Automated Cell Culture Systems Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
The Automated Cell Culture Systems Market is segmented by Product (Process Robots, Automated Bioreactors, Media Exchange Systems, Culture Analyzers, Cell Counters, Storage Platforms, Management Software), Cell Culture Type (Infinite Cell Lines and Finite Cell Lines), Application (Cell Therapy, Drug Discovery, Stem Cell Research, Regenerative Medicine), End User (Biopharma Companies, CDMOs, Research Organizations, Academic Institutes, Hospitals), Workflow Mode (Closed Systems, Open Systems, Hybrid Systems), and Automation Level (Fully Automated Systems, Semi Automated Systems, Modular Systems). Forecast for 2026 to 2036.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Automated Cell Culture Systems Market Size, Market Forecast and Outlook By FMI
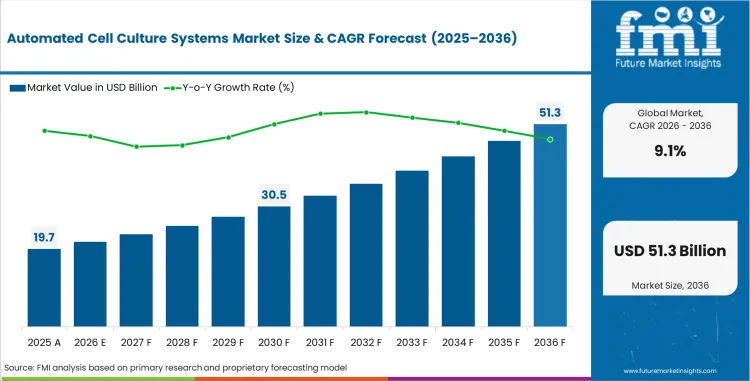
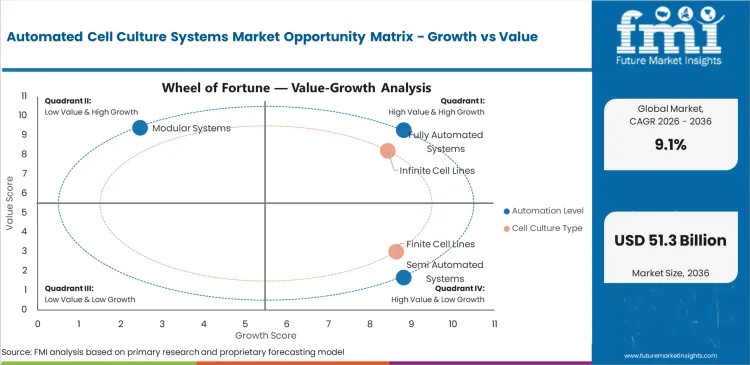
The Automated Cell Culture Systems Market is valued at USD 18.1 billion in 2025. The industry is projected to reach USD 19.7 billion by 2026 and USD 47.1 billion by 2036 at 9.1% CAGR from 2026 to 2036. Fully automated systems are estimated to account for 62.0% of automation level share in 2026, while infinite cell line cultures are projected to hold 58.0% of cell culture type share.
Summary of the Automated Cell Culture Systems Market
- Demand and Growth Drivers
- Automated cell culture systems see steady demand as biologics manufacturers reduce manual culture handling across research and production workflows.
- Cell therapy production supports adoption as developers need closed systems and traceable culture records.
- GMP documentation needs favor software linked platforms that help operators monitor culture steps with less direct contact.
- Product and Segment View
- Fully automated systems lead because sealed workflows reduce contamination risk in sterile production areas.
- Infinite cell line cultures hold demand as biologics and screening programs need repeatable culture output.
- Process robots increase adoption as laboratories combine seeding and feeding steps in one controlled platform.
- Geography and Competitive Outlook
- Japan records demand as iPSC research and regenerative medicine programs need precise culture control.
- China and South Korea see rising adoption as biologics capacity and therapy manufacturing clusters expand.
- Companies with robotics and validation support can improve acceptance through closed culture workflow expertise.
- Analyst Opinion
- Sabyasachi Ghosh, Associate Vice President at FMI says, "I see automated cell culture systems moving from lab support tools into core production assets. Cell therapy developers need sterile handling. Biologics teams need repeatable culture output. Research centers need clear digital records for every culture step. Cost pressure is still real, so suppliers must prove lower labor load and lower contamination risk."
- Automated Cell Culture Systems Market Value Analysis
- The automated cell culture systems market is moving from lab support equipment into core culture control.
- Adoption is rising as biologics manufacturers reduce manual handling across culture workflows.
- Closed systems in cell therapy production are increasing demand.
- Demand comes from controlled handling and traceable culture records.
Automated Cell Culture Systems Market Definition
The automated cell culture systems market covers robotic platforms and controlled equipment used to grow cells with less manual handling. The scope includes automated bioreactors, media exchange systems, culture analyzers, counters, storage equipment, and software. The systems support biologics, cell therapy, stem cell work, drug screening, and clinical-grade culture tasks. The scope excludes manual lab tools, standalone reagents, basic culture plates, and non-cell laboratory automation.
Automated Cell Culture Systems Market Inclusions
Market scope includes process robots, automated bioreactors, media exchange systems, culture analyzers, cell counters, storage platforms, software, finite cultures, infinite cultures, cell therapy, drug discovery, stem cell work, regenerative medicine, biopharma companies, CDMOs, hospitals, research centers, academic labs, and all major regions.
Automated Cell Culture Systems Market Exclusions
The scope excludes manual incubators without automation, basic pipettes, general lab furniture, culture media sold alone, single-use consumables without system control, diagnostic instruments outside cell culture, and non-life-science automation tools.
Automated Cell Culture Systems Market Research Methodology
- Primary Research: FMI analysts talked to biopharma production teams, CDMO process managers, hospital therapy units, and laboratory automation suppliers in key markets.
- Desk Research: Combined data from regulator guidance, cell therapy activity, biologics capacity, company releases, and public life science manufacturing sources.
- Market sizing and forecasting: Added up system demand from cell therapy, drug discovery, stem cell research, regenerative medicine, and biopharma production applications from the bottom up, with regional adoption curves.
- Data validation: Cross-checked every three months against biologics capacity expansion, cell therapy trial activity, supplier launches, and inputs from manufacturers.
Why is the Automated Cell Culture Systems Market Growing?
- Biologics production needs repeatable cell output, so process teams are shifting feeding and passaging into robotic systems.
- Cell therapy manufacturing needs closed handling, which supports adoption of automated cell therapy processing systems across clinical and commercial sites.
- GMP traceability pressure favors software-linked systems that record culture steps and reduce open handling in sterile areas.
- Japan leads country growth at 10.6% CAGR through 2036 due to iPSC research and clinical regenerative medicine activity.
The market is expanding as manual culture work creates uneven output across long studies and therapy batches. Automated platforms standardize media exchange, cell seeding, passaging, and harvest with controlled timing. FDA issued CAR-T guidance in January 2024, and this raised focus on process records in clinical cell therapy work. That shift helps suppliers that can connect robotics with audit-ready software and service support.
Biopharma firms use automation to lower contamination risk during repeated culture tasks. CDMOs use these platforms to handle parallel client programs without adding the same labor load. Hospitals use closed systems for therapy preparation near clinical teams. As cell therapy systems scale across oncology and rare disease programs, culture automation moves closer to required production infrastructure.
Market Segmentation Analysis
- Fully automated systems are estimated to account for 62.0% automation level share in 2026 as users reduce manual culture handling.
- Infinite cell line cultures are projected to hold 58.0% cell culture type share due to repeated use in biologics and screening work.
- Cell therapy is expected to represent 36.0% application share as clinical batches need closed workflows and traceable records.
- Process robots are estimated at 34.6% product share as users combine seeding, feeding, and harvest tasks in one platform.
The market for automated cell culture systems is divided into groups based on product, cell culture type, application, end user, workflow mode, and automation level. Products include process robots, automated bioreactors, media exchange systems, culture analyzers, cell counters, storage platforms, and management software. Cell culture types include infinite cell lines and finite cell lines. Applications include cell therapy, drug discovery, stem cell research, and regenerative medicine. End users include biopharma companies, CDMOs, research organizations, academic institutes, and hospitals. Workflow modes include closed systems, open systems, and hybrid systems. Automation levels include fully automated systems, semi automated systems, and modular systems.
Insights into the automation level segment

- Fully automated systems hold 62.0% share in 2026 because biopharma operators need limited open handling steps and clear process records.
- Automated workflows reduce operator contact and improve batch consistency.
Insights into the cell culture type segment
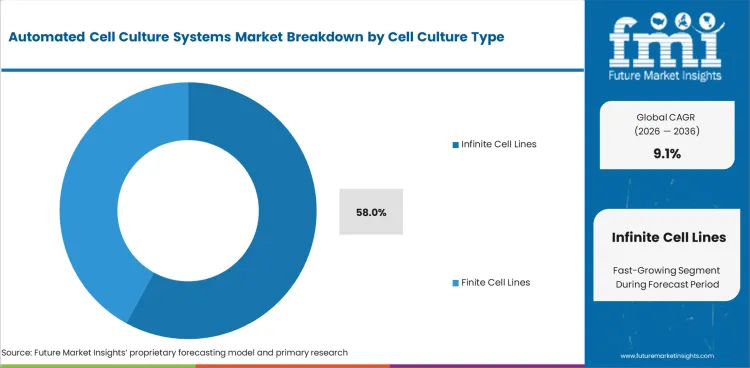
- Infinite cell line cultures account for 58.0% share in 2026. Stable cell lines support repeated biologics studies and screening work.
- Their use fits programs needing consistent culture behavior across repeated runs.
Insights into the application segment
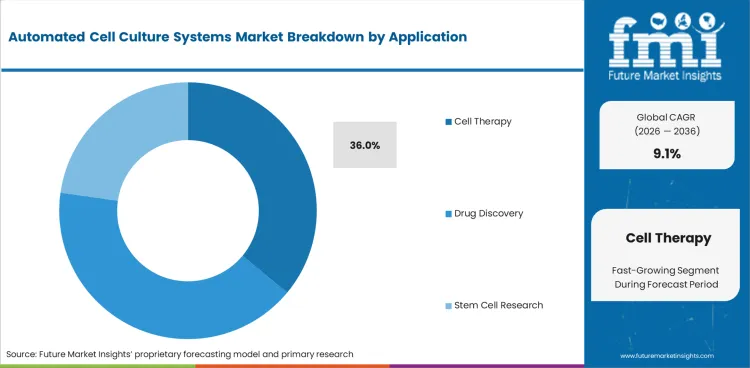
- Cell therapy accounts for 36.0% share in 2026. Personalized batches need closed culture control and documented handling.
- Drug discovery laboratories use 3D cell culture and screening tools to test cell response with limited manual steps.
Insights into the product segment
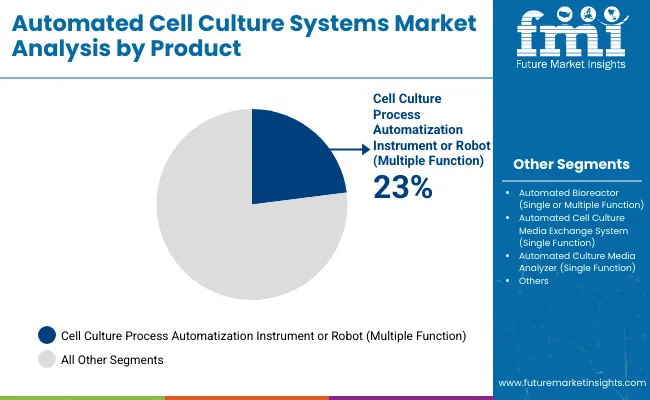
- Process robots represent 34.6% share in 2026. One platform can link seeding and monitoring in a controlled workflow.
- Automated bioreactors and automated cell shakers support high throughput culture work during process development.
Insights into the end user segment
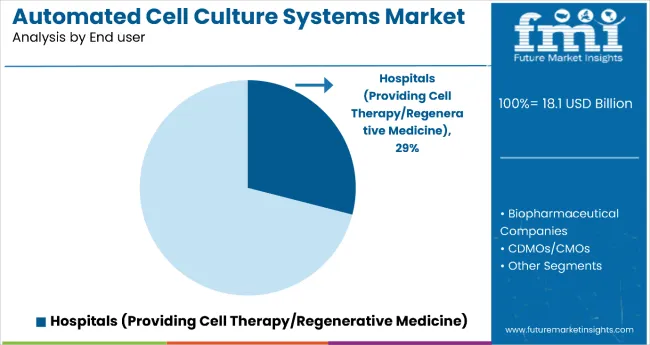
- Biopharma companies hold 31.5% share in 2026 because biologics pipelines need expanded culture capacity and validation support.
- These companies need culture systems that can move from research work into validated production.
Insights into the workflow mode segment
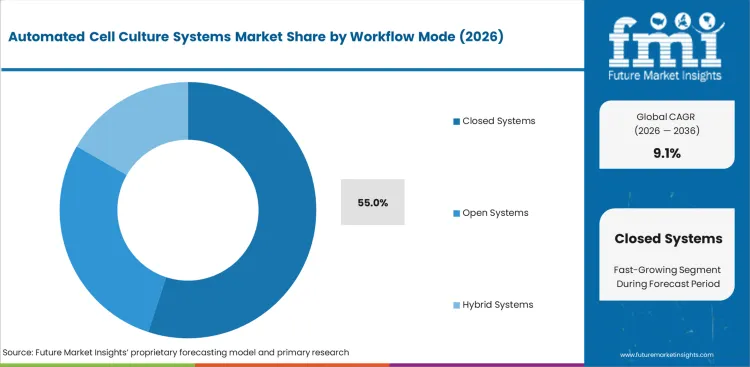
- Closed systems account for 55.0% share in 2026. Sterile culture tasks favor sealed handling and reduced contamination risk.
- Closed workflows pair with cell culture incubator platforms to protect cells during long culture cycles.
Automated Cell Culture Systems Market Drivers, Restraints, and Opportunities
- Closed culture systems reduce operator contact, which supports adoption in sterile therapy and biologics production suites.
- High upfront cost limits small lab adoption, so suppliers are selling modular systems with staged capacity additions.
- Real-time sensors create new opportunity as users want live control of pH, glucose, oxygen, and cell growth.
- Demand for cell culture sampling devices rises as automated processes need cleaner sample collection and data capture.
Closed culture control
Closed culture control is the main driver for users handling sensitive cells. Open culture steps increase contamination risk and make records harder to manage. EU sterile manufacturing rules emphasize contamination control strategy, and this places more value on sealed systems. Suppliers with strong hardware and software links are better placed for clinical-grade demand.
Capital cost pressure
High system cost creates a slow buying path for academic labs and smaller therapy firms. Buyers often start with one module before a full culture line. This supports vendors that offer service packages and validation support. The same pressure keeps cell culture waste aspirator and media handling upgrades tied to larger automation plans.
Sensor-based process control
Sensor-based process control is moving from premium feature to buying requirement in advanced labs. Hamilton launched GlucoSense in December 2025 for real-time glucose sensing in mammalian cell culture. Live data helps users adjust feeding and protect culture health before a batch drifts. This strengthens demand for systems with monitoring depth.
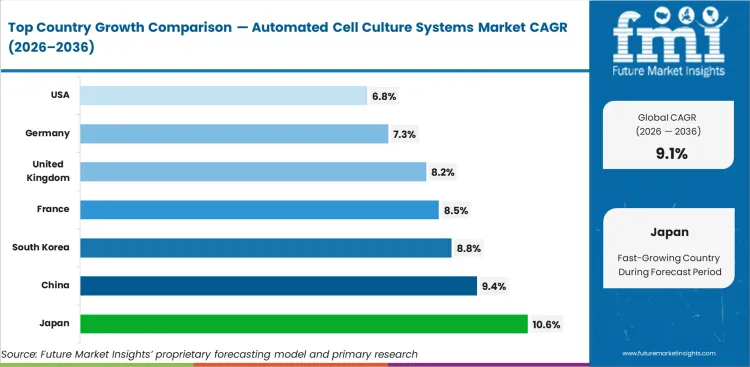
Analysis of Automated Cell Culture Systems Market By Key Countries
.webp)
| Country | CAGR |
|---|---|
| United States | 6.8% |
| United Kingdom | 8.2% |
| France | 8.5% |
| Germany | 7.3% |
| Japan | 10.6% |
| China | 9.4% |
| South Korea | 8.8% |
Source: FMI analysis based on primary research and proprietary forecasting model.
Automated Cell Culture Systems Market CAGR Analysis By Country
- Japan is projected to record 10.6% CAGR through 2036 as iPSC and regenerative medicine centers use robotic culture systems.
- China is expected to expand at 9.4% CAGR through 2036 as local biologics capacity uses more GMP cell culture tools.
- South Korea is forecast to grow at 8.8% CAGR through 2036 as biomanufacturing parks support automated culture lines.
- France is expected to advance at 8.5% CAGR through 2036 as bioproduction grants support clinical-grade culture capacity.
- The United Kingdom is projected to rise at 8.2% CAGR through 2036 as ATMP centers add digital and automated workflows.
- Germany is forecast at 7.3% CAGR through 2036 as engineering depth supports precision bioreactors and robotic handling.
- The United States is expected to post 6.8% CAGR through 2036 as many large sites already use advanced cell culture systems.
The global industry is forecast to expand at 9.1% CAGR from 2026 to 2036. Country differences reflect therapy translation speed, bioproduction capacity, research funding, and supplier access.
Demand Outlook for Automated Cell Culture Systems Market in the United States
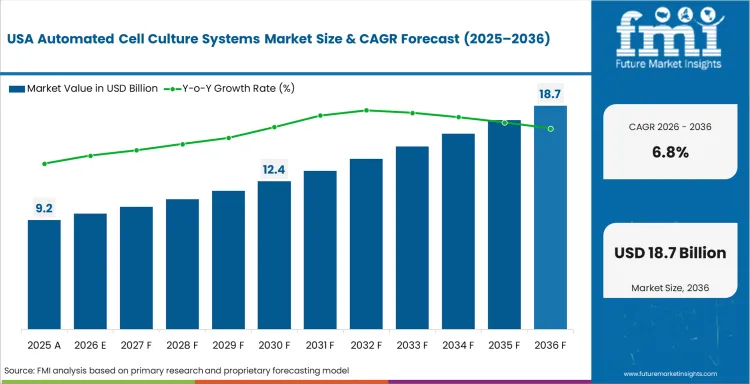
The United States is expected to post 6.8% CAGR through 2036 as FDA cell therapy guidance raises focus on repeatable culture records. Large biopharma and CDMO sites use automation to manage cell therapy and biologics work. US demand is mature compared with Asia, so growth tracks below the global pace. Demand for USA cell therapy processing systems supports closed handling and batch documentation.
- FDA CAR-T guidance from January 2024 supports better documentation in clinical cell therapy work.
- Large CDMO networks use automated bioreactors for parallel client programs.
- Hospital-linked therapy centers buy closed systems to cut contamination risk.
Future Outlook for Automated Cell Culture Systems Market in the United Kingdom
The United Kingdom is projected to rise at 8.2% CAGR from 2026 to 2036 as ATMP infrastructure supports automated therapy production. Shared manufacturing centers help smaller firms access equipment before full plant buildout. UK growth favors platforms that support cell harvesting systems and digital records.
- Stevenage and Braintree sites support therapy makers that need scalable culture workflows.
- NHS-linked research pathways support clinical-grade cell handling.
- Digital manufacturing projects encourage system records and automated feeding.
Opportunity Analysis of Automated Cell Culture Systems Market in France
France is expected to advance at 8.5% CAGR through 2036 as bioproduction programs support local biologics and cell therapy capacity. French bioclusters use automation to shorten development work and reduce manual batch variation. Hospital research groups need controlled systems for immunotherapy and stem cell preparation. This creates steady demand for cell culture media bags and automated culture platforms.
- Biotherapy clusters around Paris and Lyon support clinical-grade cell culture work.
- Hospital research networks use closed culture systems for immunotherapy preparation.
- Public backing for bioproduction helps startups buy modular automation early.
In-depth Analysis of Automated Cell Culture Systems Market in Germany
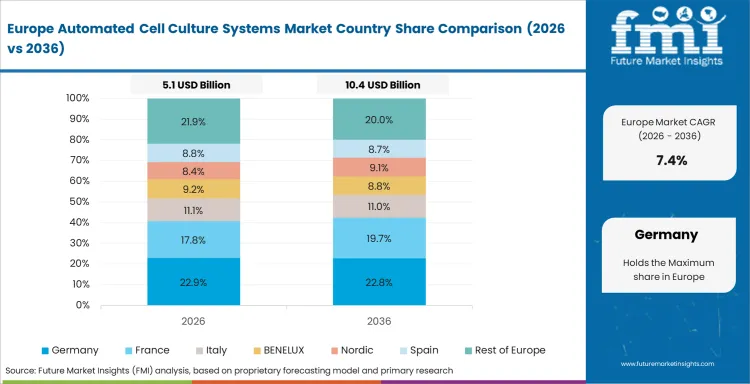
Germany is forecast at 7.3% CAGR through 2036 as precision engineering supports automated bioreactors and robotic handling systems. CDMOs in Germany use digital tracking for audit preparation and batch control. Research groups use organoid and immunotherapy models that need stable environmental control. This supports 3D cell culture plasticware and automated culture setup demand.
- Darmstadt and other life science hubs support bioprocess equipment use.
- Fraunhofer and university labs support applied cell culture process work.
- German buyers favor validation depth before full line expansion.
In-depth Analysis of Automated Cell Culture Systems Market in Japan
Japan is projected to record 10.6% CAGR through 2036 as iPSC research and regenerative therapy work need precise culture control. Hospitals and drug makers use robotic incubators and seeding systems to lower human handling. Japan’s clinical focus on age-related disease supports patient-specific therapy production. This strengthens demand for Japan cell therapy processing systems across research and care settings.
- Kobe and Osaka support regenerative medicine translation and cell processing activity.
- Robotic culture systems suit compact cleanroom layouts in Japanese facilities.
- AI imaging helps advanced labs track cell health during long culture cycles.
Sales Analysis of Automated Cell Culture Systems Market in China
China is expected to expand at 9.4% CAGR through 2036 as biologics capacity and local cell therapy trials raise system demand. Merck started commercial production at its first GMP cell culture media line in China in July 2024. Local supply of China cell culture media bags supports faster setup for automated culture lines. Domestic producers favor systems that combine scale and GMP documentation.
- Shanghai and Suzhou bioparks support automated cell culture demand from biologics firms.
- Local GMP media supply lowers lead time for culture workflow setup.
- Chinese therapy firms use automated systems to manage trial-scale batch volume.
In-depth Analysis of Automated Cell Culture Systems Market in South Korea
South Korea is forecast to grow at 8.8% CAGR through 2036 as biomanufacturing clusters expand culture capacity. Songdo and Osong host biologics and therapy manufacturing activity that needs automated process control. Local firms favor systems that connect cell expansion with data capture. Demand for cell culture demand supports related equipment and media supply.
- Songdo biomanufacturing sites support higher use of automated bioreactor systems.
- Government-backed bio clusters help expand cleanroom and lab capacity.
- Export-focused biologics producers need culture systems with repeatable output.
Competitive Landscape and Strategic Positioning
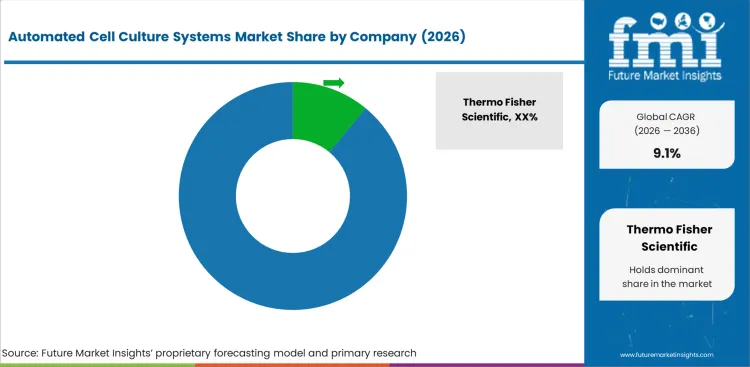
- Thermo Fisher Scientific holds a strong position through Gibco inputs, workflow software, and bioproduction system support.
- Sartorius depend on Ambr automated bioreactors and digital tools for small-scale process work.
- Lonza gains strength from closed therapy manufacturing platforms and CDMO experience in advanced therapies.
- Tecan and Hamilton compete in robotic handling, liquid transfer, and automation-ready lab platforms.
Competition centers on validated automation, closed culture control, sensor depth, and service support. Thermo Fisher launched the Gibco CHOvantage GS Cell Line Development Kit in April 2026, which supports faster biologics development and cell line work. Tecan introduced Veya in January 2025 and acquired Wako Automation assets in December 2025. These actions show supplier focus on scalable robotic workcells and digital scheduling.
Platform depth matters most for users moving from research to GMP production. Lonza received FDA Advanced Manufacturing Technologies designation for the Cocoon Platform in December 2025. Hamilton launched GlucoSense in December 2025 for real-time mammalian cell culture glucose monitoring. BioSpherix and Cellbox Solutions announced a July 2025 partnership for closed cell handling and transport. These moves show competition shifting toward sealed culture workflows and live process data.
Key Companies in the Automated Cell Culture Systems Market
Key global companies leading the automated cell culture systems industry include:
- Thermo Fisher Scientific, Merck KGaA, Sartorius AG, and Lonza Group Ltd have strong bioprocess portfolios and global service reach.
- Corning Incorporated, Tecan Trading AG, and Hamilton support culture vessels, robotic workcells, and liquid handling systems.
- BioSpherix Ltd and PromoCell GmbH address specialist use cases in closed incubation, primary cells, and translational research.
Competitive Benchmarking: Automated Cell Culture Systems Market
| Company | Closed Culture Control | Robotic Workflow Depth | Validation Support | Footprint |
|---|---|---|---|---|
| Thermo Fisher Scientific | High | High | Strong | Global |
| Sartorius AG | High | High | Strong | Global |
| Lonza Group Ltd | High | Medium | Strong | Global |
| Merck KGaA | Medium | Medium | Strong | Global |
| Corning Incorporated | Medium | Medium | Moderate | Global |
| Tecan Trading AG | Medium | High | Moderate | Global |
| Hamilton | Medium | High | Moderate | Global |
| BioSpherix Ltd | High | Medium | Moderate | North America |
| PromoCell GmbH | Medium | Low | Moderate | Europe |
Source: Future Market Insights competitive analysis, 2026.
Key Developments in Automated Cell Culture Systems Market
- In April 2026, Thermo Fisher Scientific launched the Gibco CHOvantage GS Cell Line Development Kit for biologics development workflows.
- In December 2025, Lonza received FDA Advanced Manufacturing Technologies designation for its Cocoon Platform.
Key Players in the Automated Cell Culture Systems Market
Major Global Players:
- Thermo Fisher Scientific
- Corning Incorporated
- Merck KGaA
- Lonza Group Ltd
- Sartorius AG
- Tecan Trading AG
- Hamilton
Specialist and Regional Players:
- BioSpherix Ltd
- PromoCell GmbH
- Cellbox Solutions
- Cellular Origins
- Akadeum Life Sciences
Report Scope and Coverage
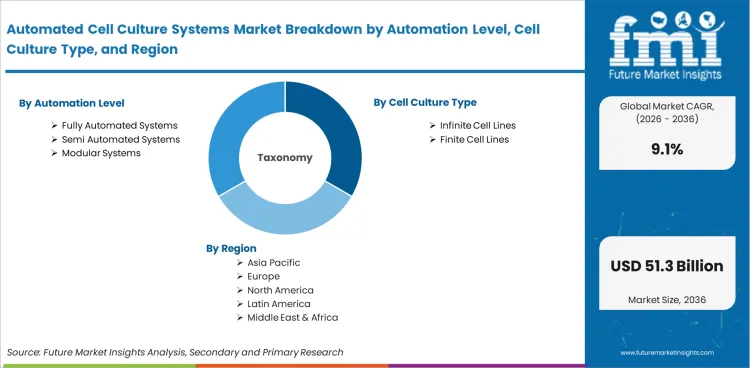
| Parameter | Details |
|---|---|
| Quantitative Units | USD 19.7 billion to USD 47.1 billion, at 9.1% CAGR |
| Market Definition | Automated systems used to grow, feed, monitor, and handle cells with reduced manual contact. |
| Product Segments | Process Robots, Automated Bioreactors, Media Exchange Systems, Culture Analyzers, Cell Counters, Storage Platforms, Management Software |
| Cell Culture Type | Infinite Cell Lines, Finite Cell Lines |
| Applications | Cell Therapy, Drug Discovery, Stem Cell Research, Regenerative Medicine |
| End Users | Biopharma Companies, CDMOs, Research Organizations, Academic Institutes, Hospitals |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | United States, United Kingdom, France, Germany, Japan, China, South Korea, 30 plus countries |
| Key Companies Profiled | Thermo Fisher Scientific, Corning Incorporated, Merck KGaA, Lonza Group Ltd, Sartorius AG, Tecan Trading AG, Hamilton, BioSpherix Ltd, PromoCell GmbH |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down methodology using system pricing, user adoption, and regional therapy capacity checks. |
Automated Cell Culture Systems Market by Segments
Automated Cell Culture Systems Market Segmented by Product:
- Process Robots
- Automated Bioreactors
- Media Exchange Systems
- Culture Analyzers
- Cell Counters
- Storage Platforms
- Management Software
Automated Cell Culture Systems Market Segmented by Cell Culture Type:
- Infinite Cell Lines
- Finite Cell Lines
Automated Cell Culture Systems Market Segmented by Application:
- Cell Therapy
- Drug Discovery
- Stem Cell Research
- Regenerative Medicine
Automated Cell Culture Systems Market Segmented by End User:
- Biopharma Companies
- CDMOs
- Research Organizations
- Academic Institutes
- Hospitals
Automated Cell Culture Systems Market Segmented by Workflow Mode:
- Closed Systems
- Open Systems
- Hybrid Systems
Automated Cell Culture Systems Market Segmented by Automation Level:
- Fully Automated Systems
- Semi Automated Systems
- Modular Systems
Automated Cell Culture Systems Market by Region:
- North America
- United States
- Canada
- Mexico
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia and New Zealand
- Middle East and Africa
- GCC Countries
- South Africa
Research Sources and Bibliography
- 1. Alliance for Regenerative Medicine. (2025, November). Q3 2025 sector snapshot.
- 2. American Society of Gene & Cell Therapy. (2025, November). Gene, cell, & RNA therapy landscape report.
- 3. Thermo Fisher Scientific. (2026, April 1). Thermo Fisher Scientific launches integrated platform to accelerate biologics development.
- 4. Lonza. (2025, December 11). Lonza’s Cocoon Platform receives Advanced Manufacturing Technologies designation.
- 5. BioSpherix & Cellbox Solutions. (2025, July 17). BioSpherix and Cellbox Solutions announce strategic partnership.
This Report Answers
- What is the current and future size of the automated cell culture systems market?
- How fast is the automated cell culture systems market expected to grow between 2026 and 2036?
- Which automation level segment is projected to lead by 2026?
- Which cell culture type is expected to account for the highest share by 2026?
- What factors are driving demand for automated cell culture systems globally?
- How are closed culture workflows influencing adoption in cell therapy production?
- Why is Japan expected to be the fastest-growing country through 2036?
- Who are the key companies active in automated cell culture systems?
- How does FMI estimate and validate the automated cell culture systems forecast?
Frequently Asked Questions
What is the global market demand for Automated Cell Culture Systems in 2026?
In 2026, global demand for automated cell culture systems is expected to be worth USD 19.7 billion across research and production use.
How big will the Automated Cell Culture Systems Market be in 2036?
By 2036, the automated cell culture systems market is projected to reach USD 47.1 billion with stronger biologics and therapy production.
How fast will demand for Automated Cell Culture Systems grow between 2026 and 2036?
Between 2026 and 2036, demand for automated cell culture systems is expected to expand at 9.1% CAGR across global end users.
Which automation level segment is expected to lead by 2026?
Fully automated systems are expected to hold 62.0% share in 2026 as users reduce manual culture handling and contamination risk.
What is driving Automated Cell Culture Systems demand in Japan?
Japan is projected at 10.6% CAGR through 2036 as iPSC research and regenerative medicine programs need precise culture control.
What does this report mean by Automated Cell Culture Systems Market definition?
The market includes robotic culture platforms, automated bioreactors, media exchange tools, analyzers, counters, storage systems, and software for controlled cell growth.
How does FMI make the Automated Cell Culture Systems forecast and check it?
FMI uses system pricing, adoption curves, supplier checks, therapy capacity data, and regional validation to estimate the 2026 to 2036 forecast.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Automation Level
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Automation Level , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Automation Level , 2026 to 2036
- Fully Automated Systems
- Semi Automated Systems
- Modular Systems
- Fully Automated Systems
- Y-o-Y Growth Trend Analysis By Automation Level , 2021 to 2025
- Absolute $ Opportunity Analysis By Automation Level , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Cell Culture Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Cell Culture Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Cell Culture Type, 2026 to 2036
- Infinite Cell Lines
- Finite Cell Lines
- Infinite Cell Lines
- Y-o-Y Growth Trend Analysis By Cell Culture Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Cell Culture Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Cell Therapy
- Drug Discovery
- Stem Cell Research
- Cell Therapy
- Y-o-Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product, 2026 to 2036
- Process Robots
- Automated Bioreactors
- Media Exchange Systems
- Process Robots
- Y-o-Y Growth Trend Analysis By Product, 2021 to 2025
- Absolute $ Opportunity Analysis By Product, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Biopharma Companies
- CDMOs
- Hospitals
- Biopharma Companies
- Y-o-Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Workflow Mode
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Workflow Mode, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Workflow Mode, 2026 to 2036
- Closed Systems
- Open Systems
- Hybrid Systems
- Closed Systems
- Y-o-Y Growth Trend Analysis By Workflow Mode, 2021 to 2025
- Absolute $ Opportunity Analysis By Workflow Mode, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- By Country
- Market Attractiveness Analysis
- By Country
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- By Country
- Market Attractiveness Analysis
- By Country
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- By Country
- Market Attractiveness Analysis
- By Country
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- By Country
- Market Attractiveness Analysis
- By Country
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- By Country
- Market Attractiveness Analysis
- By Country
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- By Country
- Market Attractiveness Analysis
- By Country
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- By Country
- Market Attractiveness Analysis
- By Country
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Automation Level
- By Cell Culture Type
- By Application
- By Product
- By End User
- By Workflow Mode
- Competition Analysis
- Competition Deep Dive
- Thermo Fisher Scientific
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Corning Incorporated
- Merck KGaA
- Lonza Group Ltd
- Sartorius AG
- Tecan Trading AG
- Hamilton
- Thermo Fisher Scientific
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Automation Level , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Cell Culture Type, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 7: Global Market Value (USD Million) Forecast by Workflow Mode, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Automation Level , 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Cell Culture Type, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 13: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 14: North America Market Value (USD Million) Forecast by Workflow Mode, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Automation Level , 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Cell Culture Type, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: Latin America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 20: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 21: Latin America Market Value (USD Million) Forecast by Workflow Mode, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Automation Level , 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Cell Culture Type, 2021 to 2036
- Table 25: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 26: Western Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 27: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 28: Western Europe Market Value (USD Million) Forecast by Workflow Mode, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Automation Level , 2021 to 2036
- Table 31: Eastern Europe Market Value (USD Million) Forecast by Cell Culture Type, 2021 to 2036
- Table 32: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 33: Eastern Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 34: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 35: Eastern Europe Market Value (USD Million) Forecast by Workflow Mode, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: East Asia Market Value (USD Million) Forecast by Automation Level , 2021 to 2036
- Table 38: East Asia Market Value (USD Million) Forecast by Cell Culture Type, 2021 to 2036
- Table 39: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 40: East Asia Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 41: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 42: East Asia Market Value (USD Million) Forecast by Workflow Mode, 2021 to 2036
- Table 43: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: South Asia and Pacific Market Value (USD Million) Forecast by Automation Level , 2021 to 2036
- Table 45: South Asia and Pacific Market Value (USD Million) Forecast by Cell Culture Type, 2021 to 2036
- Table 46: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 47: South Asia and Pacific Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 48: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 49: South Asia and Pacific Market Value (USD Million) Forecast by Workflow Mode, 2021 to 2036
- Table 50: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 51: Middle East & Africa Market Value (USD Million) Forecast by Automation Level , 2021 to 2036
- Table 52: Middle East & Africa Market Value (USD Million) Forecast by Cell Culture Type, 2021 to 2036
- Table 53: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 54: Middle East & Africa Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 55: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 56: Middle East & Africa Market Value (USD Million) Forecast by Workflow Mode, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Automation Level , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Automation Level , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Automation Level
- Figure 6: Global Market Value Share and BPS Analysis by Cell Culture Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Cell Culture Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Cell Culture Type
- Figure 9: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Application
- Figure 12: Global Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Product
- Figure 15: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by End User
- Figure 18: Global Market Value Share and BPS Analysis by Workflow Mode, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Workflow Mode, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Workflow Mode
- Figure 21: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 22: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 23: Global Market Attractiveness Analysis by Region
- Figure 24: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 29: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 30: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 31: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 32: North America Market Value Share and BPS Analysis by Automation Level , 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Automation Level , 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Automation Level
- Figure 35: North America Market Value Share and BPS Analysis by Cell Culture Type, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Cell Culture Type, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Cell Culture Type
- Figure 38: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Application
- Figure 41: North America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Product
- Figure 44: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 45: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 46: North America Market Attractiveness Analysis by End User
- Figure 47: North America Market Value Share and BPS Analysis by Workflow Mode, 2026 and 2036
- Figure 48: North America Market Y-o-Y Growth Comparison by Workflow Mode, 2026-2036
- Figure 49: North America Market Attractiveness Analysis by Workflow Mode
- Figure 50: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 51: Latin America Market Value Share and BPS Analysis by Automation Level , 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Automation Level , 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Automation Level
- Figure 54: Latin America Market Value Share and BPS Analysis by Cell Culture Type, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Cell Culture Type, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Cell Culture Type
- Figure 57: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Application
- Figure 60: Latin America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 61: Latin America Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 62: Latin America Market Attractiveness Analysis by Product
- Figure 63: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 64: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 65: Latin America Market Attractiveness Analysis by End User
- Figure 66: Latin America Market Value Share and BPS Analysis by Workflow Mode, 2026 and 2036
- Figure 67: Latin America Market Y-o-Y Growth Comparison by Workflow Mode, 2026-2036
- Figure 68: Latin America Market Attractiveness Analysis by Workflow Mode
- Figure 69: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 70: Western Europe Market Value Share and BPS Analysis by Automation Level , 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Automation Level , 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Automation Level
- Figure 73: Western Europe Market Value Share and BPS Analysis by Cell Culture Type, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Cell Culture Type, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Cell Culture Type
- Figure 76: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 77: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 78: Western Europe Market Attractiveness Analysis by Application
- Figure 79: Western Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 80: Western Europe Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 81: Western Europe Market Attractiveness Analysis by Product
- Figure 82: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 83: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 84: Western Europe Market Attractiveness Analysis by End User
- Figure 85: Western Europe Market Value Share and BPS Analysis by Workflow Mode, 2026 and 2036
- Figure 86: Western Europe Market Y-o-Y Growth Comparison by Workflow Mode, 2026-2036
- Figure 87: Western Europe Market Attractiveness Analysis by Workflow Mode
- Figure 88: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Automation Level , 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Automation Level , 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Automation Level
- Figure 92: Eastern Europe Market Value Share and BPS Analysis by Cell Culture Type, 2026 and 2036
- Figure 93: Eastern Europe Market Y-o-Y Growth Comparison by Cell Culture Type, 2026-2036
- Figure 94: Eastern Europe Market Attractiveness Analysis by Cell Culture Type
- Figure 95: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 96: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 97: Eastern Europe Market Attractiveness Analysis by Application
- Figure 98: Eastern Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 99: Eastern Europe Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 100: Eastern Europe Market Attractiveness Analysis by Product
- Figure 101: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 102: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 103: Eastern Europe Market Attractiveness Analysis by End User
- Figure 104: Eastern Europe Market Value Share and BPS Analysis by Workflow Mode, 2026 and 2036
- Figure 105: Eastern Europe Market Y-o-Y Growth Comparison by Workflow Mode, 2026-2036
- Figure 106: Eastern Europe Market Attractiveness Analysis by Workflow Mode
- Figure 107: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 108: East Asia Market Value Share and BPS Analysis by Automation Level , 2026 and 2036
- Figure 109: East Asia Market Y-o-Y Growth Comparison by Automation Level , 2026-2036
- Figure 110: East Asia Market Attractiveness Analysis by Automation Level
- Figure 111: East Asia Market Value Share and BPS Analysis by Cell Culture Type, 2026 and 2036
- Figure 112: East Asia Market Y-o-Y Growth Comparison by Cell Culture Type, 2026-2036
- Figure 113: East Asia Market Attractiveness Analysis by Cell Culture Type
- Figure 114: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 115: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 116: East Asia Market Attractiveness Analysis by Application
- Figure 117: East Asia Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 118: East Asia Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 119: East Asia Market Attractiveness Analysis by Product
- Figure 120: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 121: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 122: East Asia Market Attractiveness Analysis by End User
- Figure 123: East Asia Market Value Share and BPS Analysis by Workflow Mode, 2026 and 2036
- Figure 124: East Asia Market Y-o-Y Growth Comparison by Workflow Mode, 2026-2036
- Figure 125: East Asia Market Attractiveness Analysis by Workflow Mode
- Figure 126: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 127: South Asia and Pacific Market Value Share and BPS Analysis by Automation Level , 2026 and 2036
- Figure 128: South Asia and Pacific Market Y-o-Y Growth Comparison by Automation Level , 2026-2036
- Figure 129: South Asia and Pacific Market Attractiveness Analysis by Automation Level
- Figure 130: South Asia and Pacific Market Value Share and BPS Analysis by Cell Culture Type, 2026 and 2036
- Figure 131: South Asia and Pacific Market Y-o-Y Growth Comparison by Cell Culture Type, 2026-2036
- Figure 132: South Asia and Pacific Market Attractiveness Analysis by Cell Culture Type
- Figure 133: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 134: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 135: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 136: South Asia and Pacific Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 137: South Asia and Pacific Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 138: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 139: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 140: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 141: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 142: South Asia and Pacific Market Value Share and BPS Analysis by Workflow Mode, 2026 and 2036
- Figure 143: South Asia and Pacific Market Y-o-Y Growth Comparison by Workflow Mode, 2026-2036
- Figure 144: South Asia and Pacific Market Attractiveness Analysis by Workflow Mode
- Figure 145: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 146: Middle East & Africa Market Value Share and BPS Analysis by Automation Level , 2026 and 2036
- Figure 147: Middle East & Africa Market Y-o-Y Growth Comparison by Automation Level , 2026-2036
- Figure 148: Middle East & Africa Market Attractiveness Analysis by Automation Level
- Figure 149: Middle East & Africa Market Value Share and BPS Analysis by Cell Culture Type, 2026 and 2036
- Figure 150: Middle East & Africa Market Y-o-Y Growth Comparison by Cell Culture Type, 2026-2036
- Figure 151: Middle East & Africa Market Attractiveness Analysis by Cell Culture Type
- Figure 152: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 153: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 154: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 155: Middle East & Africa Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 156: Middle East & Africa Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 157: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 158: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 159: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 160: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 161: Middle East & Africa Market Value Share and BPS Analysis by Workflow Mode, 2026 and 2036
- Figure 162: Middle East & Africa Market Y-o-Y Growth Comparison by Workflow Mode, 2026-2036
- Figure 163: Middle East & Africa Market Attractiveness Analysis by Workflow Mode
- Figure 164: Global Market - Tier Structure Analysis
- Figure 165: Global Market - Company Share Analysis

