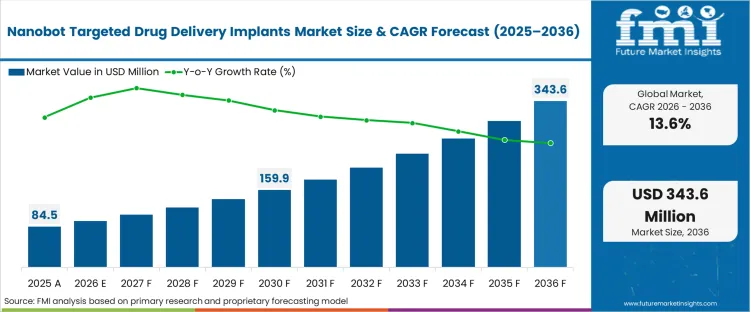
The nanobot targeted drug delivery implants market was valued at USD 84.5 million in 2025. The sector is set to cross USD 96.0 million in 2026 at a CAGR of 13.60% during the forecast period. Long-term funding fuels a rise in market valuation to USD 343.8 million through 2036 as clinical pipelines for narrow-therapeutic-index biopharmaceuticals mandate localised, zero-order elution profiles to prevent systemic toxicity.
Oncologists and neurosurgeons are moving away from relying on patient adherence to oral regimens or tolerating the severe off-target effects of systemic infusions. The decision now centers on qualifying active implantable drug delivery systems that guarantee a multi-month therapeutic window precisely at the tumour resection margin or neural target. Delaying this transition leaves clinical trial sponsors vulnerable to higher relapse rates and elevated systemic toxicity profiles that compromise drug approval pathways. Practitioners operating with translational nanomedicine devices understand that the barrier is no longer payload compatibility, but the integration of these implantable nanobot drug delivery systems into established medical device technologies without extending surgical duration.
Before localised nanomedicine scales commercially, the change from explantable hardware to fully bioresorbable nanofluidic structures must be realized. Materials science developers trigger this gate when they achieve controlled, multi-phase degradation that matches the exact duration of the injectable nanomedicines payload life. Once secondary retrieval surgeries are eliminated from the clinical pathway, adoption among ambulatory surgical centers and specialty clinics accelerates, cementing the rise of implantable targeted nanomedicine.
China is poised to advance at 15.8%, followed by India recording a 15.1% expansion rate. South Korea tracks closely at 14.0%, while the United States is anticipated to post a 13.2% CAGR. Germany is expected to expand at 12.6%, the United Kingdom will follow at 12.2%, and Japan will garner an 11.9% rate. This divergence across geographic lines reflects a transition from regions heavily burdened by legacy infusion infrastructure constraints to Asian markets actively bypassing traditional clinical setups to scale precision oncology workflows.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 96.0 million |
| Industry Value (2036) | USD 343.8 million |
| CAGR (2026-2036) | 13.60% |
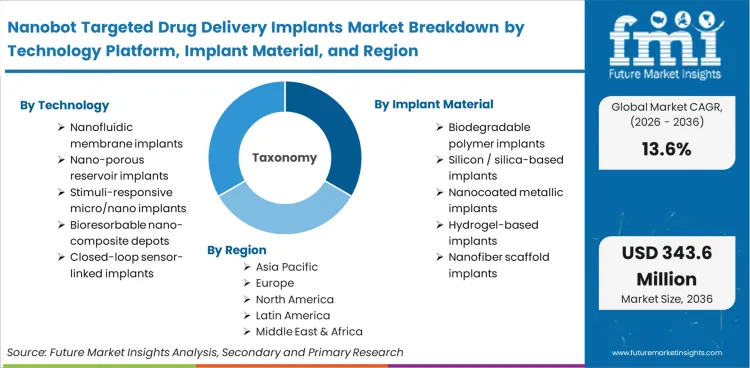
The targeted drug delivery implants market encompasses sub-millimeter and nanoscale device platforms surgically or minimally invasively placed within the body to release therapeutic payloads at localized sites. These systems utilize nanofluidic implant drug delivery channels, porous architectures, or stimuli-responsive degradation to control exact elution rates. The market boundary strictly includes implants engineered at the nanoscale for molecular delivery control and excludes macro-scale infusion pumps.
Scope includes nanofluidic membranes, bioresorbable nanocomposites, long acting implant and ocular drug delivery polymer systems, and stimuli-triggered reservoirs. Included materials cover synthesized biodegradable polymers, silicon/silica constructs, and hydrogel scaffolds designed for localized payload housing. The category also captures advanced drug delivery technology mechanisms operating via passive diffusion, external triggers, or programmable implantable drug delivery phase release within a physiological environment.
External wearable infusion pumps, traditional intra-venous catheters, and macroscopic orthopedic implants lacking active drug-eluting nanoscale architectures fall explicitly outside this scope. Standard oral nanomedicines, non-implantable circulating nanocarriers, and unguided drug delivery micro robots are excluded because they do not require surgical or site-specific physical implantation. These parameters distinguish fixed, localized therapeutic hardware from generalized systemic delivery mechanisms.
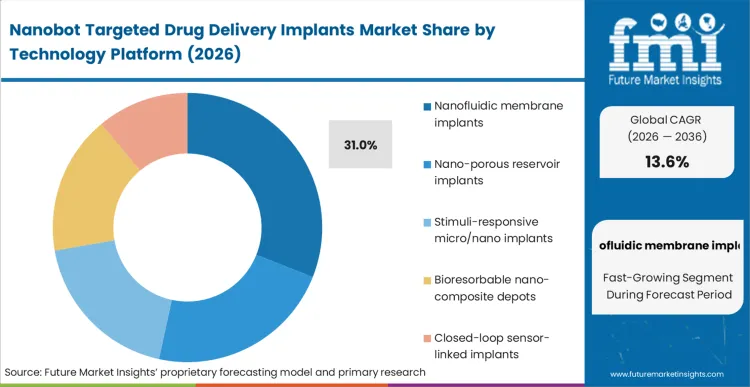
Legacy porous silicon matrices often fail to constrain the initial burst release of highly concentrated biologics, forcing researchers to evaluate the internal geometry of the implant. The reason nanofluidic implant drug delivery holds 31.0% of this sector is its ability to physically restrict molecular diffusion through precisely etched channels, guaranteeing true zero-order kinetics.
According to FMI's estimates, clinical pharmacologists utilise this architectural certainty to deliver otherwise toxic concentrations directly into glioblastoma resection cavities without breaching systemic safety thresholds. The qualification of this pharmaceutical drug delivery platform moves the development burden from chemical formulation back to mechanical engineering. Biopharma engineers who specify variable nano-porous implant drug delivery legacy systems face consistent regulatory pushback during Phase II trials due to unpredictable pharmacokinetic spikes.
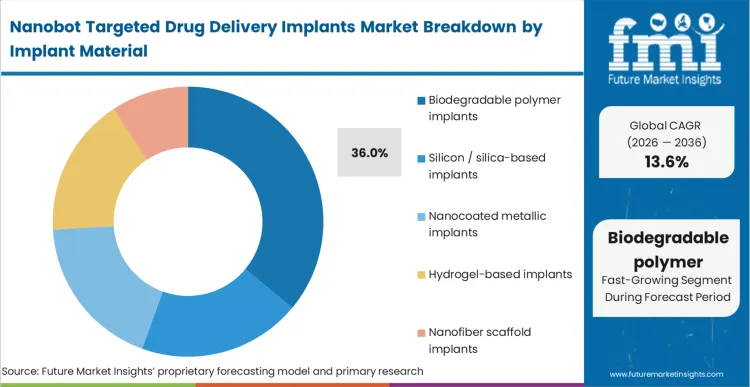
The core decision facing combination product architects today is whether to optimize for absolute rigidity or complete physiological clearance. Biodegradable polymer implants lead this dimension with 36.0% share because clinical procurement networks refuse to adopt any system that mandates a secondary explantation surgery for a depleted device.
FMI analysts estimate that polymer chemists actively tune the molecular weight of these matrices to ensure the collapse aligns perfectly with the depletion of the final peptide based nanomaterials payload. This exact synchronization prevents the formation of fibrous capsules around inert, empty casings that complicate subsequent imaging or re-dosing efforts. Formulators who cling to durable but permanent silica housings forfeit access to lucrative chronic care pathways where repeated interventions using precision medicine implant platforms are commercially unviable.
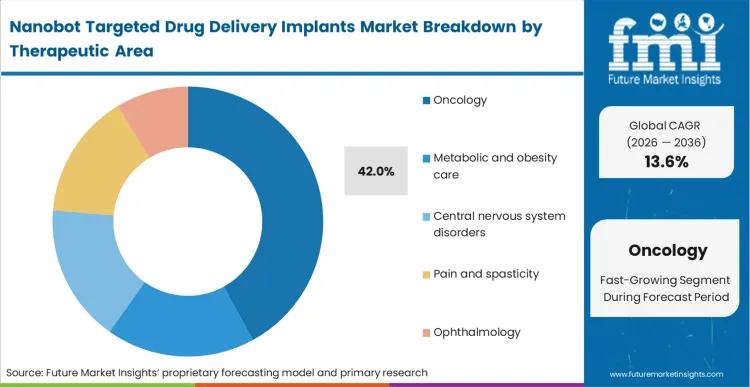
The requirement to maintain continuous, localised toxicity without triggering a systemic immune collapse explains why Oncology captures 42.0% of the total application space. Based on FMI's assessment, solid tumour microenvironments actively repel circulating nanocarriers through high interstitial fluid pressure, neutralising standard IV therapies before they penetrate the core. Surgically placing active drug delivery solutions directly into the resection margin bypasses this pressure gradient entirely, allowing continuous diffusion into adjacent malignant cells. Oncologists using oncology implantable targeted drug delivery transition from managing systemic side-effects to monitoring localised tumour regression via imaging. Facilities that delay integration of these site-specific localized products into their surgical workflows limit their capability to treat aggressive localized recurrences.
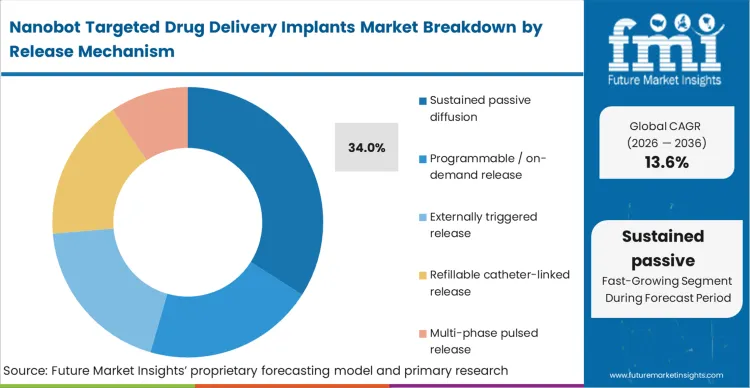
The commercial reality of medical device regulation severely penalizes systems requiring internal electronics or external battery penalisesactuation. Sustained passive diffusion retains a 34.0% share by deliberately removing mechanical and electronic failure points from the nanoparticle technology equation. Pharmaceutical manufacturers select passive diffusion to streamline the FDA combination product approval process, proving chemical kinetics rather than software reliability for smart implants drug delivery. This mechanism transforms a highly complex smart-device concept into a reliable, predictable material science execution that functions independently of patient interaction or external hardware. Developers opting for highly complex programmable electronic valves or closed-loop implant drug delivery devices face qualification timelines that routinely outlast the patent life of their primary biologic payload.
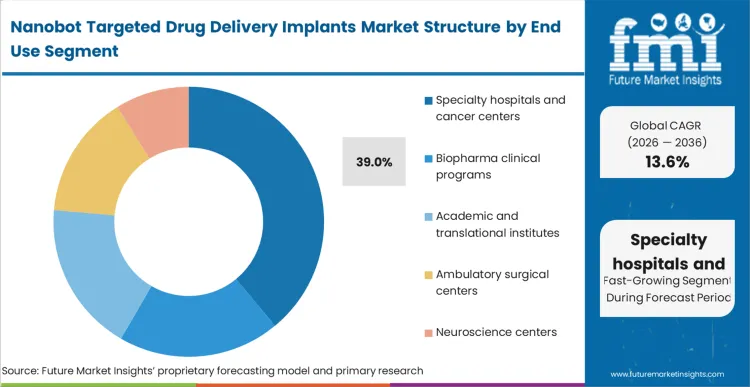
General hospital procurement networks lack the immediate clinical justification to inventory highly specialized, payload-specific combination devices. Specialty hospitals and cancer centers secure 39.0% share because they concentrate the specific patient populations, advanced imaging suites, and trained surgical oncologists required to execute these protocols. These centres internalise the cost of advanced connected drug delivery devices because the localized treatment pathway directly reduces the severe inpatient readmissions associated with traditional systemic chemotherapy toxicity. The workflow moves from outpatient infusion chairs to rapid, image-guided interventional suites executing oncology medtech implants. Institutions that fail to establish dedicated interventional oncology workflows cannot qualify for the limited distribution networks established by tier-1 combination product developers.
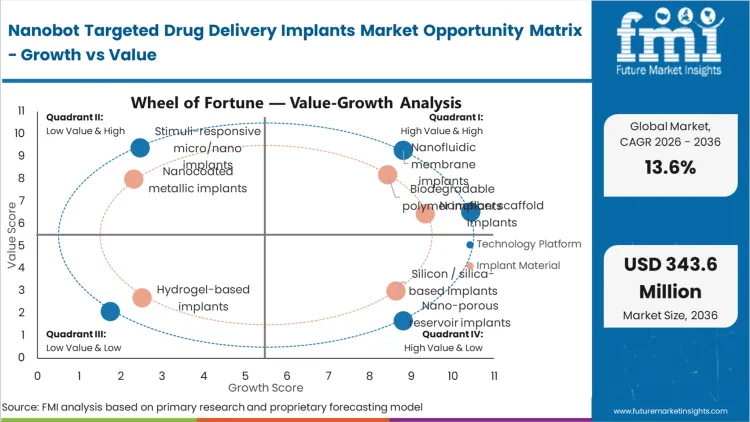
The high clinical failure rate of next-generation biologic and genetic therapies due to dose-limiting systemic toxicity forces clinical pipeline directors to adopt localized hardware. Rather than abandoning potent molecules that destroy hepatic or renal function during intravenous transit, biopharma engineers enclose these assets in nanofluidic titanium or polymer casings. This shift completely redefines the pharmacokinetic boundaries in medtech targeted drug delivery implants, allowing oncologists to maintain continuous, highly concentrated therapeutic pressure directly at a solid tumor site without compromising patient survival metrics. Delaying this architectural pivot leaves potent IP stranded in Phase II trials, unable to clear basic systemic safety thresholds.
Translational delays stem directly from the complex regulatory ambiguity surrounding combination products. Pipeline directors face a friction where the timeline to optimize a controlled release drug delivery technology under the stringent regulatory pathway for implantable drug delivery devices exceeds the development window of the active pharmaceutical ingredient itself. Furthermore, mitigating foreign body reaction in implantable drug delivery systems requires massive upfront toxicology investments. Because regulatory bodies require simultaneous validation of both the molecular stability and the mechanical reliability of the nanoscale casing, trials stretch across multiple disparate engineering disciplines.
Opportunities in the Nanobot Targeted Drug Delivery Implants Market
The regional assessment divides the Nanobot Targeted Drug Delivery Implants Market into Asia Pacific, North America, Europe, and additional regions spanning more than 40 countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 15.8% |
| India | 15.1% |
| South Korea | 14.0% |
| United States | 13.2% |
| Germany | 12.6% |
| United Kingdom | 12.2% |
| Japan | 11.9% |
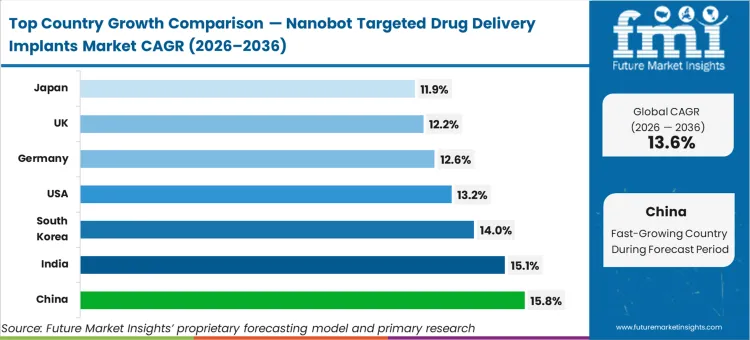
Rapidly scaling medtech miniaturization infrastructure across Asia Pacific accelerates the translation of precision device commercialization far beyond mature Western baselines. Rather than retrofitting legacy infusion networks, tier-1 research hospitals in this region are aggressively integrating localized ocular drug delivery system architectures and continuous oncology depots directly into standard practice. Based on FMI's assessment, local clinical sponsors move combination products through localized regulatory pathways optimized for advanced medical hardware, compressing the timeline between biomaterial validation and patient application.
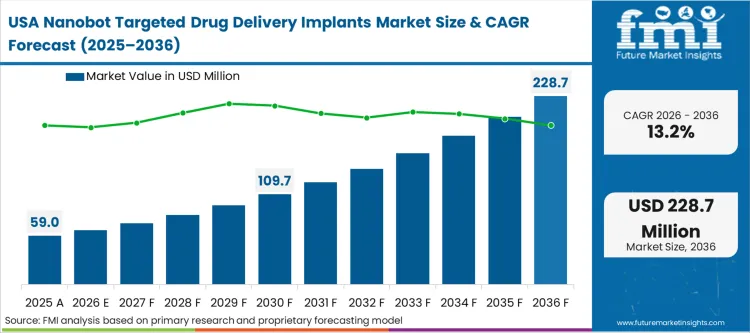
Deep clinical trial infrastructure and the heavy concentration of incumbent implantable-device intellectual property define the North American trajectory. The ecosystem revolves around massive biopharma players licensing advanced nanofluidic and drug integrated polymer fibers capabilities to rescue their own stalled biologic pipelines. As per FMI's projection, the presence of an established, rigid regulatory gating process for combination therapies moderates the pace of absolute commercial volume expansion compared to Asian markets.
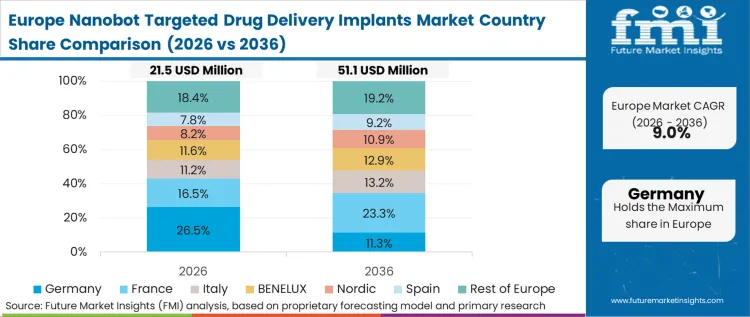
Europe's transition toward localized nanomedicine is governed heavily by established medical device frameworks and staggered, country-by-country reimbursement negotiations. Procurement networks evaluate new combinations based on strict health economic outcomes, requiring definitive proof that a highly priced localized depot offsets the downstream costs of systemic toxicity management. According to FMI's estimates, European developers focus intensely on engineering biodegradable polymers to satisfy regional demands for sustainable, intervention-free patient pathways.
FMI's report includes comprehensive analysis across other highly regulated medical device markets not explicitly detailed above. The unifying trend across these secondary regions is the rapid formulation of specialized regulatory task forces designed specifically to handle the influx of drug-device combination products, replacing outdated, fragmented review protocols. These local task forces utilize bio implants data from primary markets to accelerate their own domestic approvals.
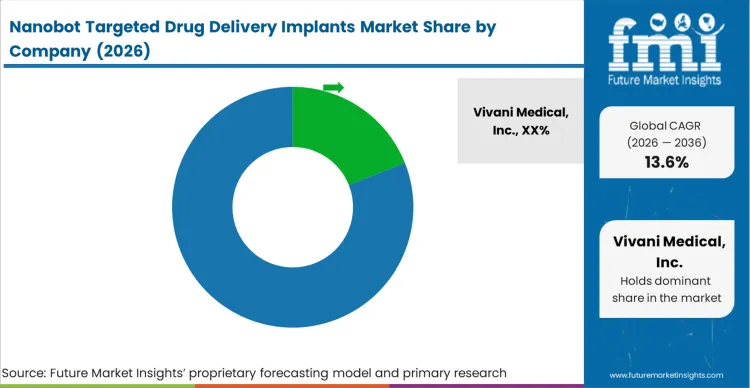
The nanobot targeted drug delivery sector operates under extreme concentration at the core technology platform level, despite a fragmented landscape of clinical applications. Massive biopharmaceutical players do not build proprietary micro robots or nanofluidic IP in-house; they hunt for validated, de-risked delivery mechanisms to rescue potent molecules that failed systemic safety checks. Procurement teams evaluate nanobot drug delivery implant suppliers and implantable drug delivery device manufacturers based almost entirely on the mechanical reliability of the zero-order elution profile. A developer secures leverage not by marketing directly to hospitals, but by embedding their proprietary delivery hardware deeply into the multi-year clinical trial pathways of blockbuster biologic assets, establishing themselves as long-acting implant drug delivery partners.
Device manufacturers dominating the legacy infusion space possess vast clinical networks, yet they often lack the specialized material science required for nanoscale fluidics. These large players must pursue implantable nanomedicine licensing deals or heavily license biocompatible polymers capability from implantable targeted drug delivery platform companies. The advantage held by early innovators is the vast repository of biocompatibility data accumulated during Phase I/II trials. For a challenger to secure nanofluidic implant technology licensing or contract development for implantable drug delivery systems, they must fund the exhaustive multi-year in-vivo toxicology studies required to prove their housing degrades without triggering localized inflammation.
Through 2036, the tension between biopharma sponsors desiring universal, modular implant casings and device developers pushing highly customised, payload-specific architectures will dictate market consolidation. As complex biologics require highly specific localized microenvironments, the generic "off-the-shelf" reservoir model becomes unviable, driving demand for the best long-acting implant platform for targeted delivery. The market accelerates toward deep, exclusive co-development partnerships, binding the fate of the medical device directly to the clinical success of the single therapeutic payload it was engineered to deliver.
| Metric | Value |
|---|---|
| Quantitative Units | USD 96.0 million to USD 343.8 million, at a CAGR of 13.60% |
| Market Definition | Sub-millimeter and nanoscale device platforms surgically placed to release therapeutic payloads at localized sites utilizing nanofluidic channels or stimuli-responsive degradation for molecular delivery control. |
| Technology platform Segmentation | Nanofluidic membrane implants, Nano-porous reservoir implants, Stimuli-responsive micro/nano implants, Bioresorbable nano-composite depots, Closed-loop sensor-linked implants |
| Implant material Segmentation | Biodegradable polymer implants, Silicon / silica-based implants, Nanocoated metallic implants, Hydrogel-based implants, Nanofiber scaffold implants |
| Therapeutic area Segmentation | Oncology, Metabolic and obesity care, Central nervous system disorders, Pain and spasticity, Ophthalmology |
| Release mechanism Segmentation | Sustained passive diffusion, Programmable / on-demand release, Externally triggered release, Refillable catheter-linked release, Multi-phase pulsed release |
| Regions Covered | Asia Pacific, North America, Europe, Latin America, Middle East & Africa |
| Countries Covered | United States, China, India, Germany, United Kingdom, Japan, South Korea, and 40 plus countries |
| Key Companies Profiled | Vivani Medical, Inc., Delpor, Inc., DelSiTech Ltd., Daré Bioscience, Inc., Medtronic plc, Renishaw plc, Flowonix Medical, Inc. |
| Forecast Period | 2026 to 2036 |
| Approach | Primary research integrates insights from translational researchers and specialty hospital procurement heads. Baseline volume anchors to verified combination product trial outputs and specialized implant material metrics. Forecasts undergo cross-validation against independent biomaterial supply volume data and registered surgical procedure counts. |
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
What are nanobot targeted drug delivery implants?
These are sub-millimeter, engineered medical devices implanted surgically or minimally invasively to deliver concentrated therapeutics precisely to a localised anatomical site. They eliminate systemic toxicity by bypassing intravenous circulation.
Explain the nanobot targeted drug delivery implants market in simple terms?
It is a fast-growing medical device sector where pharmaceutical companies partner with materials science engineers to place powerful drugs directly into tumours or neural targets using nanoscale structures. The market will cross USD 96.0 million in 2026 as biopharma developers rescue highly toxic biologic drugs by localizing their delivery.
Are nanobot drug delivery implants commercially available?
Yes, but primarily in specialized, high-acuity clinical trials rather than general commercial use. Early forms of nano-enabled resorbable polylocalisingts are currently being utilized in late-stage oncology and chronic metabolic disease trials.
How do nanofluidic drug implants work?
They utilize physically etched micro-channels at the nanoscale that act as mechanical gates. These constraints ensure molecules diffuse out of the internal reservoir at an exact, unvarying zero-order rate, preventing dangerous pharmacokinetic spikes.
Which diseases use implantable targeted drug delivery?
The technology is predominantly deployed in oncology to maintain high localized toxicity at tumour resection margins. It is also rapidly expanding into chronic weight management, psychiatric care, and central nervous system disorders.
What is the difference between nanobots and nano-enabled implants?
Medical "nanobots" often conceptually refer to autonomous, circulating microscopic robots, which remain largely experimental. Nano-enabled implants, however, are static, surgically placed depots that utilize nanoscale physics (like nanofluidic channels) to control drug release, and they represent the credible, investable segment of this market.
Are biodegradable drug implants safer than refillable implants?
From a chronic care perspective, yes. Biodegradable systems utilize natural enzymatic breakdown pathways to dissolve entirely, eliminating the surgical risk and fibrous capsule formation associated with leaving an empty, permanent titanium or silica pump in the body.
Which companies are developing implantable nanobot drug delivery platforms?
Key innovators include materials science and specialty medtech developers such as Vivani Medical, Inc., DelSiTech Ltd., Delpor, Inc., and Daré Bioscience, Inc., alongside massive legacy incumbents like Medtronic plc.
Is nanobot drug delivery closer to implants or injectable nanomedicine?
It is closer to an implant. While it manages nanoscale therapeutic payloads similar to injectable nanomedicines, the delivery mechanism requires surgical or localized physical placement of hardware, classifying it functionally and regulatorily as a combination medical device.
Give me forecast drivers for implantable targeted nanomedicine through 2036?
The market expands at a 13.60% CAGR driven by the absolute clinical necessity to localize highly toxic next-generation biologics, the shift toward outpatient ambulatory oncology care, and the urgent need to solve patient non-adherence in chronic psychiatric and metabolic treatments.
What are real examples of nano-enabled drug implants in development?
Vivani Medical’s NPM-139 is a prime example; it is a miniature, subdermal semaglutide implant entering clinical studies for once or twice-yealocalisenic weight management dosing.
Summarize risks for commercializing nanobot drug delivery implants?
The primary risk is the immense capital burden of dual-pathway regulatory approval. Developers must simultaneously prove the molecular stability of the drug and the mechanical safety of the casing, requiring exhausting toxicology studies to rule out localized foreign body reactions.
Nanobot drug delivery implants vs implantable infusion pumps?
Infusion pumps rely on macro-scale electronic motors and external batteries to push fluids, making them bulky and prone to mechanical failure. Nanobot implants rely on passive nanoscale fluidics or polymer degradation, achieving zero-power operation in a fraction of the physical footprint.
Nano-enabled implants vs injectable nanomedicines?
Injectable nanomedicines circulate systemically through the bloodstream and rely on chemical targeting to find tumours, often losing efficacy to hepatic clearance. Nano-enabled implants are physically placed directly at the disease site, completely bypassing systemic circulation.
Biodegradable drug implants vs refillable pumps?
Refillable pumps offer multi-year lifespans but require permanent indwelling hardware and regular clinical visits for transcutaneous refilling. Biodegradable implants offer shorter durations (3 to 12 months) but dissolve completely, freeing the patient from secondary explantation surgeries.
Nanofluidic implants vs polymer depots?
Polymer depots rely on the chemical degradation of the matrix to release the drug, which can sometimes be affected by local enzymatic variations. Nanofluidic implants rely on rigid, physically etched channels that guarantee an exact elution rate regardless of the surrounding physiological environment.
Smart implants vs passive diffusion implants?
Smart implants utilize integrated biosensors and external triggers to alter dosing on demand, which complicates FDA approval due to software verification requirements. Passive diffusion implants rely solely on osmotic gradients, ensuring reliable, intervention-free operation that regulators approve much faster.
Localized cancer therapy implants vs systemic therapy?
Systemic therapy floods the entire body with cytotoxic chemicals, causing severe collateral damage to healthy organs. Localized cancer therapy implants restrict the cytotoxic payload strictly to the tumour microenvironment, allowing oncologists to use vastly higher concentrations safely.
Can implantable biosensors control drug release automatically?
Yes, but they are subject to extreme regulatory scrutiny. While closed-loop systems exist in prototype phases that read local biomarkers and actuate drug release autonomously, the near-term commercial volume remains heavily anchored in reliable, passive zero-order diffusion architectures.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.