Whole Plasmid Sequencing Market
Whole Plasmid Sequencing Market is segmented by Drug Class (Direct oral anticoagulants (DOACs), Low molecular weight heparins (LMWHs), Vitamin k antagonists (VKAs), Factor Xa inhibitors, Anticoagulants, Antiplatelet agents), Application (Venous thromboembolism (VTE), Atrial fibrillation (AF) and stroke prevention, Post-surgical prophylaxis, Cancer-associated thrombosis (CAT), Pregnancy-associated thrombosis, Thromboprophylaxis in cardiovascular diseases, Others), End User (Hospitals, Ambulatory surgical centers (ASCs), Clinics, Home care settings, Specialty pharmacies), and Region. Forecast for 2026 to 2036.
Whole Plasmid Sequencing Market Size, Market Forecast and Outlook By FMI
Summary of the Whole Plasmid Sequencing Market
- Demand and Growth Drivers
- Growth in gene therapy and mRNA vaccine manufacturing is likely to sustain demand for whole plasmid sequencing services that verify vector integrity and sequence accuracy in biopharmaceutical production.
- Regulatory requirements for complete plasmid characterization in biologic drug submissions are expected to increase procurement of whole plasmid sequencing from CDMOs and CROs during the forecast period.
- Advancement in long-read sequencing technology is likely to reduce per-plasmid sequencing costs and expand adoption across smaller biotech companies through 2036.
- Product and Segment View
- Direct oral anticoagulants (DOACs) is likely to account for 39.1% of the drug class segment, reflecting sustained procurement across primary end-use categories.
- Venous thromboembolism (VTE) is likely to account for 34.7% of the application segment, reflecting sustained procurement across primary end-use categories.
- Hospitals is likely to account for 61.8% of the end user segment, reflecting sustained procurement across primary end-use categories.
- Geography and Competitive Outlook
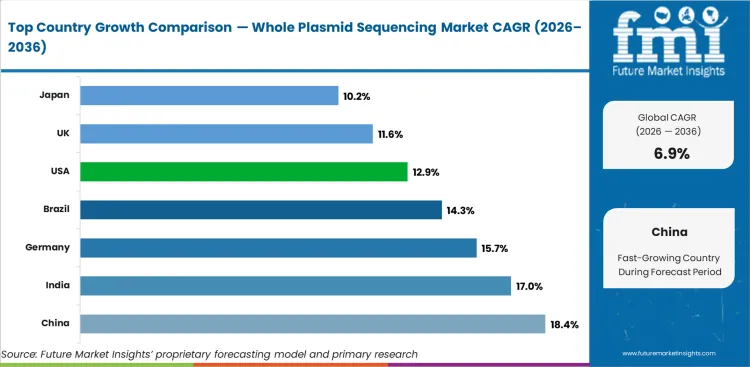
- China is likely to remain a key market at 18.4%, supported by rapid biopharmaceutical manufacturing expansion.
- India is likely to sustain growth at 17.0%, driven by expanding pharmaceutical manufacturing capacity.
- Companies that combine product breadth with customer access and distribution reach are likely to strengthen competitive positioning during the forecast period.
- Analyst Opinion
- Nandini Roy Choudhury, Principal Consultant at FMI says, 'The whole plasmid sequencing market reflects biopharmaceutical quality control requirements and gene therapy manufacturing growth. Service providers that can deliver rapid, cost-effective, and regulatory-compliant whole plasmid characterization are positioned to capture long-term pharmaceutical accounts during the forecast period.'
- The whole plasmid sequencing market is evolving from research-grade service toward regulatory-compliant pharmaceutical quality control.
- Adoption is increasing due to gene therapy manufacturing growth and regulatory requirements for complete vector characterization.
- Demand is further supported by long-read sequencing cost reduction expanding access for smaller biotech companies.
- Service providers with validated workflows, regulatory expertise, and rapid turnaround are likely to capture pharmaceutical accounts.
Whole Plasmid Sequencing Market Definition
The whole plasmid sequencing market includes sequencing services and platforms that provide complete, end-to-end characterization of circular plasmid DNA molecules used in biopharmaceutical manufacturing, gene therapy vector production, vaccine development, and recombinant protein expression systems.
Whole Plasmid Sequencing Market Inclusions
Market scope encompasses all commercially traded whole plasmid sequencing products and services categorized by service type (sequencing and analysis, library preparation, data interpretation), technology (long-read sequencing, short-read assembly, hybrid approaches), end user (biopharmaceutical companies, CROs and CDMOs, academic research institutions), application (gene therapy QC, vaccine manufacturing, recombinant protein production, synthetic biology), and delivery model (service-based, platform licensing). The range of revenue sizes is from 2026 to 2036.
Whole Plasmid Sequencing Market Exclusions
The scope does not include general whole-genome sequencing, targeted gene panel sequencing, or RNA sequencing services not specifically designed for circular plasmid DNA characterization.
Whole Plasmid Sequencing Market Research Methodology
- Primary Research: FMI analysts conducted interviews with manufacturers, end-use procurement teams, and distribution partners across key markets.
- Desk Research: Combined data from industry associations, regulatory filings, trade publications, and manufacturer disclosures.
- Market sizing and forecasting: Bottom-up aggregation across product types, end-use applications, and regional adoption curves, validated against industry benchmarks.
- Data validation: Cross-checked every quarter against production data, trade statistics, and manufacturer-reported figures.
Why is the Whole Plasmid Sequencing Market Growing?
- Gene therapy and vaccine manufacturing growth are creating sustained demand for whole plasmid sequencing services that verify vector integrity and sequence fidelity in biopharmaceutical production.
- Regulatory requirements for complete plasmid characterization in biologic drug filings are increasing outsourcing of sequencing services to specialized providers.
- China (18.4%) and India (17.0%) lead growth, reflecting expanding biopharmaceutical manufacturing capacity and growing adoption of quality control sequencing protocols.
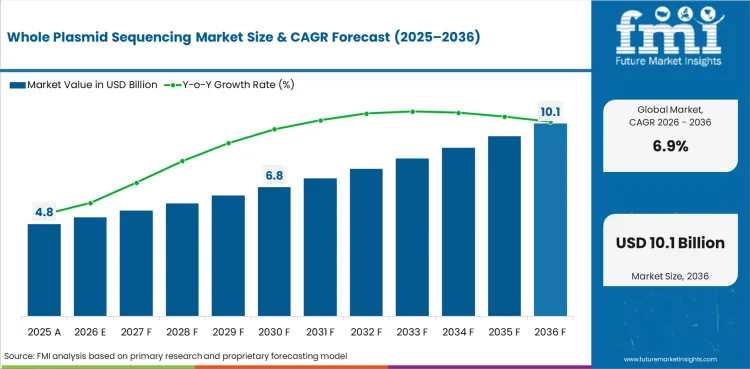
The whole plasmid sequencing market is expanding at 6.9% CAGR, reflecting biopharmaceutical quality control demand, gene therapy manufacturing growth, and long-read sequencing technology advancement. Sequencing services and analysis account for the primary revenue stream as biopharmaceutical companies outsource plasmid characterization to specialized sequencing providers with validated workflows.
Gene therapy vector manufacturing represents the highest-growth application, where regulatory submissions require complete sequence verification of plasmid DNA used for viral vector production. Each gene therapy program in clinical development or commercial manufacturing requires ongoing plasmid sequencing for batch release and stability monitoring.
Long-read sequencing platforms from providers like Oxford Nanopore and Pacific Biosciences enable complete plasmid reads in single passes, eliminating assembly artifacts and providing higher confidence in sequence accuracy compared to short-read approaches. Cost reduction in long-read sequencing is expanding adoption from large pharma to mid-size biotech companies.
Market Segmentation Analysis
- Direct oral anticoagulants (DOACs) accounts for 39.1% of the drug class segment, reflecting consistent demand across primary applications.
- Venous thromboembolism (VTE) accounts for 34.7% of the application segment, reflecting consistent demand across primary applications.
- Hospitals accounts for 61.8% of the end user segment, reflecting consistent demand across primary applications.
The whole plasmid sequencing market is segmented by drug class, application, end user, product.
Insights into the Direct oral anticoagulants (DOACs) Drug Class Segment
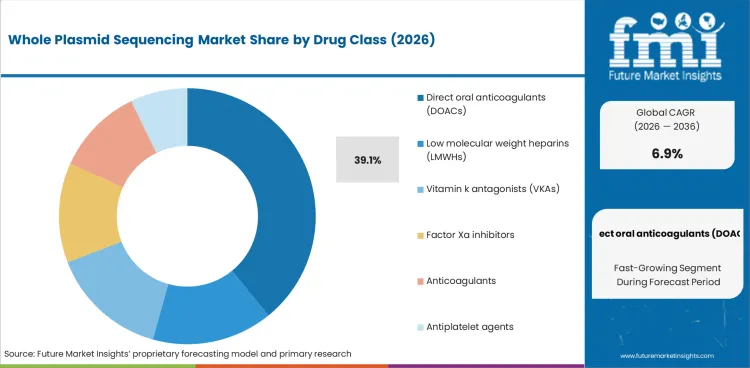
In 2026, direct oral anticoagulants (doacs) is expected to account for 39.1% of the drug class segment. Demand reflects procurement preferences where performance and specification compliance are critical.
Competitive positioning depends on production capability, quality certification, and the ability to meet evolving requirements. Suppliers with established relationships and proven performance are likely to maintain share.
Insights into the Venous thromboembolism (VTE) Application Segment
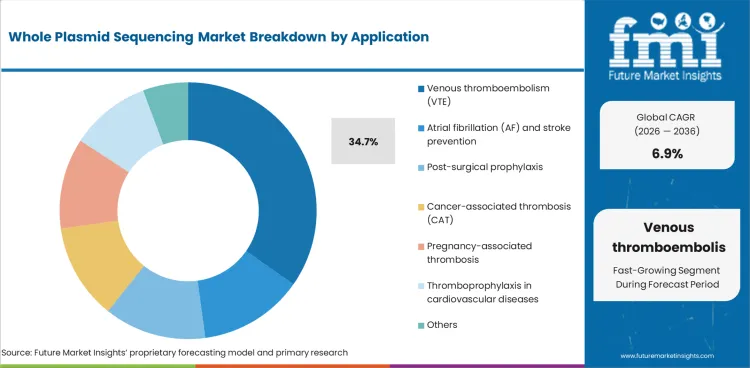
In 2026, venous thromboembolism (vte) is expected to account for 34.7% of the application segment. Demand reflects procurement preferences where performance and specification compliance are critical.
Competitive positioning depends on production capability, quality certification, and the ability to meet evolving requirements. Suppliers with established relationships and proven performance are likely to maintain share.
Whole Plasmid Sequencing Market Drivers, Restraints, and Opportunities
- Gene therapy manufacturing growth and regulatory requirements for complete plasmid characterization are creating sustained demand for whole plasmid sequencing services.
- Sequencing cost per plasmid and turnaround time constraints limit adoption among cost-sensitive academic and early-stage biotech clients.
- Long-read sequencing technology advancement is reducing per-plasmid costs and expanding the addressable market for routine quality control sequencing.
The whole plasmid sequencing market reflects biopharmaceutical quality control requirements, gene therapy manufacturing growth, and sequencing technology advancement.
Gene Therapy and Vaccine Manufacturing QC Demand
Biopharmaceutical companies require complete plasmid sequence verification for regulatory submissions and batch release. Gene therapy programs in clinical and commercial stages create recurring sequencing demand for each manufacturing campaign.
Sequencing Cost and Throughput Limitations
Per-plasmid sequencing costs and laboratory turnaround times influence adoption velocity. Service providers that can deliver rapid, cost-effective whole plasmid sequencing gain competitive advantage in high-volume pharmaceutical accounts.
Long-Read Technology Cost Reduction
Advancement in nanopore and single-molecule sequencing is reducing per-read costs and enabling single-pass complete plasmid sequencing. Cost reduction expands adoption from large pharmaceutical to mid-size biotech and academic clients.
Analysis of Whole Plasmid Sequencing Market By Key Countries
.webp)
| Country | CAGR |
|---|---|
| China | 18.4% |
| India | 17.0% |
| Germany | 15.7% |
| Brazil | 14.3% |
| USA | 12.9% |
| UK | 11.6% |
| Japan | 10.2% |
- China leads with 18.4%, supported by rapid biopharmaceutical manufacturing expansion.
- India at 17.0%, reflecting expanding pharmaceutical manufacturing capacity.
- Germany at 15.7%, reflecting advanced biopharmaceutical manufacturing.
The global whole plasmid sequencing market is expected to grow at a rate of 6.9% per year from 2026 to 2036. The study covers more than 30 countries, and the main markets are listed below.
Demand Outlook for Whole Plasmid Sequencing Market in China
China is expected to grow at 18.4% through 2036, reflecting rapid biopharmaceutical manufacturing expansion, gene therapy development programs, and growing adoption of sequencing-based quality control.
- Biopharma expansion drives sequencing demand.
- Gene therapy development increases plasmid QC needs.
- Manufacturing growth sustains service procurement.
Future Outlook for Whole Plasmid Sequencing Market in India
India is expected to grow at 17.0% through 2036, reflecting expanding pharmaceutical manufacturing capacity, growing biosimilar production, and increasing adoption of advanced quality control technologies.
- Pharma manufacturing growth creates QC demand.
- Biosimilar production increases sequencing needs.
- Technology adoption accelerates service procurement.
Opportunity Analysis of Whole Plasmid Sequencing Market in Germany
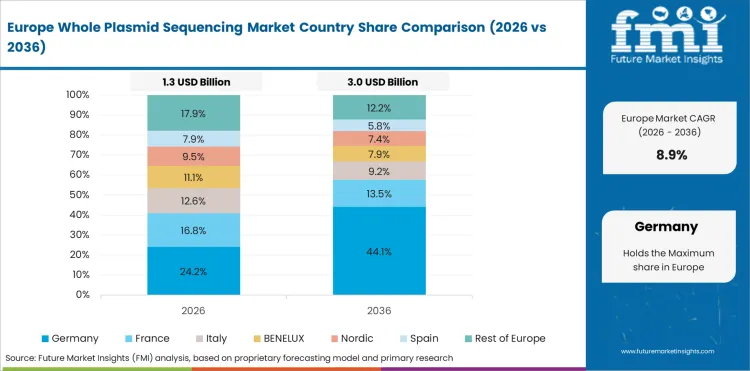
Germany is expected to grow at 15.7% through 2036, reflecting advanced biopharmaceutical manufacturing, strict regulatory compliance, and leadership in pharmaceutical quality control standards.
- Advanced manufacturing sustains QC sequencing demand.
- Regulatory compliance drives characterization requirements.
- Quality standards support premium service procurement.
In-depth Analysis of Whole Plasmid Sequencing Market in Brazil
Brazil is expected to grow at 14.3% through 2036, reflecting pharmaceutical manufacturing growth, biologics production expansion, and increasing adoption of molecular quality control.
- Pharma growth creates quality control demand.
- Biologics expansion increases sequencing needs.
- Technology adoption supports service procurement.
Sales Analysis of Whole Plasmid Sequencing Market in USA
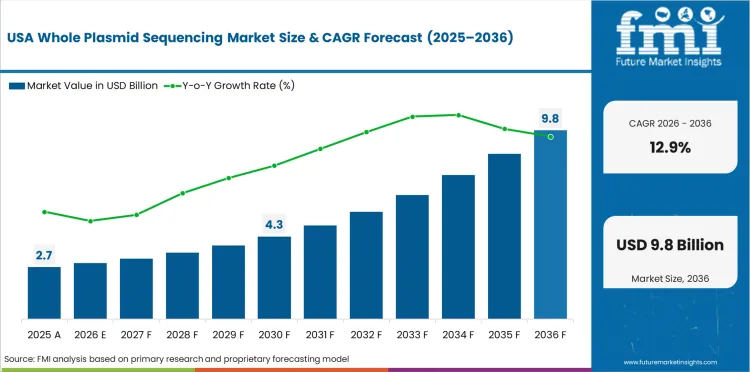
USA is expected to grow at 12.9% through 2036, reflecting largest biopharmaceutical market, gene therapy manufacturing leadership, and established sequencing service provider infrastructure.
- Largest biopharma market sustains sequencing demand.
- Gene therapy leadership drives plasmid QC volumes.
- Established service infrastructure supports adoption.
Growth Analysis of Whole Plasmid Sequencing Market in UK
UK is expected to grow at 11.6% through 2036, reflecting biopharmaceutical R&D strength, MHRA regulatory requirements, and growing gene therapy development activity.
- Biopharma R&D strength drives sequencing demand.
- Regulatory requirements sustain QC procurement.
- Gene therapy growth increases characterization needs.
Market Outlook for Whole Plasmid Sequencing Market in Japan
Japan is expected to grow at 10.2% through 2036, reflecting advanced pharmaceutical manufacturing, precision quality standards, and growing adoption of sequencing-based characterization.
- Advanced manufacturing sustains precision QC demand.
- Quality standards drive sequencing specification.
- Technology adoption supports service procurement.
Competitive Landscape and Strategic Positioning
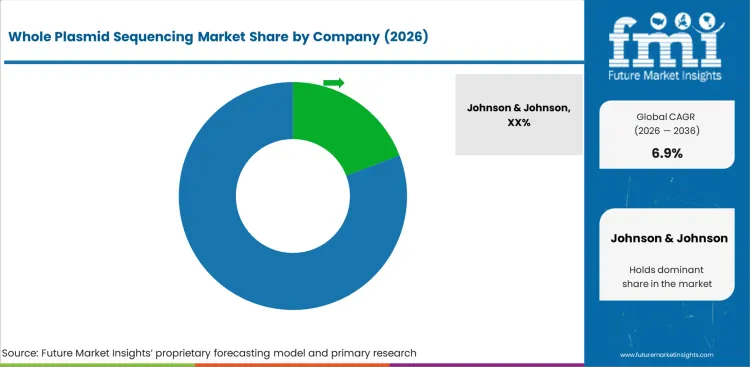
- Johnson & Johnson and Boehringer Ingelheim GmbH lead through broad portfolios and established customer relationships.
- Mid-tier participants compete through regional specialization and targeted product development.
- New entrants focus on technology-differentiated products and specialized applications.
Johnson & Johnson holds a strong position through technology leadership and established relationships. Boehringer Ingelheim GmbH competes through complementary capabilities.
Sanofi SA and Pfizer Inc. maintain positions through specialized offerings.
Barriers include established relationships, regulatory requirements, technology expertise, and manufacturing scale. Strategic priorities include technology advancement, compliance, and geographic expansion.
Key Companies in the Whole Plasmid Sequencing Market
Key global companies leading the whole plasmid sequencing market include:
- Johnson & Johnson, Boehringer Ingelheim GmbH, Sanofi SA hold leading positions through technology leadership and established relationships.
- Pfizer Inc., Bayer AG have established positions through specialized offerings.
- Plasmidsaurus Inc., Eurofins Genomics, Azenta Life Sciences represent emerging participants with growth potential.
Competitive Benchmarking: Whole Plasmid Sequencing Market
| Company | Sequencing Technology | Throughput Capacity | Regulatory Expertise | Geographic Reach |
|---|---|---|---|---|
| Johnson & Johnson | High | High | High | Global |
| Boehringer Ingelheim GmbH | High | Medium | High | Global |
| Sanofi SA | Medium | Medium | Medium | Global |
| Pfizer Inc. | Medium | Moderate | Medium | Regional |
| Bayer AG | Moderate | Moderate | Moderate | Regional |
| Bristol-Myers Squibb | Moderate | Low | Moderate | Regional |
| Plasmidsaurus Inc. | Low | Low | Low | Niche |
| Eurofins Genomics | Low | Low | Low | Niche |
| Azenta Life Sciences | Low | Low | Low | Niche |
| Genewiz (Azenta) | Low | Low | Low | Niche |
Source: Future Market Insights competitive analysis, 2026.
Key Developments in Whole Plasmid Sequencing Market
- In 2025, Thermo Fisher Scientific expanded its whole plasmid sequencing service capacity to address growing demand from gene therapy and vaccine manufacturers requiring complete vector characterization.
- In 2025, a leading sequencing provider launched a rapid-turnaround whole plasmid sequencing service targeting biopharmaceutical batch release and stability testing applications.
Key Players in the Whole Plasmid Sequencing Market
Major Global Players
- Johnson & Johnson
- Boehringer Ingelheim GmbH
- Sanofi SA
- Pfizer Inc.
- Bayer AG
- Bristol-Myers Squibb
- AstraZeneca
Emerging Players/Startups
- Plasmidsaurus Inc.
- Eurofins Genomics
- Azenta Life Sciences
- Genewiz (Azenta)
- Eton Bioscience
Report Scope and Coverage
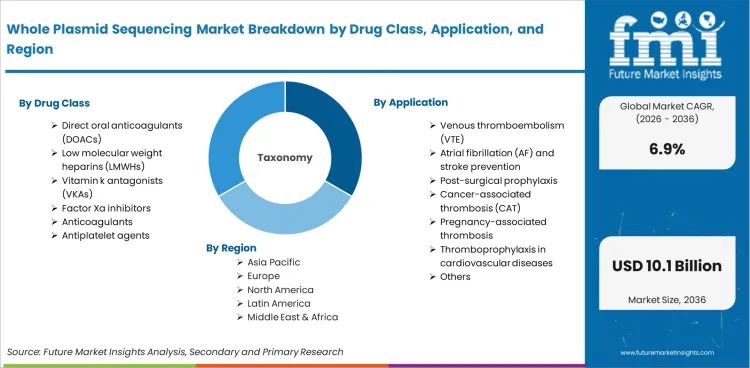
| Quantitative Units | USD 5.17 Billion to USD 10.08 Billion, at a CAGR of 6.9% |
|---|---|
| Market Definition | The whole plasmid sequencing market encompasses whole plasmid DNA sequencing services and platforms used for complete vector characterization in gene therapy, vaccine, and recombinant protein biopharmaceutical manufacturing quality control. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | China, India, Germany, Brazil, USA, UK, Japan, 30 plus countries |
| Key Companies Profiled | Johnson & Johnson, Boehringer Ingelheim GmbH, Sanofi SA, Pfizer Inc., Bayer AG, Bristol-Myers Squibb, AstraZeneca, Sun Pharmaceutical Industries, Daiichi Sankyo, Portola Pharmaceuticals, Laurus Labs |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down methodology starting with verified transaction data, projecting adoption velocity across segments and regions. |
Segmentation
Whole Plasmid Sequencing Market Segmented by Drug Class:
- Direct oral anticoagulants (DOACs)
- Low molecular weight heparins (LMWHs)
- Vitamin k antagonists (VKAs)
- Factor Xa inhibitors
- Anticoagulants
- Antiplatelet agents
Whole Plasmid Sequencing Market Segmented by Application:
- Venous thromboembolism (VTE)
- Atrial fibrillation (AF) and stroke prevention
- Post-surgical prophylaxis
- Cancer-associated thrombosis (CAT)
- Pregnancy-associated thrombosis
- Thromboprophylaxis in cardiovascular diseases
- Others
Whole Plasmid Sequencing Market Segmented by End User:
- Hospitals
- Ambulatory surgical centers (ASCs)
- Clinics
- Home care settings
- Specialty pharmacies
Whole Plasmid Sequencing Market Segmented by Product:
- Sequencing Services
- Short Read Sequencing Services
- Illumina Based Sequencing
- Ion Semiconductor Sequencing
- Long Read Sequencing Services
- Nanopore Sequencing
- Single Molecule Real Time Sequencing
- Short Read Sequencing Services
- Sequencing Kits And Reagents
- Library Preparation Kits
- Fragmentation Kits
- Adapter Ligation Kits
- Sequencing Reagents
- Polymerase Reagents
- Buffer Solutions
- Library Preparation Kits
- Instruments
- Benchtop Sequencers
- Low Throughput Systems
- Medium Throughput Systems
- High Throughput Sequencers
- Core Lab Systems
- Production Scale Systems
- Benchtop Sequencers
Whole Plasmid Sequencing Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Research Sources and Bibliography
- USA Food and Drug Administration. (2024). FDA Guidance on Chemistry, Manufacturing, and Controls for Gene Therapy Products. FDA.
- European Medicines Agency. (2024). EMA Guidelines on Quality Requirements for Plasmid DNA Vectors. EMA.
- International Council for Harmonisation. (2024). ICH Q5B: Quality of Biotechnological Products, Analysis of Expression Construct. ICH.
- World Health Organization. (2024). WHO Technical Report on Gene Therapy Product Quality Standards. WHO.
- International Organization for Standardization. (2024). ISO 20395: Biotechnology, Requirements for Evaluating Performance of Quantification Methods for Nucleic Acid Sequences. ISO.
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with publication dates, URLs, and supporting data for all cited works.
This Report Answers
- Estimating the size of the market and revenue from 2026 to 2036.
- Segmentation analysis across all product, application, and end-use categories.
- Regional insights covering more than 30 markets worldwide.
- Technology and material analysis relevant to market applications.
- Competitive landscape assessment including company benchmarking.
- Identification of investment opportunities across high-growth segments and regions.
- Supply chain analysis and distribution channel assessment.
- Data delivery in PDF and Excel formats.
Frequently Asked Questions
What is the global market demand for Whole Plasmid Sequencing Market in 2026?
In 2026, the global whole plasmid sequencing market is expected to be worth USD 5.17 billion.
How big will the Whole Plasmid Sequencing Market be in 2036?
By 2036, the whole plasmid sequencing market is expected to be worth USD 10.08 billion.
How much is demand expected to grow between 2026 and 2036?
Between 2026 and 2036, demand is expected to grow at a CAGR of 6.9%.
Which End User segment is likely to lead globally in 2026?
Hospitals is expected to account for 61.8% of the end user segment in 2026, reflecting strong end-use demand.
What is causing demand to rise in China?
China is expected to grow at 18.4% through 2036, reflecting rapid biopharmaceutical manufacturing expansion, gene therapy development programs, and growing adoption of sequencing-based quality control.
What is causing demand to rise in India?
India is expected to grow at 17.0% through 2036, reflecting expanding pharmaceutical manufacturing capacity, growing biosimilar production, and increasing adoption of advanced quality control technologies.
What does this report mean by 'Whole Plasmid Sequencing Market definition'?
The whole plasmid sequencing market encompasses whole plasmid DNA sequencing services and platforms used for complete vector characterization in gene therapy, vaccine, and recombinant protein biopharmaceutical manufacturing quality control.
How does FMI make the forecast and validate it?
Forecasting models use a hybrid bottom-up and top-down approach, starting with verified transaction data and validating against industry statistics and manufacturer disclosures.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Drug Class
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Drug Class , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Drug Class , 2026 to 2036
- Direct oral anticoagulants (DOACs)
- Low molecular weight heparins (LMWHs)
- Vitamin k antagonists (VKAs)
- Factor Xa inhibitors
- Anticoagulants
- Antiplatelet agents
- Direct oral anticoagulants (DOACs)
- Y to o to Y Growth Trend Analysis By Drug Class , 2021 to 2025
- Absolute $ Opportunity Analysis By Drug Class , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Venous thromboembolism (VTE)
- Atrial fibrillation (AF) and stroke prevention
- Post-surgical prophylaxis
- Cancer-associated thrombosis (CAT)
- Pregnancy-associated thrombosis
- Thromboprophylaxis in cardiovascular diseases
- Others
- Venous thromboembolism (VTE)
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- Ambulatory surgical centers (ASCs)
- Clinics
- Home care settings
- Specialty pharmacies
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product, 2026 to 2036
- Sequencing Services
- Short Read Sequencing Services
- _Illumina Based Sequencing
- _Ion Semiconductor Sequencing
- Long Read Sequencing Services
- _Nanopore Sequencing
- _Single Molecule Real Time Sequencing
- Sequencing Kits And Reagents
- Library Preparation Kits
- _Fragmentation Kits
- _Adapter Ligation Kits
- Sequencing Reagents
- _Polymerase Reagents
- Instruments
- Benchtop Sequencers
- _Low Throughput Systems
- _Medium Throughput Systems
- High Throughput Sequencers
- _Core Lab Systems
- Sequencing Services
- Y to o to Y Growth Trend Analysis By Product, 2021 to 2025
- Absolute $ Opportunity Analysis By Product, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Drug Class
- By Application
- By End User
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Application
- By End User
- By Product
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Drug Class
- By Application
- By End User
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Application
- By End User
- By Product
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Drug Class
- By Application
- By End User
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Application
- By End User
- By Product
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Drug Class
- By Application
- By End User
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Application
- By End User
- By Product
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Drug Class
- By Application
- By End User
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Application
- By End User
- By Product
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Drug Class
- By Application
- By End User
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Application
- By End User
- By Product
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Drug Class
- By Application
- By End User
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Application
- By End User
- By Product
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Application
- By End User
- By Product
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Application
- By End User
- By Product
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Application
- By End User
- By Product
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Application
- By End User
- By Product
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Application
- By End User
- By Product
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Application
- By End User
- By Product
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Application
- By End User
- By Product
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Application
- By End User
- By Product
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Application
- By End User
- By Product
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Application
- By End User
- By Product
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Application
- By End User
- By Product
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Application
- By End User
- By Product
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Application
- By End User
- By Product
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Application
- By End User
- By Product
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Application
- By End User
- By Product
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Application
- By End User
- By Product
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Application
- By End User
- By Product
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Application
- By End User
- By Product
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Application
- By End User
- By Product
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Application
- By End User
- By Product
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Application
- By End User
- By Product
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Application
- By End User
- By Product
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Drug Class
- By Application
- By End User
- By Product
- Competition Analysis
- Competition Deep Dive
- Johnson & Johnson
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Boehringer Ingelheim GmbH
- Sanofi SA
- Pfizer Inc.
- Bayer AG
- Bristol-Myers Squibb
- AstraZeneca
- Sun Pharmaceutical Industries
- Daiichi Sankyo
- Portola Pharmaceuticals
- Laurus Labs
- Johnson & Johnson
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Drug Class , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Drug Class , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Drug Class , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by Drug Class , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by Drug Class , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by Drug Class , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by Drug Class , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by Drug Class , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by Product, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Drug Class , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Drug Class , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Drug Class
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Product
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by Drug Class , 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Drug Class , 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Drug Class
- Figure 29: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Application
- Figure 32: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by End User
- Figure 35: North America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Product
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by Drug Class , 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Drug Class , 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Drug Class
- Figure 42: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 44: Latin America Market Attractiveness Analysis by Application
- Figure 45: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by End User
- Figure 48: Latin America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Product
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by Drug Class , 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by Drug Class , 2026-2036
- Figure 54: Western Europe Market Attractiveness Analysis by Drug Class
- Figure 55: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 57: Western Europe Market Attractiveness Analysis by Application
- Figure 58: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 60: Western Europe Market Attractiveness Analysis by End User
- Figure 61: Western Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Product
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by Drug Class , 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by Drug Class , 2026-2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by Drug Class
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by Application
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by End User
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by Product
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by Drug Class , 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by Drug Class , 2026-2036
- Figure 80: East Asia Market Attractiveness Analysis by Drug Class
- Figure 81: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 83: East Asia Market Attractiveness Analysis by Application
- Figure 84: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 86: East Asia Market Attractiveness Analysis by End User
- Figure 87: East Asia Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 89: East Asia Market Attractiveness Analysis by Product
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by Drug Class , 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by Drug Class , 2026-2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by Drug Class
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by Drug Class , 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by Drug Class , 2026-2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by Drug Class
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

