
- Formulation expertise supports technical participation because experienced laboratories can absorb higher-spec analytical workflows.
- Translational activity keeps demand steady because research programs continue to move into structured development settings.
Future Outlook for Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments in Japan
Japan is expected to expand at a steady rate in this market because analytical practice remains precise and manufacturing culture remains quality-sensitive. Japan is projected to post a CAGR of 4.5% in nanoparticle drug delivery size and zeta potential measurement instruments by 2036 because pharma development remains quality-focused. Installed-base maturity is higher than India and China and that keeps volume expansion moderate.
- Analytical discipline supports quality-sensitive workflows because laboratories prioritize repeatability and controlled method execution.
- Select therapy work supports niche demand because some programs require specialized particle characterization steps.
- Mature installed bases cap wider volume growth because many laboratories already operate particle measurement systems.
Competitive Landscape and Strategic Positioning
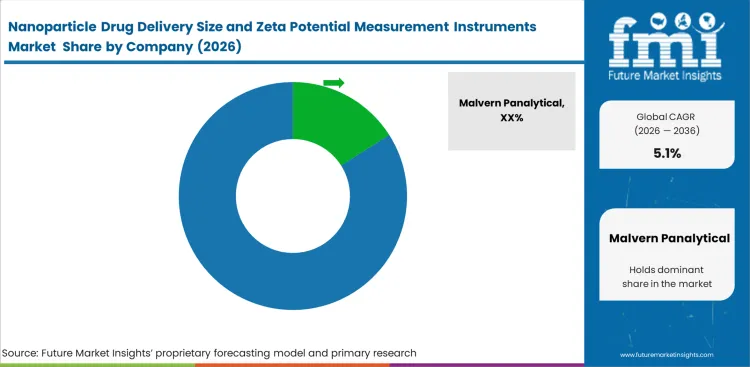
- Competition is shaped by installed-base trust and particle-sizing capability. Zeta-potential workflow fit and software support influence supplier choice as well.
- Large suppliers gain advantage from broader platform families and technical literature visibility. Multi-workflow supply access adds another competitive benefit.
- New entrants face a higher barrier because size accuracy and zeta-potential repeatability must hold under regulated analytical use. Sample handling and method continuity must work at the same time.
Malvern Panalytical and HORIBA and Anton Paar and Waters form the largest capability group because particle characterization depth and biopharma workflow visibility remain strong. Malvern positions NanoSight Pro for LNP and viral vector measurements from early R&D to batch release. Zetasizer Ultra supports particle size and zeta potential and concentration analysis with low sample use.
Brookhaven Instruments and Microtrac MRB and Otsuka Electronics hold meaningful positions in specialist particle analysis and regional laboratory demand and platform-specific supply. Adjacent work across cell and gene therapy analytics and wider biopharma characterization reflects movement toward integrated measurement platforms.
Key Companies in the Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments Market
Key global companies leading the nanoparticle drug delivery size and zeta potential measurement instruments market include:
- Broad particle characterization leaders: Malvern Panalytical, Anton Paar, HORIBA
- Biopharma and formulation analytics specialists: Waters Corporation, Brookhaven Instruments, Microtrac MRB
- Specialist precision particle analysis suppliers: Otsuka Electronics
Competitive Benchmarking: Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments
| Company |
Particle Sizing Capability |
Zeta Potential Workflow Depth |
Regulated Workflow Support |
Geographic Footprint |
| Malvern Panalytical |
High |
High |
Strong |
Global |
| Anton Paar |
High |
High |
Medium |
Global |
| HORIBA |
Medium |
Medium |
Medium |
Global |
| Waters Corporation |
Medium |
Low |
Strong |
Global |
| Brookhaven Instruments |
Medium |
High |
Medium |
North America |
| Microtrac MRB |
Medium |
Medium |
Medium |
Global |
| Otsuka Electronics |
Medium |
Medium |
Medium |
Asia |
Source: Future Market Insights competitive analysis, 2026. Ratings reflect relative positioning based on particle sizing capability, zeta potential workflow depth, regulated workflow support, and stated market reach.
Key Developments in Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments
- In March 2025, HORIBA announced the Veloci BioPharma Analyzer for biopharma and pharma applications. This is a biopharma analytical product launch that broadens HORIBA’s commercial reach in advanced therapeutic workflows.
- In April 2025, Particle Metrix GmbH announced the launch of ZetaView® Evolution, a next‑generation nanoparticle tracking analysis (NTA) system designed for advanced biopharma and life‑science applications.
Key Players in the Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments:
Major Global Players:
- Malvern Panalytical
- Anton Paar
- HORIBA
- Waters Corporation
Emerging Players/Startups
- Brookhaven Instruments
- Microtrac MRB
- Otsuka Electronics
Report Scope and Coverage
| Parameter |
Details |
| Quantitative Units |
USD Million |
| Market Definition |
Analytical instruments used for particle size, size distribution, and zeta potential measurement in nanoparticle drug delivery workflows |
| Regions Covered |
North America, Europe, Asia Pacific, Rest of World |
| Countries Covered |
India, China, South Korea, Germany, United States, United Kingdom, Japan |
| Key Companies Profiled |
Malvern Panalytical, Anton Paar, HORIBA, Waters Corporation, Brookhaven Instruments, Microtrac MRB, Otsuka Electronics |
| Forecast Period |
2026 - 2036 |
|
Approach
|
Primary research, desk research, segment modeling, and proprietary forecasting
|
Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments Market by Segments
Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments Market Segmented by Instrument Type:
- DLS Systems
- Zeta Potential Systems
- NTA Systems
- Multi-Mode Systems
- Particle Characterization Platforms
Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments Market Segmented by Application:
- Size Measurement
- Zeta Potential Measurement
- Concentration Analysis
- Stability Screening
- Formulation Comparison
Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments Market Segmented by Workflow Stage:
- Discovery
- Preclinical Development
- Process Development
- QC Testing
- Stability Studies
Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments Market Segmented by Particle Class:
- Lipid Nanoparticles
- Liposomes
- Polymeric Nanoparticles
- Nanocrystals
- Micelles
Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments Market Segmented by End User:
- Pharma Companies
- CDMOs
- CROs
- Academic Institutes
- Public Laboratories
Nanoparticle Drug Delivery Size and Zeta Potential Measurement Instruments Market Segmented by Region:
- North America
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Research Sources and Bibliography
- HORIBA, Ltd. (2025, March 4). HORIBA Announces New Veloci BioPharma Analyzer for Biopharma and Pharma Applications.
- Kıvılcım Öztürk, Meryem Kaplan, and Sema Çalış (2024, December). Effects of nanoparticle size, shape, and zeta potential on drug delivery.
- Aulia Fikri Hidayat, Yoga Windhu Wardhana, Suwendar Suwendar, Ahmed Fouad Abdelwahab Mohammed, Safwat A. Mahmoud, Khaled M. Elamin, and Nasrul Wathoni (2025, July). A Review on QbD-Driven Optimization of Lipid Nanoparticles for Oral Drug Delivery: From Framework to Formulation.
- Jiaxing Di, Yuhong Xu, and Tonglei Li (2025, December). Critical quality attributes of lipid nanoparticles and in vivo fate.
- Sarah Lindsay, Muattaz Hussain, Burcu Binici, and Yvonne Perrie (2025, March). Exploring the Challenges of Lipid Nanoparticle Development: The In Vitro–In Vivo Correlation Gap.
- Helia Nafar, Pouya Mahdavi Sharif, and Nima Rezaei (2025, December). Advances in nanomedicine-based retinal drug delivery: mechanisms and translational applications.
- Particle Metrix GmbH. (2025, April). Particle Metrix unveils ZetaView® Evolution: A new era in nanoparticle tracking analysis.
This Report Answers
- How large is the nanoparticle drug delivery size and zeta potential measurement instruments market in 2025?
- How large can the market become by 2036?
- Which instrument type leads the market in 2026?
- Why do DLS systems hold the top instrument share?
- Which end user contributes the largest demand base?
- Why do lipid nanoparticles lead particle-class demand?
- Which countries post faster-than-global growth?
- Which factors slow wider use of new particle characterization platforms?
- Why does low sample volume carry more weight in this market?
- Which companies hold the strongest particle-analysis capability?
- How do FDA and ISO guidance shape supplier selection?
- Which opportunity pocket offers better value: replacement demand or fresh advanced-therapy lab build-out?
Frequently Asked Questions
What is driving this market?
Formal characterization requirements in liposome, LNP, and related nanoparticle programs support size and zeta potential instrument demand.
Which segment leads by instrument type?
DLS systems lead because routine particle size screening is used across the widest share of workflows.
Why are pharma companies ahead of CDMOs?
Internal sponsor programs carry more repeated analytical work across development, comparability, and stability studies.
Which particle class creates the most demand?
Lipid nanoparticles lead because RNA delivery and LNP formulation programs require dense analytical support.
Why do labs still favor established platforms?
Method continuity, software familiarity, and measurement confidence often carry more weight than novelty in regulated laboratories.
Which country is expected to grow fastest?
India is projected to post the fastest CAGR among key countries covered in this study.
Are zeta potential systems enough on their own?
In some workflows, yes. In many advanced programs, size, concentration, and zeta potential are used together.
What do pharma labs screen first?
Particle size accuracy, sample volume, workflow fit, and analytical repeatability usually rank ahead of headline feature count.







.webp)





