The colorimetric fructosamine assay kits market is projected to reach USD 298.2 million in 2026, with an anticipated value of USD 433.0 million by 2036, reflecting a compound annual growth rate (CAGR) of 3.80%. This assay kit is crucial for laboratories aiming to assess short-term glycemic control, especially in scenarios where HbA1c measurements may be unreliable or not readily accessible. The choice of method depends heavily on factors such as the compatibility of the analyzer, turnaround times, and the assay’s resistance to interferences. Once a laboratory has validated its reagent system, switching to an alternative method necessitates conducting parallel runs and maintaining method comparison files.
When selecting assay kits, buyers prioritize consistency across batches, stability of calibration, and adequate shelf life, rather than the breadth of items available in catalogs. Laboratories prefer kits that can be integrated into existing chemistry analyzers with minimal additional handling.
The dynamics of reagent supply are closely tied to laboratory operations rather than retail patterns. Suppliers of these reagents focus on ensuring a consistent supply, high-quality documentation, and robust technical support, particularly during audit processes. Effective production planning hinges on the stability of reagents, precise filling of bottles, and protective packaging that guards against light and temperature variations. The release of these products is contingent on stringent recovery controls and linearity assessments over the ranges utilized in standard testing panels.
Distributors often position their inventory strategically near hospital networks to maintain service levels, while instrument vendors play a significant role in influencing reagent selection through their menu designs and service agreements. The formation of profit margins largely depends on yield rates, complaint frequency, and steady demand driven by ongoing testing programs, rather than short-term promotions or a quick expansion of testing menus.
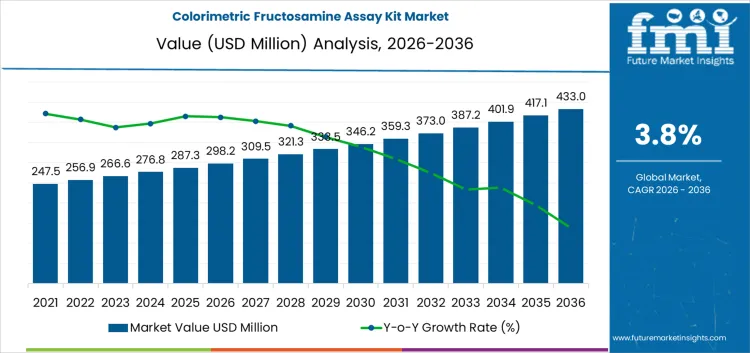
Testing protocol and laboratory workflow design shape demand for the colorimetric fructosamine assay kit market more than overall diabetes screening volumes. In 2026, at about USD 298.2 million, use is concentrated in hospital laboratories, reference labs, and specialty clinics where short term glycemic control monitoring is part of routine panels. Kit selection is tied to analyzer compatibility, turnaround time targets, and reagent handling practice, which fixes purchasing patterns across long validation periods. Volume growth follows expansion of test menus and wider inclusion in follow up protocols rather than rapid changes in patient volumes. The steady rise reflects gradual extension of testing into more care pathways and laboratory networks.
Quality control practice and supply continuity guide the later phase of the colorimetric fructosamine assay kit market. As value approaches roughly USD 433.0 million by 2036, buyers focus on lot to lot consistency, shelf life management, and stable calibration behavior. Suppliers invest more in documentation, internal controls, and distribution reliability as installed bases expand. The move from just under three hundred million into the low four hundred reflects accumulated placements across many laboratories rather than a single change in clinical guidance. Companies that maintain dependable reagent performance and predictable delivery schedules tend to secure long term positions in routine testing portfolios.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 298.2 million |
| Forecast Value (2036) | USD 433.0 million |
| Forecast CAGR 2026 to 2036 | 3.8% |
Colorimetric fructosamine assay kits measure glycated serum proteins to indicate average glucose control over recent weeks. Clinical laboratories use them when HbA1c results are unreliable or when faster feedback is needed after therapy changes. Earlier testing workflows depended mainly on fasting glucose and periodic HbA1c, which left gaps during pregnancy, anemia, or rapid treatment adjustment. These kits fit routine chemistry analyzers and provide results within standard lab turnaround times. Buyers assess them by analytical range, interference tolerance, reagent stability, and calibration requirements. Use is common in diabetes clinics, hospital labs, and research studies that track short term control. Test volume follows outpatient monitoring patterns and inpatient medication adjustment protocols, not population screening programs or one time diagnostic campaigns.
Selection and purchasing decisions are led by laboratory directors and quality managers because assay performance affects clinical interpretation and accreditation results. Earlier sourcing accepted wider method variation, which complicated result comparison between sites. Current networks seek method harmonization, stable lot performance, and clear traceability to reference materials. Kit suppliers compete on precision, ease of automation, and documentation support rather than on unit price alone. Laboratories evaluate total cost through repeat testing rates, control consumption, and instrument downtime, not through reagent cost per test. Distributors add value by managing cold chain, lot tracking, and recall communication. Over time, demand follows growth in diabetes management programs and in situations where rapid therapy adjustment requires short interval feedback, while routine long term control remains anchored to established markers.
Specification in Colorimetric Fructosamine Assay Kit Market is driven by laboratory workflow design, quality control protocols, and accreditation requirements rather than by test price alone. Once an analytical method and reporting pathway are approved, the assay format becomes embedded in the laboratory procedure set. This ties demand to platform standardization, audit cycles, and test menu rationalization programs. Segment structure reflects differences in throughput, handling steps, and integration with existing analyzers. Buyers prioritize result reproducibility, calibration stability, and documentation support because fructosamine results are used in clinical decision pathways. Volume development therefore follows laboratory consolidation, automation investment, and testing protocol updates rather than short term fluctuations in patient visit counts.
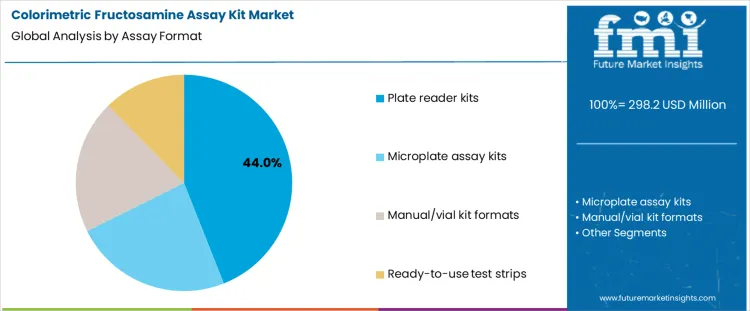
Plate reader kits represent about 44% of demand in the Colorimetric Fructosamine Assay Kit Market because many laboratories operate centralized analyzers and batch testing workflows. These formats support high sample throughput and consistent timing control across runs. Microplate assay kits serve similar environments but are often chosen where flexibility in batch size and layout is required. Manual or vial based kits remain in use in settings with lower test volumes or limited automation, though they increase operator dependency and procedural variability. Ready to use test strips address rapid assessment needs, where minimal preparation and short turnaround time are prioritized over batch efficiency. Each format establishes different staffing patterns, equipment utilization, and control procedures.
From a quality management perspective, format choice defines inspection and documentation workload. Automated plate based systems rely on instrument calibration and reagent lot tracking to maintain consistency. Manual formats require tighter procedural oversight and operator training to control variability. Test strip systems introduce additional controls around storage conditions and reader compatibility. Once a laboratory standardizes on a format, changes are avoided because method validation, reference range confirmation, and staff training must be repeated. Demand by assay format therefore follows laboratory automation strategies and accreditation planning rather than short term reagent purchasing considerations.
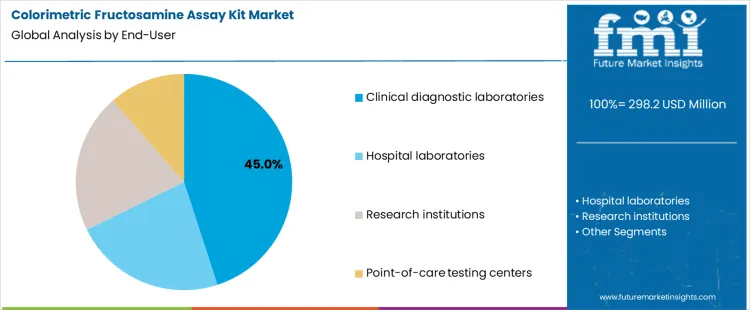
Clinical diagnostic laboratories account for about 45% of demand in the Colorimetric Fructosamine Assay Kit Market because they process large numbers of routine samples under standardized operating procedures. These facilities require stable supply, consistent performance, and predictable turnaround times. Hospital laboratories operate within integrated clinical workflows, where test selection and frequency are linked to inpatient and outpatient care protocols. Research institutions use fructosamine assays for study specific purposes, resulting in smaller volumes and variable formats. Point of care testing centers apply rapid formats for immediate decision support, which increases emphasis on simplicity and speed over batch efficiency.
End user mix also shapes procurement and approval pathways. Large diagnostic networks purchase through centralized tenders and enforce strict validation and lot release procedures. Hospitals align purchases with laboratory information systems and clinical governance structures. Research organizations acquire kits on a project basis, producing irregular demand patterns. Point of care operators require compact systems with simplified quality control. The resulting structure concentrates volume in centralized laboratories while compliance and validation requirements intensify in clinical environments. Segment shares therefore move with laboratory network expansion, hospital testing protocols, and decentralization of diagnostics rather than with short term changes in disease screening rates.
The Colorimetric Fructosamine Assay Kit Market is shaped by clinical demand for indicators that reflect short-term glycemic control and by how easily tests fit into routine laboratory operations. Fructosamine testing is used when shorter monitoring windows are needed or when other markers are less suitable, which makes turnaround time, reagent stability, and compatibility with existing analyzers critical. Buyers evaluate kits based on result consistency, ease of calibration, and workflow integration. Selection decisions involve laboratory managers and clinicians rather than procurement alone. This positions assay kits as part of diagnostic workflow reliability rather than as interchangeable consumables.
Clinical laboratories increasingly aim to standardize testing procedures across instruments and sites to simplify training, quality control, and result comparison. This is changing expectations in the Colorimetric Fructosamine Assay Kit Market toward kits with stable performance, clear instructions, and predictable lot-to-lot behavior. Laboratories prefer reagents that require minimal manual adjustment and that align with existing quality assurance protocols. As a result, suppliers are judged on documentation clarity, consistency of performance, and long-term availability. The buying decision focuses more on operational stability and audit readiness than on marginal differences in analytical specifications.
Routine demand is being built in settings that manage large populations of patients with diabetes or conditions that affect protein turnover. Hospitals, diagnostic laboratories, and specialty clinics create steady testing volume where fructosamine is used alongside other markers for patient follow-up and therapy monitoring. The Colorimetric Fructosamine Assay Kit Market also benefits from multi-site laboratory networks that adopt a single kit across their testing platforms. There is opportunity for suppliers that can support training, quality control materials, and consistent supply across locations, creating repeat purchasing patterns tied to ongoing patient monitoring rather than to sporadic testing needs.
Many clinicians and laboratories rely primarily on more established markers and may use fructosamine testing only in specific cases. This limits how often the test is ordered in routine practice. The Colorimetric Fructosamine Assay Kit Market also faces constraints from reimbursement structures and internal testing menus, which can make it difficult to justify adding or expanding use of the assay. In addition, laboratories are cautious about changing or adding methods because of validation work and staff training requirements. These factors slow broader adoption and keep usage concentrated in defined clinical scenarios.
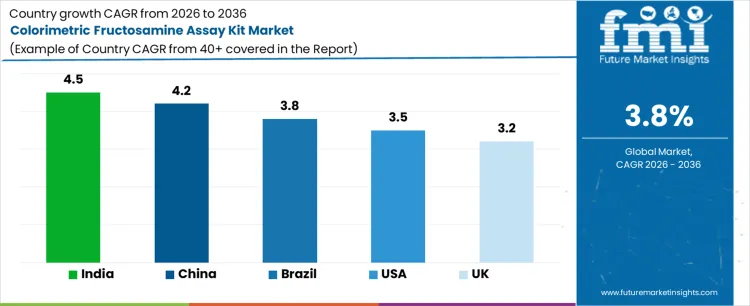
| Country | CAGR |
|---|---|
| USA | 3.5% |
| UK | 3.2% |
| China | 4.2% |
| India | 4.5% |
| Brazil | 3.8% |
Demand for colorimetric fructosamine assay kits is growing as clinical laboratories and hospitals expand intermediate term glycemic monitoring alongside routine HbA1c testing. India leads with a 4.5% CAGR, supported by rising diabetes screening volumes, growth in diagnostic chains, and wider use in outpatient monitoring. China follows at 4.2%, driven by expanding hospital testing capacity and higher chronic disease surveillance. Brazil records 3.8%, reflecting broader access to laboratory diagnostics and public health programs. The USA grows at 3.5%, shaped by stable testing volumes and use in specific clinical indications. The UK, at 3.2%, reflects a mature diagnostics environment with incremental adoption.
Clinical testing expansion keeps the colorimetric fructosamine assay kit market in the United States on a 3.5% CAGR path. Hospital laboratories, diagnostic chains, and reference centers represent the main users. Test selection focuses on reproducibility, calibration stability, and compatibility with existing chemistry analyzers. Procurement follows annual reagent contracts rather than ad hoc purchasing. Quality managers track lot to lot variation and control performance during routine runs. Replacement of approved kits occurs rarely due to validation effort. Inventory planning aligns with patient testing volumes. Training programs support consistent handling and result interpretation. Regulatory inspections influence documentation practices. Distribution networks ensure regional availability. Budget planning follows laboratory utilization trends. Commercial position depends on inclusion in approved test menus and reliable technical support rather than competition for one time reagent orders from small independent laboratories.
Service consolidation places the colorimetric fructosamine assay kit market in the United Kingdom on a 3.2% CAGR trajectory. Regional pathology hubs and hospital trusts account for most consumption. Method approval follows analytical performance checks and verification studies. Central procurement teams manage reagent frameworks covering multiple sites. Laboratory managers focus on turnaround time, analyzer compatibility, and quality control stability. Usage volumes follow diabetic clinic workloads and monitoring schedules. Stock policies reflect contract delivery timetables. Training programs influence consistency of result reporting. Replacement of established assays requires governance approval. External quality assessment participation shapes confidence in method choice. Import reliance influences logistics planning. Commercial access depends on framework participation and audit history rather than competition for small volume reagent purchases by individual laboratories.
Large scale diagnostic deployment places the colorimetric fructosamine assay kit market in China on a 4.2% growth path. Public hospitals, private laboratory chains, and screening centers generate most demand. Central laboratory groups define assay menus and validation requirements. Local manufacturing supports volume supply and stable pricing. Procurement aggregates reagent needs through group purchasing platforms. Analyzer integration and throughput capacity guide kit selection. Quality teams monitor precision and control trends during routine operation. Inventory planning follows continuous testing schedules. Replacement activity links to platform upgrades or method revisions. Training programs support standardized procedures across sites. Reporting systems archive results for clinical review. Commercial success depends on placement within hospital networks and laboratory chains rather than fragmented sales to small independent clinics.
Rising test volumes fix the colorimetric fructosamine assay kit market in India at a 4.5% CAGR. Hospital laboratories, diagnostic chains, and specialty clinics represent the main users. Procurement decisions focus on reliability, service support, and compatibility with installed analyzers. Many sites operate under centralized test menus defined by head offices. Incoming lot verification remains mandatory before routine use. Storage conditions receive attention due to temperature sensitivity. Quality control programs track precision and drift. Replacement demand follows analyzer changes or menu rationalization. Distributor coverage shapes regional availability. Training programs influence handling consistency. Inventory planning follows patient throughput patterns. Commercial results depend on inclusion in chain level contracts and maintaining technical support rather than competition for one-time reagent supply to individual laboratories.
Expansion of diagnostic services keeps the colorimetric fructosamine assay kit market in Brazil near a 3.8% CAGR. Private laboratory networks, hospitals, and outpatient centers form the main customer base. Test selection reflects analytical performance, ease of use, and documentation quality. Procurement often occurs through annual supply agreements. Import procedures influence delivery schedules and stock policies. Laboratory managers track control stability and result consistency. Training programs support standardized operation across sites. Replacement cycles follow platform updates or method reviews. Inventory management aligns with patient testing volumes. Distributor networks manage regional service and logistics. Participation in quality programs supports method credibility. Commercial position depends on approval within laboratory networks and consistent supply performance rather than competition for short term reagent sales to individual testing sites.
Colorimetric fructosamine assay kit selection follows laboratory workflow design and analyzer compatibility rather than catalog comparison. Clinical chemistry managers define throughput targets, calibration routines, reagent stability windows, and reporting ranges before naming suppliers. Abbott, Randox, BioVision, DiaSys, Sekisui Diagnostics compete within those method selection exercises. Once a kit enters standard operating procedures, method validation files and quality control charts lock the choice. Abbott benefits from placement inside its analyzer ecosystems and bundled reagent contracts. Randox supplies multi analyte quality control and reagent programs across hospital networks. DiaSys focuses on open channel chemistry systems and standardized reagent formats. Sekisui supports laboratories that require long reagent stability and consistent lot supply. BioVision serves research and specialty testing settings that value flexible pack sizes and method development access.
Competition reflects reliability in daily operation, documentation discipline, and service response rather than headline sensitivity claims. Laboratories examine linearity, interference profiles, calibration drift, and control recovery during verification. Inventory planning favors kits with predictable shelf life and simple storage conditions. Network laboratories prefer suppliers that support harmonized methods across multiple sites. Abbott secures positions where analyzer standardization governs procurement. Randox gains share through quality control programs linked to routine chemistry menus. DiaSys competes where open systems and cost control guide decisions. Sekisui holds accounts that prioritize stability and supply continuity. BioVision remains present in smaller volume environments and research linked laboratories. Market positions differ by laboratory size, analyzer mix, accreditation regime, and purchasing structure.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD million |
| Assay Format | Plate reader kits, Microplate assay kits, Manual and vial kit formats, Ready-to-use test strips |
| End-User | Clinical diagnostic laboratories, Hospital laboratories, Research institutions, Point-of-care testing centers |
| Region | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, United States, Canada, Mexico, Brazil, Chile, Saudi Arabia, Turkey, South Africa, and other regional markets |
| Key Companies Profiled | Abbott Laboratories, Randox Laboratories, BioVision, DiaSys Diagnostic Systems, Sekisui Diagnostics |
| Additional Attributes | Dollar by sales by assay format and end-user; demand driven by diabetes follow-up testing and laboratory workflow standardization; selection based on analyzer compatibility, lot consistency, and calibration stability; procurement through annual reagent contracts; focus on shelf life management, quality control performance, and documentation for audits and accreditation. |
How big is the colorimetric fructosamine assay kit market in 2026?
The global colorimetric fructosamine assay kit market is estimated to be valued at USD 298.2 million in 2026.
What will be the size of colorimetric fructosamine assay kit market in 2036?
The market size for the colorimetric fructosamine assay kit market is projected to reach USD 433.0 million by 2036.
How much will be the colorimetric fructosamine assay kit market growth between 2026 and 2036?
The colorimetric fructosamine assay kit market is expected to grow at a 3.8% CAGR between 2026 and 2036.
What are the key product types in the colorimetric fructosamine assay kit market?
The key product types in colorimetric fructosamine assay kit market are plate reader kits , microplate assay kits, manual/vial kit formats and ready‑to‑use test strips.
Which end‑user segment to contribute significant share in the colorimetric fructosamine assay kit market in 2026?
In terms of end‑user, clinical diagnostic laboratories segment to command 45.0% share in the colorimetric fructosamine assay kit market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.