Automated External Defibrillator Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
Automated External Defibrillator Market is segmented by Product (Automated External Defibrillator (AED), Semi Automated External Defibrillator (Semi-AED)), Modality (Public Access AED, Professional Use AED), End User (Hospitals, Clinics, Long Term Care Centers, Ambulatory Surgical Centers, Emergency Medical Services EMS, Urgent Care Centers, Independent Catheterization Labs, Public Access, Airports, Train Stations, Shopping Malls, Hypermarkets And Supermarkets, Sports Arenas And Stadiums, Schools And Colleges, Gymnasiums, Corporate Office Buildings, Home Care Settings, Parks, Hotels And Restaurants, Others), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Automated External Defibrillator Market Size, Market Forecast and Outlook By FMI
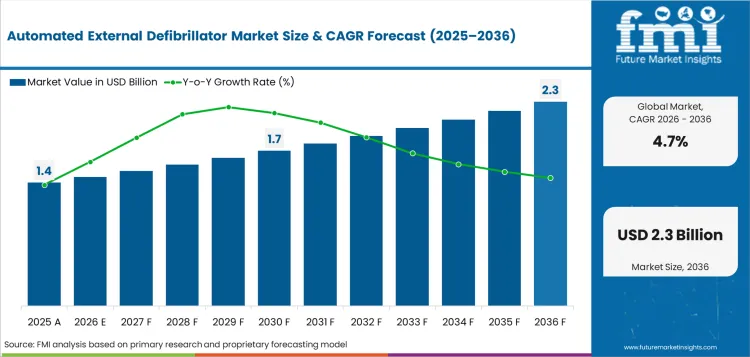
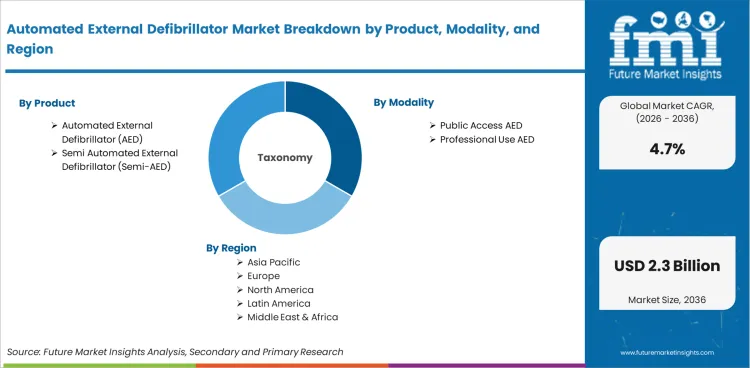
The automated external defibrillator market was valued at USD 1.4 billion in 2025. The market is set to reach USD 1.5 billion by 2026-end and grow at a CAGR of 4.7% between 2026-2036 to reach USD 2.3 billion by 2036. Automated External Defibrillator (AED) will dominate with a 72.0% share, while Public Access AED will lead with a 54.0% share.
Summary of the Automated External Defibrillator Market
- Demand and Growth Drivers
- Regulatory approvals and hospital capital expenditure cycles in developed and emerging markets support clinical adoption of automated external defibrillator market products.
- Purchasing is focused in tertiary care facilities, where demand mirrors the move toward early diagnosis and treatment protocols.
- In China and India, we expect the expansion of reimbursement coverage to support volume growth in the forecast period.
- Product and Segment View
- The Automated External Defibrillator (AED) is expected to capture 72.0% of the product segment in 2026, which is in line with the established demand patterns.
- Public Access AED commands 54.0% share of the modality segment, driven by end-use requirements and specification adherence.
- Product mix is moving towards higher specification variants with premium segments growing faster than commodity alternatives.
- Geography and Competitive Outlook
- the expansion of hospital infrastructure and government funding of health programs.
- India 8.3%. Investment in healthcare infrastructure, Ayushman Bharat coverage and increasing private hospital networks.
- “Companies with existing distribution reach and product breadth across several segments should be able to capture incremental demand.”
- Analyst Opinion
- The automated external defibrillator market is transitioning from volume-led growth toward value-driven procurement, where product performance, compliance, and cost efficiency determine supplier selection.
- Adoption is increasing due to structural demand from primary end-use sectors, with procurement budgets reflecting longer planning cycles and specification requirements.
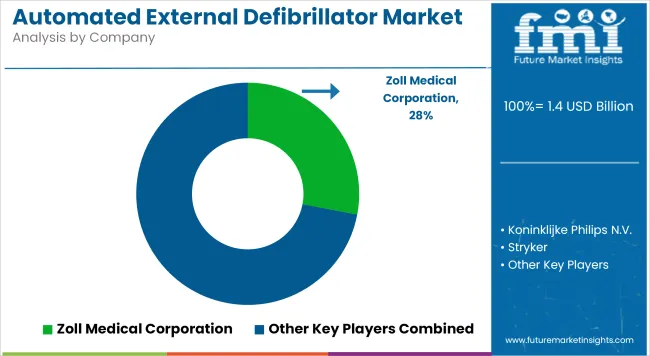
- Zoll Medical Corporation maintains the leading position, with competitive advantages in product breadth and geographic reach.
- Companies that can balance cost competitiveness with technical performance across China and India are positioned to strengthen market share over the forecast period.
- Capital allocation toward capacity expansion and downstream integration is expected to define competitive positioning through 2036.
Automated External Defibrillator Market Definition
The Automated External Defibrillator Market is segmented into product, modality, end user, technology and distribution channel. Market scope is the revenue generated from the manufacturing, distribution, and procurement by the end-users of automated external defibrillator market across the globe during the forecast period of 2026 to 2036.
Automated External Defibrillator Market Inclusions
Market scope covers all the commercialized automated external defibrillator market products, segmented on the basis of product, modality, end user, technology, distribution channel. Revenue is tracked from 2026-2036 across China, India, South Korea, UK, Germany, Canada, USA and 30 plus additional countries.
Automated External Defibrillator Market Exclusions
The scope does not cover raw materials that are not processed, internal consumption that is not commercially traded, and products that are in adjacent market categories. Captive production consumed within vertically integrated operations is excluded unless sold on a commercial basis.
Automated External Defibrillator Market Research Methodology
- Primary Research: FMI analysts spoke with manufacturers, distributors and end-use procurement teams in China, India and other key markets.
- Desk Research: Data from industry associations, regulatory databases, trade publications and manufacturer disclosures.
- Market Sizing and Forecasting: Bottom-up aggregation from product, end-use and regional adoption curves, triangulated with top-down industry benchmarks.
- Data Validation: Quarterly cross-check against production data, trade statistics and manufacturer financial disclosures.
Why is the Automated External Defibrillator Market Growing?
- Structured demand for automated external defibrillator market products is emerging from the rising clinical adoption and regulatory approvals in hospital and ambulatory care settings.
- China was the best performer with 8.9% growth, spurred by investment in healthcare infrastructure and rising patient numbers.
- Addressable market is expanding in emerging economies with reimbursement coverage expansion and government health programs.
The growth of the automated external defibrillator market is driven by clinical adoption in hospital networks and ambulatory care settings. Growth is concentrated in China and India, where investments in healthcare infrastructure and growth in patient volumes are driving ongoing procurement cycles. Hospital capital expenditure budgets have longer planning horizons and procurement teams will specify products that meet both clinical performance requirements and cost-efficiency requirements. Regulatory approvals and reimbursement coverage are the main gates to market access. Companies with a well-established regulatory portfolio have structural advantages.
Demand is also shaped by trends in the population, including the aging of developed markets and the increasing access to health care in emerging economies. Procurement is moving towards integrated solutions that reduce the total cost of care, with buyers assessing product performance in conjunction with training, service and lifecycle support. Structural demand drivers are anticipated to drive the market to grow at a CAGR of 4.7% between 2026 and 2036.
Market Segmentation Analysis
- The product segment, Automated External Defibrillator (AED accounted for 72.0% demand patterns of concentrated end-use.
- Specification requirements from primary buyers dominate the Public Access AED market share of the modality segment.
- The market is segmented by product, modality, end user, technology and distribution channel and revenue (USD Million) is tracked from 2026 to 2036.
Automated External Defibrillator Market Segmentation The automated external defibrillator market is segmented on the basis of product, modality, end user, technology, and distribution channel. Each segmentation dimension is shaped by different procurement patterns and end-use requirements, which in turn shape competitive dynamics and the distribution of revenue.
Insights into the Automated External Defibrillator (AED) Product Segment
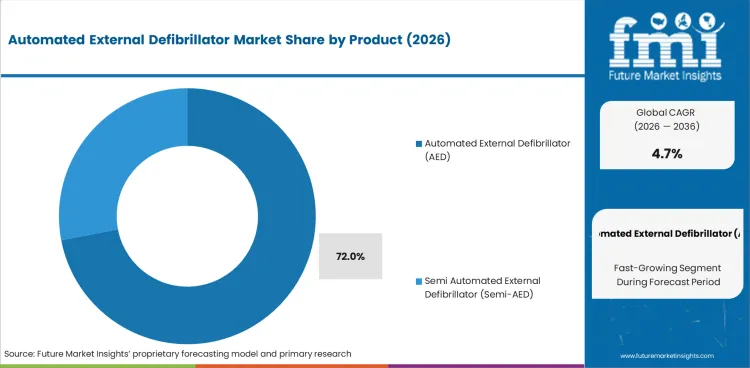
account for 72.0% of the product segment in 2026. Demand is concentrated on established end-use applications where automated external defibrillator (aed) products meet specification requirements and procurement standards. The behaviour of the buyers in this segment is a reflection of the performance expectations and the cost-sensitivity. The procurement cycles are associated with the investment patterns of the enduse industries.
Other segments including semi automated external defibrillator (semi-aed) contribute to the rest of the market share. Growth in these segments is due to diversification of the end-use applications and specification requirements that favour the specialized product variations over the standard alternative.
Insights into the Public Access AED Modality Segment
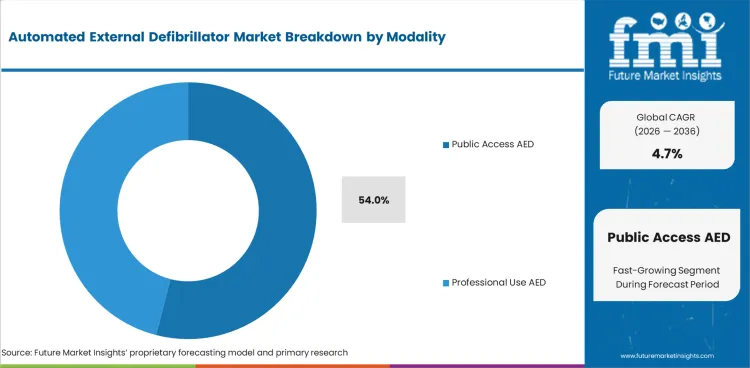
Public access aed is expected to be 54.0% of the modality segment in 2026. Demand is focused on established end-use applications where the public can access aed products that meet specification requirements and procurement standards. Performance-oriented and cost-conscious are the buyers in this segment. Their procurement cycles are linked to the investment cycles in the end-use industries.
Other segments including professional use AED account for the remaining market share. Diversification of end-use applications and specifications supporting specialized variants of products over the standard alternatives are driving the growth in these segments.
Automated External Defibrillator Market Drivers, Restraints, and Opportunities
- Structural demand from primary end-use industries and regulatory compliance requirements are supporting adoption of automated external defibrillator market products across key markets.
- Cost competitiveness relative to alternative solutions and scalability of production capacity remain constraints for smaller suppliers.
- Government investment programs in China and India are creating incremental demand beyond baseline consumption patterns.
The automated external defibrillator market is shaped by demand-side structural drivers, supply-side constraints, and policy-driven opportunities. Growth at 4.7% CAGR from 2026 to 2036 reflects the balance of these forces across segments and regions.
Clinical Adoption and Regulatory Expansion
Demand reflects broadening clinical protocols and regulatory approvals that expand the addressable patient population. Hospital procurement teams are incorporating automated external defibrillator market products into standard care pathways, with reimbursement coverage acting as the primary adoption gate.
Cost and Reimbursement Constraints
Adoption is constrained by product pricing relative to reimbursement rates, particularly in emerging markets where healthcare budgets face competing priorities. Procurement decisions balance clinical benefit against per-unit cost and total treatment expenditure.
Government Health Programs
Public health investment programs in China and India are expanding access and creating structured procurement channels. These programs generate multi-year demand visibility for manufacturers with approved product portfolios.
Technology Advancement and Product Differentiation
Product development is focused on improved clinical outcomes, ease of use, and compatibility with existing clinical workflows. Companies investing in next-generation platforms are positioning for specification changes expected during the forecast period.
Analysis of Automated External Defibrillator Market By Key Countries
.webp)
| Country | CAGR |
|---|---|
| China | 8.9% |
| India | 8.3% |
| South Korea | 7.9% |
| UK | 6% |
| Germany | 3.7% |
| Canada | 2.8% |
| USA | 2.5% |
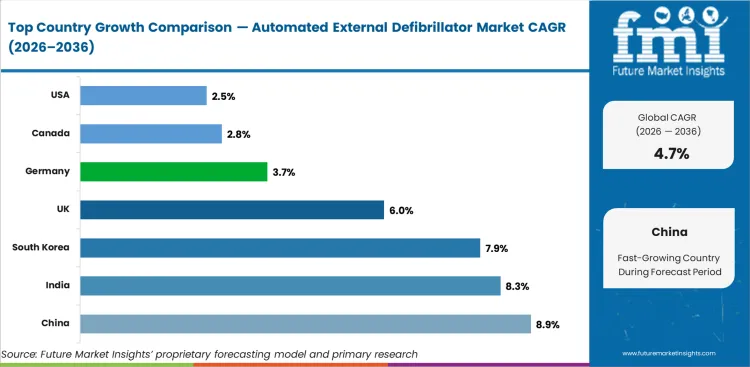
- China at 8.9% CAGR reflects hospital infrastructure expansion and government health program funding.
- India at 8.3% CAGR reflects healthcare infrastructure investment, Ayushman Bharat coverage, and growing private hospital networks.
- South Korea at 7.9% CAGR reflects advanced healthcare system and government health technology adoption programs.
- UK at 6.0% CAGR reflects NHS modernization programs and procurement framework updates.
The global automated external defibrillator market is estimated at USD 1.46 billion in 2026 and is projected to expand at 4.7% CAGR from 2026 to 2036. The analysis covers more than 30 countries, with key markets detailed below.
Demand Outlook for Automated External Defibrillator Market in China
China is projected to expand at 8.9% CAGR through 2036. Growth is supported by hospital infrastructure expansion and government health program funding.
- Hospital infrastructure expansion creates multi-year procurement cycles for clinical equipment.
- Government health programs are broadening patient coverage and increasing procedure volumes.
- Domestic manufacturers are gaining qualification for products previously sourced from imports.
Future Outlook for Automated External Defibrillator Market in India
India is projected to expand at 8.3% CAGR through 2036. Growth is supported by healthcare infrastructure investment, Ayushman Bharat coverage, and growing private hospital networks.
- Ayushman Bharat and private healthcare investment are expanding the addressable patient population.
- Urban tertiary care growth creates demand for advanced clinical products and solutions.
- Cost-effectiveness requirements shape procurement specifications toward value-oriented offerings.
Opportunity Analysis of Automated External Defibrillator Market in South Korea
South Korea is projected to expand at 7.9% CAGR through 2036. Growth is supported by advanced healthcare system and government health technology adoption programs.
- Advanced healthcare system supports rapid adoption of new clinical technologies.
- Government health technology programs create structured procurement channels.
- Quality and after-sales support requirements shape supplier selection criteria.
In-depth Analysis of Automated External Defibrillator Market in UK
UK is projected to expand at 6.0% CAGR through 2036. Growth is supported by NHS modernization programs and procurement framework updates.
- NHS modernization programs create procurement frameworks for updated clinical equipment.
- Post-Brexit regulatory independence is creating separate qualification requirements.
- Private healthcare investment supplements public sector procurement volumes.
Sales Analysis of Automated External Defibrillator Market in Germany
Germany is projected to expand at 3.7% CAGR through 2036. Growth is supported by EU regulatory standards, hospital modernization, and reimbursement system coverage.
- EU regulatory standards and hospital modernization programs drive procurement specification compliance.
- Reimbursement system coverage supports sustained demand for approved products.
- Clinical research infrastructure generates early adoption of advanced solutions.
Growth Dynamics for Automated External Defibrillator Market in Canada
Canada is projected to expand at 2.8% CAGR through 2036. Growth is supported by universal healthcare system and hospital capital expenditure cycles.
- Universal healthcare system creates structured procurement frameworks.
- Hospital capital expenditure cycles generate multi-year demand visibility.
- Proximity to US markets influences product specification alignment.
Competitive Landscape and Strategic Positioning
- Zoll Medical Corporation is a major player in the automated external defibrillator market, with product breadth and geographic distribution infrastructure.
- The top 5-7 companies hold the lion’s share of the market, with the rest split between regional and specialized players.
- New entrants are targeting niche applications and underserved geographies to gain a foothold in the market.
Integrated product portfolios and well-established end-use relationships sustain the Zoll Medical Corporation’s leading position. The company’s distribution reach in China, India and other key markets provides structural procurement access advantages.
Second-tier players compete on regional strength, specialized product capabilities and cost positioning. Where companies have strong positions in specific end-use segments or geographies, they have viable competitive positions, even with small overall share.
Barriers to entry include product qualification requirements, development of a distribution network, regulatory approval time and established buyer-supplier relationships. The strategic priorities of market participants are focused on geographic expansion, broadening of product portfolio and cost reduction.
Key Companies in the Automated External Defibrillator Market
Key global companies leading the automated external defibrillator market include:
- Zoll Medical Corporation, Koninklijke Philips N.V., Stryker, NIHON KOHDEN CORPORATION hold leading positions through broad product portfolios and established end-use relationships.
- Shenzhen Mindray Bio-Medical Electronics Co., Ltd., FUKUDA DENSHI, Schiller AG maintain regional strength through specialized product capabilities and local market knowledge.
- Metrax GmbH, Avive Solutions, Inc., MEDIANA CO., Ltd. represent emerging participants targeting niche applications and growth segments.
Competitive Benchmarking: Automated External Defibrillator Market
| Company | Product Breadth | Geographic Reach | End-Use Access | Technology Capability |
|---|---|---|---|---|
| Zoll Medical Corporation | High | Strong | Strong | High |
| Koninklijke Philips N.V. | High | Strong | Strong | High |
| Stryker | High | Strong | Strong | High |
| NIHON KOHDEN CORPORATION | Medium | Moderate | Moderate | Medium |
| Shenzhen Mindray Bio-Medical Electronics Co., Ltd. | Medium | Moderate | Moderate | Medium |
| FUKUDA DENSHI | Medium | Moderate | Moderate | Medium |
| Schiller AG | Medium | Moderate | Low | Medium |
| Metrax GmbH | Medium | Moderate | Low | Medium |
| Avive Solutions, Inc. | Medium | Moderate | Low | Medium |
| MEDIANA CO., Ltd. | Medium | Moderate | Low | Medium |
Source: Future Market Insights competitive analysis, 2026.
Key Developments in Automated External Defibrillator Market
- In 2025, Koninklijke Philips N.V. expanded its automated external defibrillator portfolio with advanced connected emergency response and public access defibrillation solutions targeting healthcare and public safety applications.
- In 2025, Stryker Corporation continued manufacturing and distribution expansion initiatives for LIFEPAK defibrillator systems to support growing emergency care demand across North America and international markets.
Key Players in the Automated External Defibrillator Market
Major Global Players
- Zoll Medical Corporation
- Koninklijke Philips N.V.
- Stryker
- NIHON KOHDEN CORPORATION
- Shenzhen Mindray Bio-Medical Electronics Co., Ltd.
- FUKUDA DENSHI
- Schiller AG
Emerging Players/Startups
- Metrax GmbH
- Avive Solutions, Inc.
- MEDIANA CO., Ltd.
- Progetti Medical Equipment Solutions
- Bexen
- Corpuls
- CU Medical Systems, Inc.
- Promed Technology Co., Limited
- INNOMED MEDICAL INC.
Report Scope and Coverage
| Parameter | Details |
|---|---|
| Quantitative Units | USD 1.46 billion to USD 2.3 billion, at a CAGR of 4.7% |
| Market Definition | The automated external defibrillator market encompasses commercially traded products and solutions across product, modality, end user, technology, distribution channel. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | China, India, South Korea, UK, Germany, Canada, USA, 30 plus countries |
| Key Companies Profiled | Zoll Medical Corporation, Koninklijke Philips N.V., Stryker, NIHON KOHDEN CORPORATION, Shenzhen Mindray Bio-Medical Electronics Co., Ltd., FUKUDA DENSHI, Schiller AG, Metrax GmbH, Avive Solutions, Inc., MEDIANA CO., Ltd., Progetti Medical Equipment Solutions, Bexen |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down methodology starting with verified transaction data, projecting adoption velocity across segments and regions. |
Segmentation
Automated External Defibrillator Market Segmented by Product:
- Automated External Defibrillator (AED)
- Fully Automated Defibrillator
- Automatic Shock Delivery
- Minimal User Intervention
- Smart AED Systems
- Real Time ECG Analysis
- Advanced Safety Algorithms
- Fully Automated Defibrillator
- Semi Automated External Defibrillator (Semi-AED)
- Manual Shock Advised Devices
- Healthcare Professional Use
- Controlled Emergency Response
- User Guided Defibrillation
- Step By Step Voice Assistance
- Basic Rhythm Analysis
- Manual Shock Advised Devices
Automated External Defibrillator Market Segmented by Modality:
- Public Access AED
- Public Safety AED Units
- Airports And Transit Hubs
- Shopping And Commercial Spaces
- Layperson Operated Devices
- Voice Guided Operation
- Emergency Public Response Systems
- Public Safety AED Units
- Professional Use AED
- Hospital Grade AED Systems
- Emergency Room Use
- ICU And Critical Care Units
- Medical Staff Operated Devices
- Paramedic And EMS Use
- Clinical Environment Deployment
- Hospital Grade AED Systems
Automated External Defibrillator Market Segmented by End User:
- Hospitals
- Emergency Departments
- Cardiac Arrest Response Units
- Critical Care Support Systems
- Operation Theaters
- Surgical Emergency Backup
- Inpatient Cardiac Care
- Emergency Departments
- Clinics
- Outpatient Clinics
- Cardiac Monitoring Units
- Primary Care Emergency Support
- Outpatient Clinics
- Long Term Care Centers
- Nursing Homes
- Elderly Care Facilities
- Chronic Patient Monitoring
- Nursing Homes
- Ambulatory Surgical Centers
- Same Day Surgery Units
- Post Operative Emergency Care
- Cardiac Safety Equipment
- Same Day Surgery Units
- Emergency Medical Services EMS
- Ambulance Services
- Pre Hospital Cardiac Response
- Field Emergency Care
- Ambulance Services
- Urgent Care Centers
- Fast Response Clinics
- Minor Emergency Treatment
- Walk In Emergency Care
- Fast Response Clinics
- Independent Catheterization Labs
- Cardiac Procedure Labs
- Interventional Cardiology Units
- Procedure Backup Safety Systems
- Cardiac Procedure Labs
- Public Access
- Airports
- International Airports
- Passenger Terminal Safety Systems
- Emergency Response Stations
- International Airports
- Train Stations
- Railway Hubs
- Passenger Safety Installations
- Transit Emergency Equipment
- Railway Hubs
- Shopping Malls
- Retail Complexes
- High Footfall Emergency Systems
- Public Safety Installations
- Retail Complexes
- Hypermarkets And Supermarkets
- Retail Emergency Preparedness
- Customer Safety Systems
- On Site Medical Response
- Retail Emergency Preparedness
- Sports Arenas And Stadiums
- Sports Venues
- Athlete Emergency Response
- Crowd Safety Systems
- Sports Venues
- Schools And Colleges
- Educational Institutions
- Student Safety Programs
- Campus Emergency Systems
- Educational Institutions
- Gymnasiums
- Fitness Centers
- Athletic Emergency Response
- Member Safety Equipment
- Fitness Centers
- Corporate Office Buildings
- Workplace Safety Systems
- Employee Emergency Preparedness
- Office Health Infrastructure
- Workplace Safety Systems
- Home Care Settings
- Residential Emergency Kits
- Home Cardiac Safety Devices
- Personal Emergency Preparedness
- Residential Emergency Kits
- Parks
- Public Recreational Areas
- Outdoor Emergency Access
- Community Safety Installations
- Public Recreational Areas
- Hotels And Restaurants
- Hospitality Sector Safety
- Guest Emergency Systems
- Tourist Safety Infrastructure
- Hospitality Sector Safety
- Others
- Public Institutions
- Government Buildings
- Event Venues
- Public Institutions
Automated External Defibrillator Market Segmented by Technology:
- Biphasic Waveform Technology
- Truncated Exponential Waveform
- Efficient Shock Delivery
- Reduced Energy Consumption
- Rectilinear Biphasic Waveform
- Improved Patient Safety
- Better Cardiac Conversion Rates
- Truncated Exponential Waveform
- Monophasic Waveform Technology
- Traditional Shock Systems
- Legacy AED Devices
- High Energy Output Systems
- Simple Circuit Defibrillation
- Basic Emergency Response Use
- Traditional Shock Systems
- Real Time ECG Analysis
- Automated Rhythm Detection
- Shock Advisory Algorithms
- AI Assisted Cardiac Monitoring
- Patient Signal Processing
- Noise Reduction Systems
- Improved Accuracy Detection
- Automated Rhythm Detection
- Voice And Visual Guidance Systems
- Step By Step Instructions
- Layperson Friendly Operation
- Emergency Response Support
- Smart Display Interfaces
- Real Time Feedback Screens
- Instructional Alerts
- Step By Step Instructions
- Connected AED Systems
- IoT Enabled Devices
- Remote Monitoring Systems
- Device Status Tracking
- Cloud Connected Platforms
- Emergency Network Integration
- Maintenance Alerts
- IoT Enabled Devices
Automated External Defibrillator Market Segmented by Distribution Channel:
- Direct Sales
- Manufacturer To Hospital Sales
- Bulk Institutional Procurement
- Government Tender Supply
- B2B Direct Contracts
- EMS And Healthcare Networks
- Long Term Supply Agreements
- Manufacturer To Hospital Sales
- Distributor And Wholesaler
- Medical Equipment Distributors
- Regional Supply Chains
- Multi Brand Distribution
- Healthcare Wholesalers
- Bulk Medical Device Supply
- Inventory Based Distribution
- Medical Equipment Distributors
- Retail Pharmacies And Medical Stores
- Pharmacy Chains
- Over The Counter AED Sales
- Emergency Device Availability
- Medical Equipment Stores
- Local Healthcare Retail
- Small Scale Purchases
- Pharmacy Chains
- Online Channels
- E Commerce Platforms
- Digital Medical Device Sales
- Home Delivery AED Kits
- Company Owned Websites
- Direct Brand Sales Portals
- Product Information And Ordering
- E Commerce Platforms
Automated External Defibrillator Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Research Sources and Bibliography
- 1. World Health Organization. (2025). WHO Global Health Observatory Data Repository. WHO.
- 2. USA Food and Drug Administration. (2025). FDA Medical Device Databases. FDA.
- 3. European Medicines Agency. (2025). EMA Annual Activity Report. EMA.
- 4. Centers for Disease Control and Prevention. (2025). CDC National Health Statistics. CDC.
- 5. Organisation for Economic Co-operation and Development. (2025). OECD Health Statistics. OECD.
- 6. International Medical Device Regulators Forum. (2025). IMDRF Regulatory Convergence Reports. IMDRF.
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with publication dates, URLs, and supporting data for all cited works.
This Report Answers
- Estimating the size of the market and revenue projections from 2026 to 2036.
- Segmentation by product, modality, end user, technology, distribution channel.
- Insights across more than 30 regional markets.
- Technology and product analysis across key segments.
- Assessment of the competitive landscape and company positioning.
- Identification of investment opportunities across product, modality, end user categories.
- Supply chain tracking and procurement pattern analysis.
- Delivery of data in PDF and Excel formats.
Frequently Asked Questions
What is the global market demand for Automated External Defibrillator Market in 2026?
In 2026, the global automated external defibrillator market is estimated at USD 1.46 billion.
How big will the Automated External Defibrillator Market be in 2036?
By 2036, the automated external defibrillator market is projected to reach USD 2.3 billion.
How much is demand for Automated External Defibrillator Market expected to grow between 2026 and 2036?
Between 2026 and 2036, the automated external defibrillator market is projected to expand at a CAGR of 4.7%.
Which product segment is expected to lead in the Automated External Defibrillator Market in 2026?
Automated External Defibrillator (AED) is expected to hold 72.0% of the product segment in 2026, reflecting concentrated end-use demand.
What is causing demand to rise in China?
China is expected to grow at 8.9% CAGR through 2036, supported by hospital infrastructure expansion and government health program funding.
What is causing demand to rise in India?
India is expected to grow at 8.3% CAGR through 2036, supported by healthcare infrastructure investment, Ayushman Bharat coverage, and growing private hospital networks.
What does this report mean by Automated External Defibrillator Market definition?
The automated external defibrillator market includes all commercially traded products and solutions categorized by product, modality, end user, technology, distribution channel, covering manufacturing, distribution, and end-use procurement across the 2026 to 2036 forecast period.
How does FMI make the Automated External Defibrillator Market forecast and validate it?
Forecasting models use a hybrid bottom-up and top-down approach, starting with verified transaction data and validating against global production statistics and manufacturer disclosures.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2026 to 2036
- Automated External Defibrillator (AED)
- Fully Automated Defibrillator
- Automatic Shock Delivery
- Minimal User Intervention
- Smart AED Systems
- Real Time ECG Analysis
- Advanced Safety Algorithms
- Semi Automated External Defibrillator (Semi-AED)
- Manual Shock Advised Devices
- Healthcare Professional Use
- Controlled Emergency Response
- User Guided Defibrillation
- Step By Step Voice Assistance
- Automated External Defibrillator (AED)
- Y-o-Y Growth Trend Analysis By Product , 2021 to 2025
- Absolute $ Opportunity Analysis By Product , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Modality
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Modality, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Modality, 2026 to 2036
- Public Access AED
- Public Safety AED Value (USD Million)s
- Airports And Transit Hubs
- Shopping And Commercial Spaces
- Layperson Operated Devices
- Voice Guided Operation
- Emergency Public Response Systems
- Professional Use AED
- Hospital Grade AED Systems
- Emergency Room Use
- ICU And Critical Care Value (USD Million)s
- Medical Staff Operated Devices
- Paramedic And EMS Use
- Public Access AED
- Y-o-Y Growth Trend Analysis By Modality, 2021 to 2025
- Absolute $ Opportunity Analysis By Modality, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- Emergency Departments
- Cardiac Arrest Response Value (USD Million)s
- Critical Care Support Systems
- Operation Theaters
- Surgical Emergency Backup
- Inpatient Cardiac Care
- Clinics
- Outpatient Clinics
- Cardiac Monitoring Value (USD Million)s
- Primary Care Emergency Support
- Long Term Care Centers
- Nursing Homes
- Elderly Care Facilities
- Chronic Patient Monitoring
- Ambulatory Surgical Centers
- Same Day Surgery Value (USD Million)s
- Post Operative Emergency Care
- Cardiac Safety Equipment
- Emergency Medical Services EMS
- Ambulance Services
- Pre Hospital Cardiac Response
- Field Emergency Care
- Urgent Care Centers
- Fast Response Clinics
- Minor Emergency Treatment
- Walk In Emergency Care
- Independent Catheterization Labs
- Cardiac Procedure Labs
- Interventional Cardiology Value (USD Million)s
- Procedure Backup Safety Systems
- Public Access
- Airports
- International Airports
- Passenger Terminal Safety Systems
- Emergency Response Stations
- Train Stations
- Railway Hubs
- Passenger Safety Installations
- Transit Emergency Equipment
- Shopping Malls
- Retail Complexes
- High Footfall Emergency Systems
- Public Safety Installations
- Hospitals
- Y-o-Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- Biphasic Waveform Technology
- Truncated Exponential Waveform
- Efficient Shock Delivery
- Reduced Energy Consumption
- Rectilinear Biphasic Waveform
- Improved Patient Safety
- Better Cardiac Conversion Rates
- Monophasic Waveform Technology
- Traditional Shock Systems
- Legacy AED Devices
- High Energy Output Systems
- Simple Circuit Defibrillation
- Basic Emergency Response Use
- Real Time ECG Analysis
- Automated Rhythm Detection
- Shock Advisory Algorithms
- AI Assisted Cardiac Monitoring
- Patient Signal Processing
- Noise Reduction Systems
- Voice And Visual Guidance Systems
- Step By Step Instructions
- Layperson Friendly Operation
- Emergency Response Support
- Smart Display Interfaces
- Real Time Feedback Screens
- Connected AED Systems
- IoT Enabled Devices
- Remote Monitoring Systems
- Device Status Tracking
- Cloud Connected Platforms
- Emergency Network Integration
- Biphasic Waveform Technology
- Y-o-Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Direct Sales
- Manufacturer To Hospital Sales
- Bulk Institutional Procurement
- Government Tender Supply
- B2B Direct Contracts
- EMS And Healthcare Networks
- Long Term Supply Agreements
- Distributor And Wholesaler
- Medical Equipment Distributors
- Regional Supply Chains
- Multi Brand Distribution
- Healthcare Wholesalers
- Bulk Medical Device Supply
- Retail Pharmacies And Medical Stores
- Pharmacy Chains
- Over The Counter AED Sales
- Emergency Device Availability
- Medical Equipment Stores
- Local Healthcare Retail
- Online Channels
- E Commerce Platforms
- Digital Medical Device Sales
- Home Delivery AED Kits
- Company Owned Websites
- Direct Brand Sales Portals
- Direct Sales
- Y-o-Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Modality
- By End User
- By Technology
- By Distribution Channel
- Competition Analysis
- Competition Deep Dive
- Zoll Medical Corporation
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Koninklijke Philips N.V.
- Stryker
- NIHON KOHDEN CORPORATION
- Shenzhen Mindray Bio-Medical Electronics Co., Ltd.
- FUKUDA DENSHI
- Schiller AG
- Metrax GmbH
- Avive Solutions, Inc.
- MEDIANA CO., Ltd.
- Progetti Medical Equipment Solutions
- Bexen
- Corpuls
- CU Medical Systems, Inc.
- Promed Technology Co., Limited
- INNOMED MEDICAL INC.
- Zoll Medical Corporation
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Modality, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Modality, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Modality, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Modality, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Modality, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Modality, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Modality, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Modality, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by Modality, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Modality, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Modality
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Technology
- Figure 15: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Distribution Channel
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Product
- Figure 32: North America Market Value Share and BPS Analysis by Modality, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Modality, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Modality
- Figure 35: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by End User
- Figure 38: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Technology
- Figure 41: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Distribution Channel
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Product
- Figure 48: Latin America Market Value Share and BPS Analysis by Modality, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Modality, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Modality
- Figure 51: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by End User
- Figure 54: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Technology
- Figure 57: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Product
- Figure 64: Western Europe Market Value Share and BPS Analysis by Modality, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Modality, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Modality
- Figure 67: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by End User
- Figure 70: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Technology
- Figure 73: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Product
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Modality, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Modality, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Modality
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by End User
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Product
- Figure 96: East Asia Market Value Share and BPS Analysis by Modality, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Modality, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Modality
- Figure 99: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by End User
- Figure 102: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Technology
- Figure 105: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Modality, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Modality, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Modality
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Modality, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Modality, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Modality
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

