Appendage Management Market
Appendage Management Market Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 to 2023 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2026 to 2035
Appendage Management Market Size and Share Forecast Outlook 2025 to 2035
The Appendage Management Market is estimated to be valued at USD 290.4 million in 2025 and is projected to reach USD 550.2 million by 2035, registering a compound annual growth rate (CAGR) of 6.6% over the forecast period.
Quick Stats for Appendage Management Market
- Appendage Management Market Industry Value (2025): USD 290.4 million
- Appendage Management Market Forecast Value (2035): USD 550.2 million
- Appendage Management Market Forecast CAGR: 6.6%
- Leading Segment in Appendage Management Market in 2025: Endocardial LAA Closure Device (61.4%)
- Key Growth Region in Appendage Management Market: North America, Asia-Pacific, Europe
- Top Key Players in Appendage Management Market: Boston Scientific Corporation, AtriCure, Inc., Abbott Laboratories, Cardia, Inc., Medtronic plc, SentreHEART, Inc., Lifetech Scientific Co., Ltd, Occlutech International AB, Johnson & Johnson Company
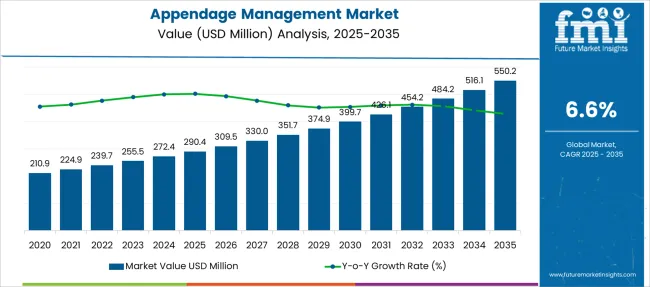
| Metric | Value |
|---|---|
| Appendage Management Market Estimated Value in (2025 E) | USD 290.4 million |
| Appendage Management Market Forecast Value in (2035 F) | USD 550.2 million |
| Forecast CAGR (2025 to 2035) | 6.6% |
Rationale for Segmental Growth in the Appendage Management Market
The appendage management market is expanding steadily due to rising global incidences of atrial fibrillation, increasing procedural volumes for left atrial appendage (LAA) closure, and technological advancements in cardiac rhythm management. Growth is further supported by the shift toward minimally invasive interventions, which is reshaping clinical decision-making in stroke prevention.
Increased awareness among clinicians about embolic stroke risks, along with positive clinical outcomes published in peer-reviewed journals, is influencing adoption. Additionally, aging populations and higher comorbidity rates are prompting hospitals and cardiac centers to invest in specialized appendage management solutions.
Favorable reimbursement frameworks and ongoing innovations in device delivery mechanisms are also reinforcing confidence among interventional cardiologists and electrophysiologists. Looking forward, broader indications, next-generation device miniaturization, and clinical guideline integration are expected to strengthen the market’s clinical and commercial footprint.
Segmental Analysis
The market is segmented by Product Type and End User and region. By Product Type, the market is divided into Endocardial LAA Closure Device and Epicardial LAA Closure Devices. In terms of End User, the market is classified into Hospitals and Cath Labs. Regionally, the market is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the Endocardial LAA Closure Device Product Type Segment:
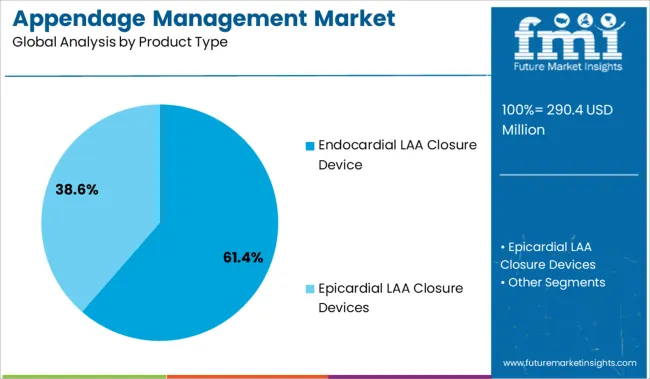
Endocardial LAA closure devices are expected to account for 61.40% of the total market revenue by 2025, making them the leading product type. Their dominance is being driven by favorable safety and efficacy data in high-risk atrial fibrillation patients, as well as ease of percutaneous implantation.
The minimally invasive nature of these devices has reduced the need for open-heart procedures, significantly lowering patient recovery time and procedural risk. Enhanced imaging compatibility with transesophageal echocardiography and fluoroscopy has further improved precision during deployment.
Continuous clinical trial support, regulatory approvals, and device innovations including enhanced seal mechanisms and retrievability have positioned endocardial devices as the preferred choice among interventional cardiologists seeking to reduce long-term stroke risk without permanent anticoagulation.
Insights into the Hospitals End User Segment:
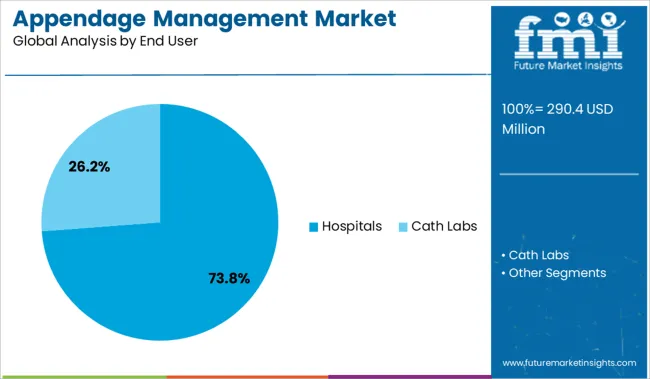
Hospitals are projected to hold 73.80% of the appendage management market share by 2025, solidifying their role as the primary point of care for these procedures. This leadership is being reinforced by the availability of specialized cardiac catheterization labs, experienced electrophysiology teams, and integrated patient care systems that support perioperative and post-procedural management.
Hospitals also serve as the principal centers for early technology adoption, clinical research, and training, making them the preferred location for LAA closure procedures. The presence of reimbursement infrastructure, access to emergency response systems, and high patient throughput enables cost-effective deployment of these interventions.
As procedural volumes increase and device approvals expand, hospitals remain at the forefront of appendage management implementation, particularly for high-risk and comorbid patient groups.
How The Market Progressed Till June 2022?
| Particulars | Details |
|---|---|
| H1, 2024 | 6.68% |
| H1, 2025 Projected | 6.64% |
| H1, 2025 Outlook | 6.54% |
| BPS Change - H1, 2025 (O) - H1, 2025 (P) | (-) 10 ↓ |
| BPS Change - H1, 2025 (O) - H1, 2024 | (-) 14 ↓ |
Future Market Insights presents a comparative analysis about the market growth rates and development prospects in the global appendage management market. According to FMI analysis, the appendage management market is expected to drop by 14 Basis Point Share (BPS) in H1-2025 (O) compared to H1-2024.
Further comparison between the values for H1-2025 outlook and H1-2025 projected showed a dip of 10 BPS. Key reasons for this dip in BPS is attributed to the risks associated with improper implantation of the medical device.
In Asia-Pacific region, the developing countries have moderately updated healthcare infrastructure when compared to those highly developed regions which will indirectly hamper the growth of appendage management market. Also frequent product recalls will tend to restrict the market growth.
Major risks associated with improper implantation include blood clot formation, bleeding, infection and even allergic reaction to the device and irregular heartbeat as well. These factors will impact the overall market growth.
Despite these negative prospects, there are some plus points that stay ahead for the industry including increased adoption rate of closure devices, increasing reimbursement policies and higher adoption of minimally invasive surgeries in non-traditional markets. The market, thus, is expected to show a reduced compounded growth rate in H1-2025 as compared with H1-2024.
2020 to 2025 Appendage management Market Demand Outlook Compared to 2025 to 2035 Forecast
Sales of appendage management market grew at a CAGR of 5.7% between 2013 and 2024.
In 2024, the global market of appendage management accounts for approx. 10.9% of the overall cardiac surgery devices market that accounts around USD 2060.1 Million.
Traditional methods of open heart surgery has various complications associated with the procedure. Implantation of left atrial appendage closure device is done by a non-surgical approach or by causing minimum invasion.
Non-surgical approach, usually helps in faster recovery due to minimally invasive nature. The patient is discharged in a day, thus, reducing the hospital days and aiding better patient outcome.
Decreasing surgical and implantation complications subsequently increases the adoption rate of LAA closure devices, which further contributes in driving the expansion of the appendage management market.
Appendage management procedures are a one-time procedures and has a notable success rate. The complications associated with the surgical procedure of appendage management has been significantly reduced post product approvals. The only unsuccessful surgeries are incomplete or not attempted procedures. This goes to show that there are very few, if any complications associated with the surgical procedure. As a result, the growing adoption of closure devices owing to their success rates drives the growth appendage management market.
The global appendage management market is expected to grow at a CAGR of 6.6% through the forecast period 2025 to 2035.
Which Factors Are Restraining Demand for Appendage management market?
Frequent Product recalls is expected to hamper the growth of market over the forecast period. Product recalls hampers the company image as well as doubts the reliability of the product. This factor is considered as a restraint for the appendage management market.
In February 2020, AtriCure, Inc.’s Epicardial left atrial appendage closure device, AtriClip was recalled.
Such recalls affect the consumer perception of the product, brand image of the company and doubts the quality of the product casing a restraint in appendage management market.
Moreover, some of the threats encountered during the implantation include damage to structures in the heart, dislodging of the device, incomplete closure of the left atrial appendage, bruising, bleeding, infection, and blood clot formation on device, allergic reaction to the device, irregular heartbeat, stroke or even rarely death. Risks associated with improper implantation of the device is other restring factor for market.
Country-wise Insights
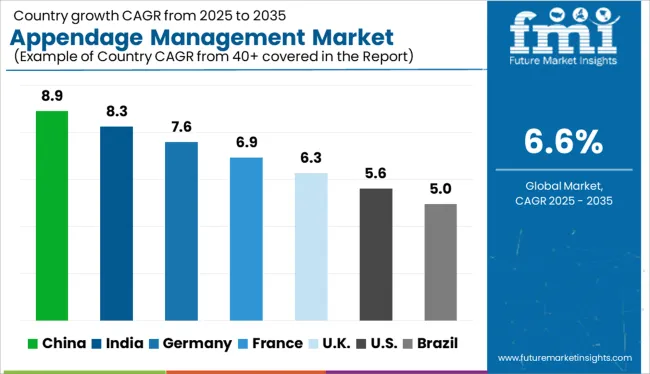
What Makes the USA a Large Market for Appendage management market?
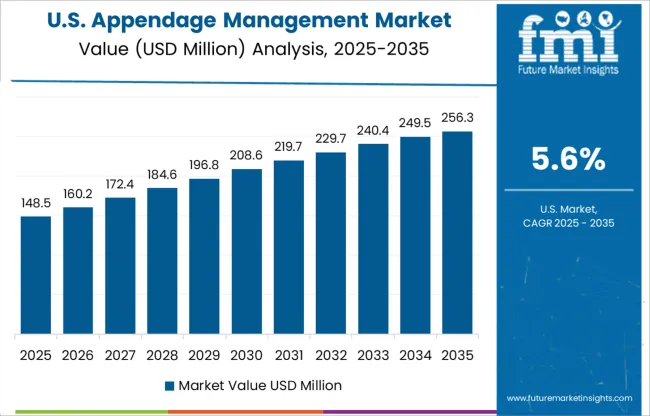
The USA is estimated to account for around 86.3% of the North America appendage management market in 2025 owing to the increasing adoption of left atrial appendage closure devices in attempt to eliminate the risks of side effects of drugs and favorable reimbursement policies which are luring the manufactures in appendage management market. Health insurance in the USA is a reimbursement program by Medicare and Medicaid insurance or a social welfare program funded by the government.
Moreover, focus on new technology development with advancements in medical device industry exhibiting innovative product design with enhanced patient safety is expected to create a huge platform for innovative technologies in USA
Why is Germany Considered a Lucrative in Measurement technology in downstream process market?
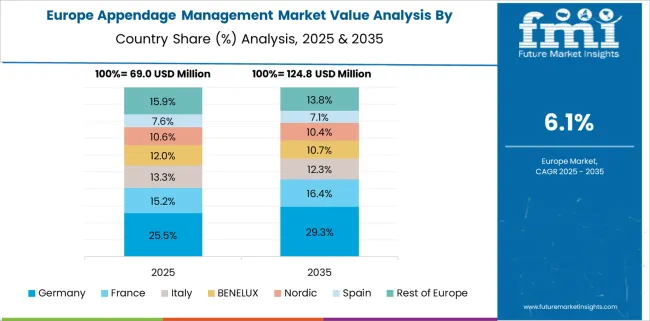
Germany is estimated to account for 20.7% of market revenue share in Europe in 2025. Rising geriatric population who are on risk due to high cholesterol, high blood pressure, obesity, physical inactivity are some of the major factors boosting the demand for appendage management market in Germany.
The risk associated with stroke by atrial fibrillation increases by age. The growing geriatric population is considered an important factor driving the appendage management market.
For instance, According to the World Health Aging 2024 highlights by the United Nations Department of Economic and Social Affairs, in 2024, population aged 65 years and above for Germany was 21.7%. Population ages 65 and above as a percentage of the total population.
As per reported data in 2020 by European Society of Cardiology in Germany, Age group of 75-84 years show a non-valvular atrial fibrillation (NVAF) incidence of 18.6 per 1,000 person-years. The prevalence of NVAF was found to be between 10% and 17% of those aged 80 years or older.
What are the Factors Fueling the Demand for Appendage management market in UK?
UK having huge growth due to technological advances and high healthcare expenditure is expected a growth rate of CAGR 5.3% by 2035.
Additionally, rapid technological advancement, following rise in diseases and the increase in disposable income can boost the demand for appendage management.
Presence of various manufacturers and awareness about cardiovascular disease is predicted to drive the expansion of Europe appendage management market and drive the focus of the manufacturers in these markets.
How will the Appendage management market growth Across Japan?
Demand for appendage management market in Japan has been rising recently, thereby, estimated to contribute nearly 27.2% of market revenue share in the APAC region in 2025.
Developing healthcare infrastructure with improved services and affordability is expected to drive the growth of the market for appendage management in Japan.
Japan appendage management market is lucrative due to rising adoption of LAA closure devices. Key players are determined to establish their company footprints in these emerging markets by increasing distribution channels and partnering with the regional players and distributors. This is expected to drive the demand for appendage management in the Japan.
Category-wise Insights
Demand for Endocardial LAA Closure Device to continue gaining Traction by product type?
Endocardial LAA closure device is expected to account for the highest share of nearly 66.2% in 2025 due to advanced technology with ease of implantation and high specificity, thus, driving the market.
Endocardial LAA closure devices has been widely used and has a good success rate. However, patient are adopting this devices due to it effective product feature. In spite of new innovative products for LAA closure, endocardial LAA closure devices are expected to continue to gain maximum market revenue share over the forecasted years.
Which End User will hold the most of the market share?
By end user, Cath Labs lead the market with market value share of 74.7% in 2025 owing to high walk-in patient in Cath lab for catheterization procedures. More skilled healthcare professionals and technologically advanced services are easily available in Cath lab.
Additionally, appendage management and implantation can be easily performed in Cath lab. Thus this segment is expected to witness significant growth in recent years.
Competitive Landscape
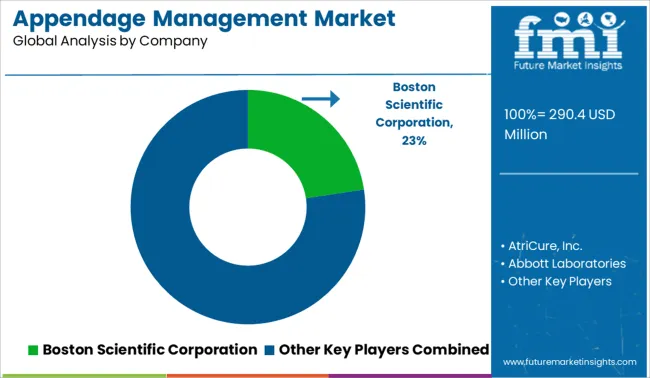
The appendage management market is an extremely consolidated market, with major competitors such as Boston Scientific Corporation, AtriCure, Inc., Abbott Laboratories, Cardia, Inc., Medtronic plc, DePuy Synthes (Johnson & Johnson Services, Inc.), SentreHEART, Inc., Lifetech Scientific Co., Ltd, Occlutech International AB, Johnson & Johnson.
Company operating in every region of the globe. To attract more customers, major players are focusing on mergers and acquisition and launching novel products in order to boost their market position and expand their product portfolios.
- In April 2024, AtriCure, Inc. got USA Food and Drug Administration (FDA) approval of the EPi-Sense® System to treat patients diagnosed with long-standing persistent Afib.
- In Jun 2024, Abbott received European and Canadian approval for amplatzer™ steerable delivery sheath to optimize left atrial appendage closure procedures for people at risk of stroke.
Scope of the Report
| Attribute | Details |
|---|---|
| Forecast Period | 2025 to 2035 |
| Historical Data Available for | 2013 to 2024 |
| Market Analysis | USD Million for Value and Volume in Unit |
| Key Countries Covered | USA, Canada, Brazil, Mexico, Argentina, Germany, UK, France, Spain, Russia, China, Japan, South Korea, India, Australia, New Zealand, GCC Countries, South Africa |
| Key Segments Covered | Product Type, End User and Region |
| Key Companies Profiled | Boston Scientific Corporation; AtriCure, Inc.; Abbott Laboratories; Cardia, Inc.; Medtronic plc; DePuy Synthes (Johnson & Johnson Services, Inc.); SentreHEART, Inc.; Lifetech Scientific Co., Ltd; Occlutech International AB; Johnson & Johnson company |
| Report Coverage | Market Forecast, Competition Intelligence, DROT Analysis, Market Dynamics and Challenges, Strategic Growth Initiatives |
| Customization & Pricing | Available upon Request |
Appendage management market by Category
By Product Type:
- Epicardial LAA Closure Devices
- Endocardial LAA Closure Device
By End User:
- Hospitals
- Cath Labs
By Region:
- North America
- Latin America
- Europe
- APAC
- Middle East & Africa (MEA)
Frequently Asked Questions
How big is the appendage management market in 2025?
The global appendage management market is estimated to be valued at USD 290.4 million in 2025.
What will be the size of appendage management market in 2035?
The market size for the appendage management market is projected to reach USD 550.2 million by 2035.
How much will be the appendage management market growth between 2025 and 2035?
The appendage management market is expected to grow at a 6.6% CAGR between 2025 and 2035.
What are the key product types in the appendage management market?
The key product types in appendage management market are endocardial laa closure device and epicardial laa closure devices.
Which end user segment to contribute significant share in the appendage management market in 2025?
In terms of end user, hospitals segment to command 73.8% share in the appendage management market in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2025 to 2035
- Endocardial LAA Closure Device
- Epicardial LAA Closure Devices
- Y-o-Y Growth Trend Analysis By Product Type , 2020 to 2024
- Absolute $ Opportunity Analysis By Product Type , 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2025 to 2035
- Hospitals
- Cath Labs
- Y-o-Y Growth Trend Analysis By End User, 2020 to 2024
- Absolute $ Opportunity Analysis By End User, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2020 to 2024
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- Key Takeaways
- Latin America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- Key Takeaways
- Western Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- Key Takeaways
- East Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Type
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By End User
- Competition Analysis
- Competition Deep Dive
- Boston Scientific Corporation
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- AtriCure, Inc.
- Abbott Laboratories
- Cardia, Inc.
- Medtronic plc
- SentreHEART, Inc.
- Lifetech Scientific Co., Ltd
- Occlutech International AB
- Johnson & Johnson Company
- Boston Scientific Corporation
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2020-2035
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2020-2035
- Table 3: Global Market Value (USD Million) Forecast by End User, 2020-2035
- Table 4: North America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 5: North America Market Value (USD Million) Forecast by Product Type , 2020-2035
- Table 6: North America Market Value (USD Million) Forecast by End User, 2020-2035
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 8: Latin America Market Value (USD Million) Forecast by Product Type , 2020-2035
- Table 9: Latin America Market Value (USD Million) Forecast by End User, 2020-2035
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 11: Western Europe Market Value (USD Million) Forecast by Product Type , 2020-2035
- Table 12: Western Europe Market Value (USD Million) Forecast by End User, 2020-2035
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2020-2035
- Table 15: Eastern Europe Market Value (USD Million) Forecast by End User, 2020-2035
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2020-2035
- Table 17: East Asia Market Value (USD Million) Forecast by Product Type , 2020-2035
- Table 18: East Asia Market Value (USD Million) Forecast by End User, 2020-2035
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020-2035
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2020-2035
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2020-2035
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2020-2035
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2020-2035
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by End User, 2020-2035
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020-2035
- Figure 3: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 4: Global Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 5: Global Market Attractiveness Analysis by Region
- Figure 6: North America Market Incremental Dollar Opportunity, 2025-2035
- Figure 7: Latin America Market Incremental Dollar Opportunity, 2025-2035
- Figure 8: Western Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 9: Eastern Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 10: East Asia Market Incremental Dollar Opportunity, 2025-2035
- Figure 11: South Asia and Pacific Market Incremental Dollar Opportunity, 2025-2035
- Figure 12: Middle East & Africa Market Incremental Dollar Opportunity, 2025-2035
- Figure 13: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 14: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 15: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 16: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 17: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 18: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 19: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 20: Global Market - Tier Structure Analysis

