ASEAN Cosmetic Chemicals Market
ASEAN Cosmetic Chemicals Market Growth - Trends & Forecast 2026 to 2036
ASEAN Cosmetic Chemicals Market Forecast and Outlook 2026 to 2036
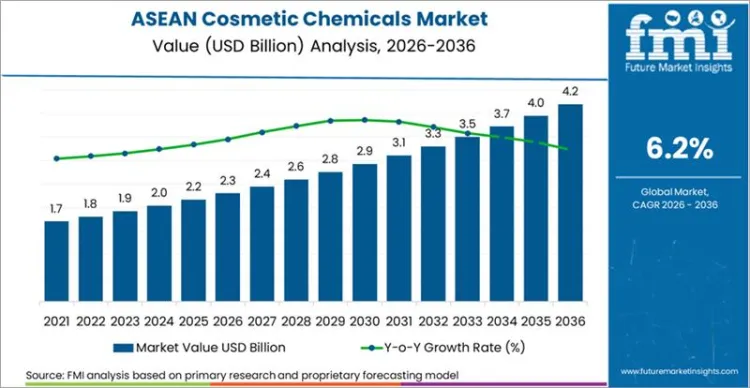
The ASEAN cosmetic chemicals market is projected to grow from USD 2.3 Billion in 2026 to USD 4.1 Billion by 2036, advancing at a 6.2% CAGR. Investment activity by ingredient suppliers is shifting production closer to Southeast Asian formulators, improving lead times and supply continuity for high-volume inputs such as surfactants. BASF announced an expansion of its Alkyl Polyglucosides footprint in Asia with a new plant at its Bangpakong site in Thailand, explicitly linking the investment to growing demand for sustainable surfactants and regional supply agility.
R&D and claim substantiation capability is also being built in-region. Singapore’s Economic Development Board reported that Evonik established the Evonik Skin Institute to meet demand for well substantiated active ingredients and cosmetic solutions, positioning it as a platform for co-creation projects that rely on biological mechanisms, biomarkers, and preclinical or clinical capabilities. Evonik’s Yann d’Hervé stated: ‘We believe that life sciences are completely redefining innovation in cosmetics.’
On the demand side, harmonised regulation reduces technical barriers across ASEAN, but it also raises documentation intensity. Singapore’s Health Sciences Authority describes the ASEAN Cosmetic Directive as an agreement to harmonise requirements and reduce technical barriers to trade, which increases cross-border product flow and, by extension, ingredient pull-through for compliant formulations.
ASEAN Cosmetic Chemicals Market Key Takeaways
| Metric | Value (USD Billion) |
|---|---|
| Market Size (2026) | 2.3 |
| Forecast Value (2036) | 4.1 |
| CAGR (2026 to 2036) | 6.2% |
Summary of the ASEAN Cosmetic Chemicals Market
What is Growth Outlook for ASEAN Cosmetic Chemicals Market as per Future Market Insights Projection?
The market is projected to expand at a 6.2% CAGR, from USD 2.3 Billion in 2026 to USD 4.1 Billion by 2036.
FMI Research Approach: Modeled using finished cosmetics output, regulatory intensity for notifications and dossiers, and regional capacity additions by ingredient suppliers.
Which product type is structurally central to volume growth in ASEAN cosmetic chemicals?
Surfactants lead with a 34.0% share because cleansing and emulsification systems are the largest repeat-volume chemical load across skin and hair formats, and suppliers are adding Asia capacity in surfactant chemistries.
FMI Research Approach: Segment share aligned to typical formulation load factors and disclosed supplier investments in surfactants for personal care.
Which application category anchors chemical demand across ASEAN?
Skin care leads with a 43.0% share, reflecting the category’s high ingredient intensity across emulsions, serums, and active-led claims that require stronger substantiation support.
FMI Research Approach: Share allocation based on product mix, claim substantiation requirements, and active ingredient platform investments.
Which country shows the highest growth rate within ASEAN for cosmetic chemicals?
Vietnam leads at a 6.7% CAGR over 2026 to 2036, supported by formalised cosmetic management rules that govern notification, labeling, safety, and advertising, increasing compliance-led ingredient demand.
FMI Research Approach: Country scoring based on regulatory formalisation, local manufacturing scale-up, and cross-border trade readiness.
Which company holds the largest share among leading suppliers in ASEAN cosmetic chemicals?
BASF leads with a 12.0% share, reinforced by disclosed regional surfactant capacity expansion and broad formulation portfolio coverage.
FMI Research Approach: Competitive weighting based on disclosed capacity, portfolio breadth, and repeated regional investment signals.
What is driving demand for cosmetic chemicals across ASEAN supply chains?
Regulatory harmonisation is expanding cross-border cosmetics throughput, which increases pull-through demand for compliant ingredients and documentation-ready suppliers. Singapore’s Health Sciences Authority describes the ASEAN Cosmetic Directive as a framework to harmonise requirements and reduce technical barriers to trade, which supports wider multi-country launches and higher ingredient volumes per formulation platform. Supply is responding with regionalised capacity. BASF announced an expanded APG surfactant footprint in Thailand, explicitly positioning the investment around faster, more flexible service for customers and growing demand for sustainable surfactants. At the same time, claim substantiation and safety expectations are becoming a procurement gate. Singapore’s Economic Development Board reported Evonik’s establishment of the Evonik Skin Institute to support claim-substantiated cosmetics through biological mechanisms and clinical capabilities, increasing demand for higher-value actives and testing-supported ingredient systems.
How is the ASEAN Cosmetic Chemicals Market Segmented?
The ASEAN cosmetic chemicals market is segmented by product type, application, and country to reflect how ingredient demand is generated and scaled across regional formulation ecosystems. By product type, the market includes surfactants, emollients and film formers, polymers, preservatives, thickening agents, additives, and colorants that form the functional backbone of cosmetic formulations. By application, demand spans skin care, hair care, make-up, oral care, perfumes and fragrances, and other personal care categories, with skin care acting as the primary amplifier due to its higher ingredient load and claim substantiation intensity. By country, adoption is shaped by Thailand, Indonesia, Vietnam, the Philippines, and Malaysia, reflecting differing levels of regulatory formalisation, notification systems, and manufacturing scale-up across ASEAN.
FMI analysis suggests that while ingredient portfolios are expanding in complexity, the market structure remains heavily anchored to high-volume functional chemistries and compliance-ready supply. This segmentation highlights a dual-track market where repeat-volume inputs such as surfactants provide scale and throughput, while application-led complexity, particularly in skin care, drives demand for higher-value ingredients supported by documentation, testing, and regulatory alignment.
Why do surfactants command the largest product type share in ASEAN cosmetic chemicals?
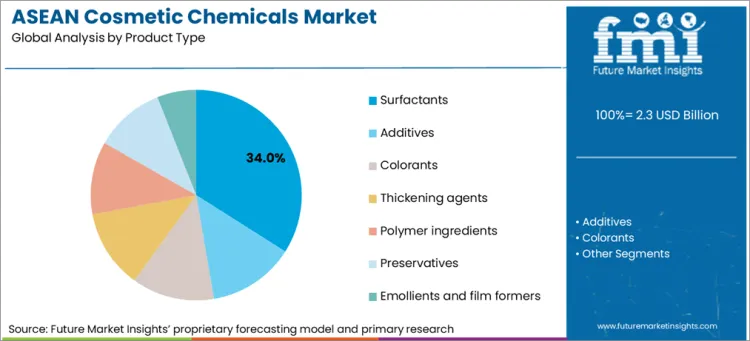
Surfactants lead with a 34.0% share because cleansing and emulsification systems sit at the center of daily-use product volumes and regional supply chains are being rebuilt around local availability. The commercial signal is capacity investment aimed at surfactant chemistries used in personal care formulations. BASF announced the expansion of its Alkyl Polyglucosides footprint in Asia with a new plant at the Bangpakong site in Thailand and tied the move to strengthening regional supply capabilities and meeting growing demand for sustainable surfactants. Mary Kurian, President Care Chemicals, framed the investment as improving agility and flexibility for customers across personal care and adjacent markets. This matters for ASEAN because notification-led compliance and retail launch calendars penalise ingredient shortages and long lead times. Harmonised requirements under the ASEAN Cosmetic Directive reduce technical barriers to trade, which increases the number of multi-country launches that rely on consistent base surfactant systems and validated supplier documentation. The segment’s lead is therefore a function of repeat-volume chemistry, higher execution penalties for stockouts, and supplier capital allocation that is explicitly moving surfactant production closer to ASEAN formulation hubs.
What makes skin care the dominant application driver for cosmetic chemical demand?
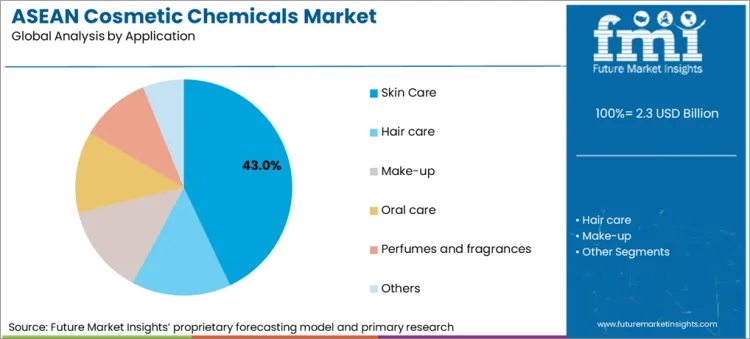
Skin care leads with a 43.0% share because it carries the highest ingredient complexity in mainstream categories, combining emulsions, polymers, preservatives, and actives that require stronger performance substantiation. The structural signal is supplier investment into evaluation, clinical capability, and biology-led claims support, which is disproportionately relevant to skin care positioning. Singapore’s Economic Development Board reported that Evonik established the Evonik Skin Institute to support claim-substantiated cosmetics using biological mechanisms, biomarkers, and preclinical or clinical capabilities, framing it as a stronger customer support platform for science-driven co-creation. This shifts procurement toward ingredient systems that can be defended in dossiers, claims files, and post-market surveillance. At the regulatory layer, the ASEAN Cosmetic Directive aims to harmonise requirements and reduce barriers across ASEAN, which increases the commercial payoff for skin care platforms that can be replicated across multiple national markets with consistent ingredient and label compliance. The category’s dominance is therefore driven by claim intensity, documentation burden, and supplier capability investments that prioritise skin biology validation.
Why Is Regionalised Ingredient Supply Reshaping ASEAN Cosmetic Chemicals?
Regionalisation is fundamentally reshaping the ASEAN cosmetic chemicals market by moving high-volume chemistry closer to formulation hubs, reducing execution risk tied to lead times and supply discontinuity. Large suppliers are reallocating capital toward Southeast Asia to support surfactants and core formulation inputs that sit at the center of daily-use cosmetic volumes. BASF’s expansion of Alkyl Polyglucosides capacity in Thailand illustrates how suppliers are prioritising responsiveness and reliability as competitive advantages. This shift directly addresses the operational penalties imposed by notification-driven launch calendars, where ingredient delays can stall multi-country rollouts. As a result, regionalised supply is no longer optional but a structural requirement for scaling across ASEAN.
How Is Claim Substantiation Infrastructure Changing Procurement Behavior?
The ASEAN cosmetic chemicals market is pivoting from materials-only sourcing toward integrated ingredient systems supported by scientific validation. Investment in regional science infrastructure is increasing demand for higher-value actives, polymers, and formulation systems that can be defended with biological, preclinical, or clinical data. The establishment of in-region platforms dedicated to skin biology and evaluation capabilities signals that claim substantiation is becoming a procurement gate rather than a downstream marketing exercise. This transition shifts purchasing preference toward suppliers that can provide both functional ingredients and the data packages required to support dossiers, claims files, and post-market scrutiny.
What Role Does Regulatory Tightening Play as a Market Restraint?
Tightening notification, labeling, and dossier expectations across ASEAN markets are creating compliance friction that raises cost-to-serve and slows portfolio agility, particularly for smaller suppliers. National frameworks such as Thailand’s Cosmetics Act B.E. 2558 (2015) formalise regulatory control and increase the value of consistent specifications, stability data, and documentation support. Malaysia’s control guidelines, adapted from the ASEAN Cosmetic Directive, further reinforce post-market surveillance and documentation intensity. This environment concentrates demand toward suppliers capable of sustaining compliance across multiple jurisdictions, limiting experimentation while reinforcing scale advantages for documentation-ready ingredient providers.
ASEAN Cosmetic Chemicals Market Analysis by Key Countries
| Country | CAGR (2026 to 2036) |
|---|---|
| Thailand | 6.4% |
| Indonesia | 6.6% |
| Vietnam | 6.7% |
| Philippines | 6.1% |
| Malaysia | 5.9% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research.
How does Thailand’s legal control framework translate into sustained ingredient demand growth?
Thailand is projected to grow at a 6.4% CAGR from 2026 to 2036 because its Cosmetics Act B.E. 2558 (2015) formalises market controls and raises the compliance value of ingredient suppliers with stable quality and documentation. The Act establishes the legal basis for rules, orders, and announcements governing cosmetic oversight, which increases the need for consistent formulations and compliant raw materials as brands scale portfolios. Supply localisation reinforces the mechanism. BASF announced an expansion of its APG surfactant footprint with a new plant at Bangpakong, Thailand, explicitly framing it around strengthening regional supply and faster service for customers. Together, tighter regulatory perimeter plus localised surfactant capacity increases the probability that Thailand becomes a higher-throughput formulation base, sustaining pull-through for surfactants, preservatives, polymers, and emollients over the forecast window.
Why does Indonesia’s system modernisation lift demand for compliant cosmetic chemicals?
Indonesia will expand at a 6.6% CAGR from 2026 to 2036 because notification system changes are shifting compliance from paper-based processes toward digital access and file governance, increasing the value of dossier-ready ingredient supply. The WTO TBT trade concerns portal notes that Indonesia implemented regulations where BPOM would start using a new cosmetics certification system from August 19, 2024, signalling procedural change that affects market access execution. The USA International Trade Administration reported that BPOM introduced version 3.0 of its cosmetics notification application on July 12, 2024, changing how product information files are managed through digital access requirements. This increases demand for ingredient suppliers that can support traceable specifications, safety data, and consistent documentation for fast notifications and renewals, particularly for surfactant systems and preservatives that sit under higher scrutiny.
What is structurally lifting Vietnam to the highest growth rate within ASEAN?
Vietnam’s 6.7% CAGR is supported by formalised cosmetic management rules that standardise how products are announced, documented, labeled, and advertised, which increases compliance-driven ingredient demand and improves repeatability of launches. Vietnam’s trade portal describes the cosmetic management framework under Circular 06/2011/TT-BYT, covering product announcement, product information dossiers, safety, labeling, and advertising. This framework raises the operating premium on suppliers that can support INCI-aligned ingredient documentation and formulation stability requirements across skin care and hair care. As brands broaden assortments and increase frequency of product notifications, ingredient pull-through grows most in high-intensity segments such as surfactants and skin-care actives where documentation and performance claims are central to approvals and commercial success.
How do Philippines ASEAN directive updates influence chemical demand through compliance cycles?
The Philippines is projected to grow at a 6.1% CAGR because ASEAN directive updates translated into national circulars create recurring compliance cycles that drive reformulation, relabeling, and ingredient requalification demand. The Philippines FDA published Circular No. 2023-011 on updates and amendments to the ASEAN Cosmetic Directive adopted during ASEAN Cosmetic Committee meetings, explicitly linking domestic implementation to ASEAN-level changes. These updates increase the need for ingredient suppliers that can provide updated documentation and support reformulation when restricted substances or labeling requirements change. In practice, this lifts demand for compliant preservatives, surfactants, and additives that can be defended in product information files while maintaining performance in skin and hair formats.
Why does Malaysia’s compliance and surveillance posture shape a steadier growth curve?
Malaysia’s 5.9% CAGR reflects a mature compliance posture that prioritises control and post-market surveillance, which supports steady ingredient demand but increases barriers for lower-capability suppliers. Malaysia’s National Pharmaceutical Regulatory Agency guideline states it is adapted from the ASEAN Cosmetic Directive and is designed to be read alongside national laws governing cosmetics, framing a structured control environment. This pushes procurement toward suppliers that can support stability, specification discipline, and documentation alignment with ASEAN expectations, sustaining demand for core functional chemicals such as surfactants, thickeners, and film formers. The trade-off is slower experimentation for smaller brands due to compliance overhead, which moderates growth versus faster-transition markets.
How is competition evolving in the cosmetic chemicals market, and what does it imply for ASEAN?
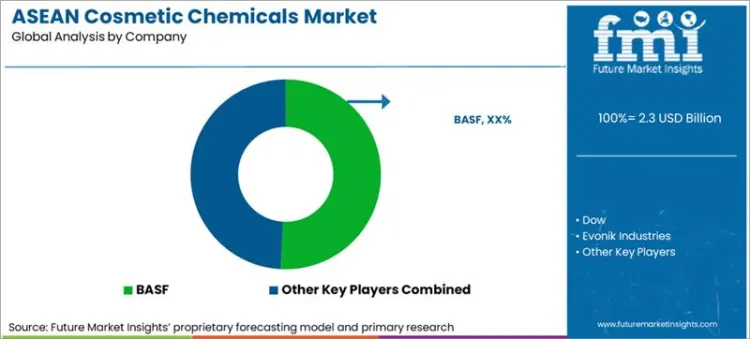
Competition is led by integrated chemical groups that can supply high-volume surfactants, polymers, and preservatives while also supporting regulatory documentation and claims substantiation. Scope includes emollients and film formers, surfactants, additives, colorants, thickening agents, polymers, and preservatives sold into cosmetics manufacturing. Scope excludes finished cosmetics, packaging materials, equipment, and pharmaceutical active ingredients used primarily in drugs. BASF is the largest named supplier in the ASEAN competitive set and is reinforcing leadership through disclosed regional surfactant capacity expansion in Thailand, positioned around reliability and faster service for personal care customers. In North America, leadership is influenced by multinational scale and tight retailer specifications. In Europe, leadership is reinforced by integrated specialty chemicals portfolios and regulatory discipline. In Asia, global leadership does not automatically translate into Japan leadership because Japan’s Ministry of Health, Labour and Welfare sets Standards for Cosmetics, including ingredient restrictions under a national framework, shaping different compliance economics versus ASEAN. ASEAN competition therefore rewards suppliers that localise supply, deliver compliant data packages, and support multi-country launches under the ASEAN Cosmetic Directive framework.
Recent Developments
- In November 2025, BASF announced a new Alkyl Polyglucosides production plant at Bangpakong, Thailand, expanding its Asia footprint to strengthen regional supply, improve customer agility, and support growing demand for sustainable, bio-based surfactants across personal, home, and industrial care sectors markets.
- In March 2024, Evonik launched the Evonik Skin Institute, uniting biotech platforms and global evaluation labs to support science-driven, claim-substantiated cosmetic solutions, strengthen customer collaboration, and expand its biosolutions portfolio, building on decades of expertise in active and cosmetic ingredients.
Key Players
- BASF
- Dow
- Evonik Industries
- Clariant
- Croda International
- Ashland
- DSM-Firmenich
- Lubrizol
- Symrise
- SEPPIC (Air Liquide)
Market Definition
ASEAN cosmetic chemicals are formulation inputs sold to cosmetics manufacturers in ASEAN, including surfactants, emollients and film formers, preservatives, polymers, thickeners, additives, colorants, and specialty actives used in skin care, hair care, make-up, oral care, perfumes and fragrances, and other cosmetic applications. The market tracks B2B sales of chemical ingredients used to create compliant cosmetic products under ASEAN-aligned regulatory expectations. The ASEAN Cosmetic Directive is treated as the governing regional framework that harmonises requirements and reduces technical barriers, shaping supplier documentation needs and multi-country platform launches.
Market Inclusion
Included revenues cover ingredients and blends sold for use in cosmetic formulations across ASEAN, including surfactants, emollients, rheology modifiers, polymers, preservatives, additives, and colorants supplied into skin care and hair care as the largest chemical load applications. Included are materials supplied for product information files and compliance documentation aligned to national rules derived from ASEAN harmonisation. Malaysia’s NPRA guideline explicitly states it is adapted from the ASEAN Cosmetic Directive, reinforcing inclusion of compliance-ready ingredient supply into the market scope. BASF is treated as the largest named supplier in this ASEAN set based on provided share and its disclosed surfactant capacity expansion in Thailand.
Market Exclusion
Excluded are finished cosmetic products sold to consumers, contract manufacturing fees that do not separate ingredient value, packaging materials, capital equipment, and pharmaceutical actives used primarily in medicines. Also excluded are ingredients sold primarily for detergents or industrial cleaning unless explicitly supplied into cosmetic-grade personal care applications. Leadership does not translate uniformly across regions. Japan is explicitly treated as a market where national ingredient standards and restrictions set by the Ministry of Health, Labour and Welfare shape a distinct compliance pathway and supplier mix versus ASEAN harmonised rules.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD 2.3 Billion |
| Product Type Segments | Surfactants; Emollients & Film Formers; Polymers; Preservatives; Thickening Agents; Additives; Colorants |
| Application Categories | Skin Care; Hair Care; Make-Up; Oral Care; Perfumes & Fragrances; Other Personal Care |
| Value Chain Position | Cosmetic Formulation Ingredients (B2B Chemical Inputs) |
| Distribution / Supply Structure | Direct Supply to Cosmetic Manufacturers; Regional Distributors; Contract Supply for Multinational Formulators |
| Regions Covered | Southeast Asia (ASEAN) |
| Key Countries | Thailand, Indonesia, Vietnam, Philippines, Malaysia |
| Key Companies Profiled | BASF; Dow; Evonik Industries; Clariant; Croda International; Ashland; DSM-Firmenich; Lubrizol; Symrise; SEPPIC (Air Liquide) |
| Regulatory Framework Reference | ASEAN Cosmetic Directive (ACD) with country-level implementation (Thailand FDA, Indonesia BPOM, Vietnam MOH, Philippines FDA, Malaysia NPRA) |
| Additional Attributes | Dollar sales measured for cosmetic formulation chemicals supplied into ASEAN manufacturing; demand driven by notification-led regulatory harmonisation under ACD; surfactant-led volume scaling supported by regional capacity expansion; rising importance of dossier-ready and documentation-compliant ingredient supply; claim-substantiation infrastructure influencing procurement of actives and polymers; regionalisation of supply chains to reduce lead times and execution risk; competitive positioning based on portfolio breadth, Asia-based production footprint, and regulatory support capability |
ASEAN Cosmetic Chemicals Market by Key Segments
By Product Type
- Emollients and film formers
- Surfactants
- Additives
- Colorants
- Thickening agents
- Polymer ingredients
- Preservatives
By Application Type
- Skin care
- Hair care
- Make-up
- Oral care
- Perfumes and fragrances
- Others
Bibliography
- BASF. (2025). BASF inaugurates the expansion of production capacity for Alkyl Polyglucosides (APGs) in Bangpakong, Thailand [Press release].
- Economic Development Board, Singapore. (2024). Evonik establishes a new institute dedicated to skin sciences: The Evonik Skin Institute.
- Food and Drug Administration, Philippines. (2023). FDA Circular No. 2023-011: Updates and amendments to the ASEAN Cosmetic Directive (ACD).
- Ministry of Health, Labour and Welfare, Japan. (2000). Standards for cosmetics (Notification No. 331 of 2000, provisional translation).
- National Pharmaceutical Regulatory Agency (NPRA), Malaysia. (2022, August 1). Guidelines for control of cosmetic products in Malaysia.
Frequently Asked Questions
What is the ASEAN Cosmetic Chemicals Market size in 2026 and 2036, and what is the CAGR?
The ASEAN cosmetic chemicals market is valued at USD 2.3 billion in 2026 and is projected to reach USD 4.1 billion by 2036, expanding at a 6.2% CAGR.
Which product type anchors volume growth in the ASEAN Cosmetic Chemicals Market in 2026?
Surfactants lead with a 34.0% share in 2026 because cleansing and emulsification systems represent the highest repeat-volume chemical load across skin and hair formulations.
Which application segment drives the highest demand for cosmetic chemicals across ASEAN?
Skin care dominates with a 43.0% share in 2026 due to its higher ingredient complexity and stronger claim substantiation requirements.
Which ASEAN country is expected to grow the fastest in the cosmetic chemicals market through 2036?
Vietnam leads growth at a 6.7% CAGR from 2026 to 2036 as formalised cosmetic management rules increase compliance-led ingredient demand.
Which supplier holds the largest share in the ASEAN Cosmetic Chemicals Market?
BASF holds the largest share at 12.0%, supported by regional surfactant capacity expansion and broad formulation portfolio coverage.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual Reports and Integrated Reports
- Peer-reviewed Medical and Clinical Journals
- Corporate Websites and Product Technical Literature
- Investor Presentations and Earnings Communications
- Regulatory Filings and Medical Device Standards
- Clinical Guidelines and Public Health Publications
- Trade Journals and Wound Care Industry Magazines
- Conference Proceedings and Clinical Congress Materials
- Government Health Statistics and Surveillance Data
- Press Releases and Verified Media Coverage
- Specialist Healthcare Newsletters
- Sector Databases and Medical Device Repositories
- FMI Internal Knowledge Base
- Subscription-based Healthcare Databases
- Hospital Procurement and Tendering Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews
- Quantitative Surveys
- Hybrid Validation Approach
- Why Primary Evidence is Used
- Field Techniques
- Hospital and Clinic Interviews
- Procurement and Formulary Surveys
- Wound Care Specialist Workshops
- Care Pathway Observations
- Policy and Payment Reform Consultations
- Stakeholder Universe Engaged
- C-suite Executives
- Hospital Procurement Heads
- Clinical Directors and Wound Care Specialists
- Quality and Infection Control Leads
- Regulatory and Compliance Officers
- Home Care and Post-acute Care Coordinators
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Healthcare Utilisation and Treatment Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Billion) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Product Type, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Product Type, 2026 to 2036
- Y to o to Y Growth Trend Analysis By Product Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Y to o to Y Growth Trend Analysis By Application
- Absolute $ Opportunity Analysis By Application
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Billion) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Billion) Analysis and Forecast By Region, 2026 to 2036
- ASEAN
- Market Attractiveness Analysis By Region
- Key Countries Market Analysis
- Thailand
- Indonesia
- Vietnam
- Philippines
- Malaysia
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Product Type
- By Application
- By Country
- Competition Analysis
- Competition Deep Dive
- Recent Developments
- Key Players
- Scope Controls
- Market Definition
- Market Inclusion
- Market Exclusion
- Scope of Report
- ASEAN Cosmetic Chemicals Market by Key Segments
- By Product Type
- By Application Type
- Bibliography
- Frequently Asked Questions
- Assumptions & Acronyms Used
List of Tables
- ASEAN Cosmetic Chemicals Market Key Takeaways
- Global ASEAN Cosmetic Chemicals Market Size, 2021 to 2036
- ASEAN Cosmetic Chemicals Market Size by Product Type
- ASEAN Cosmetic Chemicals Market Size by Application Type
- ASEAN Cosmetic Chemicals Market Size by Region
- ASEAN Cosmetic Chemicals Market CAGR by Country
- Scope of Report
List of Figures
- ASEAN Cosmetic Chemicals Market Growth Trajectory, 2026 to 2036
- ASEAN Cosmetic Directive Compliance and Dossier Readiness Framework
- Product Type Share Analysis
- Application Mix Analysis
- ASEAN Country CAGR Comparison
- Regional Market Attractiveness Analysis
- Competitive Landscape Overview
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

