In 2025, the global neuromuscular transmission monitor market is valued at USD 2,025.2 million and is projected to reach approximately USD 2,829.3 million by 2035, expanding at a CAGR of 3.4%. This steady rise is influenced by expanding surgical volumes, increasing preference for objective neuromuscular monitoring, and stricter anesthesia safety protocols across healthcare systems. Regional growth patterns show distinctive adoption curves shaped by healthcare infrastructure, regulatory focus on perioperative safety, and hospital automation trends.
North America leads the neuromuscular transmission monitor market with strong penetration of quantitative neuromuscular monitoring systems in hospitals and surgical centers. The United States exhibits widespread integration of train-of-four (TOF) and electromyography (EMG)-based monitors in operating rooms, driven by compliance with American Society of Anesthesiologists (ASA) guidelines. Continuous monitoring adoption is reinforced by a focus on preventing residual neuromuscular blockade and post-operative respiratory complications. Canada follows similar patterns, supported by standardized anesthesia protocols and investments in digital monitoring systems that improve clinical workflow and data accuracy.
Europe maintains a structured regulatory and technological ecosystem that promotes the adoption of objective neuromuscular transmission monitors. Markets such as Germany, France, and the United Kingdom demonstrate consistent demand for integrated anesthesia monitoring platforms. Hospitals across Western Europe are transitioning from qualitative to quantitative monitoring techniques to comply with safety directives and minimize postoperative complications. Scandinavian nations are recognized for early adoption, emphasizing patient outcome optimization and healthcare digitization. The growing replacement of acceleromyography with EMG-based systems is improving monitoring precision and patient recovery standards across European healthcare networks.
Asia-Pacific is projected to register the fastest expansion in the forecast period, attributed to the rising number of surgical procedures, healthcare infrastructure modernization, and improved anesthesia training practices. China and India represent significant growth contributors due to the rapid establishment of tertiary hospitals, medical device localization policies, and adoption of perioperative safety standards. Japan, South Korea, and Australia are advancing the integration of real-time neuromuscular transmission monitoring within automated anesthesia systems. Increasing government funding for advanced surgical infrastructure and preference for quantifiable patient safety indicators continue to enhance regional uptake. Latin America shows gradual progress led by Brazil and Mexico, where healthcare modernization initiatives and rising private hospital investments support neuromuscular monitoring adoption. The Middle East and Africa present emerging opportunities, with Gulf countries upgrading anesthesia departments and South Africa focusing on training-led procedural safety improvements.
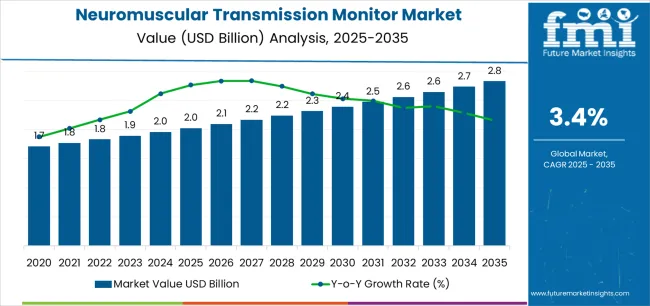
| Metric | Value |
|---|---|
| Market Value (2025) | USD 2,025.2 million |
| Market Forecast Value (2035) | USD 2,829.3 million |
| Forecast CAGR (2025-2035) | 3.4% |
| Patient Safety Enhancement | Anesthesia Quality Standards | Clinical Outcome Optimization |
|---|---|---|
| Global Patient Safety Focus Continuous expansion of perioperative safety protocols across established and emerging healthcare markets driving demand for objective monitoring solutions. Residual Blockade Prevention Growing emphasis on postoperative respiratory complication reduction creating demand for quantitative neuromuscular monitoring. Objective Assessment Requirements Superior precision and reliability characteristics making neuromuscular transmission monitors essential for evidence-based anesthesia practice. | Sophisticated Monitoring Requirements Modern surgical anesthesia requires monitoring devices delivering precise blockade assessment and enhanced patient safety outcomes. Protocol Standardization Demands Anesthesia departments investing in quantitative monitoring offering consistent assessment performance while maintaining clinical efficiency. Quality and Evidence Standards Certified monitoring systems with proven clinical validation required for advanced perioperative care applications. | Anesthesia Safety Standards Regulatory and professional requirements establishing performance benchmarks favoring objective neuromuscular monitoring solutions. Recovery Quality Standards Clinical practice standards requiring superior blockade reversal assessment and resistance to subjective evaluation errors. Complication Prevention Requirements Diverse surgical requirements and safety standards driving need for sophisticated quantitative monitoring capabilities. |
| Category | Segments Covered |
|---|---|
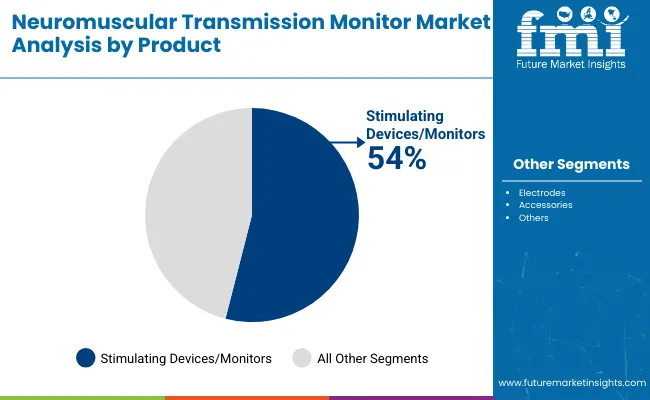
| By Product | Nerve Stimulating Devices/Monitors, Electrodes, Accessories |
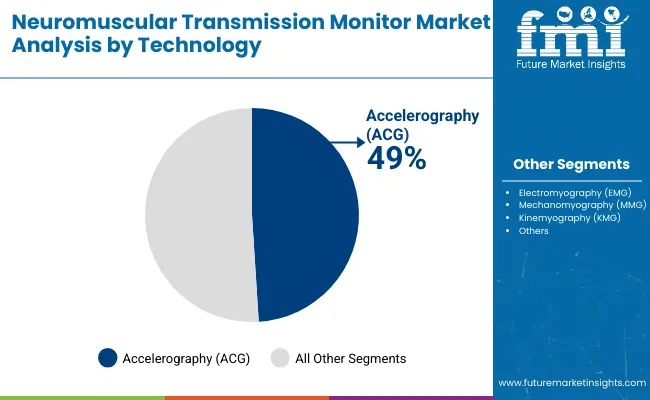
| By Technology | Accelerography (ACG), Electromyography (EMG), Mechanomyography (MMG), Kinemyography (KMG) |
| By End User | Hospitals, Specialty Clinics, Ambulatory Surgical Centers |
| By Region | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Segment | 2025 to 2035 Outlook |
|---|---|
| Nerve Stimulating Devices/Monitors | Leader in 2025 with 58.7% market share; likely to maintain leadership through 2035. Broadest use across operating rooms, intensive care units, and post-anesthesia care applications with mature technology base and established clinical protocols. Core monitoring equipment enabling train-of-four stimulation and quantitative assessment of neuromuscular blockade depth. Essential for surgical anesthesia management and residual paralysis detection. Momentum: steady growth driven by patient safety initiatives, residual blockade awareness, and hospital quality improvement programs. Watchouts: capital equipment budget constraints in cost-conscious healthcare systems and competition from integrated multiparameter monitoring platforms. |
| Electrodes | Essential consumable segment with 26.8% share, representing recurring revenue stream with consistent replacement requirements. Surface electrodes for nerve stimulation and response detection requiring regular replacement for hygiene and performance maintenance. Diverse configurations for peripheral nerve stimulation including ulnar, facial, and posterior tibial nerve applications. Momentum: steady growth aligned with monitoring device adoption and procedure volumes. Recurring purchase patterns providing stable revenue visibility. Watchouts: price competition from generic electrode manufacturers and potential reusable electrode development reducing consumable volumes. |
| Accessories | Supporting segment with 14.5% share, encompassing stimulation cables, adapters, positioning aids, and maintenance supplies. Critical for monitor functionality but lower revenue impact per unit versus core devices. Fragmented market with original equipment manufacturer and third-party accessories competing. Momentum: moderate growth tied to installed monitor base and replacement cycles. Opportunities in enhanced accessories improving workflow efficiency. Watchouts: commoditization pressures and limited differentiation opportunities constraining premium pricing strategies. |
| Segment | 2025 to 2035 Outlook |
|---|---|
| Accelerography (ACG) | Leading technology with 46.3% share in 2025, utilizing piezoelectric sensors measuring acceleration of thumb movement in response to ulnar nerve stimulation. Dominant quantitative monitoring approach offering objective train-of-four ratio measurement without subjective interpretation. Clinically validated with extensive evidence base supporting residual blockade detection. Easy to apply with minimal setup requirements supporting operating room workflow efficiency. Momentum: steady-to-strong growth driven by evidence-based medicine adoption, clinical practice guideline updates recommending quantitative monitoring, and hospital safety initiatives. Watchouts: sensor positioning sensitivity affecting measurement accuracy and requirement for unrestricted thumb movement limiting applicability in certain surgical positions. |
| Electromyography (EMG) | Established technology with 28.4% share, measuring electrical muscle activity through surface electrodes overlying target muscles. Provides quantitative assessment without movement requirements enabling use in various patient positions and clinical scenarios. Technology increasingly integrated into multiparameter anesthesia monitors. Momentum: rising through 2030 driven by integration advantages into comprehensive monitoring platforms and applicability in challenging positioning scenarios. Watchouts: electrical interference susceptibility in operating room environments and electrode placement technique sensitivity affecting signal quality. |
| Mechanomyography (MMG) | Gold standard technology with 14.7% share, measuring force of muscle contraction using force transducers. Considered reference standard for neuromuscular monitoring research but limited clinical adoption due to complexity and practical constraints. Primarily utilized in research settings and pharmaceutical trials for neuromuscular blocking agent development. Momentum: stable but limited growth constrained to specialized research applications. Clinical impracticality limiting broader adoption despite superior measurement accuracy. Watchouts: device complexity and setup requirements preventing routine clinical implementation outside academic medical centers. |
| Kinemyography (KMG) | Emerging technology with 10.6% share, utilizing piezoelectric sensors measuring bending motion of thumb in response to nerve stimulation. Novel approach attempting to address accelerography limitations while maintaining ease of use. Technology validation ongoing with growing but limited clinical evidence base. Momentum: selective growth driven by specific device manufacturer adoption and differentiation strategies. Technology advancement required for broader market acceptance. Watchouts: limited independent clinical validation versus established accelerography and mechanomyography methods. Market penetration dependent on single manufacturer advocacy limiting technology standardization. |
| Segment | 2025 to 2035 Outlook |
|---|---|
| Hospitals | At 63.5%, dominant end-user segment in 2025 with established presence in operating rooms, intensive care units, and post-anesthesia care units. Core institutional market driving neuromuscular monitoring adoption through quality improvement initiatives and patient safety programs. Capital equipment purchasing power and clinical protocol standardization supporting market development. Momentum: steady growth driven by surgical volume expansion in emerging markets, evidence-based medicine adoption, and anesthesia quality initiatives in developed healthcare systems. Watchouts: hospital budget constraints affecting capital equipment procurement and value analysis committees scrutinizing clinical utility versus cost for monitoring technology investments. |
| Specialty Clinics | Growing segment with 21.8% share, encompassing ambulatory anesthesia clinics, pain management centers, and specialized surgical facilities providing outpatient procedures. Increasing shift toward outpatient surgical care driving monitoring equipment demand in non-hospital settings. Quality standards and accreditation requirements extending to outpatient facilities. Momentum: rising through 2030 driven by healthcare cost containment policies favoring outpatient care, specialty clinic proliferation, and regulatory standards requiring equivalent monitoring capabilities versus inpatient settings. Watchouts: cost sensitivity in outpatient environments prioritizing value-based purchasing and potential for shared equipment models reducing per-facility procurement. |
| Ambulatory Surgical Centers | Expanding segment with 14.7% share, representing dedicated outpatient surgical facilities performing same-day procedures across multiple specialties. Anesthesia care requiring neuromuscular monitoring for procedures utilizing muscle relaxants and rapid patient discharge protocols necessitating residual blockade exclusion. Momentum: moderate-to-strong growth driven by ambulatory surgery market expansion, payor preferences for outpatient care, and patient convenience factors. Quality standards increasingly aligned with hospital operating room requirements. Watchouts: price-conscious purchasing environment and procedure mix with varying neuromuscular blocking agent utilization affecting monitoring necessity across all cases. |
| Drivers | Restraints | Key Trends |
|---|---|---|
| Residual Blockade Awareness Continuing expansion of clinical evidence demonstrating postoperative respiratory complication risks from inadequate neuromuscular recovery driving demand for quantitative monitoring. Evidence-Based Practice Adoption Increasing recognition of subjective clinical assessment limitations in detecting residual paralysis importance in patient safety outcomes. Surgical Volume Growth Growing demand for monitoring solutions that support both patient safety and operational efficiency in high-volume surgical environments. | Capital Budget Constraints Healthcare cost containment pressures affecting monitoring equipment procurement and competing priorities for capital allocation. Clinical Practice Inertia Established practice patterns relying on subjective assessment and variation in neuromuscular monitoring adoption across institutions and practitioners. Reimbursement Limitations Absence of specific procedural reimbursement for neuromuscular monitoring limiting economic incentives for adoption. Technology Learning Curves Device utilization training requirements and workflow integration challenges influencing adoption rates and consistent utilization. | Miniaturization and Integration Integration of wireless connectivity, compact form factors, and incorporation into multiparameter monitoring platforms enabling improved workflow efficiency. Artificial Intelligence Application Enhanced automated interpretation algorithms, trend analysis capabilities, and clinical decision support compared to basic numerical display. Point-of-Care Testing Expansion Development of simplified monitoring devices and rapid assessment technologies providing accessible monitoring in resource-limited settings. Outcome Registry Development Integration of monitoring data analytics and population-level outcome tracking for quality improvement and research applications. |
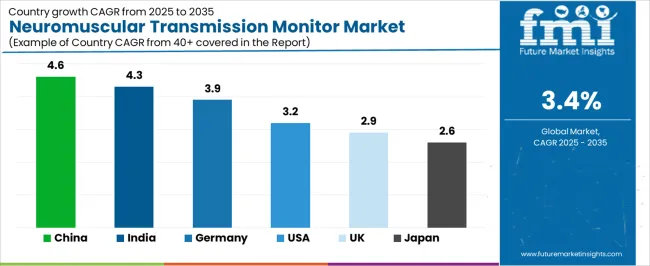
| Country | CAGR (2025-2035) |
|---|---|
| China | 4.6% |
| India | 4.3% |
| Germany | 3.9% |
| United States | 3.2% |
| United Kingdom | 2.9% |
| Japan | 2.6% |
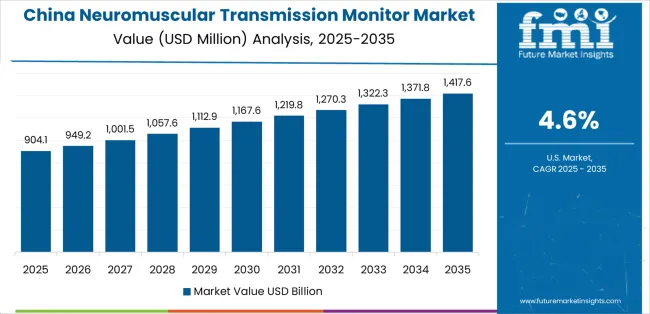
Revenue from Neuromuscular Transmission Monitor in China is projected to exhibit strong growth with a market value of USD 129.9 million by 2035, driven by expanding tertiary hospital surgical capacity and comprehensive anesthesia quality improvement initiatives creating substantial opportunities for monitoring equipment suppliers across major medical centers, university hospitals, and specialized surgical facilities. The country's rapidly modernizing healthcare infrastructure and increasing focus on perioperative patient safety are creating significant demand for advanced anesthesia monitoring technologies. Major hospital systems and medical device distributors are establishing comprehensive equipment procurement programs to support surgical volume growth and meet rising quality standards for anesthesia care delivery.
Revenue from Neuromuscular Transmission Monitor in India is expanding to reach USD 120.3 million by 2035, supported by extensive private hospital expansion and comprehensive surgical capacity development creating sustained demand for anesthesia monitoring equipment across diverse healthcare facility categories and emerging corporate hospital chains. The country's growing private healthcare sector and expanding medical tourism industry are driving demand for monitoring technologies that support international quality standards while addressing cost-effectiveness requirements. Medical equipment suppliers and hospital groups are investing in technology infrastructure to support surgical program development and quality differentiation strategies.
Demand for Neuromuscular Transmission Monitor in Germany is projected to reach USD 110.6 million by 2035, supported by the country's leadership in perioperative medicine excellence and advanced anesthesia technologies requiring sophisticated monitoring systems for patient safety optimization. German hospitals and anesthesia departments are implementing evidence-based monitoring protocols that support quantitative assessment, quality documentation, and comprehensive safety standards. The neuromuscular transmission monitor market is characterized by focus on clinical excellence, guideline adherence, and compliance with stringent medical device and patient safety standards.
Revenue from Neuromuscular Transmission Monitor in United States is growing to reach USD 91.4 million by 2035, driven by patient safety initiatives and increasing evidence-based medicine adoption creating sustained opportunities for monitoring equipment suppliers serving both academic medical centers and community hospital operating rooms. The country's extensive healthcare infrastructure and focus on perioperative quality metrics are creating demand for neuromuscular monitoring that supports objective assessment and complications reduction. Hospital systems and anesthesia practices are developing quality improvement strategies incorporating quantitative monitoring for enhanced patient outcomes.
Demand for Neuromuscular Transmission Monitor in United Kingdom is projected to reach USD 81.8 million by 2035, expanding at a CAGR of 2.9%, driven by National Health Service quality improvement priorities and anesthesia guideline recommendations supporting quantitative neuromuscular monitoring adoption. The country's established anesthesia safety culture and professional society leadership are creating demand for monitoring technologies that support evidence-based practice and patient outcome optimization. NHS trusts and private hospitals are maintaining equipment modernization programs to support clinical guideline compliance.
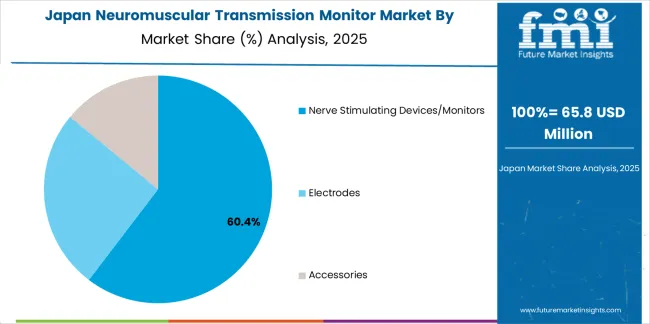
Demand for Neuromuscular Transmission Monitor in Japan is projected to reach USD 72.2 million by 2035, expanding at a CAGR of 2.6%, driven by meticulous clinical practice standards and sophisticated perioperative care capabilities supporting comprehensive anesthesia monitoring. The country's established quality culture and aging population surgical needs are creating demand for monitoring technologies that support precise neuromuscular management and safety assurance. Academic hospitals and advanced medical centers are maintaining rigorous monitoring capabilities to support demanding Japanese clinical practice expectations.
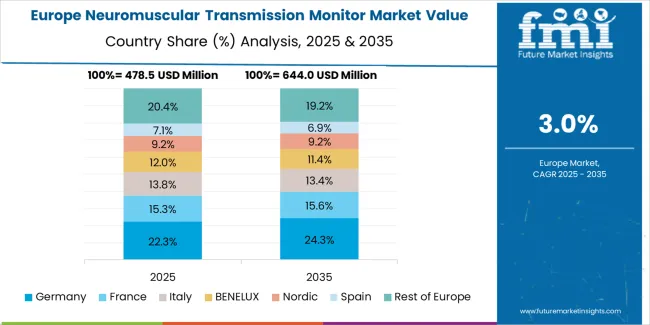
The neuromuscular transmission monitor market in Europe is projected to grow from USD 79.2 million in 2025 to USD 110.6 million by 2035, registering a CAGR of 3.4% over the forecast period. Germany is expected to maintain its leadership position with a 31.5% market share in 2025, supported by its advanced anesthesia practice standards and comprehensive perioperative medicine infrastructure across major university hospitals and surgical centers throughout Bavaria, North Rhine-Westphalia, and Baden-Württemberg regions.
France follows with a 24.7% share in 2025, projected to reach 25.1% by 2035, driven by comprehensive anesthesia society guidelines and teaching hospital leadership in quantitative monitoring adoption. United Kingdom holds a 20.6% share in 2025, expected to maintain 20.3% by 2035 due to Royal College of Anaesthetists recommendations and NHS quality improvement initiatives. Italy commands a 13.8% share, while Spain accounts for 9.4% in 2025. The Rest of Europe region is anticipated to maintain steady momentum, attributed to increasing neuromuscular monitoring adoption in Nordic countries and emerging Eastern European healthcare facilities implementing anesthesia quality programs.
European neuromuscular transmission monitor operations are increasingly defined by strong anesthesia professional society leadership and evidence-based practice culture. German and French anesthesia departments lead quantitative monitoring adoption, leveraging academic medical center clinical research and guideline development that establish best practice standards for European anesthesiology community. German university hospitals maintain leadership in perioperative outcome research and monitoring technology clinical validation, with anesthesia departments driving evidence-based protocols that influence international practice.
British anesthesia community demonstrates strong professional society engagement with Royal College of Anaesthetists and Association of Anaesthetists providing clinical guidance supporting quantitative neuromuscular monitoring. NHS quality improvement programs and patient safety initiatives create institutional drivers for monitoring technology adoption beyond individual practitioner preferences. Teaching hospitals and specialty surgical centers lead implementation with community hospital diffusion following academic center experience.
Southern European anesthesia practices in Italy and Spain show variable monitoring adoption influenced by institutional resources and individual practitioner preferences. Academic medical centers demonstrate higher technology utilization aligned with international guidelines while community hospitals may maintain traditional clinical assessment approaches. Economic constraints and capital equipment availability affect monitoring technology penetration rates.
Eastern European healthcare systems in Poland, Czech Republic, and Hungary demonstrate growing anesthesia monitoring capabilities as healthcare infrastructure modernizes and EU alignment drives quality standard adoption. International medical device companies increasingly target these markets with educational programs supporting quantitative monitoring awareness and clinical practice evolution.
Regulatory environment through European Union medical device regulations establishes safety and performance standards for neuromuscular monitors while national healthcare systems determine reimbursement and procurement policies affecting market access. Professional society guidelines from European Society of Anaesthesiology provide clinical recommendations influencing institutional protocols though adoption remains variable across diverse European healthcare contexts.
Japanese neuromuscular transmission monitor operations reflect the country's meticulous clinical practice culture and comprehensive patient safety emphasis. Major university hospitals including University of Tokyo Hospital, Kyoto University Hospital, and leading medical centers maintain sophisticated anesthesia monitoring capabilities that often exceed international norms, requiring comprehensive documentation and quality assurance processes that support Japan's patient safety reputation.
The Japanese market demonstrates unique preferences for domestic medical device manufacturers with long-established relationships and proven clinical support capabilities. Nihon Kohden and other Japanese companies maintain strong market positions through trusted brand reputations and responsive technical service networks. International manufacturers require local partnerships and extensive clinical validation to gain market acceptance.
Regulatory oversight through Pharmaceuticals and Medical Devices Agency establishes comprehensive approval processes for neuromuscular monitors requiring clinical data and manufacturing quality documentation. The approval timeline and requirements create market entry barriers but ensure device safety and performance meeting Japanese quality expectations.
Supply chain dynamics emphasize long-term relationships with medical equipment distributors providing clinical education and technical support throughout device lifecycle. Japanese hospitals value comprehensive service including training, maintenance, and responsive troubleshooting that influences purchasing decisions beyond initial device pricing considerations.
The neuromuscular transmission monitor market faces constraints from conservative practice patterns in some institutions, the continued use of traditional clinical assessment approaches, and limited economic incentives for investing in monitoring technology. The aging population's surgical needs and academic medical center leadership in evidence-based practice support gradual monitoring adoption expansion across the Japanese healthcare system.
South Korean neuromuscular transmission monitor operations reflect the country's technology-forward healthcare system and quality improvement culture. Major university hospitals including Seoul National University Hospital, Asan Medical Center, and Samsung Medical Center drive anesthesia monitoring adoption through clinical research programs and quality initiatives emphasizing patient safety and outcome optimization.
The Korean market demonstrates strong preference for advanced medical technologies with rapid adoption of innovations supported by government healthcare modernization policies. Large corporate hospitals invest comprehensively in perioperative monitoring equipment as competitive differentiation strategy and quality positioning for domestic and international patients.
Regulatory frameworks through Ministry of Food and Drug Safety establish medical device approval processes aligned with international standards while National Health Insurance Service determines reimbursement policies affecting hospital procurement decisions. Recent healthcare quality assessment programs by Health Insurance Review and Assessment Service create institutional incentives for enhanced monitoring capabilities.
Supply chain infrastructure includes both domestic medical device distributors and direct relationships with international manufacturers. Korean hospitals increasingly pursue group purchasing strategies through hospital associations achieving volume pricing while maintaining equipment standardization across member institutions.
The neuromuscular transmission monitor market faces challenges from cost containment pressures in the national health insurance system, affecting hospital operating margins and equipment investment capacity. Competition between hospitals and procedure volume concentration in major medical centers influences market dynamics. The continued healthcare quality emphasis and surgical volume growth support sustained demand for anesthesia monitoring technologies meeting international practice standards and supporting Korea's medical tourism positioning.
Profit pools are consolidating around established medical device companies integrating proprietary monitoring technologies with comprehensive clinical evidence and global commercial infrastructure. Value is migrating from standalone monitoring devices toward integrated perioperative platforms where data connectivity, electronic health record integration, and analytics capabilities command premium positioning. Several archetypes set the pace: global medical device conglomerates defending share through broad product portfolios and established hospital relationships; specialized neuromuscular monitoring companies with proprietary technologies and clinical validation; anesthesia equipment manufacturers integrating monitoring into comprehensive anesthesia workstations; and emerging digital health companies pursuing wireless connectivity and data analytics differentiation. Switching costs—capital investment, clinician training, workflow integration, and disposable electrode lock-in—stabilize incumbent positions while technology advancement and evidence generation create opportunities for differentiated solutions. Market consolidation continues as major medical device companies acquire specialized monitoring technology firms while clinical practice guideline evolution and patient safety initiatives shape adoption trajectories. Do now: secure comprehensive clinical evidence through investigator-initiated research and outcomes registry participation; hard-wire hospital group purchasing organization relationships and value analysis committee engagement; option: develop integrated perioperative data platforms with predictive analytics and co-branded enhanced recovery protocol partnerships.
| Stakeholder Type | Primary Advantage | Repeatable Plays |
|---|---|---|
| Global medical device conglomerates | Product portfolio breadth, commercial infrastructure, financial resources | GPO contracts, integrated solutions, global reach |
| Specialized neuromuscular monitoring companies | Technology differentiation, clinical expertise, market focus | Clinical evidence generation, key opinion leader partnerships, specialized sales force |
| Anesthesia equipment manufacturers | Integrated workstation platforms, established anesthesia relationships, workflow integration | Equipment replacement cycles, integrated purchasing, comprehensive service contracts |
| Emerging digital health companies | Data analytics, connectivity innovation, user experience | Wireless solutions, cloud platforms, mobile applications |
| Consumable electrode suppliers | Recurring revenue, installed base leverage, manufacturing scale | Disposable electrode contracts, aftermarket positioning, value-based pricing |
| Items | Values |
|---|---|
| Quantitative Units | USD 2,025.2 Million |
| Product | Nerve Stimulating Devices/Monitors, Electrodes, Accessories |
| Technology | Accelerography (ACG), Electromyography (EMG), Mechanomyography (MMG), Kinemyography (KMG) |
| End User | Hospitals, Specialty Clinics, Ambulatory Surgical Centers |
| Application | Surgical Anesthesia, Intensive Care, Post-Anesthesia Care, Emergency Medicine, Others |
| Regions Covered | North America, Latin America, Europe, Asia Pacific, Middle East & Africa |
| Country Covered | United States, Germany, United Kingdom, China, India, Japan, and other 40+ countries |
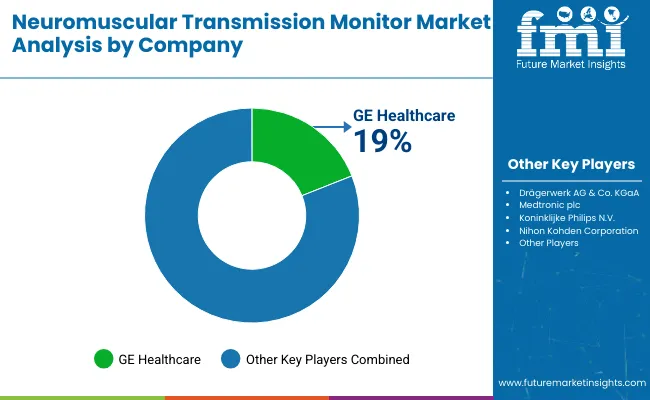
| Key Companies Profiled | GE HealthCare (Applied Precision), Nihon Kohden Corporation, Medtronic plc, Drägerwerk AG & Co. KGaA, Koninklijke Philips N.V., Becton Dickinson and Company, Senzime AB, Organon & Co., Masimo Corporation, Ambu A/S |
| Additional Attributes | Dollar sales by product/technology/end user/application, regional demand (NA, EU, APAC), competitive landscape, quantitative vs. qualitative monitoring adoption, technology evolution integration, and clinical outcome innovations driving residual blockade prevention, patient safety enhancement, and evidence-based anesthesia practice |
By Product
How big is the neuromuscular transmission monitor market in 2025?
The global neuromuscular transmission monitor market is estimated to be valued at USD 2.0 billion in 2025.
What will be the size of neuromuscular transmission monitor market in 2035?
The market size for the neuromuscular transmission monitor market is projected to reach USD 2,829.3 billion by 2035.
How much will be the neuromuscular transmission monitor market growth between 2025 and 2035?
The neuromuscular transmission monitor market is expected to grow at a 3.4% CAGR between 2025 and 2035.
What are the key product types in the neuromuscular transmission monitor market?
The key product types in neuromuscular transmission monitor market are nerve stimulating devices/monitors, electrodes and accessories.
Which technology segment to contribute significant share in the neuromuscular transmission monitor market in 2025?
In terms of technology, accelerography (acg) segment to command 46.3% share in the neuromuscular transmission monitor market in 2025.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.