Medical Device Technology Market
Covering the medical device technology market in detail, this report addresses market valuation, share breakdown, revenue projections, price benchmarking, company positioning, competitive dynamics, demand patterns, expansion drivers, market challenges, ai integration trends, artificial intelligence integration trends, clinical adoption patterns, channel performance, supply chain evolution, regulatory influence, opportunity assessment, and strategic recommendations.
Medical Device Technology Market Size, Market Forecast and Outlook By FMI
The medical device technology market was valued at USD 575.60 billion in 2025, projected to reach USD 603.23 billion in 2026, and is forecast to expand to USD 964.04 billion by 2036 at a 4.8% CAGR. Hospital capital expenditure cycles and the migration of complex procedures from inpatient to ambulatory settings are reshaping device procurement patterns across orthopedic, ophthalmology, endoscopy, and diabetes care categories. Healthcare systems allocating capital budgets for facility modernization now specify connected, software-enabled medical devices that integrate with electronic health record platforms and clinical decision support systems as standard procurement criteria.
Summary of Medical Device Technology Market
- Market valuation is expected to climb to USD 964.04 billion by 2036.
- The industry is expected to expand at 4.8% annually from 2026 through 2036.
- The 2025 baseline valuation came in at USD 575.60 billion.
- The ten-year outlook presents a cumulative growth opportunity of USD 360.81 billion.
- Orthopedic Devices lead by device type with 26.3% share in 2026, and Hospitals lead by end user with 32.1% share in 2026.
- The market is a capital-intensive healthcare technology category where procurement is governed by facility modernization budgets, clinical workflow integration requirements, and the shift toward software-enabled, connected device platforms.
- Among key countries, China leads at 6.5% CAGR, followed by India at 6.0%, Germany at 5.5%, France at 5.0%, the UK at 4.6%, the USA at 4.1%, and Brazil at 3.6%.
The absolute dollar opportunity of USD 360.81 billion between 2026 and 2036 positions this as one of the largest addressable markets in healthcare capital equipment. Orthopedic devices maintain category leadership as aging population demographics sustain joint replacement, spinal fixation, and trauma management procedure volumes across mature and developing healthcare markets. FMI analysts observe that diabetes care device technology is experiencing the fastest intra-category growth as continuous glucose monitoring systems and insulin delivery platforms transition from specialty prescriptions to primary care standards. As per FMI, the integration of artificial intelligence into diagnostic imaging, endoscopy navigation, and respiratory monitoring is differentiating next-generation devices from legacy product lines in hospital procurement evaluations.
China leads at 6.5% CAGR as its healthcare infrastructure modernization programs and domestic medical device manufacturing incentives drive technology adoption across tier-2 and tier-3 city hospital networks. India follows at 6.0% driven by expanding private hospital chains and government health insurance coverage extension. Germany at 5.5% reflects its medical device engineering leadership and hospital digitization mandates. France at 5.0% benefits from public healthcare system equipment renewal programs. The UK at 4.6% sustains demand through NHS technology modernization cycles. The USA at 4.1% maintains the largest absolute market value, while Brazil at 3.6% benefits from expanding diagnostic center networks in urban centers.
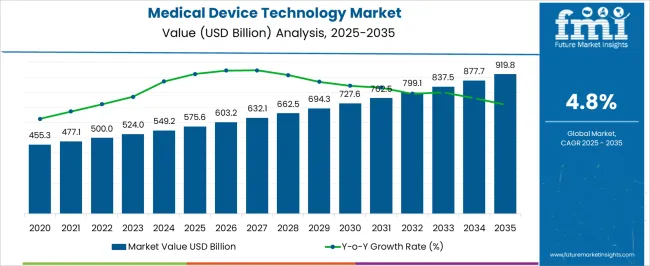
Medical Device Technology Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 603.23 billion |
| Industry Value (2036) | USD 964.04 billion |
| CAGR (2026-2036) | 4.8% |
Source: Future Market Insights, 2026
Medical Device Technology Market Definition
Medical device technology encompasses the engineered instruments, apparatus, machines, and implants used in clinical diagnosis, treatment, monitoring, and rehabilitation across healthcare delivery settings. The category includes orthopedic implants and instruments, ophthalmology surgical and diagnostic devices, endoscopy systems, diabetes care monitoring and delivery platforms, wound management technologies, dialysis equipment, and anesthesia and respiratory care devices. These products serve hospitals, clinics, diagnostic centers, ambulatory surgical centers, and research institutions.
Medical Device Technology Market Inclusions
Market scope includes orthopedic, ophthalmology, endoscopy, diabetes care, wound management, kidney/dialysis, and anesthesia and respiratory care device technologies. Coverage includes global and regional market sizes, segment breakdowns by device type and end user, and forecast projections for 2026 to 2036.
Medical Device Technology Market Exclusions
The scope excludes pharmaceutical drug products, health IT software-only platforms without integrated hardware components, laboratory reagent consumables, and hospital furniture and non-clinical infrastructure equipment.
Medical Device Technology Market Research Methodology
- Primary Research: Analysts engaged with hospital procurement directors, clinical department heads, and medical device regulatory affairs managers to map technology adoption patterns, capital budget allocation frameworks, and device specification decision criteria.
- Desk Research: Data collection aggregated medical device regulatory clearance databases, hospital capital expenditure surveys, and clinical technology adoption studies from health authorities and medical device industry associations.
- Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of medical device procurement volumes across healthcare facility categories, applying capital budget growth rates, procedure volume projections, and technology replacement cycles to forecast demand.
- Data Validation and Update Cycle: Projections are tested against publicly reported expenditure data, regulatory compliance spending trends, and industry association procurement survey results to confirm forecast alignment with observable market activity.
Why is the Medical Device Technology Market Growing?
The medical device technology market is witnessing steady growth, driven by increasing demand for innovative healthcare solutions and advanced medical equipment across hospitals, clinics, and outpatient care centers. Rising investments in healthcare infrastructure, coupled with the growing prevalence of chronic diseases and aging populations, are boosting the adoption of medical devices.
Technological advancements in diagnostic, therapeutic, and monitoring devices are enhancing clinical outcomes and improving operational efficiency in healthcare facilities. Integration of digital health solutions, artificial intelligence, and connected devices is further expanding the capabilities of medical devices, enabling real-time monitoring, predictive analytics, and personalized treatment plans.
Regulatory approvals and standards for safety and efficacy are supporting the adoption of high-quality devices, while healthcare providers are increasingly prioritizing devices that improve patient care and workflow efficiency As healthcare spending continues to rise globally, and hospitals seek scalable, interoperable, and precise medical technologies, the market is expected to maintain robust growth over the coming decade.
Segmental Analysis
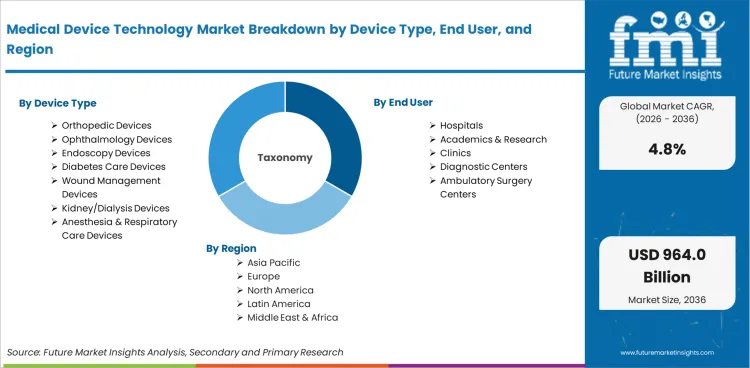
The medical device technology market is segmented by device type, end user, and geographic regions. By device type, medical device technology market is divided into Orthopedic Devices, Ophthalmology Devices, Endoscopy Devices, Diabetes Care Devices, Wound Management Devices, Kidney/Dialysis Devices, and Anesthesia & Respiratory Care Devices. In terms of end user, medical device technology market is classified into Hospitals, Academics & Research, Clinics, Diagnostic Centers, and Ambulatory Surgery Centers. Regionally, the medical device technology industry is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the Orthopedic Devices Segment
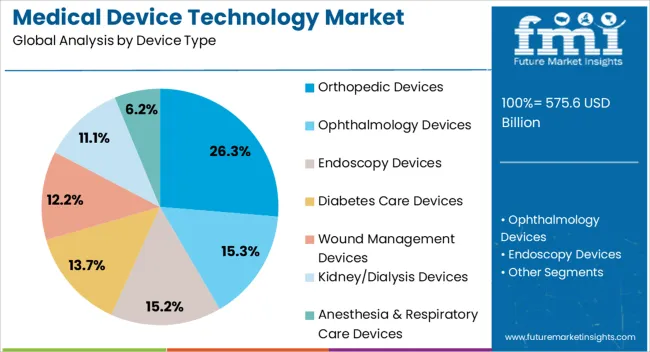
The orthopedic devices segment is projected to hold 26.3% of the medical device technology market revenue share in 2026, making it the leading device type. This dominance is being driven by the rising prevalence of musculoskeletal disorders, including fractures, arthritis, and osteoporosis, which have created strong demand for implants, prosthetics, and surgical instruments. Technological advancements in minimally invasive procedures, smart implants, and customized orthopedic solutions are enhancing patient outcomes and recovery times.
The segment benefits from the ability of orthopedic devices to improve mobility and quality of life, which is particularly valued in aging populations. Hospitals and specialized orthopedic centers are increasingly investing in advanced devices that support precision surgery and efficient post-operative care.
The segment’s growth is also supported by ongoing research and development, allowing manufacturers to offer innovative materials, design improvements, and digital integration for enhanced monitoring These factors collectively reinforce the segment’s leadership position in the medical device technology market.
Insights into the Hospitals End-User Segment
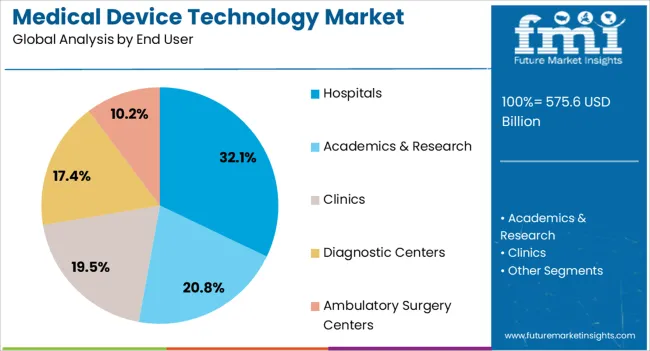
The hospitals end-user segment is expected to account for 32.1% of the medical device technology market revenue share in 2026, establishing it as the leading end-user category. This is being driven by the concentration of critical care, surgical, and diagnostic services in hospitals, which require high-performance, reliable, and interoperable medical devices. Hospitals are prioritizing devices that enhance patient safety, operational efficiency, and treatment precision, which increases demand for technologically advanced solutions.
Investments in hospital infrastructure, modernization of surgical suites, and adoption of connected medical systems are further boosting market growth. Additionally, hospitals are leveraging digital health integration, real-time monitoring, and advanced imaging and surgical technologies to optimize clinical outcomes and streamline workflows.
Regulatory compliance, accreditation standards, and increasing patient expectations for high-quality care are also driving hospitals to adopt state-of-the-art medical devices As global healthcare infrastructure expands and hospitals continue to invest in advanced technologies, this end-user segment is anticipated to maintain its leadership in market revenue share.
Market Overview
Medical Device Technology Market Dynamics
The primary driver of the industry is the aging population and the increasing prevalence of chronic diseases. As populations age globally, the incidence of chronic conditions such as cardiovascular diseases, diabetes, and orthopedic disorders rises.
Medical devices play a crucial role in the diagnosis, treatment, and management of the conditions, driving demand for innovative technologies that can address the unique needs of aging patients.
Advancements in medical device technologies, such as implantable devices, remote monitoring systems, and minimally invasive surgical tools, cater to the specific healthcare requirements of elderly populations, contributing to medical device technology market growth.
A significant restraint in the industry is the stringent regulatory requirements imposed by regulatory bodies worldwide. Regulatory compliance is essential for ensuring the safety, efficacy, and quality of medical devices, but the complex and lengthy approval processes can delay market entry and increase development costs for manufacturers.
Frequent changes in regulations and varying requirements across different regions pose challenges for companies seeking to navigate the regulatory landscape. Compliance with regulatory standards necessitates substantial investments in research, testing, and documentation, which can deter smaller companies from entering the industry and limit innovation.
An opportunity in the medical device technology market lies in the rise of telemedicine and remote monitoring solutions. The increasing adoption of telehealth services, spurred by the COVID 19 pandemic and the need for remote healthcare delivery, creates a demand for medical devices that enable virtual consultations, remote patient monitoring, and home based diagnostics.
Devices such as wearable sensors, remote monitoring systems, and telemedicine platforms offer healthcare providers real time access to patient data, facilitating timely interventions and continuity of care.
The expansion of telemedicine presents opportunities for medical device manufacturers to develop innovative technologies tailored to the growing telehealth industry, enhancing patient outcomes and expanding access to healthcare services, opening new opportunities in the industry.
A notable trend in the medical device technology market is the integration of artificial intelligence and machine learning algorithms into medical devices. Artificial intelligence powered technologies enhance the capabilities of medical devices by enabling automated data analysis, predictive analytics, and personalized treatment recommendations.
Medical devices equipped with artificial intelligence algorithms can improve diagnostic accuracy, optimize treatment plans, and streamline workflow processes for healthcare providers. Artificial intelligence driven medical devices facilitate remote monitoring and telemedicine initiatives, enabling more efficient and cost effective healthcare delivery.
The integration of artificial intelligence and machine learning represents a transformative trend in the medical device technology market, driving innovation, improving patient care, and shaping the future of healthcare delivery industry.
Analysis of Medical Device Technology Market By Key Countries
.webp)
| Country | CAGR |
|---|---|
| China | 6.5% |
| India | 6.0% |
| Germany | 5.5% |
| France | 5.0% |
| UK | 4.6% |
| USA | 4.1% |
| Brazil | 3.6% |
Source: FMI analysis based on primary research and proprietary forecasting model
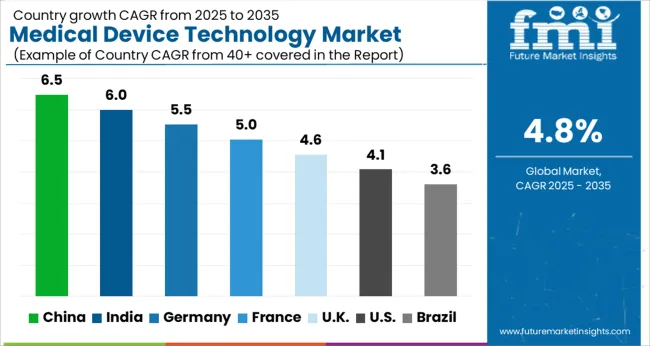
Country-wise Analysis
The Medical Device Technology Market is expected to register a CAGR of 4.8% during the forecast period, exhibiting varied country level momentum. China leads with the highest CAGR of 6.5%, followed by India at 6.0%. Developed markets such as Germany, France, and the Kcontinue to expand steadily, while the USA is likely to grow at consistent rates. Brazil posts the lowest CAGR at 3.6%, yet still underscores a broadly positive trajectory for the global Medical Device Technology Market. In 2024, Germany held a dominant revenue in the Western Europe market and is expected to grow with a CAGR of 5.5%. The USA Medical Device Technology Market is estimated to be valued at USD 199.7 billion in 2026 and is anticipated to reach a valuation of USD 297.9 billion by 2036. Sales are projected to rise at a CAGR of 4.1% over the forecast period between 2026 and 2036. While Japan and South Korea markets are estimated to be valued at USD 29.9 billion and USD 15.9 billion respectively in 2026.
Key Players in the Medical Device Technology Market
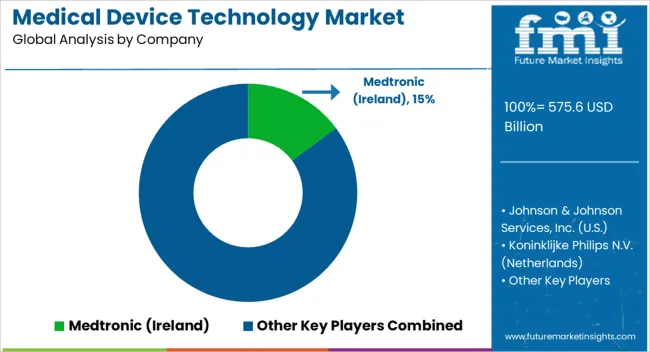
- Medtronic (Ireland)
- Johnson & Johnson Services, Inc. (USA)
- Koninklijke Philips N.V. (Netherlands)
- F. Hoffmann-La Roche Ltd. (Switzerland)
- Boston Scientific Corporation (USA)
- Fresenius Medical Care AG (Germany)
- GE Healthcare (USA)
- Siemens Healthineers AG (Germany)
- Stryker (USA)
- Abbott (USA)
- BD (USA)
- Cardinal Health (USA)
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 603.23 billion to USD 964.04 billion, at a CAGR of 4.8% |
| Market Definition | Engineered clinical instruments, apparatus, and implants used in diagnostic, therapeutic, monitoring, and rehabilitation applications across healthcare settings. |
| Segmentation | Device Type: Orthopedic Devices, Ophthalmology Devices, Endoscopy Devices, Diabetes Care Devices, Wound Management Devices, Kidney/Dialysis Devices, Anesthesia and Respiratory Care Devices; End User: Hospitals, Academics and Research, Clinics, Diagnostic Centers, Ambulatory Surgery Centers |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | India, China, USA, Canada, UK, Germany, Japan, and 40 plus countries |
| Key Companies Profiled | Medtronic, Johnson & Johnson Services, Inc., Koninklijke Philips N.V., F. Hoffmann-La Roche Ltd., Boston Scientific Corporation, Fresenius Medical Care AG, GE Healthcare, Siemens Healthineers AG, Stryker, Abbott, BD, Cardinal Health |
| Forecast Period | 2026 to 2036 |
| Approach | Forecasting models apply a bottom-up methodology starting with installed base metrics and procurement volume analysis, cross-validated against industry expenditure data and regulatory adoption timelines. |
Medical Device Technology Market by Segments
By Device Type:
- Orthopedic Devices
- Ophthalmology Devices
- Endoscopy Devices
- Diabetes Care Devices
- Wound Management Devices
- Kidney/Dialysis Devices
- Anesthesia & Respiratory Care Devices
By End User:
- Hospitals
- Academics & Research
- Clinics
- Diagnostic Centers
- Ambulatory Surgery Centers
By Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- World Health Organization. (2024). Global atlas of medical devices: Technology access and availability. WHO.
- USA Food and Drug Administration. (2024). Medical device classification database and 510(k) clearance summary. FDA.
- European Commission. (2024). Medical device regulation (MDR): Post-market surveillance requirements update. European Commission.
- Medtronic plc. (2024). Annual report 2024: Medical device segment performance review. Medtronic.
- Organisation for Economic Co-operation and Development. (2024). Health at a glance: Medical technology and innovation. OECD.
- Siemens Healthineers AG. (2024). Annual report 2024: Diagnostic and therapeutic device review. Siemens Healthineers.
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
Frequently Asked Questions
How large is the demand for Medical Device Technology in the global market in 2026?
Demand for Medical Device Technology in the global market is estimated to be valued at USD 603.23 billion in 2026.
What will be the market size of Medical Device Technology in the global market by 2036?
Market size for Medical Device Technology is projected to reach USD 964.04 billion by 2036.
What is the expected demand growth for Medical Device Technology between 2026 and 2036?
Demand for Medical Device Technology is expected to grow at a CAGR of 4.8% between 2026 and 2036.
Which Device Type is poised to lead global sales by 2026?
Orthopedic Devices accounts for 26.3% share in 2026 based on structural demand patterns and procurement preference data analyzed in the FMI report.
What is the China growth outlook in this report?
China is projected to grow at a CAGR of 6.5% during 2026 to 2036.
What is Medical Device Technology and what is it mainly used for?
Engineered clinical instruments, apparatus, and implants used in diagnostic, therapeutic, monitoring, and rehabilitation applications across healthcare settings.
What is included in the scope of this Medical Device Technology report?
Market scope includes orthopedic, ophthalmology, endoscopy, diabetes care, wound management, kidney/dialysis, and anesthesia and respiratory care device technologies. Coverage includes global and regional market sizes, segment breakdowns by device type and end user, and forecast projections for 2026 to 2036..
How does FMI build and validate the Medical Device Technology forecast?
Forecasting models apply a bottom-up methodology starting with installed base metrics and procurement volume analysis, cross-validated against industry expenditure data and regulatory adoption timelines.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Device Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Device Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Device Type , 2026 to 2036
- Orthopedic Devices
- Ophthalmology Devices
- Endoscopy Devices
- Diabetes Care Devices
- Wound Management Devices
- Kidney/Dialysis Devices
- Anesthesia & Respiratory Care Devices
- Orthopedic Devices
- Y to o to Y Growth Trend Analysis By Device Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Device Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- Academics & Research
- Clinics
- Diagnostic Centers
- Ambulatory Surgery Centers
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Device Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Device Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Device Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Device Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Device Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Device Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Device Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Device Type
- By End User
- Competition Analysis
- Competition Deep Dive
- Medtronic (Ireland)
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Johnson & Johnson Services, Inc. (USA)
- Koninklijke Philips N.V. (Netherlands)
- F. Hoffmann-La Roche Ltd. (Switzerland)
- Boston Scientific Corporation (USA)
- Fresenius Medical Care AG (Germany)
- GE Healthcare (USA)
- Siemens Healthineers AG (Germany)
- Stryker (USA)
- Abbott (USA)
- BD (USA)
- Cardinal Health (USA)
- Medtronic (Ireland)
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Device Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Device Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Device Type
- Figure 6: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by End User
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Device Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Device Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Device Type
- Figure 23: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by End User
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Device Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Device Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Device Type
- Figure 30: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by End User
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Device Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Device Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Device Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by End User
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Device Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Device Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Device Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by End User
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Device Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Device Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Device Type
- Figure 51: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by End User
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Device Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Device Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Device Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Device Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Device Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Device Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

