In 2025, the pyoderma gangrenosum therapeutics market was valued at USD 402.3 million. Based on Future Market Insights' analysis, demand for pyoderma gangrenosum therapeutics is estimated to grow to USD 416.4 million in 2026 and USD 567.5 million by 2036. FMI projects a CAGR of 3.5% during the forecast period, with the market expected to grow by approximately 1.41X.
An absolute dollar growth of USD 151.1 million between 2026 and 2036 reflects therapeutic intensification in rare disease management rather than volume-only patient expansion. Growth depends on increased conversion from systemic corticosteroid monotherapy to biologic combination regimens that deliver higher complete response rates and reduced recurrence. As per FMI, pharmaceutical developers are capturing value not from incremental patient starts alone but from clinical complexity embedded in TNF-α inhibitor protocols for refractory cases and IL-pathway biologics addressing corticosteroid-dependent populations.
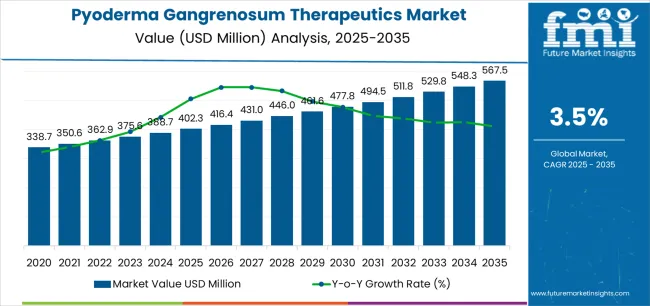
This performance reflects accelerating demand for targeted immunosuppression across hospital and specialty clinic settings. North America leads global expansion supported by reference center concentration and biologic adoption rates. Europe follows driven by specialty dermatology networks and registry participation. Asia-Pacific maintains steady growth through diagnostic awareness improvement and healthcare infrastructure development.
The pyoderma gangrenosum therapeutics market encompasses the global supply of pharmaceutical agents used to treat pyoderma gangrenosum, a rare neutrophilic dermatosis characterized by painful, rapidly progressing ulcerations. Therapeutic approaches include systemic corticosteroids for acute control, calcineurin inhibitors for steroid-sparing maintenance, TNF-α inhibitors for refractory disease, IL-pathway biologics targeting specific inflammatory cascades, JAK inhibitors for pathway-specific suppression, and adjunctive therapies supporting wound healing and infection prevention.
The report includes global and regional market size estimates in value terms, with a forecast period from 2026 to 2036. It provides segmentation by Therapy Class, Route of Administration, End User, and Region. The analysis incorporates treatment paradigm evolution, clinical trial developments, regulatory designation pathways, and competitive positioning across therapeutic categories. Regional demand dynamics, country-level growth rates, and reimbursement landscape assessments are also evaluated as per FMI methodology.
The scope excludes over-the-counter wound care products not specifically indicated for pyoderma gangrenosum, investigational compounds without regulatory approval, and revenues from surgical interventions including debridement and skin grafting. It also excludes diagnostic services and focuses strictly on the therapeutic product layer.
Future Market Insights analysis indicates the market has historically existed as a small, off-label driven category within the broader dermatology therapeutics landscape. Its baseline scale is anchored in diagnosed prevalence estimates of 3 to 10 cases per million annually, with treatment patterns varying significantly across regions and clinical settings [1]. As per FMI, the market's valuation is increasingly influenced by biologic penetration into refractory segments and the emergence of dedicated clinical development programs.
FMI analysts observe a clear stratification between first-line systemic corticosteroids optimized for acute control and targeted biologics engineered for refractory disease management. Standard therapy faces pressure where corticosteroid resistance develops, while growth concentrates in agents demonstrating sustained complete response rates through pathway-specific suppression.
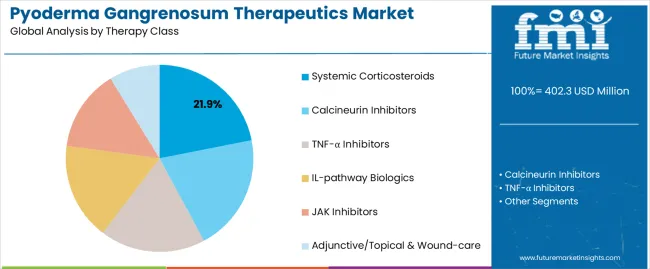
Based on FMI's pyoderma gangrenosum therapeutics market report, demand for systemic corticosteroids is estimated to lead the therapy class segment, capturing 21.9% of the market share in 2025. Systemic corticosteroids including prednisone and methylprednisolone represent first-line therapy for acute disease control due to rapid onset and broad immunosuppressive effects. Their leadership reflects established clinical guidelines and decades of clinical experience supporting empiric use at diagnosis. Growth is sustained by consistent utilization in initial management while newer agents address corticosteroid-dependent and refractory populations.
TNF-α inhibitors represent the leading biologic class, supported by randomized controlled trial data for infliximab in pyoderma gangrenosum. Adalimumab demonstrates efficacy in open-label series and case reports, driving off-label utilization in refractory disease. IL-pathway biologics including ustekinumab show promise in case series, with development programs expanding evidence base.
Calcineurin inhibitors including cyclosporine provide steroid-sparing alternatives for maintenance therapy, particularly in patients requiring long-term immunosuppression. JAK inhibitors represent emerging candidates with pathway-specific mechanisms potentially offering rapid symptom control.
Oral administration dominates systemic therapy delivery, with oral corticosteroids representing first-line treatment and oral calcineurin inhibitors supporting maintenance regimens. Patient preference for oral over injectable administration drives utilization where comparable efficacy exists. Intravenous infusion is required for infliximab and certain biologic induction regimens, typically administered in hospital outpatient settings or infusion centers.
Subcutaneous administration supports adalimumab and emerging biologics, enabling self-administration after initial training and reducing healthcare resource utilization. Topical administration addresses localized disease or adjunctive wound care, with topical corticosteroids and topical calcineurin inhibitors applied to ulcer margins.
The pyoderma gangrenosum therapeutics market is segmented geographically across North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa. Growth dynamics differ based on diagnostic awareness, biologic adoption rates, and healthcare infrastructure development.
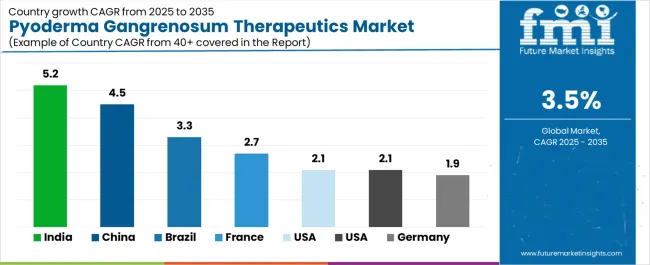
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 4.5% |
| India | 4.2% |
| Germany | 3.2% |
| Japan | 3.0% |
| United States | 2.8% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America represents the largest value market for pyoderma gangrenosum therapeutics, driven by reference center concentration, biologic adoption rates, and reimbursement frameworks supporting specialty pharmacy distribution. The region's healthcare system supports multidisciplinary care through dermatology referral networks and wound care centers.
Demand for pyoderma gangrenosum therapeutics in the United States is projected to rise at 2.8 % CAGR through 2036. The USA market benefits from the National Organization for Rare Disorders advocacy programs increasing disease awareness and supporting patient access to specialized care [12].
Academic medical centers maintain pyoderma gangrenosum registries advancing understanding of treatment outcomes and natural history. The FDA's orphan drug program provides development incentives attracting investment in dedicated clinical programs [6]. Biologic utilization exceeds other regions due to favorable reimbursement and specialty pharmacy distribution networks.
FMI's analysis of the pyoderma gangrenosum therapeutics market in North America consists of country-wise assessment including the United States and Canada. Readers can find orphan drug policy tracking, reimbursement analysis, and competitive intelligence.
Europe represents a mature market characterized by specialty dermatology networks, registry participation, and emerging biologic adoption. The region's healthcare systems support multidisciplinary care through referral networks and academic center concentration.
Demand for pyoderma gangrenosum therapeutics in Germany is projected to rise at 3.2 % CAGR through 2036. Germany's strong dermatology infrastructure and registry participation support evidence generation guiding treatment decisions. University dermatology departments maintain pyoderma gangrenosum expertise attracting referrals from community settings. The country's reimbursement framework supports biologic utilization through hospital outpatient departments and specialty clinics. German participation in European reference networks advances rare disease care coordination.
FMI's analysis of the pyoderma gangrenosum therapeutics market in Europe consists of country-wise assessment including Germany, United Kingdom, France, Italy, and Spain. Readers can find registry participation tracking, treatment guideline analysis, and competitive landscape insights.
Asia-Pacific represents the fastest-growing regional market for pyoderma gangrenosum therapeutics, driven by diagnostic awareness improvement, healthcare infrastructure development, and increasing biologic access.
Demand for pyoderma gangrenosum therapeutics in China is projected to rise at 4.5 % CAGR through 2036. China's healthcare reform initiatives expanding specialty care access create opportunities for rare disease management programs. The country's growing dermatology workforce improves diagnostic recognition of ulcerative conditions.
Biologic access expands through national reimbursement drug lists and specialty pharmacy development. Chinese participation in global clinical trials supports local investigator expertise and treatment familiarity.
FMI's analysis of the pyoderma gangrenosum therapeutics market in Asia-Pacific consists of country-wise assessment including China, India, Japan, and South Korea. Readers can find healthcare reform tracking, diagnostic awareness analysis, and market access intelligence.
Demand for pyoderma gangrenosum therapeutics in India is projected to rise at 4.2% CAGR through 2036. India's dermatology training programs emphasize rare disease recognition, improving diagnostic rates across academic and community settings.
The country's pharmaceutical manufacturing capabilities support generic immunosuppressant availability at accessible price points. Biologic access expands through patient assistance programs and specialty pharmacy development. Indian participation in international dermatology societies advances treatment protocol adoption.
Demand for pyoderma gangrenosum therapeutics in Japan is projected to rise at 3.0% CAGR through 2036. Japan's universal health insurance system provides broad access to systemic therapies and emerging biologics. The country's aging population increases prevalence of inflammatory conditions including pyoderma gangrenosum.
Japanese dermatology departments maintain expertise in neutrophilic dermatoses through academic center concentration. Regulatory pathways support rapid biologic approval following international trial completion.
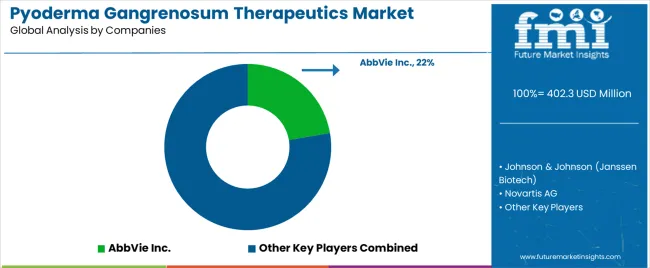
Established pharmaceutical companies with broad immunology portfolios and specialized developers pursuing orphan indications characterize the pyoderma gangrenosum therapeutics market. Leading firms are differentiating through randomized controlled trial investment supporting labeled indications and guideline inclusion.
AbbVie leverages adalimumab's extensive immunology experience and real-world evidence supporting pyoderma gangrenosum utilization. The company's specialty pharmacy network supports patient access and adherence programs. Johnson & Johnson maintains infliximab's position as the only TNF-α inhibitor with randomized controlled trial data in pyoderma gangrenosum, supporting utilization in refractory cases. Novartis pursues IL-pathway biologics with potential applications in neutrophilic dermatoses through ongoing clinical development. Pfizer maintains broad immunology portfolio with opportunities for life cycle management into rare dermatology indications. Boehringer Ingelheim advances pipeline candidates targeting inflammatory pathways relevant to pyoderma gangrenosum. InflaRx N.V. develops specific complement inhibitors with potential application in neutrophil-mediated conditions.
Buyer behavior in this market reflects specialty pharmacy distribution and payer management of biologic utilization. Hospital formularies maintain systemic corticosteroids and calcineurin inhibitors as standard options while managing biologic access through prior authorization protocols.
Recent Developments:
| Metric | Value |
|---|---|
| Quantitative Units | USD 416.4 million (2026) to USD 567.5 million (2036), at a CAGR of 3.5% |
| Market Definition | The pyoderma gangrenosum therapeutics market comprises pharmaceutical agents for treating this rare neutrophilic dermatosis across acute and maintenance settings. |
| Therapy Class Segmentation | Systemic Corticosteroids, Calcineurin Inhibitors, TNF-α Inhibitors, IL-Pathway Biologics, JAK Inhibitors, Adjunctive/Topical & Wound-Care |
| Route of Administration Segmentation | Oral, IV Infusion, Subcutaneous, Topical |
| End User Segmentation | Hospitals, Specialty Clinics, Ambulatory Surgical Centers, Others |
| Region Segmentation | North America, Latin America, Europe, Asia-Pacific, Middle East & Africa |
| Key Countries Covered | United States, Canada, Germany, United Kingdom, France, Italy, Spain, China, India, Japan, South Korea, and 40+ countries |
| Key Companies Profiled | AbbVie Inc., Johnson & Johnson, Novartis AG, Pfizer Inc., Boehringer Ingelheim, InflaRx N.V., UCB S.A., Amgen Inc., Eli Lilly and Company, Bristol Myers Squibb |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up market modeling validated through primary interviews with dermatologists and specialty pharmacy executives, supported by rare disease prevalence analysis and treatment algorithm tracking |
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
Demand for pyoderma gangrenosum therapeutics is estimated to be valued at USD 416.4 million in 2026.
Market size for pyoderma gangrenosum therapeutics is projected to reach USD 567.5 million by 2036.
Demand for pyoderma gangrenosum therapeutics is expected to grow at a 3.5 % CAGR between 2026 and 2036.
Systemic corticosteroids are estimated to lead the therapy class segment, holding 21.9 % share in 2025, reflecting first-line positioning for acute disease control.
Hospitals represent the dominant end-user segment, driven by inpatient management of severe ulcerations and multidisciplinary care coordination requirements.
Reference center concentration, biologic adoption rates, and FDA orphan drug incentives supporting development investment are driving North American demand.
The United States is projected to expand at a 2.8 % CAGR during 2026 to 2036.
Specialty dermatology networks, registry participation, and emerging biologic adoption are supporting European market growth.
Germany is projected to expand at a 3.2 % CAGR during 2026 to 2036, supported by dermatology infrastructure and registry participation.
Yes, China is included within Asia-Pacific and is a high-growth country market covered in the report.
China is projected to expand at a 4.5 % CAGR during 2026 to 2036, supported by healthcare reform and biologic access expansion.
Yes, India is included within Asia-Pacific and is covered in the country-wise assessment.
India is projected to expand at a 4.2 % CAGR during 2026 to 2036, supported by diagnostic awareness improvement and generic immunosuppressant availability.
Yes, Japan is included within Asia-Pacific under the regional scope of analysis.
Japan is projected to expand at a 3.0 % CAGR during 2026 to 2036, supported by universal health insurance coverage and academic dermatology expertise.
Diagnostic awareness improvement, healthcare infrastructure development, and increasing biologic access are supporting Asia-Pacific demand growth.
A therapeutic is included if it is used for pyoderma gangrenosum treatment across acute control, maintenance, or refractory management, whether on-label or off-label with established clinical evidence.
Scope includes market sizing and forecasting for 2026 to 2036, segmentation by therapy class, route of administration, end user, and region, along with competitive assessment and country-wise outlook.
The scope excludes over-the-counter wound care products, investigational compounds without regulatory approval, surgical interventions, and diagnostic services.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.