The ampoule clarity test apparatus market is likely to be valued at USD 190.0 million in 2026 and is forecasted to reach USD 346.7 million by 2036 at a CAGR of 6.2%. Value formation is anchored in mandatory visual inspection requirements for sterile injectable containers rather than discretionary laboratory modernization. Each ampoule batch must undergo documented clarity assessment to confirm absence of visible particulates and container defects before release. Equipment demand scales with injectable production volume inspection frequency and audit exposure. Capital allocation favors standardized optical conditions reproducible inspector positioning and traceable documentation outputs. Spending behavior reflects regulatory obligation risk mitigation and batch release continuity rather than throughput optimization alone.
Adoption remains constrained by operator dependence validation burden and limited automation tolerance within regulated quality systems. Visual inspection outcomes still rely on human acuity creating fatigue sensitivity and false rejection exposure at higher volumes. Scaling inspection capacity requires trained personnel controlled lighting environments and periodic requalification. Transition toward fully automated clarity inspection faces capital intensity software validation complexity and regulator conservatism. Procurement decisions prioritize proven designs serviceability and long-term calibration support. Demand persistence tracks injectable portfolio expansion regulatory inspection rigor and replacement of aging benches rather than rapid technology substitution across facilities globally.
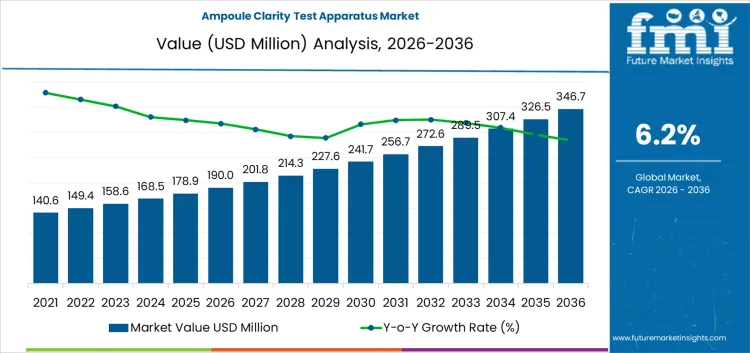
| Metric | Value |
|---|---|
| Market Value (2026) | USD 190.0 million |
| Market Forecast Value (2036) | USD 346.7 million |
| Forecast CAGR 2026 to 2036 | 6.2% |
Demand for ampoule clarity test apparatus is increasing as pharmaceutical manufacturers and quality control laboratories require precise optical inspection of sterile liquid dosage forms to ensure product safety and regulatory compliance. Ampoules used for injectable formulations must be free of particulate matter, cracks, and crystallization that could compromise sterility or dose integrity, and clarity testing apparatus provides standardized illumination, magnification, and measurement conditions to support repeatable evaluation. Quality assurance professionals specify these systems to document visual inspection results that align with pharmacopeial criteria and internal quality benchmarks. Procurement teams assess optical resolution, lighting uniformity, ergonomic design, and data capture capabilities to select equipment that integrates with laboratory workflows and document control systems.
Regulators emphasize documented evidence of container integrity assessments as part of batch release protocols, driving investment in apparatus that support audit ready records and minimize subjectivity in visual evaluation. Expansion of biologics, vaccines, and complex sterile injectables reinforces need for robust clarity testing because particulate contamination risk increases with formulation complexity and production volumes. Technical staff benefit from apparatus that offer calibrated optics, repeatable positioning fixtures, and traceable software outputs that reduce rework and operator variability. These operational and regulatory factors are contributing to sustained demand for ampoule clarity test apparatus in pharmaceutical quality environments
Demand for ampoule clarity test apparatus is shaped by injectable safety requirements, particulate contamination control, and regulatory inspection mandates. Sterile drug products require visual clarity verification to prevent patient exposure to visible particulates. Adoption aligns with enforcement of pharmacopeial standards and quality system audits. Equipment selection prioritizes inspection consistency, operator ergonomics, and documentation reliability. Segment classification reflects differentiation by inspection apparatus design, operational end use, and compliance capability. Structure highlights how inspection format, production environment, and regulatory alignment influence specification priorities, inspection throughput, and utilization patterns across sterile product quality control workflows.
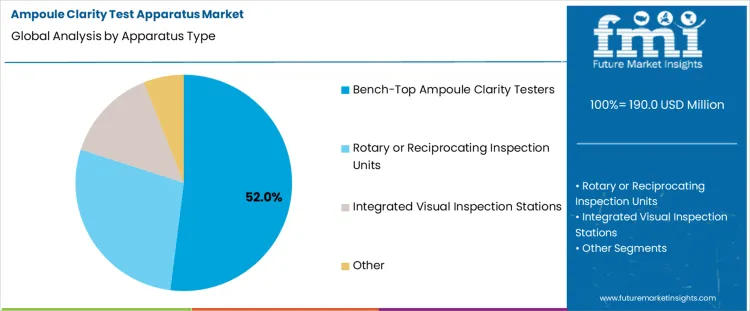
Bench-top ampoule clarity testers hold 52.0%, representing the largest share among apparatus types due to operational simplicity and inspection control. These units support manual or semi-assisted visual inspection under standardized lighting conditions. Compact footprint suits quality control laboratories with limited space. Operator-controlled handling enables inspection of varied ampoule sizes and batch volumes. Rotary and reciprocating inspection units support higher throughput with mechanical motion assistance. Integrated visual inspection stations combine clarity testing with additional checks. Apparatus type segmentation reflects preference for flexible, controlled inspection solutions supporting regulatory compliance and batch-based sterile manufacturing workflows.
Key Points
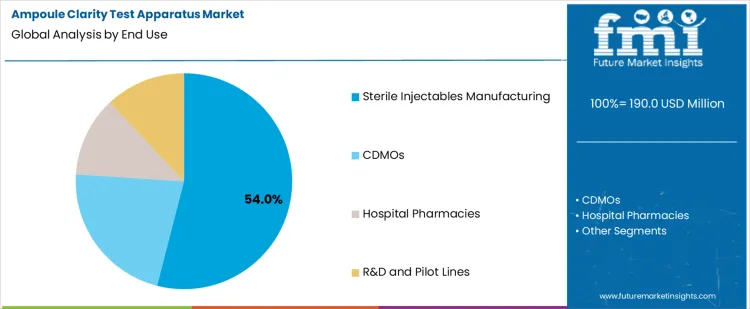
Sterile injectables manufacturing holds 54.0%, representing the largest share among end-use settings due to stringent quality requirements. Manufacturers must verify absence of visible particulates before product release. High inspection frequency aligns with large batch production and regulatory scrutiny. Clarity testing remains integral to release and in-process quality checks. Contract development and manufacturing organizations support multiple clients with varied inspection volumes. Hospital pharmacies apply testing for compounding verification. Research and pilot lines perform limited-volume inspection. End-use segmentation reflects concentration of clarity testing demand where regulatory accountability and patient safety obligations remain highest.
Key Points
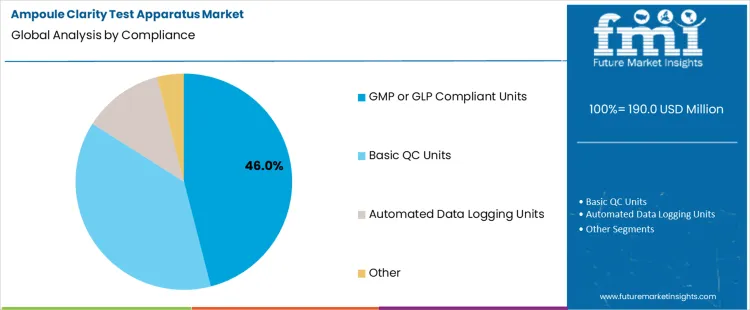
GMP or GLP compliant units hold 46.0%, representing the largest share among compliance categories due to regulatory enforcement. These systems meet documentation, validation, and operational requirements specified by quality guidelines. Compliance supports audit readiness and standardized inspection practices. Equipment qualification ensures reproducible inspection conditions. Basic quality control units serve non-regulated environments with reduced documentation needs. Automated data logging units enhance traceability with lower adoption volume. Compliance segmentation reflects preference for validated equipment where regulatory conformity and inspection credibility remain essential.
Key Points
Demand for ampoule clarity test apparatus reflects quality control requirements in sterile injectable manufacturing and pharmaceutical packaging operations. Adoption concentrates within pharmaceutical production plants, contract manufacturing organizations, and quality control laboratories handling glass ampoules and liquid injectables. Global scope aligns with parenteral drug safety expectations and visual inspection mandates. Usage centers on manual and semi-automated systems designed to detect visible particulate matter, turbidity, and cosmetic defects in sealed ampoules.
Sterile injectable products require visual clarity verification to ensure absence of particulate contamination before release. Demand increases as regulatory guidelines mandate 100 percent inspection of parenteral dosage forms. Ampoule clarity test apparatus provide controlled illumination and contrast to support consistent visual assessment by trained inspectors. Pharmaceutical manufacturers adopt standardized inspection setups to reduce subjective variation across operators. Increased production of biologics and high-potency injectables elevates sensitivity to visible and sub-visible contamination. Integration of rotating platforms improves detection of moving particles within liquid media. Adoption reflects non-discretionary compliance with product safety requirements and batch release protocols.
Manual and semi-automated clarity testing remains dependent on human visual acuity, introducing fatigue-related variability. Demand sensitivity rises where high production volumes strain inspection throughput capacity. Training and qualification requirements increase operational cost and limit rapid workforce scaling. False rejection rates affect yield and increase reinspection workload. Transition to fully automated systems faces validation complexity and high capital cost. Regulatory documentation requires strict control of inspection conditions and records. Glass ampoule variability affects inspection consistency. Space and lighting control constraints limit flexible deployment across smaller production sites.
Demand for ampoule clarity test apparatus is expanding globally due to injectable drug production growth, visual inspection compliance, and tightening quality control requirements. Pharmaceutical manufacturers require reliable detection of particulate contamination in ampoules before release. Manual and semi-automated inspection systems support batch verification under regulatory audit conditions. Sterile manufacturing scale-up across emerging regions reinforces equipment procurement. Growth rates in India at 8.6%, China at 8.1%, Vietnam at 7.5%, Indonesia at 7.1%, and Mexico at 6.4% indicate sustained expansion driven by injectable output growth, inspection standardization, and lifecycle replacement of legacy inspection setups.
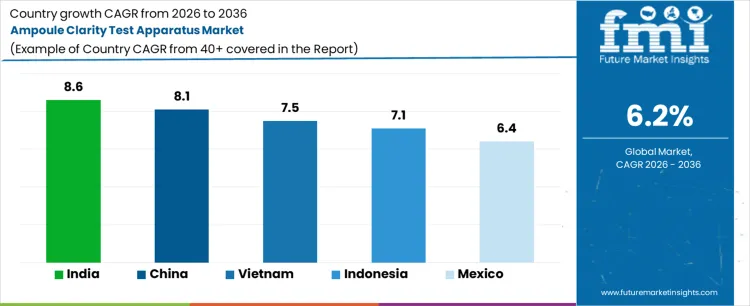
| Country | CAGR (%) |
|---|---|
| India | 8.6% |
| China | 8.1% |
| Vietnam | 7.5% |
| Indonesia | 7.1% |
| Mexico | 6.4% |
India is growing at a CAGR of 8.6%, supported by expansion of injectable pharmaceutical manufacturing. Large-scale production of antibiotics, vaccines, and critical care drugs increases inspection workload. Regulatory authorities emphasize visible particulate control and documented inspection outcomes. Contract manufacturing organizations invest in standardized inspection stations to support export compliance. Workforce variability increases preference for controlled visual inspection environments. Demand growth reflects injectable volume expansion and regulatory scrutiny rather than innovation-driven inspection upgrades.
Ampoule clarity test apparatus demand in China is expanding at a CAGR of 8.1%, driven by high-volume sterile drug production and quality system enforcement. Injectable formulations for hospitals and public health programs increase batch inspection frequency. Centralized pharmaceutical plants implement standardized visual inspection protocols. Automation integration remains selective, sustaining demand for reliable manual and semi-automated systems. Domestic equipment suppliers support rapid deployment and service coverage. Demand growth reflects manufacturing scale and inspection consistency requirements rather than transition to fully automated inspection lines.
Ampoule clarity test apparatus demand in Vietnam is growing at a CAGR of 7.5%, shaped by pharmaceutical manufacturing expansion and export compliance alignment. Injectable drug production for regional markets increases inspection needs. Facilities adopt standardized inspection benches to meet international quality expectations. Workforce training considerations favor controlled visual testing setups. Government support for pharmaceutical self-sufficiency sustains capital investment. Demand growth reflects production scaling and compliance alignment rather than replacement of automated inspection infrastructure.
Indonesia is expanding at a CAGR of 7.1%, influenced by growth in domestic injectable manufacturing and hospital supply programs. Public sector procurement increases demand for sterile injectables requiring batch inspection. Manual inspection systems remain prevalent due to cost sensitivity. Quality assurance frameworks emphasize visible contamination control. Incremental facility upgrades sustain steady equipment procurement. Demand growth reflects domestic healthcare supply needs and inspection compliance rather than advanced automation adoption.
Ampoule clarity test apparatus demand in Mexico is growing at a CAGR of 6.4%, supported by export-oriented pharmaceutical manufacturing and regulatory alignment with North American standards. Injectable drug producers require documented visual inspection for batch release. Contract manufacturers serving international clients invest in compliant inspection setups. Stable production volumes sustain predictable procurement cycles. Demand growth reflects export compliance and quality documentation needs rather than rapid expansion of injectable capacity.
Demand for ampoule clarity test apparatus is driven by stringent pharmaceutical quality control requirements, regulatory expectations for parenteral product integrity, and rising production of injectable and biologic therapies. These devices enable inspection of glass and polymer ampoules for particulate matter, glass defects, and solution clarity according to pharmacopeial standards. Buyers evaluate imaging resolution, sensitivity to fine particles, repeatability, ease of integration with inspection lines, and compliance with GMP and regulatory documentation. Procurement teams prioritize suppliers with validated performance data, global service and calibration networks, regulatory readiness, and strong application support for varied container sizes. Trend in the global market reflects expansion of injectable drug portfolios, increased automation of visual inspection processes, and emphasis on minimizing quality deviations during sterile manufacturing.
Körber Pharma (Seidenader) holds leading positioning through advanced clarity test systems and integrated inspection solutions widely adopted by global pharmaceutical manufacturers and contract manufacturing organizations. Syntegon Technology supports demand with comprehensive inspection and test apparatus offerings designed for precision clarity measurement and process line integration. Marchesini Group participates with ampoule inspection equipment and quality test systems that align with stringent pharmaceutical quality standards. OPTIMA Pharma contributes clarity and container inspection solutions integrated within broader packaging and testing platforms for sterile products. Antares Vision Group supplies digital inspection systems and analytics tools that enhance defect detection and documentation for regulatory compliance. Competitive differentiation depends on detection sensitivity, regulatory support, integration flexibility, ease of use, and ability to support high-throughput sterile production environments.
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Apparatus Type | Bench-Top Ampoule Clarity Testers; Rotary or Reciprocating Inspection Units; Integrated Visual Inspection Stations; Other |
| End Use | Sterile Injectables Manufacturing; CDMOs; Hospital Pharmacies; R&D and Pilot Lines |
| Compliance | GMP or GLP Compliant Units; Basic QC Units; Automated Data Logging Units; Other |
| Sales Channel | Pharma Equipment Distributors; Direct Manufacturer Sales; Project EPC or Line Integrators; Other |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, China, Vietnam, Indonesia, Mexico, and 40+ countries |
| Key Companies Profiled | Körber Pharma; Syntegon Technology; Marchesini Group; OPTIMA Pharma; Antares Vision Group; Stevanato Group; Dolphin Pharmacy Instruments; IKON Instruments; LabSoul India; Regional Manufacturers |
| Additional Attributes | Dollar sales by apparatus type and end use; adoption trends for automated and integrated visual inspection supporting higher throughput and regulatory documentation; detection sensitivity for visible particulates, clarity contrast, and inspection speed performance metrics; compliance alignment with pharmacopeial clarity requirements; data integrity, audit trails, and electronic records capabilities; integration with filling, washing, and packaging lines; maintenance intervals, validation effort, and lifecycle cost considerations; procurement dynamics across direct sales, distributors, and EPC-led projects influencing ampoule clarity test apparatus selection. |
How big is the ampoule clarity test apparatus market in 2026?
The global ampoule clarity test apparatus market is estimated to be valued at USD 190.0 million in 2026.
What will be the size of ampoule clarity test apparatus market in 2036?
The market size for the ampoule clarity test apparatus market is projected to reach USD 346.7 million by 2036.
How much will be the ampoule clarity test apparatus market growth between 2026 and 2036?
The ampoule clarity test apparatus market is expected to grow at a 6.2% CAGR between 2026 and 2036.
What are the key product types in the ampoule clarity test apparatus market?
The key product types in ampoule clarity test apparatus market are bench-top ampoule clarity testers, rotary or reciprocating inspection units, integrated visual inspection stations and other.
Which end use segment to contribute significant share in the ampoule clarity test apparatus market in 2026?
In terms of end use, sterile injectables manufacturing segment to command 54.0% share in the ampoule clarity test apparatus market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.