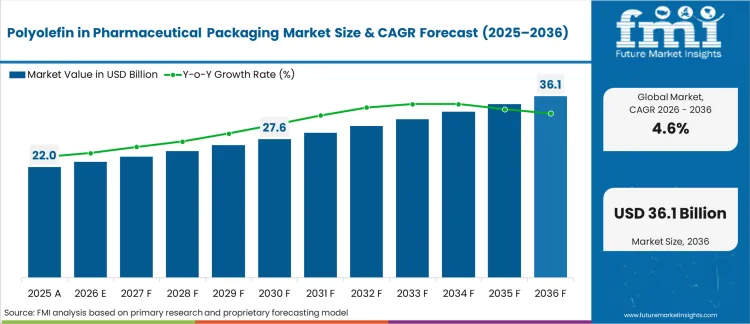
In 2025, the polyolefin in pharmaceutical packaging market was valued at USD 21,839.3 million. Based on Future Market Insights' analysis, demand for polyolefin materials in pharmaceutical packaging is estimated to grow to USD 23,016.8 million in 2026 and USD 36,001.5 million by 2036. FMI projects a CAGR of 4.6% during the forecast period.
The market is expected to generate an absolute dollar growth of USD 12,984.7 million between 2026 and 2036. This reflects steady, regulation-led expansion supported by rising pharmaceutical production, biologics growth, and increasing demand for high-barrier primary packaging. Polyethylene and polypropylene remain central to bottles, closures, blister laminates, and flexible overwraps due to chemical resistance and sterilization compatibility. Growth is reinforced by injectable and liquid dosage expansion, while resin price volatility and tightening extractables and leachable regulations continue to moderate material substitution flexibility.
“By providing high-quality and differentiated polyolefin solutions, we are enabling our partners to enhance product durability, improve recyclability, and reduce environmental impact. Borouge materials are engineered to meet the evolving needs of advanced packaging, ensuring efficiency, safety, and long-term performance. Through these partnerships, Borouge is not only strengthening supply chains but also shaping the future of sustainable manufacturing by delivering cutting-edge solutions that create real value for our customers and the industries they serve.” Said by Hazeem Sultan Al Suwaidi, CEO – Borouge.
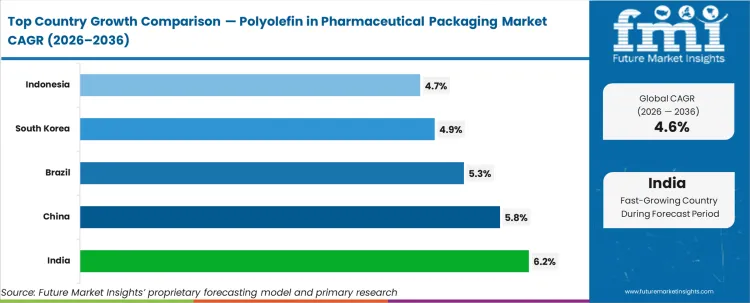
India (6.2%) is projected to register the fastest growth, driven by strong generic drug manufacturing and export capacity. China (5.8%) follows with expanding API and finished dosage production. Brazil (5.3%) benefits from rising domestic pharmaceutical consumption. South Korea (4.9%) and Indonesia (4.7%) show steady expansion supported by regional healthcare investment, while mature Western markets remain compliance-driven and replacement-oriented rather than volume-accelerated.
The polyolefin in pharmaceutical packaging market refers to the use of polyethylene, polypropylene, and related olefin-based polymers in the production of pharmaceutical packaging components. These materials are used in rigid formats such as bottles, caps, vials, syringe parts, and droppers, as well as in flexible formats such as blister packs, strip packs, pouches, and laminates. Polyolefins are selected for their chemical resistance, moisture barrier properties, lightweight structure, and compatibility with sterilization and regulatory requirements. Their primary function is to protect drug formulations from contamination, degradation, and environmental exposure. Key end users include branded pharmaceutical manufacturers, generic drug producers, biotech companies, and contract manufacturing organizations.
The report provides global and regional market size estimates for 2025 and 2026, with forecasts through 2036 and CAGR analysis. It includes segmentation by packaging format, polyolefin type, packaging function, drug type served, end user, and region. The study covers value-based sizing, regulatory impact assessment, material substitution trends, sterilization compatibility analysis, and demand patterns across pharmaceutical production categories.
The scope excludes non-polyolefin materials such as PVC, PET, glass, aluminum, and engineering plastics unless incorporated within polyolefin-based structures. It does not include finished pharmaceutical drugs or active pharmaceutical ingredients. Machinery used in pharmaceutical packaging operations is also excluded, as the analysis focuses strictly on polyolefin materials and components used in packaging systems.
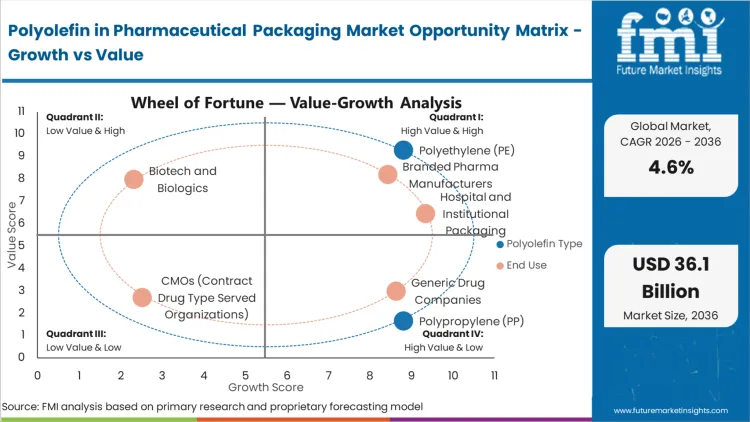
Based on FMI's Polyolefin in Pharmaceutical Packaging market report, consumption of Polyethylene (PE) is estimated to hold 50.4% share in 2026. PE dominates due to its strong chemical resistance, moisture barrier properties, and compatibility with blow molding and film extrusion processes used in pharmaceutical bottles, caps, and flexible packaging. It addresses manufacturer priorities including product stability, low extractables, lightweighting, and compliance with pharmacopeial standards. PE is widely used for solid oral dose bottles, ophthalmic containers, and liquid medication packaging.
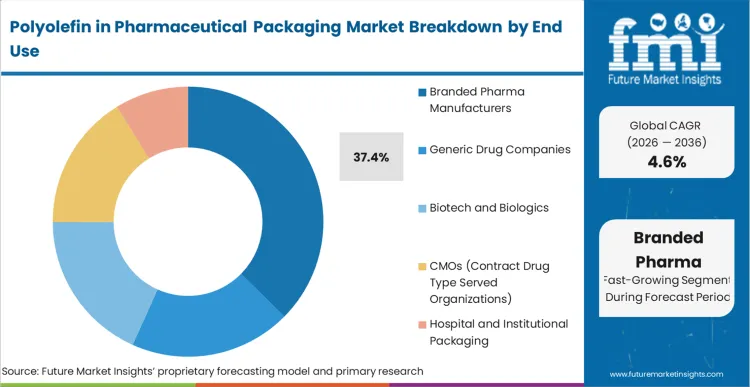
Based on FMI's Polyolefin in Pharmaceutical Packaging market report, consumption by Branded pharma manufacturers is estimated to hold 37.4% share in 2026. Branded pharmaceutical companies lead due to stringent packaging performance requirements, global regulatory obligations, and premium product positioning. Polyolefins address their need for consistent quality, barrier integrity, tamper evidence, and compatibility with serialization and track-and-trace systems.
Future Market Insights analysis indicates that the polyolefin in pharmaceutical packaging market is structurally driven by the global expansion of drug manufacturing and the increasing preference for lightweight, chemically inert packaging materials. Polyolefins such as polyethylene (PE) and polypropylene (PP) are widely used in bottles, closures, blister components, IV containers, and flexible films due to their moisture resistance, processability, and compatibility with sterilization methods. The market exists at its current scale because pharmaceutical supply chains require high-volume, cost-efficient packaging that meets strict safety and stability standards while supporting global distribution.
FMI analysts observe a transition from standard commodity-grade resins toward pharmaceutical-grade, high-purity polyolefins with enhanced barrier and extractables performance. While conventional HDPE bottles continue to dominate solid oral dose packaging, growth is increasingly concentrated in clarified polypropylene, co-extruded films, and advanced polyolefin-based blister systems designed to improve moisture protection and reduce leachables. These specialized materials command higher pricing due to documentation, validation, and regulatory compliance requirements. However, improved drug stability, extended shelf life, and reduced risk of contamination justify the premium, resulting in value-driven growth despite moderate volume expansion in mature pharmaceutical markets.
.webp)
The global polyolefin in pharmaceutical packaging market is segmented across Asia Pacific and Latin America, with strong growth pockets in South and Southeast Asia. Regional dynamics are shaped by pharmaceutical manufacturing expansion, regulatory compliance standards, sterile packaging requirements, and export-driven drug production. The full report provides comprehensive regional market attractiveness analysis.
| Country | CAGR (%) |
|---|---|
| India | 6.2% |
| China | 5.8% |
| Brazil | 5.3% |
| South Korea | 4.9% |
| Indonesia | 4.7% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia Pacific serves as the pharmaceutical manufacturing expansion hub, supported by generic drug production growth and export-oriented formulation capacity. Companies such as Uflex Limited, Huhtamaki PPL Limited, and Gerresheimer AG maintain strong presence in pharmaceutical-grade polyolefin packaging components. Competitive positioning is shaped by regulatory compliance upgrades, sterile packaging requirements, and expanding domestic drug production.
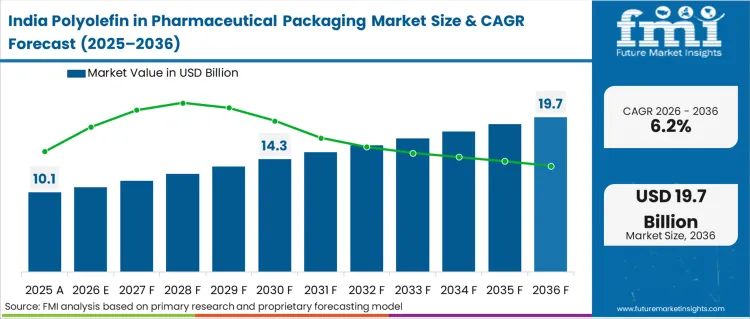
Demand for polyolefin in pharmaceutical packaging in India is projected to rise at 6.2% CAGR through 2036. Growth is closely tied to India’s expanding generic drug manufacturing base. In 2024, the Central Drugs Standard Control Organization continued strengthening Good Manufacturing Practice compliance for pharmaceutical packaging materials, reinforcing requirements for pharmaceutical-grade polypropylene and polyethylene. The Production Linked Incentive scheme for pharmaceuticals remains active, supporting expansion of bulk drug parks and formulation facilities.
In 2024, multiple state governments advanced development of pharmaceutical clusters, increasing demand for blister packaging films, bottles, and caps made from medical-grade polyolefins. Rising exports to regulated markets such as the US and EU further require high-purity and compliant packaging materials. While pricing pressure remains in generics, sustained manufacturing expansion supports steady polyolefin demand.
Demand for polyolefin in pharmaceutical packaging in China is projected to rise at 5.8% CAGR through 2036. The market benefits from rapid pharmaceutical production growth and regulatory strengthening. In 2024, the National Medical Products Administration continued enforcement of revised drug administration regulations, reinforcing packaging traceability and material compliance standards. China’s push for higher-quality domestic drug production under industrial modernization initiatives is increasing adoption of certified pharmaceutical-grade polymers. Expansion in injectable drug manufacturing and medical consumables supports demand for polyethylene bottles and polypropylene closures. Rising exports of finished formulations further strengthen the need for validated and traceable packaging materials.
FMI's analysis of polyolefin in pharmaceutical packaging market in Asia Pacific consists of country-wise assessment that includes India and China. Readers can find insights on GMP compliance impact, export-driven manufacturing, and sterile packaging demand.
Latin America represents a growing pharmaceutical packaging market driven by public healthcare procurement and domestic manufacturing expansion. Companies such as Essel Propack Limited and Constantia Flexibles Group GmbH maintain supply linkages serving regional pharmaceutical converters.
Demand for polyolefin in pharmaceutical packaging in Brazil is projected to rise at 5.3% CAGR through 2036. Growth is influenced by regulatory oversight from ANVISA, which continues to enforce Good Manufacturing Practice standards for pharmaceutical packaging materials. In 2024, public healthcare procurement under Brazil’s Unified Health System supported domestic production of essential medicines, increasing demand for compliant polymer packaging. Investments in local pharmaceutical manufacturing facilities have expanded demand for polyethylene bottles and polypropylene caps. While economic fluctuations may influence capital expenditure, public sector healthcare procurement sustains steady packaging consumption.
FMI's analysis of polyolefin in pharmaceutical packaging market in Latin America consists of country-wise assessment that includes Brazil. Readers can find insights on regulatory enforcement, public procurement impact, and domestic drug production growth.
East Asia remains a technology-driven pharmaceutical packaging market supported by advanced formulation manufacturing and export competitiveness.
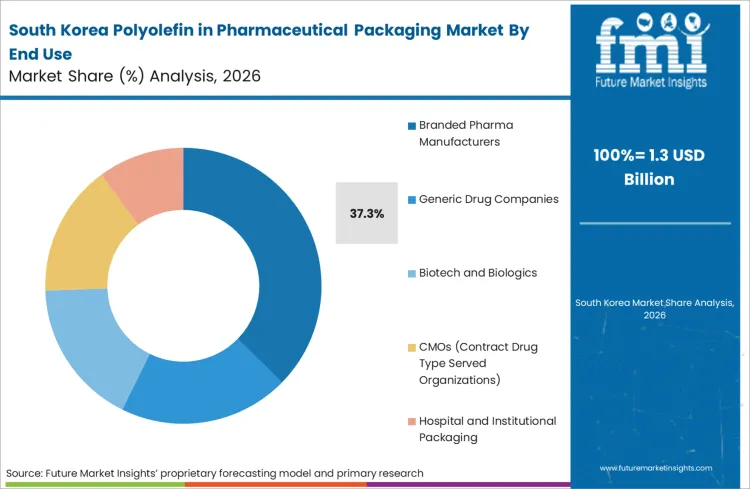
Demand for polyolefin in pharmaceutical packaging in South Korea is projected to rise at 4.9% CAGR through 2036. The country’s strong biopharmaceutical and generic drug export sector drives packaging material demand. In 2024, the Ministry of Food and Drug Safety continued enforcement of strict pharmaceutical manufacturing compliance standards, reinforcing traceability and packaging material validation. Government support for biohealth industry development has encouraged expansion of injectable and specialty drug production, increasing reliance on high-purity polymer packaging components. Sustainability considerations are also encouraging adoption of recyclable secondary packaging formats.
FMI's analysis of polyolefin in pharmaceutical packaging market in East Asia consists of country-wise assessment that includes South Korea. Readers can find insights on biohealth industry growth and compliance-driven packaging demand.
Southeast Asia functions as an emerging pharmaceutical production and packaging market.
Demand for polyolefin in pharmaceutical packaging in Indonesia is projected to rise at 4.7% CAGR through 2036. Growth is supported by government efforts to expand domestic pharmaceutical production capacity. In 2024, Indonesia continued strengthening implementation of pharmaceutical manufacturing standards under its national drug regulatory framework, reinforcing compliance for packaging materials. Public health insurance expansion and increased local drug production further stimulate demand for polyethylene bottles and polypropylene closures. Investment in local pharmaceutical manufacturing facilities is gradually reducing reliance on imports and supporting steady packaging material growth.
FMI's analysis of polyolefin in pharmaceutical packaging market in Southeast Asia consists of country-wise assessment that includes Indonesia. Readers can find insights on domestic production expansion, regulatory modernization, and healthcare demand trends.
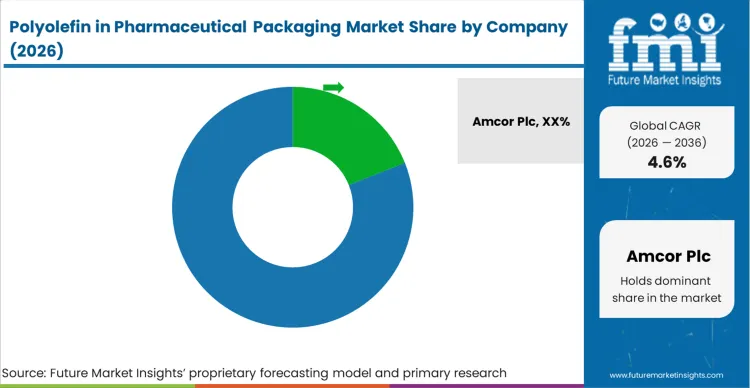
The polyolefin in pharmaceutical packaging market is moderately concentrated, led by global packaging and healthcare material specialists with established regulatory compliance frameworks. Companies such as Amcor Plc, Gerresheimer AG, AptarGroup, West Pharmaceutical Services, Schott AG, SGD Pharma, Winpak Ltd., Constantia Flexibles, and SÜDPACK hold significant positions due to their validated pharma-grade production capabilities and long-standing relationships with pharmaceutical manufacturers. Regional and niche firms including Kaufman Container, Origin Pharma Packaging, Sonic Packaging Industries, Nosco, Essel Propack, Uflex, Bilcare, Huhtamaki PPL, and CCL Healthcare compete through customization, shorter lead times, and cost efficiency. The primary competitive variables in this market are regulatory compliance (USP, EP, FDA standards), material purity, sterilization compatibility, and long-term supply reliability rather than price alone.
Structural advantages are strongest among companies with vertically integrated film extrusion, blow molding, injection molding, and cleanroom manufacturing capabilities. Firms such as Amcor, Gerresheimer, West Pharmaceutical Services, and AptarGroup benefit from dedicated healthcare divisions with controlled medical-grade polyolefin product lines and strict change management protocols. These companies maintain validated resin formulations, global GMP-certified facilities, and comprehensive documentation systems that create high entry barriers. Integration across closures, primary containers, and flexible pharma films further enhances competitive positioning, particularly for injectable, oral solid dose, and sterile packaging applications.
Buyer leverage in this market is moderated by regulatory qualification requirements. Pharmaceutical manufacturers must validate packaging materials as part of drug master files (DMFs) and regulatory submissions, making supplier switching complex and costly. While large pharmaceutical companies may dual-source to reduce risk, requalification timelines limit flexibility. As a result, approved suppliers retain relatively stable pricing power, especially during periods of feedstock volatility. However, procurement teams still negotiate long-term contracts and sustainability commitments, particularly for recyclable and mono-material polyolefin packaging solutions.
Recent Developments

| Metric | Value |
|---|---|
| Quantitative Units | USD 21,839.3 million (2025); USD 23,016.8 million (2026) to USD 36,001.5 million (2036), at a CAGR of 4.6% |
| Market Definition | The polyolefin in pharmaceutical packaging market comprises the global production and utilization of polyethylene-, polypropylene-, and specialty olefin-based materials engineered for pharmaceutical packaging applications, ensuring chemical resistance, moisture barrier protection, regulatory compliance, and compatibility with sterile and non-sterile drug formulations. |
| Form Segmentation | Rigid Packaging; Flexible Packaging |
| End Use Segmentation | Branded Pharmaceutical Manufacturers; Generic Drug Companies; Biotech & Biologics Firms; CMOs (Contract Manufacturing Organizations); Hospital & Institutional Packaging Providers |
| Application Coverage | Solid Oral Dosage Forms; Liquid Oral Dosage Forms; Injectable Formulations; Topical Formulations; Powders & Granules; Nutraceuticals |
| Regions Covered | North America; Latin America; East Asia; South Asia & Pacific; Western Europe; Eastern Europe; Middle East & Africa |
| Countries Covered | United States; Germany; France; United Kingdom; China; India; Japan; Brazil; South Korea; Italy; and 40+ countries |
| Key Companies Profiled | Amcor Plc; Gerresheimer AG; AptarGroup, Inc.; West Pharmaceutical Services, Inc.; Constantia Flexibles Group GmbH; Others |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up market modeling validated through primary interviews with pharmaceutical packaging manufacturers, polymer producers, CMOs, and drug manufacturers, supported by regulatory benchmarking (FDA, EMA, USP), plant-level capacity verification, material performance assessment, and trade flow analysis across global pharmaceutical supply chains. |
The Polyolefin in Pharmaceutical Packaging Market is valued at 23,016.8 Mn in 2026
The market was valued at 21,839.3 Million in 2025.
The market is projected to reach 36,001.5 Mn by 2036.
The Polyolefin in Pharmaceutical Packaging Market is expected to grow at a CAGR of 4.6%.
Rigid packaging leads the market with 67.0% share, while flexible packaging accounts for 33.0%.
Polyethylene (PE) dominates with 50.4% share, followed by polypropylene (PP) at 34.9%.
Branded pharma manufacturers account for 37.4% of the market, followed by generic drug companies at 27.3%.
India is projected to grow at the highest CAGR of 6.2%, followed by China at 5.8% and Brazil at 5.3%.
South Korea is projected to grow at a CAGR of 4.9%, while Indonesia is expected to expand at 4.7% during the forecast period.
The market serves solid oral dosage forms, liquid oral dosage forms, injectable formulations, topical formulations, powders and granules, and nutraceuticals.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.