Aseptic Formulation Processing Market
Aseptic Formulation Processing Market Size and Share Forecast Outlook 2025 to 2035
Aseptic Formulation Processing Market Size and Share Forecast Outlook 2025 to 2035
The Aseptic Formulation Processing Market is estimated to be valued at USD 10.5 billion in 2025 and is projected to reach USD 20.3 billion by 2035, registering a compound annual growth rate (CAGR) of 6.8% over the forecast period.
Quick Stats for Aseptic Formulation Processing Market
- Aseptic Formulation Processing Market Value (2025): USD 10.5 billion
- Aseptic Formulation Processing Market Forecast Value (2035): USD 20.3 billion
- Aseptic Formulation Processing Market Forecast CAGR: 6.8%
- Leading Segment in Aseptic Formulation Processing Market in 2025: Automated (45.0%)
- Key Growth Regions in Aseptic Formulation Processing Market: North America, Asia-Pacific, Europe
- Top Key Players in Aseptic Formulation Processing Market: Robert Bosch GmbH, Becton Dickinson and Company, Tetra Laval International S.A., E.I. du Pont de Nemours and Company, Sealed Air Corporation, GEA Group Aktiengesellschaft, Alfa Laval AB, Krones AG
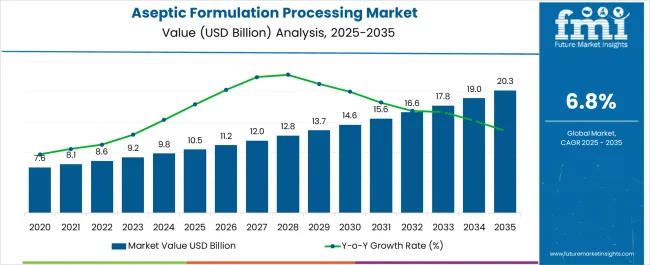
Aseptic Formulation Processing Market Key Takeaways
| Metric | Value |
|---|---|
| Aseptic Formulation Processing Market Estimated Value in (2025 E) | USD 10.5 billion |
| Aseptic Formulation Processing Market Forecast Value in (2035 F) | USD 20.3 billion |
| Forecast CAGR (2025 to 2035) | 6.8% |
Why is the Aseptic Formulation Processing Market Growing?
The Aseptic Formulation Processing market is witnessing substantial growth due to increasing demand for sterile pharmaceutical products and vaccines, driven by heightened awareness of safety and quality standards. The market is being shaped by technological advancements in automated systems, which enable consistent sterile conditions, higher throughput, and reduced human intervention, minimizing contamination risks. Rising investments in pharmaceutical infrastructure, the growing prevalence of chronic diseases, and the expansion of biologics and vaccines are contributing to market growth.
The integration of process monitoring, real-time analytics, and advanced control systems is supporting enhanced productivity and regulatory compliance. Increasing regulatory scrutiny on contamination control and stringent quality standards is pushing manufacturers toward adopting automated aseptic processing technologies.
Additionally, the need for faster product development cycles, particularly in biologics and injectable therapeutics, has created opportunities for scalable, flexible processing platforms The market is expected to continue its upward trajectory as pharmaceutical companies seek efficient, compliant, and high-quality aseptic production solutions to meet growing global demand.
Segmental Analysis
The aseptic formulation processing market is segmented by technology, product type, end user, and geographic regions. By technology, aseptic formulation processing market is divided into Automated, Semi-Automated, and Manual. In terms of product type, aseptic formulation processing market is classified into Laminar Air Flow Systems, Autoclave Devices, Dry Heat Ovens, UV- Radiation Systems, Aseptic Filling Devices, Open, Barrier, Isolators, and RABS (Restricted Access Barrier Systems). Based on end user, aseptic formulation processing market is segmented into Pharmaceutical Companies, Biotech Companies, Research Laboratories, and Others. Regionally, the aseptic formulation processing industry is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the Automated Technology Segment
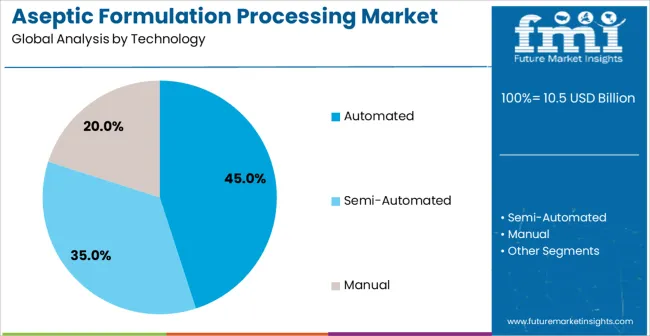
The Automated technology segment is projected to hold 45.0% of the Aseptic Formulation Processing market revenue share in 2025, making it the leading technology. This position is being attributed to the superior capability of automated systems to maintain consistent sterile conditions and reduce manual errors, which is critical for regulatory compliance and product safety.
Automation allows for real-time monitoring, precise control of environmental parameters, and efficient handling of sensitive formulations, which has driven adoption in high-volume pharmaceutical production facilities. The ability to integrate with advanced software systems for process optimization and documentation further enhances operational efficiency.
Pharmaceutical companies are increasingly favoring automated systems to meet stringent quality standards, reduce contamination risks, and improve productivity, making this segment a dominant contributor to market growth Future expansion is expected as manufacturers continue to upgrade existing facilities with advanced automation solutions to achieve higher throughput and regulatory adherence.
Insights into the Laminar Air Flow Systems Product Type Segment
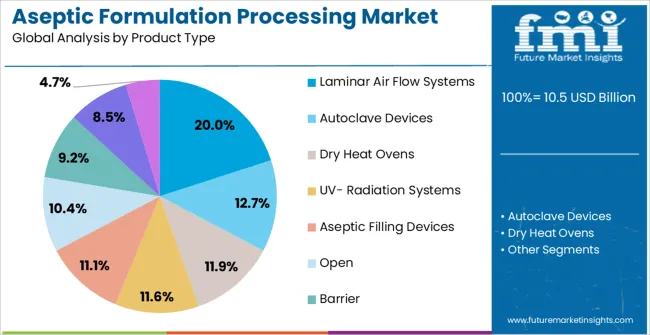
Laminar Air Flow Systems are projected to account for 20.0% of the overall Aseptic Formulation Processing market revenue share in 2025, establishing them as the leading product type. This prominence is being driven by their ability to provide a controlled sterile environment, which is critical for aseptic filling and formulation processes.
These systems ensure unidirectional airflow, reducing contamination risks and supporting compliance with regulatory standards in pharmaceutical manufacturing. The adoption of laminar airflow systems has been accelerated by the increasing production of injectable drugs, vaccines, and biologics that require stringent sterility.
The segment benefits from technological advancements that enhance airflow uniformity, energy efficiency, and integration with automation for real-time monitoring As pharmaceutical production scales up globally, the demand for reliable and efficient laminar air flow systems is expected to remain strong, supporting the segment’s leading position in the market.
Insights into the Pharmaceutical Companies End User Segment
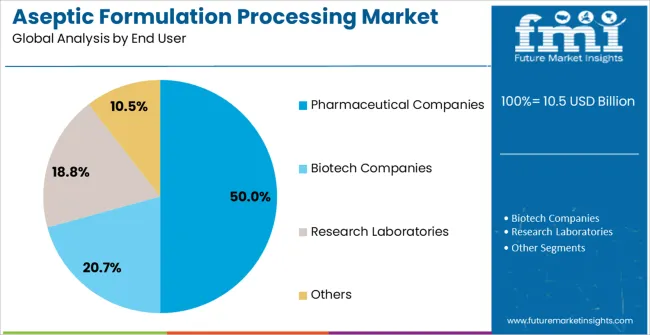
The Pharmaceutical Companies end-use industry segment is expected to hold 50.0% of the Aseptic Formulation Processing market revenue share in 2025, making it the largest end-user segment. This dominance is being driven by the growing need for sterile drug production, particularly in injectable therapies, biologics, and vaccines.
Pharmaceutical companies are increasingly adopting advanced aseptic processing technologies to ensure regulatory compliance, enhance product safety, and improve production efficiency. Investments in expanding manufacturing capacities, modernization of facilities, and integration of automation are enabling higher throughput and consistent product quality.
The segment’s growth is also supported by the rising prevalence of chronic diseases, growing demand for biologics, and global vaccination programs, which require large-scale sterile production As pharmaceutical companies prioritize cost-effective, scalable, and compliant aseptic solutions, the adoption of automated technologies and laminar air flow systems is expected to drive sustained growth in this segment.
Market Overview
Aseptic Formulation Processing Market Introduction
Aseptic formulation processing is the manipulation of sterile active pharmaceutical ingredient (APIs) or pharmaceutical product in a carefully controlled environment using aseptic technique in order to produce a sterile end product.
Aseptic formulation processing is a risk management program that ensures the pharmaceutical product's safety and integrity by confirming the transfer of formulation ingredients with high potency without any chance of contamination in the pre-sterilized vials or containers.
Maintaining a controlled environment is vital in aseptic formulation processing as the quality of the final product and sterility of the manufacturing procedure is maintained due to the sterile environment. The environmental conditions required for aseptic processing is maintained by sterile air supply through laminar air flow machines in the aseptic formulation processing facilities.
During the process of packaging of the final product, technical measures should be taken such as handling sterile materials in a controlled environment in order to control microbial and particulate contamination to acceptable levels.
This process is done manually or with the help of automated aseptic filling devices. However, for improving the aseptic formulation processing procedures in the pharmaceutical industry, the Parenteral Drug Association (PDA) published its aseptic validation technical report in 1981. After that several regulatory authorities published guidelines for aseptic processing of pharmaceutical products.
Analysis of Aseptic Formulation Processing Market By Key Countries

| Country | CAGR |
|---|---|
| China | 9.2% |
| India | 8.5% |
| Germany | 7.8% |
| Brazil | 7.1% |
| USA | 6.5% |
| UK | 5.8% |
| Japan | 5.1% |
Country-wise Analysis
The Aseptic Formulation Processing Market is expected to register a CAGR of 6.8% during the forecast period, exhibiting varied country level momentum. China leads with the highest CAGR of 9.2%, followed by India at 8.5%. Developed markets such as Germany, France, and the UK continue to expand steadily, while the USA is likely to grow at consistent rates. Japan posts the lowest CAGR at 5.1%, yet still underscores a broadly positive trajectory for the global Aseptic Formulation Processing Market. In 2024, Germany held a dominant revenue in the Western Europe market and is expected to grow with a CAGR of 7.8%. The USA Aseptic Formulation Processing Market is estimated to be valued at USD 3.6 billion in 2025 and is anticipated to reach a valuation of USD 3.6 billion by 2035. Sales are projected to rise at a CAGR of 0.0% over the forecast period between 2025 and 2035. While Japan and South Korea markets are estimated to be valued at USD 484.6 million and USD 307.0 million respectively in 2025.
Key Players in the Aseptic Formulation Processing Market

- Robert Bosch GmbH
- Becton Dickinson and Company
- Tetra Laval International S.A.
- E.I. du Pont de Nemours and Company
- Sealed Air Corporation
- GEA Group Aktiengesellschaft
- Alfa Laval AB
- Krones AG
Scope of the Report
| Item | Value |
|---|---|
| Quantitative Units | USD 10.5 Billion |
| Technology | Automated, Semi-Automated, and Manual |
| Product Type | Laminar Air Flow Systems, Autoclave Devices, Dry Heat Ovens, UV- Radiation Systems, Aseptic Filling Devices, Open, Barrier, Isolators, and RABS (Restricted Access Barrier Systems) |
| End User | Pharmaceutical Companies, Biotech Companies, Research Laboratories, and Others |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, Middle East & Africa |
| Country Covered | United States, Canada, Germany, France, United Kingdom, China, Japan, India, Brazil, South Africa |
| Key Companies Profiled | Robert Bosch GmbH, Becton Dickinson and Company, Tetra Laval International S.A., E.I. du Pont de Nemours and Company, Sealed Air Corporation, GEA Group Aktiengesellschaft, Alfa Laval AB, and Krones AG |
Aseptic Formulation Processing Market by Segments
Technology:
- Automated
- Semi-Automated
- Manual
Product Type:
- Laminar Air Flow Systems
- Autoclave Devices
- Dry Heat Ovens
- UV- Radiation Systems
- Aseptic Filling Devices
- Open
- Barrier
- Isolators
- RABS (Restricted Access Barrier Systems)
End User:
- Pharmaceutical Companies
- Biotech Companies
- Research Laboratories
- Others
Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Frequently Asked Questions
How big is the aseptic formulation processing market in 2025?
The global aseptic formulation processing market is estimated to be valued at USD 10.5 billion in 2025.
What will be the size of aseptic formulation processing market in 2035?
The market size for the aseptic formulation processing market is projected to reach USD 20.3 billion by 2035.
How much will be the aseptic formulation processing market growth between 2025 and 2035?
The aseptic formulation processing market is expected to grow at a 6.8% CAGR between 2025 and 2035.
What are the key product types in the aseptic formulation processing market?
The key product types in aseptic formulation processing market are automated, semi-automated and manual.
Which product type segment to contribute significant share in the aseptic formulation processing market in 2025?
In terms of product type, laminar air flow systems segment to command 20.0% share in the aseptic formulation processing market in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Aseptic Formulation Processing Market Analysis 2020-2024 and Forecast, 2025-2035
- Historical Market Size Value (USD Mn) Analysis, 2020-2024
- Current and Future Market Size Value (USD Mn) Projections, 2025-2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Aseptic Formulation Processing Market Pricing Analysis 2020-2024 and Forecast 2025-2035
- Global Aseptic Formulation Processing Market Analysis 2020-2024 and Forecast 2025-2035, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Mn) Analysis By Technology , 2020-2024
- Current and Future Market Size Value (USD Mn) Analysis and Forecast By Technology , 2025-2035
- Automated
- Semi-Automated
- Manual
- Y-o-Y Growth Trend Analysis By Technology , 2020-2024
- Absolute $ Opportunity Analysis By Technology , 2025-2035
- Global Aseptic Formulation Processing Market Analysis 2020-2024 and Forecast 2025-2035, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Mn) Analysis By Product Type, 2020-2024
- Current and Future Market Size Value (USD Mn) Analysis and Forecast By Product Type, 2025-2035
- Laminar Air Flow Systems
- Autoclave Devices
- Dry Heat Ovens
- UV- Radiation Systems
- Aseptic Filling Devices
- Open
- Barrier
- Isolators
- RABS (Restricted Access Barrier Systems)
- Y-o-Y Growth Trend Analysis By Product Type, 2020-2024
- Absolute $ Opportunity Analysis By Product Type, 2025-2035
- Global Aseptic Formulation Processing Market Analysis 2020-2024 and Forecast 2025-2035, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Mn) Analysis By End User, 2020-2024
- Current and Future Market Size Value (USD Mn) Analysis and Forecast By End User, 2025-2035
- Pharmaceutical Companies
- Biotech Companies
- Research Laboratories
- Others
- Y-o-Y Growth Trend Analysis By End User, 2020-2024
- Absolute $ Opportunity Analysis By End User, 2025-2035
- Global Aseptic Formulation Processing Market Analysis 2020-2024 and Forecast 2025-2035, By Region
- Introduction
- Historical Market Size Value (USD Mn) Analysis By Region, 2020-2024
- Current Market Size Value (USD Mn) Analysis and Forecast By Region, 2025-2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Aseptic Formulation Processing Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) Forecast By Market Taxonomy, 2025-2035
- By Country
- USA
- Canada
- Mexico
- By Technology
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Technology
- By Product Type
- By End User
- Key Takeaways
- Latin America Aseptic Formulation Processing Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) Forecast By Market Taxonomy, 2025-2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Technology
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Technology
- By Product Type
- By End User
- Key Takeaways
- Western Europe Aseptic Formulation Processing Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) Forecast By Market Taxonomy, 2025-2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Technology
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Technology
- By Product Type
- By End User
- Key Takeaways
- Eastern Europe Aseptic Formulation Processing Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) Forecast By Market Taxonomy, 2025-2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Technology
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Technology
- By Product Type
- By End User
- Key Takeaways
- East Asia Aseptic Formulation Processing Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) Forecast By Market Taxonomy, 2025-2035
- By Country
- China
- Japan
- South Korea
- By Technology
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Technology
- By Product Type
- By End User
- Key Takeaways
- South Asia and Pacific Aseptic Formulation Processing Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) Forecast By Market Taxonomy, 2025-2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Technology
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Technology
- By Product Type
- By End User
- Key Takeaways
- Middle East & Africa Aseptic Formulation Processing Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) Forecast By Market Taxonomy, 2025-2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Technology
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Technology
- By Product Type
- By End User
- Key Takeaways
- Key Countries Aseptic Formulation Processing Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By Technology
- By Product Type
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Technology
- By Product Type
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Technology
- By Product Type
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Technology
- By Product Type
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Technology
- By Product Type
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Technology
- By Product Type
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By Technology
- By Product Type
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Technology
- By Product Type
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Technology
- By Product Type
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Technology
- By Product Type
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Technology
- By Product Type
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Technology
- By Product Type
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Technology
- By Product Type
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Technology
- By Product Type
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Technology
- By Product Type
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Technology
- By Product Type
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Technology
- By Product Type
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Technology
- By Product Type
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Technology
- By Product Type
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Technology
- By Product Type
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Technology
- By Product Type
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Technology
- By Product Type
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Technology
- By Product Type
- By End User
- Competition Analysis
- Competition Deep Dive
- Robert Bosch GmbH
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Becton Dickinson and Company
- Tetra Laval International S.A.
- E.I. du Pont de Nemours and Company
- Sealed Air Corporation
- GEA Group Aktiengesellschaft
- Alfa Laval AB
- Krones AG
- Robert Bosch GmbH
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Aseptic Formulation Processing Market Value (USD Mn) Forecast by Region, 2020-2035
- Table 2: Global Aseptic Formulation Processing Market Value (USD Mn) Forecast by Technology , 2020-2035
- Table 3: Global Aseptic Formulation Processing Market Value (USD Mn) Forecast by Product Type, 2020-2035
- Table 4: Global Aseptic Formulation Processing Market Value (USD Mn) Forecast by End User, 2020-2035
- Table 5: North America Aseptic Formulation Processing Market Value (USD Mn) Forecast by Country, 2020-2035
- Table 6: North America Aseptic Formulation Processing Market Value (USD Mn) Forecast by Technology , 2020-2035
- Table 7: North America Aseptic Formulation Processing Market Value (USD Mn) Forecast by Product Type, 2020-2035
- Table 8: North America Aseptic Formulation Processing Market Value (USD Mn) Forecast by End User, 2020-2035
- Table 9: Latin America Aseptic Formulation Processing Market Value (USD Mn) Forecast by Country, 2020-2035
- Table 10: Latin America Aseptic Formulation Processing Market Value (USD Mn) Forecast by Technology , 2020-2035
- Table 11: Latin America Aseptic Formulation Processing Market Value (USD Mn) Forecast by Product Type, 2020-2035
- Table 12: Latin America Aseptic Formulation Processing Market Value (USD Mn) Forecast by End User, 2020-2035
- Table 13: Western Europe Aseptic Formulation Processing Market Value (USD Mn) Forecast by Country, 2020-2035
- Table 14: Western Europe Aseptic Formulation Processing Market Value (USD Mn) Forecast by Technology , 2020-2035
- Table 15: Western Europe Aseptic Formulation Processing Market Value (USD Mn) Forecast by Product Type, 2020-2035
- Table 16: Western Europe Aseptic Formulation Processing Market Value (USD Mn) Forecast by End User, 2020-2035
- Table 17: Eastern Europe Aseptic Formulation Processing Market Value (USD Mn) Forecast by Country, 2020-2035
- Table 18: Eastern Europe Aseptic Formulation Processing Market Value (USD Mn) Forecast by Technology , 2020-2035
- Table 19: Eastern Europe Aseptic Formulation Processing Market Value (USD Mn) Forecast by Product Type, 2020-2035
- Table 20: Eastern Europe Aseptic Formulation Processing Market Value (USD Mn) Forecast by End User, 2020-2035
- Table 21: East Asia Aseptic Formulation Processing Market Value (USD Mn) Forecast by Country, 2020-2035
- Table 22: East Asia Aseptic Formulation Processing Market Value (USD Mn) Forecast by Technology , 2020-2035
- Table 23: East Asia Aseptic Formulation Processing Market Value (USD Mn) Forecast by Product Type, 2020-2035
- Table 24: East Asia Aseptic Formulation Processing Market Value (USD Mn) Forecast by End User, 2020-2035
- Table 25: South Asia and Pacific Aseptic Formulation Processing Market Value (USD Mn) Forecast by Country, 2020-2035
- Table 26: South Asia and Pacific Aseptic Formulation Processing Market Value (USD Mn) Forecast by Technology , 2020-2035
- Table 27: South Asia and Pacific Aseptic Formulation Processing Market Value (USD Mn) Forecast by Product Type, 2020-2035
- Table 28: South Asia and Pacific Aseptic Formulation Processing Market Value (USD Mn) Forecast by End User, 2020-2035
- Table 29: Middle East & Africa Aseptic Formulation Processing Market Value (USD Mn) Forecast by Country, 2020-2035
- Table 30: Middle East & Africa Aseptic Formulation Processing Market Value (USD Mn) Forecast by Technology , 2020-2035
- Table 31: Middle East & Africa Aseptic Formulation Processing Market Value (USD Mn) Forecast by Product Type, 2020-2035
- Table 32: Middle East & Africa Aseptic Formulation Processing Market Value (USD Mn) Forecast by End User, 2020-2035
List of Figures
- Figure 1: Global Aseptic Formulation Processing Market Pricing Analysis
- Figure 2: Global Aseptic Formulation Processing Market Value (USD Mn) Forecast 2020–2035
- Figure 3: Global Aseptic Formulation Processing Market Value Share and BPS Analysis by Technology , 2025 and 2035
- Figure 4: Global Aseptic Formulation Processing Market Y-o-Y Growth Comparison by Technology , 2025–2035
- Figure 5: Global Aseptic Formulation Processing Market Attractiveness Analysis by Technology
- Figure 6: Global Aseptic Formulation Processing Market Value Share and BPS Analysis by Product Type, 2025 and 2035
- Figure 7: Global Aseptic Formulation Processing Market Y-o-Y Growth Comparison by Product Type, 2025–2035
- Figure 8: Global Aseptic Formulation Processing Market Attractiveness Analysis by Product Type
- Figure 9: Global Aseptic Formulation Processing Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 10: Global Aseptic Formulation Processing Market Y-o-Y Growth Comparison by End User, 2025–2035
- Figure 11: Global Aseptic Formulation Processing Market Attractiveness Analysis by End User
- Figure 12: Global Aseptic Formulation Processing Market Value (USD Mn) Share and BPS Analysis by Region, 2025 and 2035
- Figure 13: Global Aseptic Formulation Processing Market Y-o-Y Growth Comparison by Region, 2025–2035
- Figure 14: Global Aseptic Formulation Processing Market Attractiveness Analysis by Region
- Figure 15: North America Aseptic Formulation Processing Market Incremental Dollar Opportunity, 2025–2035
- Figure 16: Latin America Aseptic Formulation Processing Market Incremental Dollar Opportunity, 2025–2035
- Figure 17: Western Europe Aseptic Formulation Processing Market Incremental Dollar Opportunity, 2025–2035
- Figure 18: Eastern Europe Aseptic Formulation Processing Market Incremental Dollar Opportunity, 2025–2035
- Figure 19: East Asia Aseptic Formulation Processing Market Incremental Dollar Opportunity, 2025–2035
- Figure 20: South Asia and Pacific Aseptic Formulation Processing Market Incremental Dollar Opportunity, 2025–2035
- Figure 21: Middle East & Africa Aseptic Formulation Processing Market Incremental Dollar Opportunity, 2025–2035
- Figure 22: North America Aseptic Formulation Processing Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 23: North America Aseptic Formulation Processing Market Value Share and BPS Analysis by Technology , 2025 and 2035
- Figure 24: North America Aseptic Formulation Processing Market Y-o-Y Growth Comparison by Technology , 2025–2035
- Figure 25: North America Aseptic Formulation Processing Market Attractiveness Analysis by Technology
- Figure 26: North America Aseptic Formulation Processing Market Value Share and BPS Analysis by Product Type, 2025 and 2035
- Figure 27: North America Aseptic Formulation Processing Market Y-o-Y Growth Comparison by Product Type, 2025–2035
- Figure 28: North America Aseptic Formulation Processing Market Attractiveness Analysis by Product Type
- Figure 29: North America Aseptic Formulation Processing Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 30: North America Aseptic Formulation Processing Market Y-o-Y Growth Comparison by End User, 2025–2035
- Figure 31: North America Aseptic Formulation Processing Market Attractiveness Analysis by End User
- Figure 32: Latin America Aseptic Formulation Processing Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 33: Latin America Aseptic Formulation Processing Market Value Share and BPS Analysis by Technology , 2025 and 2035
- Figure 34: Latin America Aseptic Formulation Processing Market Y-o-Y Growth Comparison by Technology , 2025–2035
- Figure 35: Latin America Aseptic Formulation Processing Market Attractiveness Analysis by Technology
- Figure 36: Latin America Aseptic Formulation Processing Market Value Share and BPS Analysis by Product Type, 2025 and 2035
- Figure 37: Latin America Aseptic Formulation Processing Market Y-o-Y Growth Comparison by Product Type, 2025–2035
- Figure 38: Latin America Aseptic Formulation Processing Market Attractiveness Analysis by Product Type
- Figure 39: Latin America Aseptic Formulation Processing Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 40: Latin America Aseptic Formulation Processing Market Y-o-Y Growth Comparison by End User, 2025–2035
- Figure 41: Latin America Aseptic Formulation Processing Market Attractiveness Analysis by End User
- Figure 42: Western Europe Aseptic Formulation Processing Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 43: Western Europe Aseptic Formulation Processing Market Value Share and BPS Analysis by Technology , 2025 and 2035
- Figure 44: Western Europe Aseptic Formulation Processing Market Y-o-Y Growth Comparison by Technology , 2025–2035
- Figure 45: Western Europe Aseptic Formulation Processing Market Attractiveness Analysis by Technology
- Figure 46: Western Europe Aseptic Formulation Processing Market Value Share and BPS Analysis by Product Type, 2025 and 2035
- Figure 47: Western Europe Aseptic Formulation Processing Market Y-o-Y Growth Comparison by Product Type, 2025–2035
- Figure 48: Western Europe Aseptic Formulation Processing Market Attractiveness Analysis by Product Type
- Figure 49: Western Europe Aseptic Formulation Processing Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 50: Western Europe Aseptic Formulation Processing Market Y-o-Y Growth Comparison by End User, 2025–2035
- Figure 51: Western Europe Aseptic Formulation Processing Market Attractiveness Analysis by End User
- Figure 52: Eastern Europe Aseptic Formulation Processing Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 53: Eastern Europe Aseptic Formulation Processing Market Value Share and BPS Analysis by Technology , 2025 and 2035
- Figure 54: Eastern Europe Aseptic Formulation Processing Market Y-o-Y Growth Comparison by Technology , 2025–2035
- Figure 55: Eastern Europe Aseptic Formulation Processing Market Attractiveness Analysis by Technology
- Figure 56: Eastern Europe Aseptic Formulation Processing Market Value Share and BPS Analysis by Product Type, 2025 and 2035
- Figure 57: Eastern Europe Aseptic Formulation Processing Market Y-o-Y Growth Comparison by Product Type, 2025–2035
- Figure 58: Eastern Europe Aseptic Formulation Processing Market Attractiveness Analysis by Product Type
- Figure 59: Eastern Europe Aseptic Formulation Processing Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 60: Eastern Europe Aseptic Formulation Processing Market Y-o-Y Growth Comparison by End User, 2025–2035
- Figure 61: Eastern Europe Aseptic Formulation Processing Market Attractiveness Analysis by End User
- Figure 62: East Asia Aseptic Formulation Processing Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 63: East Asia Aseptic Formulation Processing Market Value Share and BPS Analysis by Technology , 2025 and 2035
- Figure 64: East Asia Aseptic Formulation Processing Market Y-o-Y Growth Comparison by Technology , 2025–2035
- Figure 65: East Asia Aseptic Formulation Processing Market Attractiveness Analysis by Technology
- Figure 66: East Asia Aseptic Formulation Processing Market Value Share and BPS Analysis by Product Type, 2025 and 2035
- Figure 67: East Asia Aseptic Formulation Processing Market Y-o-Y Growth Comparison by Product Type, 2025–2035
- Figure 68: East Asia Aseptic Formulation Processing Market Attractiveness Analysis by Product Type
- Figure 69: East Asia Aseptic Formulation Processing Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 70: East Asia Aseptic Formulation Processing Market Y-o-Y Growth Comparison by End User, 2025–2035
- Figure 71: East Asia Aseptic Formulation Processing Market Attractiveness Analysis by End User
- Figure 72: South Asia and Pacific Aseptic Formulation Processing Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 73: South Asia and Pacific Aseptic Formulation Processing Market Value Share and BPS Analysis by Technology , 2025 and 2035
- Figure 74: South Asia and Pacific Aseptic Formulation Processing Market Y-o-Y Growth Comparison by Technology , 2025–2035
- Figure 75: South Asia and Pacific Aseptic Formulation Processing Market Attractiveness Analysis by Technology
- Figure 76: South Asia and Pacific Aseptic Formulation Processing Market Value Share and BPS Analysis by Product Type, 2025 and 2035
- Figure 77: South Asia and Pacific Aseptic Formulation Processing Market Y-o-Y Growth Comparison by Product Type, 2025–2035
- Figure 78: South Asia and Pacific Aseptic Formulation Processing Market Attractiveness Analysis by Product Type
- Figure 79: South Asia and Pacific Aseptic Formulation Processing Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 80: South Asia and Pacific Aseptic Formulation Processing Market Y-o-Y Growth Comparison by End User, 2025–2035
- Figure 81: South Asia and Pacific Aseptic Formulation Processing Market Attractiveness Analysis by End User
- Figure 82: Middle East & Africa Aseptic Formulation Processing Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 83: Middle East & Africa Aseptic Formulation Processing Market Value Share and BPS Analysis by Technology , 2025 and 2035
- Figure 84: Middle East & Africa Aseptic Formulation Processing Market Y-o-Y Growth Comparison by Technology , 2025–2035
- Figure 85: Middle East & Africa Aseptic Formulation Processing Market Attractiveness Analysis by Technology
- Figure 86: Middle East & Africa Aseptic Formulation Processing Market Value Share and BPS Analysis by Product Type, 2025 and 2035
- Figure 87: Middle East & Africa Aseptic Formulation Processing Market Y-o-Y Growth Comparison by Product Type, 2025–2035
- Figure 88: Middle East & Africa Aseptic Formulation Processing Market Attractiveness Analysis by Product Type
- Figure 89: Middle East & Africa Aseptic Formulation Processing Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 90: Middle East & Africa Aseptic Formulation Processing Market Y-o-Y Growth Comparison by End User, 2025–2035
- Figure 91: Middle East & Africa Aseptic Formulation Processing Market Attractiveness Analysis by End User
- Figure 92: Global Aseptic Formulation Processing Market – Tier Structure Analysis
- Figure 93: Global Aseptic Formulation Processing Market – Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

