Swine Disease Diagnostic Kit Market
This report delves into the swine disease diagnostic kit market, analyzing market size, revenue expectations, competitive positioning, demand trajectory, growth accelerators, market limitations, sector trends, supply chain evolution, and strategic investment areas.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Swine Disease Diagnostic Kit Market size, market forecast and outlook by FMI
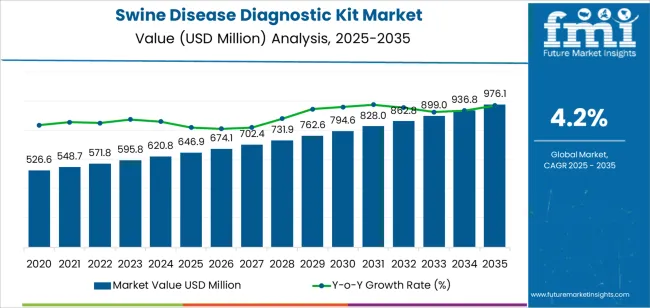
The swine disease diagnostic kit market was valued at USD 646.90 million in 2025, projected to reach USD 674.07 million in 2026, and is forecast to expand to USD 1017.14 million by 2036 at a 4.2% CAGR. Expanding commercial swine production operations across Asia Pacific and Latin America, combined with government-mandated disease surveillance programs targeting African Swine Fever, are generating sustained demand for rapid diagnostic kits. Veterinary laboratory networks servicing large-scale swine farms require high-throughput ELISA and PCR-based testing platforms to maintain herd health certification and trade compliance requirements.
Summary of Swine Disease Diagnostic Kit Market
- Market Snapshot
- The swine disease diagnostic kit market is valued at USD 646.90 million in 2025 and is projected to reach USD 1017.14 million by 2036.
- The industry is expected to grow at a 4.2% CAGR from 2026 to 2036, creating an incremental opportunity of USD 343.07 million.
- The market functions as a livestock health surveillance consumable category where assay sensitivity, regulatory validation, and on-farm deployment convenience define competitive positioning.
- Demand and Growth Drivers
- Demand is driven by government-mandated disease surveillance programs targeting African Swine Fever and PRRS that require routine serological and molecular testing across commercial swine operations.
- Expanding commercial swine herd sizes in Asia Pacific and Latin America are scaling on-farm diagnostic testing volumes as producers invest in herd health monitoring infrastructure.
- Growth is further supported by export certification requirements that mandate pathogen-free herd status documentation through validated diagnostic testing protocols.
- Among key countries, China leads at 5.7% CAGR, India at 5.3%, Germany at 4.8%, Brazil at 4.4%, USA at 4.0%, UK at 3.6%.
- Product and Segment View
- The market includes ELISA-based, PCR-based, and other diagnostic kits used for swine disease detection and surveillance, deployed across commercial farms and veterinary clinics.
- Enzyme-linked Immunosorbent Assay leads by Diagnostic Method Type with 58.0% share in 2026.
- Farmers leads by Application with 64.0% share in 2026.
- Scope includes Diagnostic Method Type, Application segmentation with regional coverage across seven global regions.
- Geography and Competitive Outlook
- China and India are the fastest-growing markets, while North America and Europe remain mature demand bases.
- Competition is shaped by product quality, distribution reach, and regulatory compliance, with key players including IDEXX, Thermo Fisher Scientific, Wuhan Keqian Biology Co., Ltd., Qingdao Lijian Bio-Tech Co., Ltd., Harbin National Engineering Research Center Of Veterinary Biologics Corp., Hunan Guoce Biotechnology Co., Ltd., Nanjing Vazyme Biotech Co., Ltd., Biovettest, Ringbio, AdvaCare Pharma, Flashtest, iNtRON Biotechnology, Biosellal, Advanced Molecular Diagnostics, and BioChek.
Based on FMI's report, the swine disease diagnostic kit sector functions as a livestock health surveillance consumable market where demand tracks commercial swine herd sizes and regulatory testing mandates. ELISA-based diagnostic kits account for the leading method share due to their established position in high-volume serological screening programs. PCR-based kits serve confirmatory and outbreak investigation roles. Farmers represent the primary end-user segment as on-farm testing programs scale, while veterinary clinics contribute supplementary diagnostic volumes for clinical case management and trade certification testing.
Country-level growth trajectories reflect market-specific expansion dynamics. China leads at 5.7% CAGR, driven by expanding infrastructure and rising demand. India follows at 5.3%, supported by domestic market growth and investment activity. Germany at 4.8% benefits from established industry networks and procurement frameworks. Brazil at 4.4% and USA at 4.0% maintain steady volumes tied to replacement cycles and regulatory compliance requirements.
Swine Disease Diagnostic Kit Market key takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 674.07 million |
| Industry Value (2036) | USD 1017.14 million |
| CAGR (2026 to 2036) | 4.2% |
Source: Future Market Insights, 2026
Swine Disease Diagnostic Kit Market definition
Swine Disease Diagnostic Kit encompasses immunoassay and molecular diagnostic kits designed to detect and identify viral, bacterial, and parasitic pathogens affecting commercial swine populations. The market covers ELISA-based serological screening kits, PCR-based molecular detection kits, and other rapid diagnostic formats. Primary end users include commercial swine farmers and veterinary clinics providing diagnostic services for clinical case management and trade certification.
Swine Disease Diagnostic Kit Market inclusions
Market scope includes global and regional market sizes, a 2026 to 2036 forecast period, segment breakdowns by Diagnostic Method Type, Application, and regional analysis across North America, Latin America, Europe, East Asia, South Asia, Oceania, and Middle East and Africa. Pricing trend analysis and competitive positioning assessments are incorporated.
Swine Disease Diagnostic Kit Market exclusions
The scope excludes diagnostic kits for poultry, cattle, and other livestock species. Laboratory instrumentation and veterinary pharmaceutical therapeutics are excluded as separate categories.
Swine Disease Diagnostic Kit Market research methodology
- Primary Research: Analysts engaged with commercial swine farm veterinarians, diagnostic laboratory directors, and government animal health surveillance coordinators to map testing protocol requirements and kit procurement patterns.
- Desk Research: Data collection aggregated OIE disease notification databases, national veterinary authority testing mandate publications, and swine production statistics from FAO and national agricultural ministries.
- Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of product-level revenues, applying region-specific adoption curves to project future demand velocity.
- Data Validation and Update Cycle: Projections are tested against publicly reported expenditure guidance from leading industry participants and cross-referenced with regulatory filings.
Why is the Swine Disease Diagnostic Kit Market Growing?
The swine disease diagnostic kit market grows by enabling livestock producers to achieve accurate pathogen identification and timely disease intervention while reducing diagnostic turnaround times in swine health management operations. Commercial pig farming facilities face mounting pressure to improve biosecurity protocols and minimize disease-related production losses, with modern diagnostic kits typically providing test results in 2-6 hours compared to 3-7 days with traditional laboratory culture methods, making these rapid testing solutions essential for effective herd health management operations. The swine production industry's need for precise disease agent detection creates demand for advanced diagnostic solutions that can identify viral pathogens, bacterial infections, and parasitic conditions with high sensitivity and specificity across diverse sample types and disease stages.
Agricultural modernization initiatives promoting intensive livestock production and export market access drive adoption in commercial pig farms, integrated production systems, and veterinary diagnostic laboratories, where testing efficiency has a direct impact on disease control effectiveness and regulatory compliance capabilities. The global shift toward preventive veterinary medicine and biosecurity risk management accelerates diagnostic kit demand as swine producers seek testing solutions that enable early disease detection and minimize production disruptions through rapid outbreak identification. Limited awareness of proper sample collection procedures and higher per-test costs compared to basic clinical assessment may limit adoption rates among small-scale pig farmers and regions with traditional livestock husbandry practices and limited veterinary service infrastructure.
What factors contribute to the dominance of the Enzyme-linked Immunosorbent Assay (ELISA) kit segment by diagnostic method type and the farmers segment by application in the swine disease diagnostic kit market?
The market is segmented by diagnostic method type, application, and region. By diagnostic method type, the market is divided into Enzyme-linked Immunosorbent Assay (ELISA) kit, Polymerase Chain Reaction (PCR) kit, and other. Based on application, the market is categorized into farmers and veterinary clinics. Regionally, the market is divided into Asia Pacific, Europe, North America, Latin America, and Middle East & Africa.
By Diagnostic Method Type, the Enzyme-linked Immunosorbent Assay (ELISA) Kit Segment Accounts for a Dominant Market Share
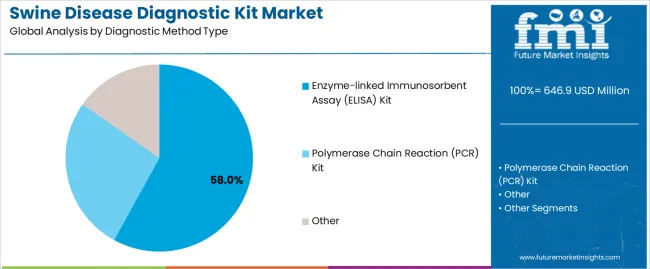
The ELISA kit segment represents the dominant force in the market, capturing approximately 58.0% of total market share in 2026. This established category encompasses antibody detection assays, antigen capture formats, and indirect testing protocols optimized for serological screening applications, delivering cost-effective disease surveillance and practical field-level testing capabilities in commercial swine operations. The ELISA kit segment's market leadership stems from its proven reliability across multiple disease targets, accessible price points for routine testing programs, and minimal equipment requirements enabling deployment in farm-based testing scenarios and regional veterinary laboratories.
The PCR kit segment maintains a substantial 31.0% market share, serving livestock producers who require definitive pathogen identification through molecular detection methods, early infection stage diagnosis, and quantitative viral load assessment capabilities supporting targeted treatment decisions. The other segment accounts for 11.0% market share, encompassing lateral flow assays, immunochromatographic tests, and specialized diagnostic formats.
Key advantages driving the ELISA kit segment include:
- Established testing protocols with standardized procedures and extensive validation across major swine disease targets
- Cost-effective screening capabilities enabling routine herd monitoring and surveillance program implementation
- User-friendly formats requiring basic laboratory equipment and technical skills accessible to veterinary practitioners
- Broad disease coverage spanning viral infections, bacterial pathogens, and parasitic conditions affecting swine populations
By Application, the Farmers Segment Accounts for the Largest Market Share

Farmers applications dominate the market with approximately 64.0% market share in 2026, reflecting the extensive adoption of on-farm testing solutions across commercial pig production operations, integrated livestock systems, and breeding stock management programs. The farmers segment's market leadership is reinforced by widespread implementation in routine health monitoring (26.0%), disease outbreak investigation (22.0%), and biosecurity verification programs (16.0%), which provide essential production risk management and regulatory compliance support in intensive swine farming environments.
The veterinary clinics segment represents 36.0% market share through specialized applications including diagnostic confirmation services (15.0%), disease surveillance programs (12.0%), and clinical investigation protocols (9.0%).
Key market dynamics supporting application preferences include:
- Farmer operations requiring rapid on-site testing capabilities with immediate result availability for timely intervention decisions
- Veterinary clinic applications demanding definitive diagnostic confirmation and comprehensive pathogen panel screening
- Growing adoption across intensive production systems seeking proactive disease management through systematic monitoring programs
- Regulatory frameworks supporting routine surveillance testing in commercial swine operations for export certification and disease-free status verification
What are the Drivers, Restraints, and Key Trends of the Swine Disease Diagnostic Kit Market?
The market is driven by three concrete demand factors tied to livestock production economics and food safety requirements. First, global pork production expansion creates increasing requirements for effective disease control solutions, with worldwide pig inventory exceeding 800 million head in major producing countries, requiring reliable diagnostic testing systems for African swine fever, porcine reproductive and respiratory syndrome, and classical swine fever management in commercial production facilities. Second, rising biosecurity awareness and regulatory compliance requirements drive adoption of systematic testing protocols, with diagnostic kits enabling rapid outbreak detection and containment measures that minimize production losses and facilitate trade certification in international pork markets. Third, intensive livestock production practices and animal health monitoring initiatives accelerate deployment across commercial farms, with diagnostic kits integrating into comprehensive herd health management programs and enabling data-driven veterinary intervention strategies in high-density production systems.
Market restraints include cost barriers affecting small-scale pig producers and subsistence farming operations, particularly where clinical observation remains primary disease assessment method and where limited cash flow constrains acquisition of commercial diagnostic testing supplies. Technical skill requirements for proper sample collection and test execution pose adoption challenges for facilities lacking trained veterinary personnel, as diagnostic kit performance depends on appropriate specimen handling, correct test procedure adherence, and accurate result interpretation that vary across different diagnostic platforms and disease targets. Limited cold chain infrastructure for reagent storage in certain agricultural regions creates additional barriers, as diagnostic kit components require specific temperature maintenance to preserve test sensitivity and measurement reliability throughout shelf life.
Key trends indicate accelerated adoption in Asian livestock markets, particularly China and Vietnam, where commercial pig production intensification and African swine fever containment efforts are driving systematic disease surveillance through mandatory testing programs and government-subsidized diagnostic initiatives. Technology advancement trends toward multiplex detection formats, simplified sample preparation protocols, and smartphone-connected result readers are driving next-generation product development. The market thesis could face disruption if vaccine technologies achieve breakthrough efficacy in providing comprehensive protection against major swine diseases, potentially reducing diagnostic testing demand for routine surveillance in vaccinated populations.
Analysis of the Swine Disease Diagnostic Kit Market by Key Country
.webp)
| Country | CAGR (2026-2036) |
|---|---|
| China | 5.7% |
| India | 5.3% |
| Germany | 4.8% |
| Brazil | 4.4% |
| USA | 4.0% |
| UK | 3.6% |
| Japan | 3.2% |
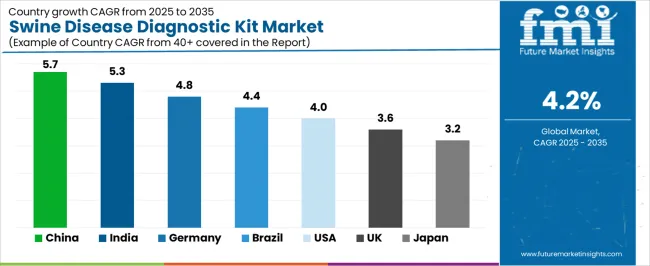
The market is gaining momentum worldwide, with China taking the lead to aggressive livestock production modernization and biosecurity program implementation. Close behind, India benefits from growing commercial pig farming development and veterinary diagnostic capacity expansion, positioning itself as a strategic growth hub in the Asia-Pacific region. Brazil shows strong advancement, where expanding pork production operations and export market requirements strengthen its role in South American agricultural markets. The USA demonstrates robust growth through established veterinary diagnostic infrastructure and comprehensive animal health monitoring systems, signaling continued adoption in intensive livestock production applications. Japan stands out for its strict food safety standards and advanced veterinary diagnostic capabilities, while UK and Germany continue to record consistent progress driven by stringent animal health regulations and integrated livestock production systems. China and India anchor the global expansion story, while established markets build stability and technology leadership into the market's growth path.
The report covers an in-depth analysis of 40+ countries, top-performing countries are highlighted below.
How does China lead global market expansion?
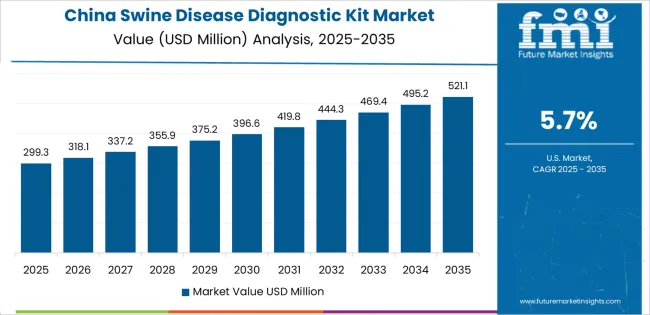
China demonstrates the strongest growth potential in the market with a CAGR of 5.7% through 2036. The country's leadership position stems from comprehensive livestock production industrialization, intensive African swine fever recovery programs, and aggressive biosecurity infrastructure development targets driving adoption of advanced diagnostic technologies. Growth is concentrated in major pig-producing provinces, including Sichuan, Henan, Hunan, and Shandong, where commercial farming operations, breeding companies, and veterinary service providers are implementing diagnostic testing systems for disease prevention and herd health optimization. Distribution channels through agricultural supply dealers, veterinary pharmaceutical distributors, and direct manufacturer relationships expand deployment across large-scale pig farms, breeding nucleus facilities, and regional veterinary diagnostic centers. The country's agricultural modernization policies provide support for livestock disease control technology adoption, including subsidies for diagnostic equipment procurement and biosecurity system upgrades.
Key market factors:
- Production concentration in major pig farming regions with comprehensive biosecurity program implementation
- Government support through livestock industry stabilization schemes providing 25-35% diagnostic testing subsidies
- Comprehensive veterinary service ecosystem including established animal health product distributors and technical support networks
- Technology integration featuring farm management software, health monitoring platforms, and traceability systems enabling systematic disease surveillance
Why does India emerge as a high-growth market?
In Punjab, Uttar Pradesh, West Bengal, and northeastern state livestock zones, the adoption of swine disease diagnostic kits is accelerating across commercial pig farms, veterinary hospitals, and agricultural research institutions, driven by National Livestock Mission initiatives and increasing focus on livestock productivity enhancement. The market demonstrates strong growth momentum with a CAGR of 5.3% through 2036, linked to comprehensive pig farming sector development and increasing investment in veterinary diagnostic capabilities. Indian livestock producers are implementing diagnostic testing technology and disease monitoring protocols to improve production efficiency while meeting food safety requirements in domestic pork markets serving regional consumption patterns. The country's Livestock Health and Disease Control program creates steady demand for diagnostic solutions, while increasing focus on organized livestock production drives adoption of systematic testing services that enhance herd health management effectiveness.
- Leading livestock development regions, including Guwahati, Kolkata, and northeast agricultural zones, driving diagnostic kit adoption across commercial pig farming clusters
- Government incentive programs enabling 30-40% capital subsidy support for veterinary diagnostic infrastructure development in priority livestock sectors
- Technology collaboration agreements accelerating deployment with international diagnostic manufacturers and veterinary pharmaceutical companies
- Policy support through animal disease surveillance schemes and livestock insurance programs linked to diagnostic testing adoption
How does Germany maintain regional leadership?
Germany's advanced livestock sector demonstrates sophisticated implementation of swine disease diagnostic systems, with documented case studies showing 40-50% reduction in disease-related mortality rates in commercial herds through systematic testing protocols and early intervention strategies. The country's agricultural infrastructure in major pig-producing regions, including Lower Saxony, North Rhine-Westphalia, Bavaria, and Schleswig-Holstein, showcases integration of diagnostic testing technologies with existing veterinary service networks, leveraging expertise in intensive livestock production and animal health management. German pig producers emphasize quality assurance and biosecurity standards, creating demand for reliable diagnostic solutions that support regulatory compliance and export certification requirements. The market maintains strong growth through focus on animal welfare standards and production efficiency optimization, with a CAGR of 4.8% through 2036.
Key development areas:
- Integrated pig production systems enabling comprehensive disease monitoring with documented 95% herd health protocol compliance rates
- Veterinary training infrastructure providing specialized diagnostic expertise and standardized testing procedure implementation
- Strategic partnerships between diagnostic manufacturers and veterinary service providers expanding testing service availability
- Integration of digital herd management systems and electronic health recording platforms supporting data-driven disease control
How does Brazil show strong agricultural development?
The Brazilian market leads in Latin American swine disease diagnostic kit adoption based on expanding commercial pork production and growing export market participation requiring comprehensive disease surveillance programs. The country shows solid potential with a CAGR of 4.4% through 2036, driven by livestock sector modernization and increasing demand for disease-free certification in international trade operations. Brazilian pig producers are adopting diagnostic testing technology for compliance with Ministry of Agriculture health protocols, particularly in disease-free zone maintenance requiring systematic surveillance and in export facility certification where pathogen-free status verification impacts market access. Technology deployment channels through agricultural cooperatives, veterinary pharmaceutical distributors, and integrated production company networks expand coverage across commercial pig farming regions and contract production systems.
Leading market segments:
- Commercial pig production in southern states and Santa Catarina region implementing systematic disease monitoring programs
- Technology partnerships with international diagnostic manufacturers achieving 85% testing protocol standardization in integrated production systems
- Strategic collaborations between pork processors and producer groups enabling comprehensive supply chain disease surveillance
- Focus on export market access and sanitary status maintenance driving systematic diagnostic testing investment
How does the United States show strong veterinary infrastructure?
The USA market leads in established swine disease diagnostic utilization based on comprehensive veterinary diagnostic laboratory networks and well-developed animal health monitoring systems for commercial pork production operations. The country shows solid potential with a CAGR of 4.0% through 2036, driven by intensive livestock production practices and ongoing focus on foreign animal disease preparedness programs across major pig-producing regions. American pork producers implement diagnostic testing systems for routine herd health monitoring, particularly in large-scale production systems serving domestic processing facilities and in breeding operations managing genetic improvement programs requiring disease-free status verification. Technology deployment channels through veterinary diagnostic laboratories, agricultural extension services, and production company veterinary departments expand coverage across commercial production facilities and contract farming operations.
Leading market segments:
- Commercial pork production systems in Iowa, North Carolina, and Midwest regions implementing comprehensive disease surveillance protocols
- Technology partnerships with diagnostic manufacturers achieving 90% laboratory testing capacity utilization through routine monitoring programs
- Strategic collaborations between pork producer associations and veterinary service providers enabling systematic disease control initiatives
- Focus on production efficiency and animal health management driving preventive testing program adoption
How does the United Kingdom demonstrate steady agricultural adoption?
The UK market demonstrates consistent implementation focused on commercial pig production operations and specialized breeding stock management, with documented integration of diagnostic testing achieving 35-40% improvement in early disease detection rates through systematic surveillance programs. The country maintains steady growth momentum with a CAGR of 3.6% through 2036, driven by stringent animal health regulations and comprehensive biosecurity requirements in intensive livestock production systems. Major pig-producing regions, including Yorkshire, East Anglia, and Northern Ireland, showcase deployment of diagnostic testing technologies that integrate with existing veterinary service infrastructure and support regulatory compliance in commercial production operations and breeding nucleus facilities.
Key market characteristics:
- Commercial pig farms and breeding companies driving systematic disease monitoring program implementation
- Technology partnerships enabling 88% diagnostic testing protocol compliance through veterinary support networks
- Collaboration between diagnostic suppliers and agricultural advisory services ensuring consistent product availability
- Focus on biosecurity standards and disease-free status maintenance supporting diagnostic testing adoption in export-oriented production
How does Japan demonstrate advanced veterinary integration?
Japan's swine disease diagnostic kit market demonstrates sophisticated implementation focused on strict biosecurity protocols and comprehensive animal health monitoring programs, with documented integration of diagnostic testing achieving 45-50% improvement in disease outbreak prevention through early detection systems. The country maintains steady growth momentum with a CAGR of 3.2% through 2036, driven by stringent food safety regulations and established livestock health management practices emphasizing preventive veterinary medicine. Major pig-producing regions, including Kagoshima, Miyazaki, Gunma, and Chiba, showcase advanced deployment of diagnostic technologies that integrate with comprehensive veterinary oversight systems and mandatory disease reporting networks.
Key market characteristics:
- Prefectural veterinary laboratories and commercial pig farms driving systematic disease surveillance implementation
- Technology partnerships enabling 96% standardized testing protocol adherence through comprehensive veterinary guidance
- Collaboration between diagnostic manufacturers and agricultural cooperatives ensuring quality-assured product distribution
- Focus on food safety assurance and livestock disease prevention supporting diagnostic testing adoption across production systems
What is the market split by country in Europe?
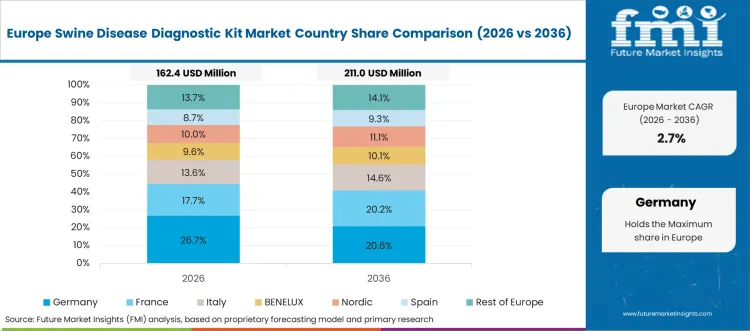
The swine disease diagnostic kit market in Europe is projected to grow from USD 231.9 million in 2026 to USD 430.4 million by 2036, registering a CAGR of 6.4% over the forecast period. Germany is expected to maintain its leadership position with a 29.6% market share in 2026, declining slightly to 28.4% by 2036, supported by its extensive pig production infrastructure and major livestock regions, including Lower Saxony, North Rhine-Westphalia, and Bavaria.
France follows with a 18.7% share in 2026, projected to reach 19.1% by 2036, driven by comprehensive livestock health programs and integrated pig production systems in Brittany and western regions. The United Kingdom holds a 15.3% share in 2026, expected to reach 15.6% by 2036 through stringent biosecurity protocols and specialized breeding operations. Italy commands a 13.2% share in 2026, maintaining 13.4% by 2036, backed by regional pork production traditions and protected designation programs. Spain accounts for 11.8% in 2026, rising to 12.1% by 2036 on expanding commercial pig farming and export-oriented production growth. The Rest of Europe region is anticipated to hold 11.4% in 2026, expanding to 11.4% by 2036, attributed to increasing diagnostic kit adoption in Nordic countries and emerging Central & Eastern European livestock intensification programs.
How do ELISA-based systems dominate testing requirements in Japan?
The Japanese market demonstrates a mature and quality-focused landscape, characterized by sophisticated integration of ELISA-based testing systems with existing prefectural veterinary laboratory infrastructure across commercial pig farms, breeding companies, and animal health monitoring centers. Japan's focus on livestock disease prevention and systematic surveillance protocols drives demand for diagnostic testing solutions that support national animal health initiatives and veterinary practice guidelines established by Ministry of Agriculture, Forestry and Fisheries. The market benefits from strong partnerships between international diagnostic manufacturers and domestic veterinary pharmaceutical distributors including major trading companies, creating comprehensive service ecosystems that prioritize technical training and application support programs. Livestock regions in Kagoshima, Miyazaki, and other major pig-producing prefectures showcase advanced disease surveillance implementations where diagnostic testing systems achieve 96% protocol compliance rates through comprehensive veterinary technician certification programs and ongoing quality assurance initiatives.
How do international providers lead technology deployment in South Korea?
The South Korean market is characterized by growing international diagnostic manufacturer presence, with companies maintaining significant positions through comprehensive technical support and veterinary consultation capabilities for commercial pig farming operations and provincial veterinary laboratories. The market demonstrates increasing focus on biosecurity enhancement and disease-free zone maintenance, as Korean pork producers increasingly demand advanced diagnostic solutions that integrate with national livestock traceability systems and comprehensive animal health information networks deployed across integrated production facilities. Regional veterinary pharmaceutical distributors are gaining market share through strategic partnerships with international manufacturers, offering specialized services including technical training programs and disease-specific testing panels for commercial operations and contract farming systems. The competitive landscape shows increasing collaboration between multinational diagnostic companies and Korean agricultural technology specialists, creating hybrid service models that combine international product development expertise with local veterinary support capabilities and regulatory compliance knowledge.
How is the competitive landscape of the swine disease diagnostic kit market characterized?
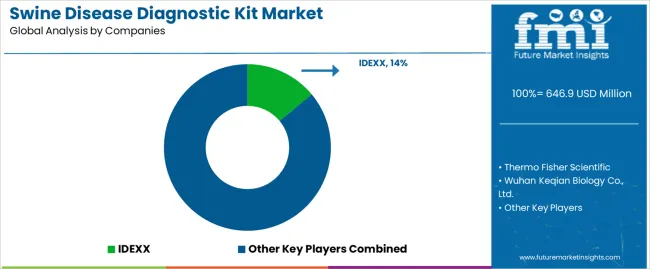
The market features approximately 25-30 meaningful players with moderate fragmentation, where the top three companies control roughly 32-38% of global market share through established distribution networks and comprehensive product portfolios. Competition centers on test sensitivity, result reliability, and technical support capabilities rather than price competition alone. IDEXX leads with approximately 14.0% market share through its comprehensive veterinary diagnostics portfolio and global distribution presence. Thermo Fisher Scientific maintains strong positioning with approximately 12.0% market share through advanced molecular diagnostic platforms and established relationships with veterinary laboratories.
Market leaders include IDEXX, Thermo Fisher Scientific, and Wuhan Keqian Biology Co., Ltd., which maintain competitive advantages through global distribution infrastructure, validated diagnostic technologies, and deep expertise in veterinary diagnostic applications across swine health management, disease surveillance programs, and biosecurity operations, creating trust and reliability advantages with commercial pig producers and veterinary diagnostic laboratories. These companies leverage research and development capabilities in assay optimization and ongoing technical support relationships to defend market positions while expanding into emerging livestock markets and specialized disease targets.
Challengers encompass Qingdao Lijian Bio-Tech Co., Ltd. and BioChek, which compete through specialized product offerings and strong regional presence in key pig-producing markets. Product specialists, including Harbin National Engineering Research Center Of Veterinary Biologics Corp., Hunan Guoce Biotechnology Co., Ltd., and Nanjing Vazyme Biotech Co., Ltd., focus on specific diagnostic formats or regional markets, offering differentiated capabilities in local disease target panels, rapid turnaround testing services, and competitive pricing structures.
Regional players and emerging veterinary diagnostic manufacturers create competitive pressure through localized production advantages and rapid response capabilities, particularly in high-growth markets including China and Vietnam, where proximity to pig farming concentrations provides advantages in technical support responsiveness and customer relationships. Market dynamics favor companies that combine proven diagnostic accuracy with comprehensive veterinary consultation offerings that address the complete disease management cycle from sample collection through result interpretation and intervention recommendations.
Global Swine Disease Diagnostic Kit Market - Stakeholder Contribution Framework
Swine disease diagnostic kits represent advanced testing solutions that enable livestock producers to achieve rapid pathogen identification and timely disease intervention, delivering superior diagnostic accuracy and operational efficiency with high sensitivity detection and reliable result delivery in critical swine health management applications. With the market projected to grow from USD 646.9 million in 2025 to USD 976.2 million by 2035 at a 4.2% CAGR, these diagnostic testing systems offer compelling advantages - early disease detection, production loss prevention, and biosecurity assurance - making them essential for farmers applications (64.0% market share), veterinary clinic operations (36.0% share), and livestock production systems seeking alternatives to conventional clinical assessment methods that compromise disease control through delayed pathogen identification. Scaling market adoption and technology deployment requires coordinated action across agricultural policy, veterinary infrastructure development, diagnostic manufacturers, livestock producers, and animal health service providers.
How Governments Could Spur Agricultural Adoption?
- Livestock Health Programs: Include diagnostic testing subsidies in animal disease control initiatives, provide equipment support for establishing farm-level testing capabilities, and establish veterinary diagnostic laboratory capacity that reduces disease surveillance gaps in commercial production regions.
- Research & Veterinary Training Funding: Fund agricultural research on emerging swine diseases, support veterinary technician training programs in diagnostic testing techniques, and invest in disease surveillance systems that advance evidence-based intervention protocols.
- Agricultural Infrastructure Incentives: Provide grants for establishing regional veterinary diagnostic centers in major pig-producing areas, offer testing service subsidies for small and medium livestock producers, and support cold chain development ensuring reliable diagnostic kit distribution.
- Standards & Quality Certification: Establish national approval programs for veterinary diagnostic products, develop training requirements for diagnostic testing personnel, and create standardized protocols facilitating consistent test performance across production systems.
How Industry Bodies Could Support Market Development?
- Diagnostic Standards: Define standardized protocols for sample collection procedures, test execution methods, and result interpretation criteria, enabling consistent diagnostic approaches and reliable disease detection outcomes.
- Quality Assurance Programs: Develop comprehensive guidelines for diagnostic kit validation, laboratory proficiency testing, and quality control procedures ensuring measurement accuracy across different testing environments.
- Professional Education: Establish certification programs for veterinary technicians, continuing education requirements for farm personnel, and training materials ensuring proper diagnostic kit utilization and sample handling.
- Disease Surveillance Frameworks: Create technical documentation standards for test result reporting, disease notification protocols, and epidemiological data collection supporting coordinated outbreak response.
How Diagnostic Manufacturers Could Strengthen the Ecosystem?
- Advanced Product Development: Develop next-generation assay formats with improved sensitivity, multiplex detection platforms with expanded pathogen panels, and simplified testing procedures enabling on-farm deployment by trained personnel.
- Testing Technology Innovation: Provide efficient detection platforms with rapid result delivery, user-friendly formats requiring minimal equipment, and quality control systems ensuring consistent performance across production batches.
- Disease-Specific Solutions: Offer diagnostic kits tailored for African swine fever detection, specialized panels for respiratory disease complexes, and customized formats addressing regional disease challenges while maintaining sensitivity standards.
- Technical Support Services: Build comprehensive customer advisory programs including sample collection training, troubleshooting assistance, and result interpretation guidance helping producers achieve reliable diagnostic outcomes.
How Livestock Producers Could Navigate Technology Adoption?
- Disease Monitoring Programs: Develop systematic testing protocols for routine herd surveillance, establish early detection systems for outbreak identification, and implement biosecurity verification procedures supporting production risk management.
- Quality Management: Execute comprehensive quality assurance including proper sample handling, cold chain maintenance, and result documentation ensuring diagnostic reliability and regulatory compliance.
- Producer Education: Create awareness programs communicating disease risk factors, testing recommendations, and biosecurity practices promoting proactive health management in commercial operations.
- Outcome Tracking: Monitor production metrics including disease incidence rates, mortality reduction, and productivity improvements validating diagnostic program effectiveness.
How Investors Could Unlock Agricultural Value?
- Diagnostic Manufacturing: Finance established veterinary diagnostic manufacturers for production capacity expansion, assay development programs, and regulatory approval processes serving growing livestock health markets.
- Veterinary Service Infrastructure: Provide capital for establishing regional diagnostic laboratories, mobile testing service programs, and farm-based testing facilities expanding diagnostic access in major production regions.
- Technology Innovation: Back biotechnology companies developing rapid detection platforms, multiplex testing systems, and digital result interpretation tools enhancing diagnostic efficiency and accessibility.
- Market Consolidation: Support strategic partnerships between diagnostic manufacturers and veterinary pharmaceutical companies, finance technical training program development, and enable market consolidation creating comprehensive animal health solution providers.
Key Players in the Swine Disease Diagnostic Kit Market
- IDEXX
- Thermo Fisher Scientific
- Wuhan Keqian Biology Co., Ltd.
- Qingdao Lijian Bio-Tech Co., Ltd.
- Harbin National Engineering Research Center Of Veterinary Biologics Corp.
- Hunan Guoce Biotechnology Co., Ltd.
- Nanjing Vazyme Biotech Co., Ltd.
- Biovettest
- Ringbio
- AdvaCare Pharma
- Flashtest
- iNtRON Biotechnology
- Biosellal
- Advanced Molecular Diagnostics
- BioChek
Scope of the report

| Metric | Value |
|---|---|
| Quantitative Units | USD 674.07 million to USD 1017.14 million, at a CAGR of 4.2% |
| Market Definition | Swine Disease Diagnostic Kit encompasses immunoassay and molecular diagnostic kits designed to detect and identify viral, bacterial, and parasitic pathogens affecting commercial swine populations. The... |
| Diagnostic Method Type Segmentation | Enzyme-linked Immunosorbent Assay, Polymerase Chain Reaction, Other |
| Application Segmentation | Farmers, Veterinary Clinics |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | China, India, Germany, Brazil, USA, UK, Japan, and 40 plus countries |
| Key Companies Profiled | IDEXX, Thermo Fisher Scientific, Wuhan Keqian Biology Co., Ltd., Qingdao Lijian Bio-Tech Co., Ltd., Harbin National Engineering Research Center Of Veterinary Biologics Corp., Hunan Guoce Biotechnology Co., Ltd., Nanjing Vazyme Biotech Co., Ltd., Biovettest, Ringbio, AdvaCare Pharma, Flashtest, iNtRON Biotechnology, Biosellal, Advanced Molecular Diagnostics, and BioChek |
| Forecast Period | 2026 to 2036 |
| Approach | Forecasting models apply a bottom-up methodology starting with global installed base metrics and projecting conversion rates to advanced product adoption. |
Swine Disease Diagnostic Kit Market by Segments
Diagnostic Method Type:
- Enzyme-linked Immunosorbent Assay (ELISA) Kit
- Polymerase Chain Reaction (PCR) Kit
- Other
Application:
- Farmers
- Veterinary Clinics
Region:
- Asia Pacific
- China
- India
- Japan
- South Korea
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Netherlands
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- 1. World Organisation for Animal Health. (2024). WOAH Terrestrial Animal Health Code. WOAH.
- 2. Food and Agriculture Organization. (2024). FAOSTAT Livestock Production Statistics. FAO.
- 3. USA Department of Agriculture. (2024). Swine Health Monitoring Program. USDA APHIS.
- 4. European Commission. (2024). Regulation (EU) 2016/429 on Animal Health. EUR-Lex.
- 5. The World Bank. (2024). World Development Indicators: Agriculture. World Bank Group.
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
Frequently Asked Questions
How large is the demand for Swine Disease Diagnostic Kit in the global market in 2026?
Demand for swine disease diagnostic kit in the global market is estimated to be valued at USD 674.07 million in 2026.
What will be the market size of Swine Disease Diagnostic Kit in the global market by 2036?
Market size for swine disease diagnostic kit is projected to reach USD 1017.14 million by 2036.
What is the expected demand growth for Swine Disease Diagnostic Kit between 2026 and 2036?
Demand for swine disease diagnostic kit is expected to grow at a CAGR of 4.2% between 2026 and 2036.
Which Diagnostic Method Type is poised to lead global sales by 2026?
Enzyme-linked Immunosorbent Assay accounts for 58.0% share in 2026 as it addresses the primary demand requirements across global markets.
How significant is the role of Farmers in driving swine disease diagnostic kit adoption in 2026?
Farmers represents 64.0% of segment share as end-user demand consolidates around established application pathways.
What is driving demand in China?
China is projected to grow at a CAGR of 5.7% during 2026 to 2036, driven by expanding infrastructure investment and market modernization.
What does market forecast mean on this page?
The market forecast represents a model-based projection built on defined industry and supply chain assumptions for strategic planning purposes.
How does FMI build and validate the swine disease diagnostic kit forecast?
Forecasting models apply a bottom-up methodology starting with global installed base metrics and cross-validate projections against quarterly industry revenue volumes.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Diagnostic Method Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Diagnostic Method Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Diagnostic Method Type , 2026 to 2036
- Enzyme-linked Immunosorbent Assay
- Polymerase Chain Reaction
- Other
- Enzyme-linked Immunosorbent Assay
- Y to o to Y Growth Trend Analysis By Diagnostic Method Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Diagnostic Method Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Farmers
- Veterinary Clinics
- Farmers
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Diagnostic Method Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Diagnostic Method Type
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Diagnostic Method Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Diagnostic Method Type
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Diagnostic Method Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Diagnostic Method Type
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Diagnostic Method Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Diagnostic Method Type
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Diagnostic Method Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Diagnostic Method Type
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Diagnostic Method Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Diagnostic Method Type
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Diagnostic Method Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Diagnostic Method Type
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Diagnostic Method Type
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Diagnostic Method Type
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Diagnostic Method Type
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Diagnostic Method Type
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Diagnostic Method Type
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Diagnostic Method Type
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Diagnostic Method Type
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Diagnostic Method Type
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Diagnostic Method Type
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Diagnostic Method Type
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Diagnostic Method Type
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Diagnostic Method Type
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Diagnostic Method Type
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Diagnostic Method Type
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Diagnostic Method Type
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Diagnostic Method Type
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Diagnostic Method Type
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Diagnostic Method Type
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Diagnostic Method Type
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Diagnostic Method Type
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Diagnostic Method Type
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Diagnostic Method Type
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Diagnostic Method Type
- By Application
- Competition Analysis
- Competition Deep Dive
- IDEXX
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Thermo Fisher Scientific
- Wuhan Keqian Biology Co., Ltd.
- Qingdao Lijian Bio-Tech Co., Ltd.
- Harbin National Engineering Research Center Of Veterinary Biologics Corp.
- Hunan Guoce Biotechnology Co., Ltd.
- Nanjing Vazyme Biotech Co., Ltd.
- Biovettest
- Ringbio
- AdvaCare Pharma
- Flashtest
- iNtRON Biotechnology
- Biosellal
- Advanced Molecular Diagnostics
- BioChek
- IDEXX
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Diagnostic Method Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Diagnostic Method Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Diagnostic Method Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Diagnostic Method Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Diagnostic Method Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Diagnostic Method Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Diagnostic Method Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Diagnostic Method Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Diagnostic Method Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Diagnostic Method Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Diagnostic Method Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Diagnostic Method Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Diagnostic Method Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Diagnostic Method Type
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Diagnostic Method Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Diagnostic Method Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Diagnostic Method Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Diagnostic Method Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Diagnostic Method Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Diagnostic Method Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Diagnostic Method Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Diagnostic Method Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Diagnostic Method Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Diagnostic Method Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Diagnostic Method Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Diagnostic Method Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Diagnostic Method Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Diagnostic Method Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Diagnostic Method Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Diagnostic Method Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Diagnostic Method Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Diagnostic Method Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

