The bilirubin analyzers market is valued at USD 555.4 million in 2026 and is projected to reach USD 1,004.0 million by 2036, expanding at a CAGR of 6.8% over the forecast period. Across the forecast horizon, purchasing behavior is shaped primarily by institutional procurement priorities rather than discretionary demand. Procurement decisions are driven by clinical accuracy, compliance with neonatal screening protocols, and reliability across high-volume maternity and NICU settings.
Strong buyer risk aversion is observed, as bilirubin measurement directly influences treatment decisions for newborns, leading hospitals to favor established vendors with proven validation and regulatory alignment. Vendor lock-in is reinforced through long equipment lifecycles, staff training investments, calibration standards, and integration with hospital information systems. Switching costs remain elevated due to retraining requirements, service contracts, and the need to revalidate diagnostic workflows, discouraging frequent supplier changes. As a result, competitive outcomes are influenced less by price competition and more by long-term trust, service continuity, and alignment with standardized neonatal care pathways.
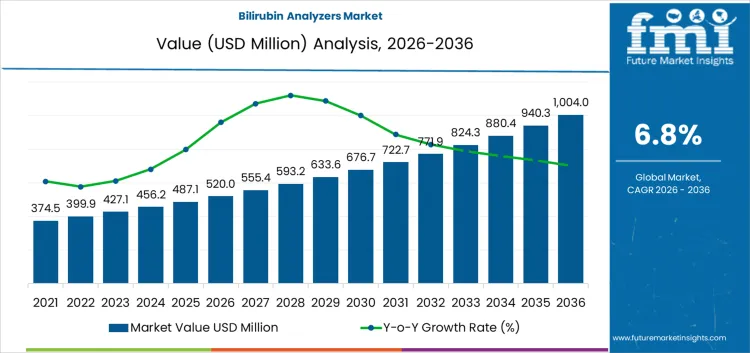
| Metric | Value |
|---|---|
| Industry Value (2026) | USD 555.4 Million |
| Forecast Value (2036) | USD 1,004.0 million |
| Forecast CAGR 2026 to 2036 | 6.80% |
Source: FMI analysis based on primary research and proprietary forecasting model
The global bilirubin analyzers market is advancing steadily, driven by the growing incidence of neonatal jaundice and the increasing emphasis on early diagnosis in neonatal and pediatric care. Bilirubin analyzers are widely used in hospitals, maternity centers, and diagnostic laboratories to measure bilirubin levels accurately, enabling timely clinical intervention and effective management of hyperbilirubinemia in newborns.
A key driver supporting market growth is the increasing adoption of non-invasive and point-of-care bilirubin measurement devices. Transcutaneous bilirubin analyzers are gaining traction as they allow quick screening without blood draws, reducing discomfort for infants and improving workflow efficiency for healthcare providers. This shift is particularly important in high-birth-rate regions and settings with limited laboratory infrastructure.
Technological advancements are further strengthening market expansion. Improvements in sensor accuracy, calibration stability, and device portability are enhancing reliability and ease of use. Integration with electronic medical records and digital monitoring systems is also improving data tracking and clinical decision-making. As neonatal care continues to prioritize early detection, patient comfort, and efficient diagnostics, the bilirubin analyzers market is expected to maintain consistent growth throughout the forecast period.
The bilirubin analyzers market is driven by the need for rapid, accurate, and minimally invasive neonatal jaundice screening. Transcutaneous bilirubin meters lead with a 58% share, reflecting strong clinical preference for non-invasive testing that reduces discomfort and enables frequent monitoring in newborns. These devices support faster decision-making in maternity wards and NICUs. On the sales channel side, hospital procurement accounts for 64% of demand, as hospitals remain the primary setting for neonatal screening and early intervention. Institutional purchasing emphasizes accuracy, clinical validation, and workflow compatibility.
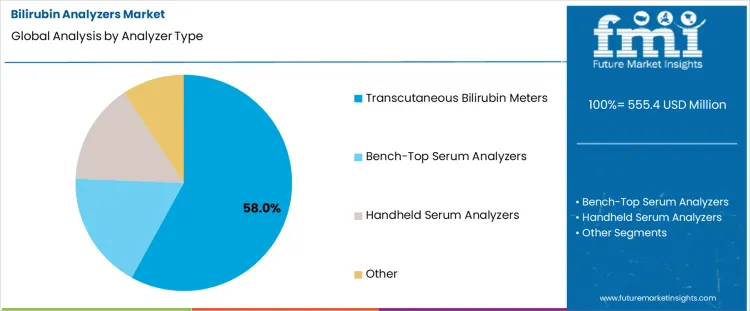
Transcutaneous bilirubin meters account for 58% of the bilirubin analyzers market due to their non-invasive nature, rapid measurement capability, and suitability for neonatal screening. These devices allow clinicians to assess bilirubin levels through the skin without requiring blood samples, reducing discomfort and infection risk in newborns. Their ease of use supports frequent monitoring in neonatal wards, maternity units, and NICUs, enabling early detection and management of hyperbilirubinemia. Transcutaneous meters also deliver immediate results, helping clinicians make faster treatment decisions and reduce reliance on laboratory-based testing. Advances in sensor accuracy, calibration algorithms, and portability have further improved clinical confidence in these devices. As neonatal care prioritizes minimally invasive diagnostics and efficient workflow, transcutaneous bilirubin meters continue to dominate analyzer adoption.
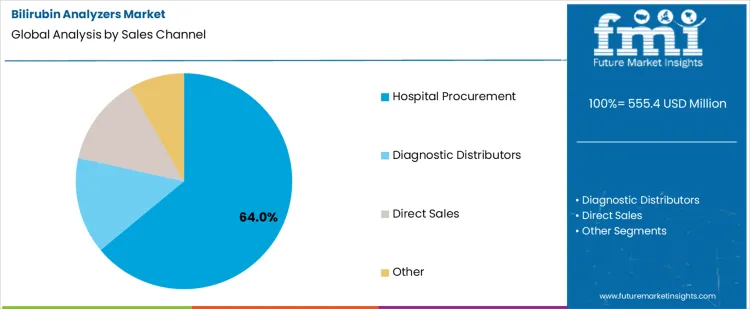
Hospital procurement represents 64% of total sales channel demand, making it the dominant purchasing route in the bilirubin analyzers market. Hospitals and maternity centers are primary points of care for newborn screening and jaundice management, requiring reliable and clinically validated diagnostic equipment. Procurement decisions are driven by compliance with clinical guidelines, device accuracy, and integration with existing neonatal care workflows. Hospitals also favor standardized equipment to support staff training, maintenance efficiency, and consistent patient outcomes. Bulk purchasing agreements and long-term supplier relationships further reinforce hospital-led procurement. Given the critical role of bilirubin monitoring in early neonatal care, hospitals remain the primary buyers, sustaining consistent demand for bilirubin analyzers across healthcare systems.
The bilirubin analyzers market is driven by the rising focus on early detection and management of jaundice and other liver-related disorders, particularly in newborns and adults. Bilirubin analyzers are diagnostic devices used in clinical laboratories, hospitals, and point-of-care settings to measure bilirubin levels in blood and support timely therapeutic decisions. Key market dynamics include increasing prevalence of liver diseases, expanding neonatal screening programs, and growing demand for rapid and accurate diagnostic tools. Advancements in analyzer technology, such as enhanced sensitivity, smaller sample volume requirements, and integration with laboratory information systems, are improving clinical workflows. Additionally, the rising emphasis on preventive healthcare and early intervention supports the adoption of bilirubin analyzers across diverse healthcare settings globally.
The bilirubin analyzers market is growing as healthcare providers prioritize early diagnosis and effective monitoring of bilirubin levels to prevent complications associated with hyperbilirubinemia, particularly in vulnerable populations like neonates. Improvements in assay precision and analyzer throughput are encouraging clinical adoption, enabling faster and more reliable results that support treatment decisions. Expanded access to healthcare services, including community clinics and rural diagnostic centers, increases the demand for point-of-care and portable bilirubin analyzers that offer rapid turnaround. Rising awareness of liver health, coupled with increasing rates of metabolic disorders and liver disease risk factors, is expanding the need for bilirubin testing. Integration of analyzers with electronic medical records also enhances operational efficiency and data management.
Key drivers shaping the bilirubin analyzers market include technological advancements that improve analyzer performance, such as enhanced optical sensors, automated calibration, and reduced sample requirements. These improvements support efficient laboratory workflows and reduce the burden on clinical staff. The increasing focus on neonatal care programs and routine newborn screening protocols is driving demand for bilirubin testing solutions that provide rapid, accurate results. Growing investment in diagnostic infrastructure in emerging regions is also expanding market reach, enabling broader adoption beyond major hospitals to community health centers. Partnerships between health systems and analyzer manufacturers enhance product availability and training, supporting effective utilization. As demand for early disease detection grows, bilirubin analyzers are becoming integral diagnostic tools for liver function evaluation and pediatric care.
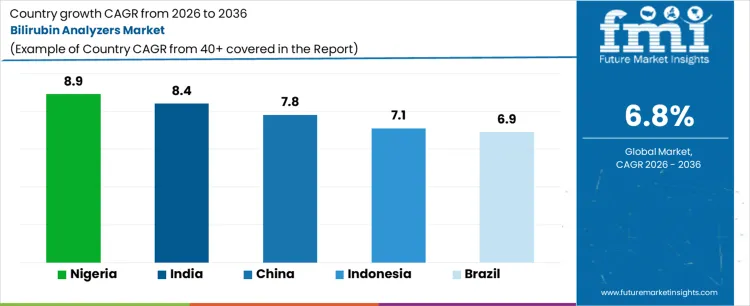
| Country | CAGR (%) |
|---|---|
| Nigeria | 8.9% |
| India | 8.4% |
| China | 7.8% |
| Indonesia | 7.1% |
| Brazil | 6.9% |
Source: FMI analysis based on primary research and proprietary forecasting model
The bilirubin analyzers market is expanding across emerging and developing healthcare systems as neonatal screening becomes a priority. Nigeria leads with an 8.9% CAGR, driven by high birth rates and increasing focus on reducing infant mortality. India follows at 8.4%, supported by expanding NICU infrastructure and institutional deliveries. China grows at 7.8%, benefiting from hospital modernization and standardized neonatal care protocols. Indonesia (7.1%) and Brazil (6.9%) show steady growth, driven by improving healthcare access and rising awareness of neonatal jaundice management.
The bilirubin analyzers market in Nigeria is growing at a CAGR of 8.9%, driven by increasing focus on neonatal care and rising awareness of early diagnosis of neonatal jaundice. Nigeria’s high birth rate and prevalence of newborn jaundice are key factors supporting demand for bilirubin testing solutions. Expansion of public and private maternity hospitals, along with gradual improvements in neonatal intensive care infrastructure, is increasing adoption of point-of-care bilirubin analyzers. Government and non-government healthcare initiatives aimed at reducing infant mortality are also improving access to basic diagnostic equipment. Portable and non-invasive bilirubin analyzers are particularly attractive in resource-constrained settings, where laboratory access may be limited. International health programs and donor-supported hospital upgrades further contribute to market growth. As neonatal screening becomes more widely adopted across healthcare facilities, demand for bilirubin analyzers in Nigeria is expected to rise steadily.
The bilirubin analyzers market in India is expanding at a CAGR of 8.4%, supported by rapid growth in neonatal healthcare services and increasing institutional deliveries. Expansion of NICUs across government and private hospitals is driving demand for reliable bilirubin measurement tools to manage neonatal jaundice. Rising awareness among healthcare professionals regarding early detection and monitoring is encouraging adoption of both transcutaneous and laboratory-based analyzers. Government programs focused on maternal and child health are improving diagnostic coverage in urban and semi-urban regions. Cost-effective, portable analyzers are gaining traction in smaller hospitals and clinics. In addition, domestic medical device manufacturing is improving affordability and availability. As India continues to strengthen neonatal screening practices and healthcare infrastructure, demand for bilirubin analyzers is expected to grow consistently.
The bilirubin analyzers market in China is growing at a CAGR of 7.8%, driven by modernization of hospital infrastructure and rising emphasis on neonatal health management. China’s expanding NICU capacity and improved standards of newborn care are increasing demand for accurate and rapid bilirubin testing. Hospitals are increasingly adopting non-invasive transcutaneous analyzers to reduce blood sampling and improve patient comfort. Strong domestic medical device manufacturing capabilities support widespread availability of advanced and mid-range analyzer systems. Increasing healthcare expenditure and government initiatives aimed at improving early childhood health outcomes further support adoption. In addition, growing parental awareness regarding neonatal jaundice and follow-up care is influencing clinical practices. As neonatal diagnostic protocols become more standardized, demand for bilirubin analyzers in China is expected to increase steadily.
The bilirubin analyzers market in Indonesia is expanding at a CAGR of 7.1%, supported by improving access to maternal and neonatal healthcare services. Growing numbers of hospital-based deliveries are increasing the need for newborn screening tools, including bilirubin analyzers. Public healthcare expansion and regional hospital upgrades are improving availability of diagnostic equipment. Portable and easy-to-use bilirubin analyzers are particularly important in Indonesia’s decentralized healthcare system, where access to centralized laboratories may be limited. International health initiatives focused on reducing neonatal complications are also contributing to adoption. Rising awareness among healthcare workers about early detection of jaundice is strengthening demand. As Indonesia continues to invest in improving neonatal outcomes, the market for bilirubin analyzers is expected to grow at a steady pace.
The bilirubin analyzers market in Brazil is growing at a CAGR of 6.9%, driven by gradual improvements in neonatal care and expansion of public healthcare facilities. Increased availability of NICUs and maternity wards is supporting adoption of bilirubin testing equipment. Healthcare providers are increasingly prioritizing early diagnosis of neonatal jaundice to reduce complications and length of hospital stays. Growth of private hospitals and diagnostic centers further supports market expansion. In addition, rising focus on standardizing neonatal screening protocols is encouraging investment in reliable analyzers. Availability of both non-invasive and laboratory-based systems allows adoption across different care settings. As Brazil continues to strengthen neonatal healthcare delivery and diagnostic practices, demand for bilirubin analyzers is expected to grow steadily.
Competition in the bilirubin analyzers market is driven by measurement accuracy, non-invasive capability, workflow efficiency, and suitability for neonatal care environments. Bilirubin monitoring is critical for early detection and management of neonatal jaundice, requiring reliable performance across varying skin tones, gestational ages, and clinical settings. Vendors differentiate through transcutaneous versus laboratory-based approaches, calibration stability, ease of use at the bedside, and integration with neonatal care workflows.
Dräger, Konica Minolta, Philips, Natus Medical, GE HealthCare, and Siemens Healthineers compete by combining clinical accuracy with neonatal-focused system design. Dräger emphasizes transcutaneous bilirubin measurement integrated into neonatal care environments, supporting quick screening without blood draws. Konica Minolta differentiates through optical sensing expertise, offering handheld transcutaneous analyzers known for consistency and ease of operation. Philips, GE HealthCare, and Siemens Healthineers leverage broad neonatal monitoring and diagnostics portfolios, positioning bilirubin analyzers as part of integrated care platforms that streamline screening, documentation, and clinical decision-making. Natus Medical focuses on neonatal-specific solutions, aligning bilirubin monitoring with hearing screening, phototherapy, and other newborn care technologies.
Roche, Abbott, Mindray, and Radiometer compete through laboratory-grade precision, system integration, and global reach. Roche and Abbott emphasize serum bilirubin testing accuracy through clinical chemistry analyzers used in centralized labs and point-of-care settings. Mindray competes with cost-effective analyzers designed for hospitals and maternity centers seeking reliable performance with lower capital investment. Radiometer differentiates through blood gas and critical care testing platforms that incorporate bilirubin measurement into broader neonatal diagnostics. Across the market, competitive advantage is shaped by accuracy, speed of results, non-invasive screening capability, ease of clinical integration, and the ability to support consistent neonatal jaundice management across diverse care settings.
| Attributes | Description |
|---|---|
| Quantitative Unit (2026) | USD Million |
| Analyzer Type | Transcutaneous Bilirubin Meters, Bench-Top Serum Analyzers, Handheld Serum Analyzers, Other |
| Sales Channel | Hospital Procurement, Diagnostic Distributors, Direct Sales, Other |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Rest of Asia Pacific, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, Rest of Europe, United States, Canada, Mexico, Brazil, Chile, Rest of Latin America, Kingdom of Saudi Arabia, Other GCC Countries, Turkey, South Africa, Other African Union, Rest of Middle East & Africa |
| Key Companies Profiled | Dräger, Konica Minolta, Philips, Natus Medical, GE HealthCare, Siemens Healthineers, Roche, Abbott, Mindray, Radiometer |
| Additional Attributes | Dollar sales by analyzer type and sales channel; regional market size and forecast analysis; growth outlook across major regions; adoption trends for bilirubin measurement technologies in neonatal and pediatric care; assessment of screening accuracy, workflow integration, and demand patterns across hospital procurement, distributor-led sales, and direct commercial channels. |
How big is the bilirubin analyzers market in 2026?
The global bilirubin analyzers market is estimated to be valued at USD 555.4 million in 2026.
What will be the size of bilirubin analyzers market in 2036?
The market size for the bilirubin analyzers market is projected to reach USD 1,072.3 million by 2036.
How much will be the bilirubin analyzers market growth between 2026 and 2036?
The bilirubin analyzers market is expected to grow at a 6.8% CAGR between 2026 and 2036.
What are the key product types in the bilirubin analyzers market?
The key product types in bilirubin analyzers market are transcutaneous bilirubin meters, bench-top serum analyzers, handheld serum analyzers and other.
Which sales channel segment to contribute significant share in the bilirubin analyzers market in 2026?
In terms of sales channel, hospital procurement segment to command 64.0% share in the bilirubin analyzers market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.