Bronchopulmonary Dysplasia Treatment Market
Bronchopulmonary Dysplasia Treatment Market Size and Share Forecast Outlook 2025 to 2035
Bronchopulmonary Dysplasia Treatment Market Size and Share Forecast Outlook 2025 to 2035
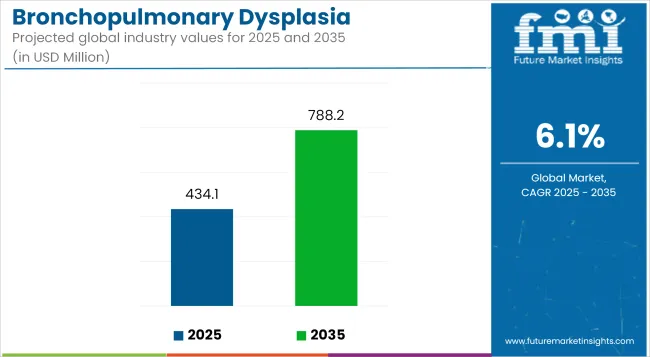
The Bronchopulmonary Dysplasia Treatment Market is anticipated to be valued at USD 434.1 million in 2025 and is expected to reach USD 788.2 million by 2035, registering a CAGR of 6.1%.
Quick Stats for Bronchopulmonary Dysplasia Treatment Market
- Industry Value (2025): USD 434.1 million
- Forecast Value (2035): USD 788.2 million
- Forecast CAGR: 6.1%
- Leading Segment in 2025: Bronchodilators (by drug type); Supplemental Oxygen Therapy (by therapy type)
- Key Growth Region: Asia-Pacific, North America
- Top Key Players: Chiesi Farmaceutici S.p.A., Medipost, Airway Therapeutics, Therabron Therapeutics, and AbbVie
| Metric | Value |
|---|---|
| Industry Size (2025E) | USD 434.1 million |
| Industry Value (2035F) | USD 788.2 million |
| CAGR (2025 to 2035) | 6.1% |
The Bronchopulmonary Dysplasia (BPD) Treatment Market is undergoing a structural evolution fueled by advancements in neonatal intensive care and a rising incidence of premature births globally. With the increasing survival rate of neonates born before 28 weeks gestation, demand for targeted therapies to manage chronic lung complications has significantly surged.
Recent annual reports from leading neonatal healthcare companies emphasize strategic investment in R&D for surfactant therapies, stem cell research, and oxygen delivery systems, all of which are enhancing patient care protocols. Industry analysts highlight the shift toward less invasive ventilation techniques and biologics as promising long-term growth avenues.
Furthermore, a multi-stakeholder approach including government policy support, specialized neonatal centers, and public-private partnerships is accelerating therapeutic innovation in this space. Over the coming years, market dynamics are expected to be driven by personalized medicine initiatives and increased clinical trials focusing on combination therapies for moderate to severe BPD cases.
Key manufacturers actively shaping the BPD treatment landscape include Chiesi Farmaceutici S.p.A., Medipost, Airway Therapeutics, Therabron Therapeutics, and AbbVie. These firms are consistently developing neonatal-focused biologics and respiratory support solutions to improve lung function in extremely low birth weight (ELBW) infants. A major driver behind market acceleration is the integration of stem cell-based therapies, with Medipost and Airway Therapeutics investing in clinical-stage development programs showing promising safety profiles.
In Oct 2024, Airway Therapeutics, Inc. announced it will launch a multinational Phase 3 clinical trial of zelpultide alfa (rhSP-D) for prevention of bronchopulmonary dysplasia (BPD) and minimization of resulting lung damage in preterm infants.
“The approval of this Phase 3 trial is based on the successful completion of our randomized blinded Phase 1b study in the USA and Europe, in which no dose limiting toxicities were found and indications of efficacy were observed, Zelpultide alfa would be the first preventive therapy to protect babies from BPD, which is a debilitating lung disease.” said Airway Chairman, CEO and Chief Medical Officer Marc Salzberg, M.D.
Analyzing Bronchopulmonary Dysplasia Treatment Market by Top Investment Segments
Bronchodilators Lead Due to Widespread Use in Airway Obstruction Management
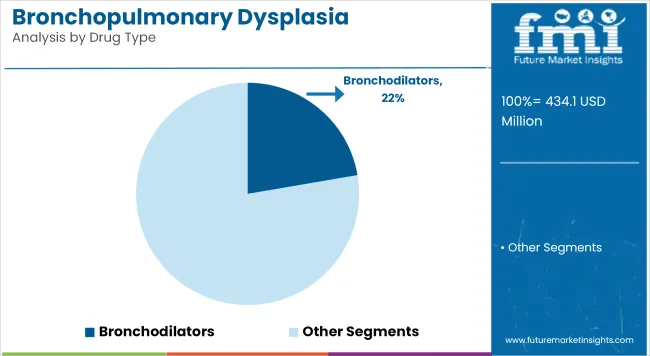
Bronchodilators are estimated to account for approximately 22.3% of total revenue in 2025, emerging as the dominant drug type in the BPD treatment landscape. Their adoption has been strongly supported by clinical protocols recommending bronchodilators for alleviating airway resistance in neonates with chronic lung disease.
The segment’s leadership has been reinforced by evidence-based inclusion in neonatal care guidelines, particularly in managing airflow limitation caused by pulmonary inflammation and smooth muscle constriction. A rising prevalence of premature births especially in low birth weight infants who are prone to lung underdevelopment has created consistent therapeutic demand.
Inhalation-based bronchodilator formulations have also been optimized for neonatal delivery, further enhancing clinical compliance. Healthcare providers have continued to favor bronchodilators for their rapid onset of action and ability to improve oxygenation, often serving as the first line of pharmacologic intervention. These factors, combined with strong physician familiarity and availability in hospital settings, have positioned bronchodilators as the most trusted and widely utilized drug category in BPD care protocols.
Supplemental Oxygen Therapy Leads Owing to Its Foundational Role in Neonatal Stabilization
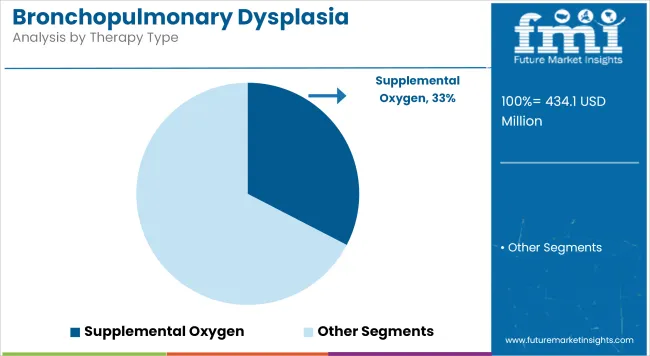
Holding an estimated 32.6% share of the BPD treatment market in 2025, supplemental oxygen therapy is considered the most widely applied therapeutic modality for managing respiratory insufficiency in neonates.
This dominance has been attributed to its critical role in stabilizing oxygen saturation levels during the acute and chronic phases of BPD. In neonatal intensive care units (NICUs), supplemental oxygen continues to be a foundational therapy used in both mechanical and non-invasive ventilation strategies.
The preference for this modality has been reinforced by its ability to immediately support gas exchange in underdeveloped lungs, thus preventing hypoxemia-related complications. Additionally, advancements in oxygen delivery systems such as high-flow nasal cannulas and pulse oximetry monitoring have improved therapy precision and minimized oxygen toxicity risk. The therapy’s broad applicability across all severity grades of BPD and its integration into both early intervention and post-discharge management protocols have further contributed to segment growth.
Hospitals Dominate Due to Availability of Advanced Neonatal Intensive Care Infrastructure
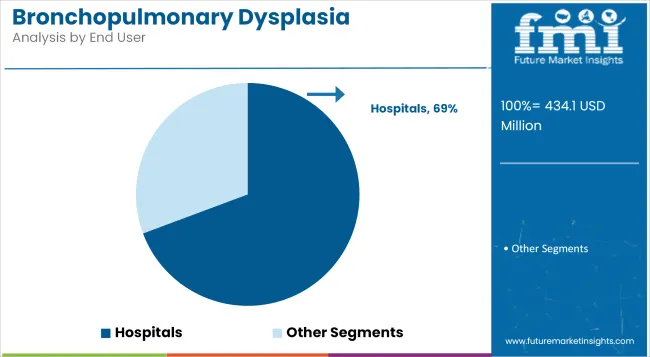
Hospitals are projected to capture nearly 69.3% of the market share in 2025, making them the leading end-user segment in the BPD treatment landscape. This dominance has been supported by the widespread presence of Level III and IV Neonatal Intensive Care Units (NICUs), which offer specialized respiratory support, surfactant administration, and ventilator-assisted therapies.
BPD treatment requires high-frequency monitoring, continuous oxygen titration, and access to pediatric pulmonologists facilities that are predominantly concentrated in hospital environments. The adoption of bundled neonatal care protocols and integrated clinical pathways in tertiary care hospitals has further strengthened the reliance on institutional settings for effective BPD management.
Additionally, hospitals are often the first point of care for premature births, which enables early initiation of evidence-based treatment plans. Technological capabilities, including access to advanced diagnostic tools and real-time respiratory monitoring, have positioned hospitals as the central hub for comprehensive BPD intervention and follow-up care.
Challenges and Opportunities
Challenges
Scalability limits of stem cell and nitric oxide therapies hinder broader adoption in developing markets
While stem cell and nitric oxide therapies are providing clinically favorable outcomes for severe cases of BPD, their application is limited by infrastructure and cost constraints. These modern interventions are largely available in tertiary care hospitals that have the capacity of specialized neonatal units but many low- and middle-income countries are not adequately catered for in this respect.
In areas where there is a high prevalence of preterm births, the lack of trained staff, storage systems, and continuous monitoring ability limits the real-world employment of such therapies. Moreover, operational restrictions in decentralized care environments impede the integration of these advanced options into routine neonatal practice.
This creates a dichotomy in the global market, with access in developing countries strongly focused on low-cost drug classes such as bronchodilators and diuretics. This gap is hampering the innovation-led expansion of the market, and is restricting treatment outcomes in high-need geographies.
Opportunities
Combinational therapy models drive long-term clinical and commercial value in BPD care
Newly emerging science on sequential and combinational treatment pathways are reshaping the BPD treatment landscape such that interventions become incremental and outcome-driven. Such regimens typically consist of a structured sequence of bronchodilators, surfactants and protein replacements, tailored to the fibrotic disease trajectory of the individual.
Such protocols have had positive impacts in terms of reducing mechanical ventilation dependency, promoting alveolar development and inflammation in both preventative and established BPD. Institutions with advanced neonatal research capabilities are pursuing testing of these strategies, with the advancement of fixed-dose combination formulations in clinical evaluation.
Growing interest from pharmaceutical players in these types of multi-action therapies are also setting the stage for commercial-scale solutions. With its unique focus on personalized, outcome-based treatment architecture, not only does this increase the chances of recovery but it also slashes hospitalization time and costs, realising long term clinical and economic value at both high-resource and tier two/three healthcare settings.
Country Wise Outlook
United States
Market Outlook
The United States leads the geographical split of the global market for bronchopulmonary dysplasia (BPD) treatment due to the high rate of preterm births and an established neonatal intensive care system as well as a robust pipeline of new innovative therapies for chronic lung disease in neonates.
Clinical management entails the application of oxygen therapy, mechanical ventilation, surfactants, corticosteroids, and, more recently, investigational biologics, with growing emphasis on personalized and preventive approaches.
Market Growth Factors
- High Survival Rate of Extremely Preterm Infants: Leading to increased BPD diagnosis and care needs.
- Advanced NICU Infrastructure with Nationwide Access to Surfactant and Ventilation Support: Standardizes treatment.
- Ongoing Trials of Regenerative Therapies (e.g., stem cells, IGF-1): Expanding therapeutic landscape.
- Established Neonatal Health Guidelines (AAP, NIH): Driving adoption of evidence-based interventions.
- Widespread Use of Postnatal Corticosteroids and Non-Invasive Ventilation Techniques: Improving outcomes.
Market Forecast
| Country | CAGR (2025 to 2035) |
|---|---|
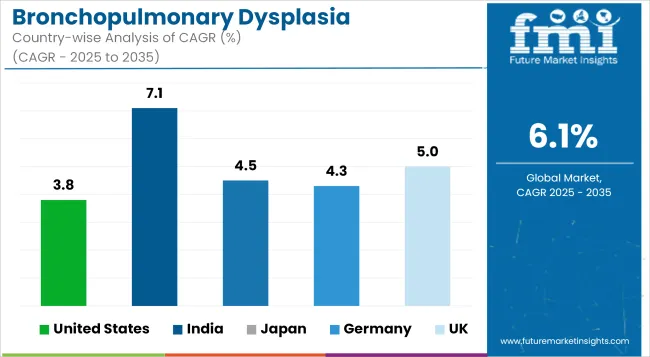
| United States | 3.8% |
India
Market Outlook
India’s BPD treatment market is growingat an increased pace owing to rising burden of preterm births, better NICU infrastructure, and rising utilization of standardized neonatal care protocols in both private and public hospitals. Rural areas have limited resources, but urban tertiary centers are up-taking non-invasive ventilation, surfactant therapy, and postnatal corticosteroids as part of the government and NGO initiatives.
Market Growth Factors
- High Preterm Birth Rate and Low Birth Weight Incidence: Sustains long-term BPD treatment demand.
- Government-Supported Neonatal Care Programs (e.g., FBNC): Improving treatment access.
- Rising Adoption of CPAP, HFNC, and Less Invasive Surfactant Methods: Enhancing survival and reducing severity.
- NGO and Donor Involvement in NICU Modernization: Facilitating access to modern therapies.
- Emerging Clinical Trials in Antioxidant and Immunomodulatory Agents: Expanding treatment horizon.
Market Forecast
| Country | CAGR (2025 to 2035) |
|---|---|
| India | 7.1% |
Japan
Market Outlook
The Japan BPD treatment market features advanced technology, particularly in reducing ventilator use, enhancing maturation of the lung, and incorporating biomarker-based monitoring. Despite a low and decreasing birth rate and an increasingly aging population, Japan continues to pursue high-quality perinatal care, leading to improved survival of such infants born prematurely with BPD.
Market Growth Factors
- Strong NICU Capacity and Neonatal Survival Rates: Drives demand for chronic lung disease management.
- Clinical Use of Caffeine, Surfactant, and Antioxidants: Well-established in first-line protocols.
- Emphasis on Early Weaning from Oxygen and Mechanical Ventilation: Aligned with global best practices.
- Government-Funded Trials in Growth Factors and Stem Cell Therapy: Boosting pipeline innovation.
- Integration of Digital Lung Function Monitoring in NICUs: Supports individualized respiratory care.
Market Forecast
| Country | CAGR (2025 to 2035) |
|---|---|
| Japan | 4.5% |
Germany
Market Outlook
The BPD treatment market in Germany is more advanced and systematized owing to infrastructure with well-equipped perinatal centers, strong emphasis on standardized protocols in NICU, and improved clinical research on lung-protective ventilation and pharmacological approaches. Antenatal steroids, surfactant therapy, and caffeine citrate are used widely and hospitals are working to reduce ventilator-induced lung injury.
Market Growth Factors
- High Level of NICU Care and Universal Neonatal Screening: Enables early diagnosis and intervention.
- Proactive Use of Lung-Protective Strategies in Preterm Infants: Reducing BPD severity.
- Government Support for Neonatal Research and Innovation: Encouraging biologic and stem cell trials.
- Strict Adherence to European Guidelines on Neonatal Respiratory Care: Ensures consistent clinical practice.
- Use of Inhaled Nitric Oxide and Minimally Invasive Surfactant Therapy (MIST): Gaining ground in tertiary centers.
Market Forecast
| Country | CAGR (2025 to 2035) |
|---|---|
| Germany | 4.3% |
UK
Market Outlook
The UK BPD treatment market is growing at a vigorous pace due to allied NHS neonatal programs, a robust focus on premature infant outcome, and the in-clinic integration of non-invasive respiratory support, pharmacotherapy, and long-term pulmonary monitoring. This adds to therapeutic diversification and also falls in line with advances in neonatal care and ongoing trials of anti-inflammatory and growth factor-based therapies.
Market Growth Factors
- Government Commitment to Neonatal Health Outcomes: Supports funding and infrastructure development.
- High Adoption of CPAP and Less Invasive Surfactant Administration (LISA): Reducing ventilation-related injuries.
- Use of National Neonatal Audit Programme (NNAP) Data for Quality Improvement: Strengthens treatment pathways.
- Increasing Clinical Research in Postnatal Steroid Use and Bronchodilators: Enhancing evidence-based care.
- Rising Demand for Home Oxygen Therapy and Community Pulmonary Support: Improves long-term follow-up.
Market Forecast
| Country | CAGR (2025 to 2035) |
|---|---|
| UK | 5.0% |
Competitive Outlook
The bronchopulmonary dysplasia (BPD) treatment market is being propelled by the growing frequency of preterm births and advancements made in neonatal intensive care. Bronchopulmonary dysplasia, or BPD, is a chronic lung disease that occurs in premature infants and often necessitates prolonged respiratory support and anti-inflammatory therapies.
Cell therapies for respiratory diseases and surfactant replacement innovations further bolster the market, along with a robust biologics pipeline. This is the most important aspect along with the types of stakeholders involved, such as pharmaceutical companies, medical device manufacturers, healthcare organization focusing on NICU facilities.
Companies
- Chiesi Farmaceutici S.p.A.,
- Medipost,
- Airway Therapeutics,
- Therabron Therapeutics,
- AbbVie
- Abbott
- GSK Plc.
- Pfizer Ltd.
- Alkem Laboratories
- Sun Pharmaceuticals
Key Segments
By Drug Type:
The market is segmented into Bronchodilators, Diuretics, Antibiotics, Steroids, Immunomodulators, and Surfactant Homeostasis.
By Therapy Type:
The market is segmented into Nitric Oxide Therapy, Protein Replacement Therapy, Stem Cell Therapy, and Supplemental Oxygen.
By End User:
The market is segmented into Hospitals, Nursing Homes, and Critical Care Centers.
By Region:
The Bronchopulmonary Dysplasia Treatment Market is analyzed across North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, and the Middle East & Africa.
Frequently Asked Questions
What was the overall size of the Bronchopulmonary Dysplasia Treatment Market in 2025?
The overall market size for bronchopulmonary dysplasia treatment market was USD 434.1 million in 2025.
How big is the Bronchopulmonary Dysplasia Treatment Market expected in 2035?
The bronchopulmonary dysplasia treatment market is expected to reach USD 788.2 million in 2035.
What will drive the demand for Bronchopulmonary Dysplasia Treatment Market during the forecast period?
Revolutionizing neonatal care with cutting-edge therapies for Bronchopulmonary Dysplasia recovery.
List the top 5 Key players of the Bronchopulmonary Dysplasia Treatment Market?
The top key players that drives the development of bronchopulmonary Chiesi Farmaceutici S.p.A., AbbVie Inc., Medtronic plc, Merck & Co., Inc. and Philips Healthcare
Which segment in drug type is expected to lead in Bronchopulmonary Dysplasia Treatment Market?
Bronchodilators is expected to command significant share over the assessment period.
Table of Content
- Executive Summary
- Industry Introduction, including Taxonomy and Market Definition
- Market Trends and Success Factors, including Macro-economic Factors, Market Dynamics, and Recent Industry Developments
- Global Market Demand Analysis 2020 to 2024 and Forecast 2025 to 2035, including Historical Analysis and Future Projections
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035
- By Drug Type
- By Therapy Type
- By End User
- By Region
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, by Drug Type
- Bronchodilators
- Diuretics
- Antibiotics
- Steroids
- Immunomodulators
- Surfactant Homeostasis
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, by Therapy Type
- Nitric Oxide Therapy
- Protein replacement therapy
- Stem Cell Therapy
- Supplemental Oxygen
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, by End User
- Hospitals
- Nursing Homes
- Critical Care Centers
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, by Region
- North America
- Latin America
- East Asia
- South Asia and Pacific
- Western Europe
- Eastern Europe
- Middle East and Africa
- North America Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Latin America Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- East Asia Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- South Asia & Pacific Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Western Europe Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Eastern Europe Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Middle East and Africa Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Sales Forecast 2025 to 2035 by Drug Type, Therapy Type, and End User for 30 Countries
- Competition Outlook, including Market Structure Analysis, Company Share Analysis by Key Players, and Competition Dashboard
- Company Profile
- Chiesi Farmaceutici S.p.A.
- AbbVie Inc.
- Medtronic plc
- Merck & Co., Inc.
- Philips Healthcare
- Drägerwerk AG & Co. KGaA
- Viatris Inc. (Mylan)
- Mallinckrodt Pharmaceuticals
- Windtree Therapeutics
- Vapotherm Inc.
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2020 to 2035
- Table 2: Global Market Value (USD Million) Forecast by Drug Type, 2020 to 2035
- Table 3: Global Market Value (USD Million) Forecast by Therapy Type, 2020 to 2035
- Table 4: Global Market Value (USD Million) Forecast by End User, 2020 to 2035
- Table 5: North America Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 6: North America Market Value (USD Million) Forecast by Drug Type, 2020 to 2035
- Table 7: North America Market Value (USD Million) Forecast by Therapy Type, 2020 to 2035
- Table 8: North America Market Value (USD Million) Forecast by End User, 2020 to 2035
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 10: Latin America Market Value (USD Million) Forecast by Drug Type, 2020 to 2035
- Table 11: Latin America Market Value (USD Million) Forecast by Therapy Type, 2020 to 2035
- Table 12: Latin America Market Value (USD Million) Forecast by End User, 2020 to 2035
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 14: Western Europe Market Value (USD Million) Forecast by Drug Type, 2020 to 2035
- Table 15: Western Europe Market Value (USD Million) Forecast by Therapy Type, 2020 to 2035
- Table 16: Western Europe Market Value (USD Million) Forecast by End User, 2020 to 2035
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Drug Type, 2020 to 2035
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Therapy Type, 2020 to 2035
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End User, 2020 to 2035
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 22: South Asia and Pacific Market Value (USD Million) Forecast by Drug Type, 2020 to 2035
- Table 23: South Asia and Pacific Market Value (USD Million) Forecast by Therapy Type, 2020 to 2035
- Table 24: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2020 to 2035
- Table 25: East Asia Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 26: East Asia Market Value (USD Million) Forecast by Drug Type, 2020 to 2035
- Table 27: East Asia Market Value (USD Million) Forecast by Therapy Type, 2020 to 2035
- Table 28: East Asia Market Value (USD Million) Forecast by End User, 2020 to 2035
- Table 29: Middle East and Africa Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 30: Middle East and Africa Market Value (USD Million) Forecast by Drug Type, 2020 to 2035
- Table 31: Middle East and Africa Market Value (USD Million) Forecast by Therapy Type, 2020 to 2035
- Table 32: Middle East and Africa Market Value (USD Million) Forecast by End User, 2020 to 2035
List of Figures
- Figure 1: Global Market Value (USD Million) by Drug Type, 2025 to 2035
- Figure 2: Global Market Value (USD Million) by Therapy Type, 2025 to 2035
- Figure 3: Global Market Value (USD Million) by End User, 2025 to 2035
- Figure 4: Global Market Value (USD Million) by Region, 2025 to 2035
- Figure 5: Global Market Value (USD Million) Analysis by Region, 2020 to 2035
- Figure 6: Global Market Value Share (%) and BPS Analysis by Region, 2025 to 2035
- Figure 7: Global Market Y-o-Y Growth (%) Projections by Region, 2025 to 2035
- Figure 8: Global Market Value (USD Million) Analysis by Drug Type, 2020 to 2035
- Figure 9: Global Market Value Share (%) and BPS Analysis by Drug Type, 2025 to 2035
- Figure 10: Global Market Y-o-Y Growth (%) Projections by Drug Type, 2025 to 2035
- Figure 11: Global Market Value (USD Million) Analysis by Therapy Type, 2020 to 2035
- Figure 12: Global Market Value Share (%) and BPS Analysis by Therapy Type, 2025 to 2035
- Figure 13: Global Market Y-o-Y Growth (%) Projections by Therapy Type, 2025 to 2035
- Figure 14: Global Market Value (USD Million) Analysis by End User, 2020 to 2035
- Figure 15: Global Market Value Share (%) and BPS Analysis by End User, 2025 to 2035
- Figure 16: Global Market Y-o-Y Growth (%) Projections by End User, 2025 to 2035
- Figure 17: Global Market Attractiveness by Drug Type, 2025 to 2035
- Figure 18: Global Market Attractiveness by Therapy Type, 2025 to 2035
- Figure 19: Global Market Attractiveness by End User, 2025 to 2035
- Figure 20: Global Market Attractiveness by Region, 2025 to 2035
- Figure 21: North America Market Value (USD Million) by Drug Type, 2025 to 2035
- Figure 22: North America Market Value (USD Million) by Therapy Type, 2025 to 2035
- Figure 23: North America Market Value (USD Million) by End User, 2025 to 2035
- Figure 24: North America Market Value (USD Million) by Country, 2025 to 2035
- Figure 25: North America Market Value (USD Million) Analysis by Country, 2020 to 2035
- Figure 26: North America Market Value Share (%) and BPS Analysis by Country, 2025 to 2035
- Figure 27: North America Market Y-o-Y Growth (%) Projections by Country, 2025 to 2035
- Figure 28: North America Market Value (USD Million) Analysis by Drug Type, 2020 to 2035
- Figure 29: North America Market Value Share (%) and BPS Analysis by Drug Type, 2025 to 2035
- Figure 30: North America Market Y-o-Y Growth (%) Projections by Drug Type, 2025 to 2035
- Figure 31: North America Market Value (USD Million) Analysis by Therapy Type, 2020 to 2035
- Figure 32: North America Market Value Share (%) and BPS Analysis by Therapy Type, 2025 to 2035
- Figure 33: North America Market Y-o-Y Growth (%) Projections by Therapy Type, 2025 to 2035
- Figure 34: North America Market Value (USD Million) Analysis by End User, 2020 to 2035
- Figure 35: North America Market Value Share (%) and BPS Analysis by End User, 2025 to 2035
- Figure 36: North America Market Y-o-Y Growth (%) Projections by End User, 2025 to 2035
- Figure 37: North America Market Attractiveness by Drug Type, 2025 to 2035
- Figure 38: North America Market Attractiveness by Therapy Type, 2025 to 2035
- Figure 39: North America Market Attractiveness by End User, 2025 to 2035
- Figure 40: North America Market Attractiveness by Country, 2025 to 2035
- Figure 41: Latin America Market Value (USD Million) by Drug Type, 2025 to 2035
- Figure 42: Latin America Market Value (USD Million) by Therapy Type, 2025 to 2035
- Figure 43: Latin America Market Value (USD Million) by End User, 2025 to 2035
- Figure 44: Latin America Market Value (USD Million) by Country, 2025 to 2035
- Figure 45: Latin America Market Value (USD Million) Analysis by Country, 2020 to 2035
- Figure 46: Latin America Market Value Share (%) and BPS Analysis by Country, 2025 to 2035
- Figure 47: Latin America Market Y-o-Y Growth (%) Projections by Country, 2025 to 2035
- Figure 48: Latin America Market Value (USD Million) Analysis by Drug Type, 2020 to 2035
- Figure 49: Latin America Market Value Share (%) and BPS Analysis by Drug Type, 2025 to 2035
- Figure 50: Latin America Market Y-o-Y Growth (%) Projections by Drug Type, 2025 to 2035
- Figure 51: Latin America Market Value (USD Million) Analysis by Therapy Type, 2020 to 2035
- Figure 52: Latin America Market Value Share (%) and BPS Analysis by Therapy Type, 2025 to 2035
- Figure 53: Latin America Market Y-o-Y Growth (%) Projections by Therapy Type, 2025 to 2035
- Figure 54: Latin America Market Value (USD Million) Analysis by End User, 2020 to 2035
- Figure 55: Latin America Market Value Share (%) and BPS Analysis by End User, 2025 to 2035
- Figure 56: Latin America Market Y-o-Y Growth (%) Projections by End User, 2025 to 2035
- Figure 57: Latin America Market Attractiveness by Drug Type, 2025 to 2035
- Figure 58: Latin America Market Attractiveness by Therapy Type, 2025 to 2035
- Figure 59: Latin America Market Attractiveness by End User, 2025 to 2035
- Figure 60: Latin America Market Attractiveness by Country, 2025 to 2035
- Figure 61: Western Europe Market Value (USD Million) by Drug Type, 2025 to 2035
- Figure 62: Western Europe Market Value (USD Million) by Therapy Type, 2025 to 2035
- Figure 63: Western Europe Market Value (USD Million) by End User, 2025 to 2035
- Figure 64: Western Europe Market Value (USD Million) by Country, 2025 to 2035
- Figure 65: Western Europe Market Value (USD Million) Analysis by Country, 2020 to 2035
- Figure 66: Western Europe Market Value Share (%) and BPS Analysis by Country, 2025 to 2035
- Figure 67: Western Europe Market Y-o-Y Growth (%) Projections by Country, 2025 to 2035
- Figure 68: Western Europe Market Value (USD Million) Analysis by Drug Type, 2020 to 2035
- Figure 69: Western Europe Market Value Share (%) and BPS Analysis by Drug Type, 2025 to 2035
- Figure 70: Western Europe Market Y-o-Y Growth (%) Projections by Drug Type, 2025 to 2035
- Figure 71: Western Europe Market Value (USD Million) Analysis by Therapy Type, 2020 to 2035
- Figure 72: Western Europe Market Value Share (%) and BPS Analysis by Therapy Type, 2025 to 2035
- Figure 73: Western Europe Market Y-o-Y Growth (%) Projections by Therapy Type, 2025 to 2035
- Figure 74: Western Europe Market Value (USD Million) Analysis by End User, 2020 to 2035
- Figure 75: Western Europe Market Value Share (%) and BPS Analysis by End User, 2025 to 2035
- Figure 76: Western Europe Market Y-o-Y Growth (%) Projections by End User, 2025 to 2035
- Figure 77: Western Europe Market Attractiveness by Drug Type, 2025 to 2035
- Figure 78: Western Europe Market Attractiveness by Therapy Type, 2025 to 2035
- Figure 79: Western Europe Market Attractiveness by End User, 2025 to 2035
- Figure 80: Western Europe Market Attractiveness by Country, 2025 to 2035
- Figure 81: Eastern Europe Market Value (USD Million) by Drug Type, 2025 to 2035
- Figure 82: Eastern Europe Market Value (USD Million) by Therapy Type, 2025 to 2035
- Figure 83: Eastern Europe Market Value (USD Million) by End User, 2025 to 2035
- Figure 84: Eastern Europe Market Value (USD Million) by Country, 2025 to 2035
- Figure 85: Eastern Europe Market Value (USD Million) Analysis by Country, 2020 to 2035
- Figure 86: Eastern Europe Market Value Share (%) and BPS Analysis by Country, 2025 to 2035
- Figure 87: Eastern Europe Market Y-o-Y Growth (%) Projections by Country, 2025 to 2035
- Figure 88: Eastern Europe Market Value (USD Million) Analysis by Drug Type, 2020 to 2035
- Figure 89: Eastern Europe Market Value Share (%) and BPS Analysis by Drug Type, 2025 to 2035
- Figure 90: Eastern Europe Market Y-o-Y Growth (%) Projections by Drug Type, 2025 to 2035
- Figure 91: Eastern Europe Market Value (USD Million) Analysis by Therapy Type, 2020 to 2035
- Figure 92: Eastern Europe Market Value Share (%) and BPS Analysis by Therapy Type, 2025 to 2035
- Figure 93: Eastern Europe Market Y-o-Y Growth (%) Projections by Therapy Type, 2025 to 2035
- Figure 94: Eastern Europe Market Value (USD Million) Analysis by End User, 2020 to 2035
- Figure 95: Eastern Europe Market Value Share (%) and BPS Analysis by End User, 2025 to 2035
- Figure 96: Eastern Europe Market Y-o-Y Growth (%) Projections by End User, 2025 to 2035
- Figure 97: Eastern Europe Market Attractiveness by Drug Type, 2025 to 2035
- Figure 98: Eastern Europe Market Attractiveness by Therapy Type, 2025 to 2035
- Figure 99: Eastern Europe Market Attractiveness by End User, 2025 to 2035
- Figure 100: Eastern Europe Market Attractiveness by Country, 2025 to 2035
- Figure 101: South Asia and Pacific Market Value (USD Million) by Drug Type, 2025 to 2035
- Figure 102: South Asia and Pacific Market Value (USD Million) by Therapy Type, 2025 to 2035
- Figure 103: South Asia and Pacific Market Value (USD Million) by End User, 2025 to 2035
- Figure 104: South Asia and Pacific Market Value (USD Million) by Country, 2025 to 2035
- Figure 105: South Asia and Pacific Market Value (USD Million) Analysis by Country, 2020 to 2035
- Figure 106: South Asia and Pacific Market Value Share (%) and BPS Analysis by Country, 2025 to 2035
- Figure 107: South Asia and Pacific Market Y-o-Y Growth (%) Projections by Country, 2025 to 2035
- Figure 108: South Asia and Pacific Market Value (USD Million) Analysis by Drug Type, 2020 to 2035
- Figure 109: South Asia and Pacific Market Value Share (%) and BPS Analysis by Drug Type, 2025 to 2035
- Figure 110: South Asia and Pacific Market Y-o-Y Growth (%) Projections by Drug Type, 2025 to 2035
- Figure 111: South Asia and Pacific Market Value (USD Million) Analysis by Therapy Type, 2020 to 2035
- Figure 112: South Asia and Pacific Market Value Share (%) and BPS Analysis by Therapy Type, 2025 to 2035
- Figure 113: South Asia and Pacific Market Y-o-Y Growth (%) Projections by Therapy Type, 2025 to 2035
- Figure 114: South Asia and Pacific Market Value (USD Million) Analysis by End User, 2020 to 2035
- Figure 115: South Asia and Pacific Market Value Share (%) and BPS Analysis by End User, 2025 to 2035
- Figure 116: South Asia and Pacific Market Y-o-Y Growth (%) Projections by End User, 2025 to 2035
- Figure 117: South Asia and Pacific Market Attractiveness by Drug Type, 2025 to 2035
- Figure 118: South Asia and Pacific Market Attractiveness by Therapy Type, 2025 to 2035
- Figure 119: South Asia and Pacific Market Attractiveness by End User, 2025 to 2035
- Figure 120: South Asia and Pacific Market Attractiveness by Country, 2025 to 2035
- Figure 121: East Asia Market Value (USD Million) by Drug Type, 2025 to 2035
- Figure 122: East Asia Market Value (USD Million) by Therapy Type, 2025 to 2035
- Figure 123: East Asia Market Value (USD Million) by End User, 2025 to 2035
- Figure 124: East Asia Market Value (USD Million) by Country, 2025 to 2035
- Figure 125: East Asia Market Value (USD Million) Analysis by Country, 2020 to 2035
- Figure 126: East Asia Market Value Share (%) and BPS Analysis by Country, 2025 to 2035
- Figure 127: East Asia Market Y-o-Y Growth (%) Projections by Country, 2025 to 2035
- Figure 128: East Asia Market Value (USD Million) Analysis by Drug Type, 2020 to 2035
- Figure 129: East Asia Market Value Share (%) and BPS Analysis by Drug Type, 2025 to 2035
- Figure 130: East Asia Market Y-o-Y Growth (%) Projections by Drug Type, 2025 to 2035
- Figure 131: East Asia Market Value (USD Million) Analysis by Therapy Type, 2020 to 2035
- Figure 132: East Asia Market Value Share (%) and BPS Analysis by Therapy Type, 2025 to 2035
- Figure 133: East Asia Market Y-o-Y Growth (%) Projections by Therapy Type, 2025 to 2035
- Figure 134: East Asia Market Value (USD Million) Analysis by End User, 2020 to 2035
- Figure 135: East Asia Market Value Share (%) and BPS Analysis by End User, 2025 to 2035
- Figure 136: East Asia Market Y-o-Y Growth (%) Projections by End User, 2025 to 2035
- Figure 137: East Asia Market Attractiveness by Drug Type, 2025 to 2035
- Figure 138: East Asia Market Attractiveness by Therapy Type, 2025 to 2035
- Figure 139: East Asia Market Attractiveness by End User, 2025 to 2035
- Figure 140: East Asia Market Attractiveness by Country, 2025 to 2035
- Figure 141: Middle East and Africa Market Value (USD Million) by Drug Type, 2025 to 2035
- Figure 142: Middle East and Africa Market Value (USD Million) by Therapy Type, 2025 to 2035
- Figure 143: Middle East and Africa Market Value (USD Million) by End User, 2025 to 2035
- Figure 144: Middle East and Africa Market Value (USD Million) by Country, 2025 to 2035
- Figure 145: Middle East and Africa Market Value (USD Million) Analysis by Country, 2020 to 2035
- Figure 146: Middle East and Africa Market Value Share (%) and BPS Analysis by Country, 2025 to 2035
- Figure 147: Middle East and Africa Market Y-o-Y Growth (%) Projections by Country, 2025 to 2035
- Figure 148: Middle East and Africa Market Value (USD Million) Analysis by Drug Type, 2020 to 2035
- Figure 149: Middle East and Africa Market Value Share (%) and BPS Analysis by Drug Type, 2025 to 2035
- Figure 150: Middle East and Africa Market Y-o-Y Growth (%) Projections by Drug Type, 2025 to 2035
- Figure 151: Middle East and Africa Market Value (USD Million) Analysis by Therapy Type, 2020 to 2035
- Figure 152: Middle East and Africa Market Value Share (%) and BPS Analysis by Therapy Type, 2025 to 2035
- Figure 153: Middle East and Africa Market Y-o-Y Growth (%) Projections by Therapy Type, 2025 to 2035
- Figure 154: Middle East and Africa Market Value (USD Million) Analysis by End User, 2020 to 2035
- Figure 155: Middle East and Africa Market Value Share (%) and BPS Analysis by End User, 2025 to 2035
- Figure 156: Middle East and Africa Market Y-o-Y Growth (%) Projections by End User, 2025 to 2035
- Figure 157: Middle East and Africa Market Attractiveness by Drug Type, 2025 to 2035
- Figure 158: Middle East and Africa Market Attractiveness by Therapy Type, 2025 to 2035
- Figure 159: Middle East and Africa Market Attractiveness by End User, 2025 to 2035
- Figure 160: Middle East and Africa Market Attractiveness by Country, 2025 to 2035
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

