ELISA POT Assay Kits Market
This report provides a comprehensive assessment of the elisa pot assay kits market, encompassing market size estimation, revenue projections, competitive dynamics, demand trends, product type trends, key growth drivers, challenges, supply chain analysis, and emerging strategic opportunities.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
ELISA POT Assay Kits Market Size, Market Forecast and Outlook By FMI
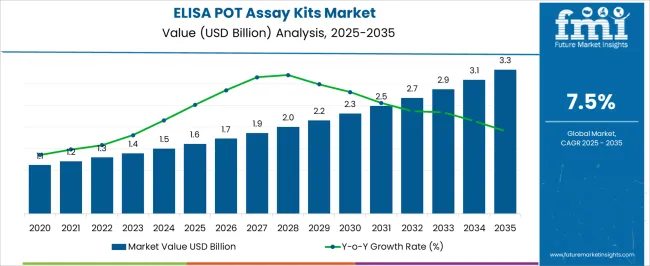
The ELISA POT assay kits market was valued at USD 1,600.00 billion in 2025, projected to reach USD 1,720.00 billion in 2026, and is forecast to expand to USD 3,544.97 billion by 2036 at a 7.50% CAGR. Expanding drug absorption and pharmacokinetics research, growing interest in proton-coupled oligopeptide transporter characterization for oral drug delivery optimization, and the increasing adoption of ELISA-based quantitative protein detection in pharmaceutical screening workflows are driving sustained demand for POT-specific assay kits. Pharmaceutical companies investigating peptide transporter-mediated prodrug delivery strategies are generating recurring assay kit procurement volumes through multi-year drug development programmes.
The absolute dollar opportunity of approximately USD 1,824.97 billion over the forecast period reflects the broadening research application base from basic transporter biology into applied pharmaceutical formulation screening and clinical pharmacokinetics studies. Multiplex ELISA kit formats enabling simultaneous quantification of multiple SLC15 family transporter proteins within single assay wells are commanding premium pricing by reducing per-sample assay costs and experimental time. Contract research organizations expanding pharmacokinetics testing capabilities are generating institutional procurement volumes for validated ELISA POT kits with documented inter-laboratory reproducibility.
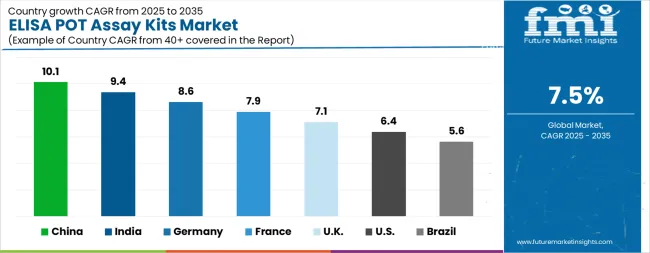
China sets the pace at a 10.1% CAGR, driven by pharmaceutical R&D investment in oral drug delivery optimization and NSFC-funded transporter biology research. India follows at 9.4% as pharmaceutical industry drug formulation research and CRO capacity expansion generate assay kit consumption. Germany registers 8.6% growth supported by pharmaceutical industry pharmacokinetics research and university transporter biology programmes. USA tracks at 6.4% driven by NIH-funded drug absorption research and pharmaceutical screening laboratory demand. Brazil maintains 5.6% with pharmaceutical research infrastructure expansion.
Summary of ELISA POT Assay Kits Market
- ELISA POT Assay Kits Market Definition
- ELISA POT assay kits are immunoassay reagents for quantitative detection of proton-coupled oligopeptide transporter proteins (SLC15 family), used in pharmaceutical drug absorption screening, transporter biology research, and clinical pharmacokinetics analysis.
- Demand Drivers in the Market
- Pharmaceutical companies investigating peptide transporter-mediated prodrug delivery strategies generate recurring multi-year assay kit procurement through drug development programmes requiring systematic SLC15 transporter expression profiling.
- Multiplex ELISA formats enabling simultaneous quantification of multiple POT family proteins command premium pricing by reducing per-sample costs and experiment duration.
- Contract research organizations expanding pharmacokinetics testing capabilities generate institutional procurement volumes for validated kits with documented inter-laboratory reproducibility.
- Key Segments Analyzed in the FMI Report
- SLC15A1 ELISA POT Assay Kits by Protein Type: 46.8% share in 2026, reflecting the primary research and pharmaceutical screening focus on PepT1, the predominant intestinal peptide transporter governing oral drug absorption and prodrug delivery.
- Pharmaceutical & Biotech Companies by End User: 52.4% share in 2026, as drug absorption screening and pharmacokinetics research generate the highest per-organization assay kit consumption volumes and the largest per-order procurement values.
- China: 10.1% compound growth driven by pharmaceutical R&D investment in oral drug delivery and NSFC transporter biology research funding expansion.
- Analyst Opinion at FMI
- The ELISA POT assay kits market serves a specialized but growing research niche where pharmaceutical interest in peptide transporter-mediated oral drug delivery is expanding the commercial application base beyond academic transporter biology into applied formulation screening. Kit manufacturers who cannot provide validated specificity data with documented cross-reactivity profiles face credibility gaps that limit adoption in pharmaceutical screening workflows requiring regulatory-grade data quality. The critical market expansion factor centers on clinical pharmacokinetics study adoption, as ELISA-based transporter quantification in clinical biopsy samples creates a higher-value application tier above preclinical research kit consumption.
- Strategic Implications / Executive Takeaways
- ELISA kit manufacturers must invest in clinical-grade validation studies demonstrating assay performance in human tissue samples to capture the growing clinical pharmacokinetics application segment.
- Pharmaceutical ADME research directors must standardize POT transporter profiling protocols across drug development programmes to enable cross-programme data comparison and assay kit procurement consolidation.
- Contract research organizations must qualify ELISA POT kits with inter-laboratory reproducibility data to support regulatory submission-quality transporter expression studies for pharmaceutical clients.
ELISA POT Assay Kits Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 1,720.00 Billion |
| Industry Value (2036) | USD 3,544.97 Billion |
| CAGR (2026-2036) | 7.50% |
Source: Future Market Insights, 2026
ELISA POT Assay Kits Market Definition
ELISA POT assay kits are enzyme-linked immunosorbent assay reagent kits designed for the quantitative detection and measurement of proton-coupled oligopeptide transporter (POT/SLC15 family) proteins in biological samples. These kits target specific transporter isoforms including SLC15A1 (PepT1), SLC15A2 (PepT2), and SLC15A4, providing researchers with validated immunoassay tools for studying peptide transporter expression, drug absorption mechanisms, and prodrug delivery pathway characterization in pharmaceutical and academic research settings.
ELISA POT Assay Kits Market Inclusions
Market scope includes SLC15A1-specific ELISA kits, SLC15A2-specific ELISA kits, SLC15A4-specific ELISA kits, and multi-target POT family assay panels. Coverage spans pharmaceutical drug absorption screening, academic transporter biology research, clinical pharmacokinetics sample analysis, and contract research organization testing services.
ELISA POT Assay Kits Market Exclusions
General-purpose ELISA kits without POT transporter specificity, western blot antibodies for SLC15 detection, and mass spectrometry-based transporter quantification reagents are excluded. Cell-based transporter functional assays without ELISA detection methodology fall outside analytical parameters.
ELISA POT Assay Kits Market Research Methodology
- Primary Research: Analysts engaged with pharmaceutical ADME research directors, academic transporter biology laboratory heads, and ELISA kit manufacturer product development managers to map the research programme and screening workflow triggers for POT assay kit procurement.
- Desk Research: Data collection phases aggregated pharmaceutical R&D pipeline data for peptide transporter-targeted drug candidates, transporter biology publication output indices, and ELISA kit manufacturer sales records.
- Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of ELISA POT kit unit sales multiplied by average per-kit prices across protein type and end user categories, applying region-specific pharmaceutical R&D investment curves.
- Data Validation and Update Cycle: Projections are tested against publicly reported life science reagent revenues from leading immunoassay kit manufacturers and pharmaceutical research tool suppliers.
Why is the ELISA POT Assay Kits Market Growing?
The ELISA POT Assay Kits market is being driven by the increasing demand for precise biomarker quantification across pharmaceutical research and clinical diagnostics. The current market scenario reflects growing investments in drug development, personalized medicine, and large-scale screening programs, which are enhancing the adoption of assay kits with high sensitivity and reproducibility. The rising prevalence of chronic diseases and metabolic disorders has also contributed to increased demand for advanced immunoassays that offer rapid and accurate protein detection.
In addition, growing collaborations between research organizations and diagnostic solution providers have supported the development of specialized assay kits tailored to emerging therapeutic targets. The availability of kits designed for specific proteins and molecular pathways has made it easier for end users to incorporate them into complex workflows, improving throughput and data reliability.
Looking forward, continuous advancements in assay optimization, automation, and multiplexing are expected to further fuel market growth The demand for robust, standardized, and scalable solutions will remain strong as research and healthcare sectors increasingly rely on precise biomarker analysis for drug discovery and patient management.
Segmental Analysis
The elisa pot assay kits market is segmented by protein type, end user, and geographic regions. By protein type, elisa pot assay kits market is divided into SLC15A1 ELISA POT Assay Kits, SLC15A2 ELISA POT Assay Kits, SLC15A4 ELISA POT Assay Kits, and Other ELISA POT Assay Kits.
In terms of end user, elisa pot assay kits market is classified into Pharmaceutical & Biotech Companies, Independent Research Labs, and Academic & Research Institutions. Regionally, the elisa pot assay kits industry is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the SLC15A1 ELISA POT Assay Kits Segment
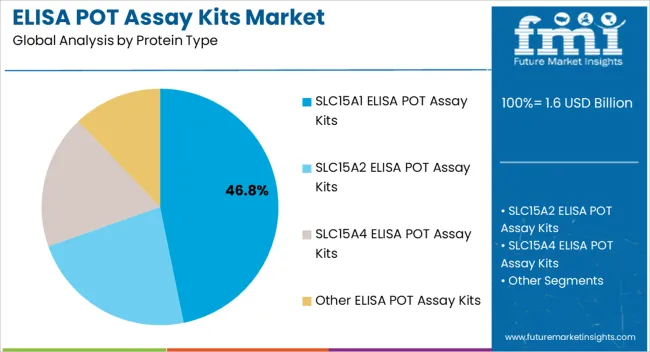
The SLC15A1 ELISA POT Assay Kits segment is estimated to account for 46.80% of the overall market revenue in 2026, establishing it as the leading protein type in this category. The dominance of this segment is attributed to its role in targeted research on peptide transport mechanisms and metabolic regulation, which have gained significant attention in both drug development and disease profiling.
The kits have been widely adopted because they provide high specificity and consistent quantification, which are essential for understanding protein expression in various biological samples. Their compatibility with high-throughput screening platforms has further encouraged their use in large-scale studies, particularly in pharmaceutical research settings.
The ease of integration into automated workflows and the availability of standardized protocols have made these kits a preferred choice among researchers Additionally, the growing focus on peptide transporters as potential therapeutic targets in oncology and metabolic diseases has reinforced the need for specialized assay kits like SLC15A1, supporting both preclinical research and clinical biomarker validation efforts.
Insights into the Pharmaceutical and Biotech Companies Segment
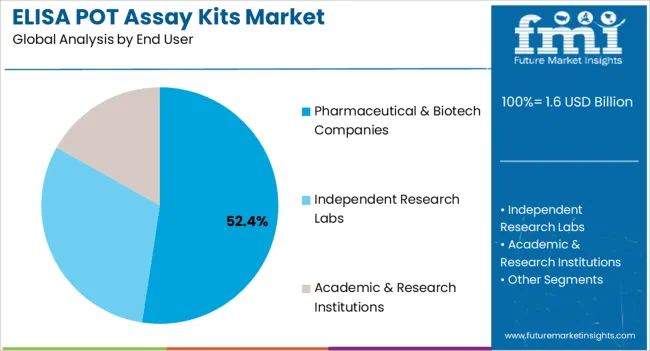
The pharmaceutical and biotech companies segment is expected to hold 52.40% of the market share in 2026, making it the largest end-user category in the ELISA POT Assay Kits market. This leadership is being driven by the sector’s growing reliance on protein quantification for drug discovery, biomarker validation, and clinical trials. The adoption of assay kits that offer high sensitivity, reproducibility, and scalability has allowed these companies to streamline research workflows and improve decision-making.
The kits have been increasingly utilized in identifying therapeutic targets, optimizing lead compounds, and evaluating disease mechanisms, which are critical steps in the development of novel drugs. Furthermore, the integration of ELISA-based assays into early-phase clinical trials has enhanced patient stratification and treatment monitoring, improving the overall efficiency of research programs.
Pharmaceutical and biotech companies are also investing in advanced technologies to accelerate drug development timelines, and assay kits tailored to specific proteins have played a central role in these efforts The continued emphasis on precision medicine and targeted therapies is expected to further strengthen the demand for ELISA POT Assay Kits within this sector.
ELISA POT Assay Kits Market Overview
Proton dependent oligopeptide transporter (POT) proteins is a family of proteins that is also known as the peptide transport (PTR) protein family. These are made up of energy-dependent transporter proteins that are commonly found in bacteria and humans. These POT proteins are nitrate permease and have the ability to transport histidine and peptides. ELISA stands for enzyme-linked immunosorbent assay. These assays are plate based and are used for quantification and detection of a number of substances including peptides, proteins, antibodies and hormones. The ELISA POT assay kits are ELISA based assay kits used to quantify and identify POT proteins and its ligands. Some of the human proteins used in the ELISA POT assay kits include SLC15A1, SLC15A2, SLC15A4 and others. The use of ELISA POT assay kits have a number of applications and can be potentially used for design and delivery of drugs. The ELISA POT assay kits can also be used for pharmacokinetic studies as well.
Analysis of ELISA POT Assay Kits Market By Key Countries
.webp)
| Country | CAGR |
|---|---|
| China | 10.1% |
| India | 9.4% |
| Germany | 8.6% |
| France | 7.9% |
| UK | 7.1% |
| USA | 6.4% |
| Brazil | 5.6% |
Source: FMI analysis based on primary research and proprietary forecasting model
Country-wise Analysis
The ELISA POT Assay Kits Market is expected to register a CAGR of 7.5% during the forecast period, exhibiting varied country level momentum. China leads with the highest CAGR of 10.1%, followed by India at 9.4%. Developed markets such as Germany, France, and the UK continue to expand steadily, while the USA is likely to grow at consistent rates.
Brazil posts the lowest CAGR at 5.6%, yet still underscores a broadly positive trajectory for the global ELISA POT Assay Kits Market. In 2024, Germany held a dominant revenue in the Western Europe market and is expected to grow with a CAGR of 8.6%.
The USA ELISA POT Assay Kits Market is estimated to be valued at USD 601.1 million in 2026 and is anticipated to reach a valuation of USD 1.1 billion by 2036. Sales are projected to rise at a CAGR of 6.4% over the forecast period between 2026 and 2036. While Japan and South Korea markets are estimated to be valued at USD 80.7 million and USD 52.2 million respectively in 2026.
Key Players in the ELISA POT Assay Kits Market
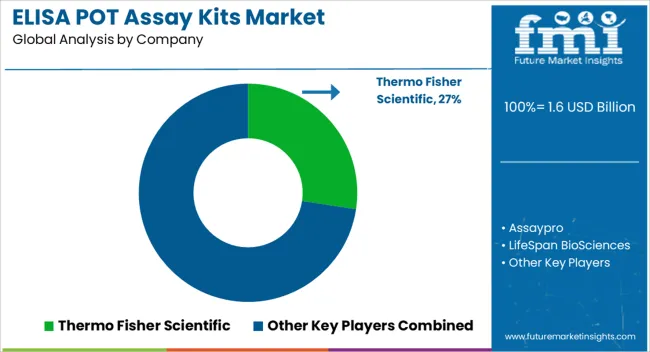
- Thermo Fisher Scientific
- Assaypro
- LifeSpan BioSciences
- Aviva Systems Biology
- Biorbyt
- Sigma-Aldrich
- Cell Signaling Technology
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 1,720.00 Billion to USD 3,544.97 Billion, at a CAGR of 7.50% |
| Market Definition | ELISA POT assay kits are immunoassay reagents for quantitative detection of proton-coupled oligopeptide transporter proteins (SLC15 family), used in pharmaceutical drug absorption screening, transporter biology research, and clinical pharmacokinetics analysis. |
| Segmentation | Protein Type: SLC15A1 ELISA POT Assay Kits, SLC15A2 ELISA POT Assay Kits, SLC15A4 ELISA POT Assay Kits, and Other ELISA POT Assay Kits; End User: Pharmaceutical & Biotech Companies, Independent Research Labs, and Academic & Research Institutions |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | China, India, Germany, USA, Brazil, and 40 plus countries |
| Key Companies Profiled | Thermo Fisher Scientific, Assaypro, LifeSpan BioSciences, Aviva Systems Biology, Biorbyt, Sigma-Aldrich, Cell Signaling Technology |
| Forecast Period | 2026 to 2036 |
| Approach | Forecasting models apply a bottom-up methodology starting with installed base metrics and projecting conversion rates to technology adoption. |
ELISA POT Assay Kits Market by Segments
Protein Type:
- SLC15A1 ELISA POT Assay Kits
- SLC15A2 ELISA POT Assay Kits
- SLC15A4 ELISA POT Assay Kits
- Other ELISA POT Assay Kits
End User:
- Pharmaceutical & Biotech Companies
- Independent Research Labs
- Academic & Research Institutions
Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- 1. USA Food and Drug Administration. (2024). Guidance for Industry: Drug Interaction Studies and Transporter-Mediated Drug Interactions. FDA.
- 2. European Medicines Agency. (2024). Guideline on Investigation of Drug Interactions: Transporter-Based Interactions. EMA.
- 3. National Natural Science Foundation of China. (2024). Drug Absorption and Transporter Biology: Research Funding Allocation Report. NSFC.
- 4. International Transporter Consortium. (2024). Updated Recommendations for Transporter Studies in Drug Development. ITC.
- 5. National Institutes of Health. (2024). NIGMS Pharmacokinetics Research: Grant Funding and Priority Areas. NIH.
- 6. Indian Pharmacopoeia Commission. (2024). Drug Absorption Testing: Transporter Assay Methodology Standards. IPC.
- This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary research documentation.
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
Frequently Asked Questions
How large is the demand for ELISA POT Assay Kits in the global market in 2026?
Demand is estimated to be valued at USD 1,720.00 billion in 2026.
What will be the market size by 2036?
Market size is projected to reach USD 3,544.97 billion by 2036.
What is the expected CAGR between 2026 and 2036?
Demand is expected to grow at a CAGR of 7.50% between 2026 and 2036.
Which Protein Type is poised to lead global sales by 2026?
SLC15A1 accounts for 46.8% share reflecting the pharmaceutical focus on PepT1 intestinal transporter for oral drug absorption screening.
How are Pharmaceutical & Biotech Companies positioned in 2026?
Pharmaceutical & Biotech Companies represents 52.4% share generating the highest consumption volumes through drug absorption and pharmacokinetics research.
What is driving demand in China?
Pharmaceutical R&D in oral drug delivery and NSFC transporter biology research fuel the fastest growth at 10.1% CAGR through 2036.
What is included in the scope of this report?
The market covers SLC15A1, SLC15A2, SLC15A4, and multi-target panel ELISA kits for pharmaceutical, CRO, and academic transporter research.
What is excluded from the scope of this report?
General ELISA kits, western blot antibodies, mass spectrometry reagents, and cell-based functional transporter assays are excluded.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Protein Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Protein Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Protein Type , 2026 to 2036
- SLC15A1 ELISA POT Assay Kits
- SLC15A2 ELISA POT Assay Kits
- SLC15A4 ELISA POT Assay Kits
- Other ELISA POT Assay Kits
- SLC15A1 ELISA POT Assay Kits
- Y to o to Y Growth Trend Analysis By Protein Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Protein Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Pharmaceutical & Biotech Companies
- Independent Research Labs
- Academic & Research Institutions
- Pharmaceutical & Biotech Companies
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Protein Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Protein Type
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Protein Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Protein Type
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Protein Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Protein Type
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Protein Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Protein Type
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Protein Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Protein Type
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Protein Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Protein Type
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Protein Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Protein Type
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Protein Type
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Protein Type
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Protein Type
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Protein Type
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Protein Type
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Protein Type
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Protein Type
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Protein Type
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Protein Type
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Protein Type
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Protein Type
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Protein Type
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Protein Type
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Protein Type
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Protein Type
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Protein Type
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Protein Type
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Protein Type
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Protein Type
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Protein Type
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Protein Type
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Protein Type
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Protein Type
- By End User
- Competition Analysis
- Competition Deep Dive
- Thermo Fisher Scientific
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Assaypro
- LifeSpan BioSciences
- Aviva Systems Biology
- Biorbyt
- Sigma-Aldrich
- Cell Signaling Technology
- Thermo Fisher Scientific
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Protein Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Protein Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Protein Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Protein Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Protein Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Protein Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Protein Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Protein Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Protein Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Protein Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Protein Type
- Figure 6: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by End User
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Protein Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Protein Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Protein Type
- Figure 23: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by End User
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Protein Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Protein Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Protein Type
- Figure 30: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by End User
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Protein Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Protein Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Protein Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by End User
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Protein Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Protein Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Protein Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by End User
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Protein Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Protein Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Protein Type
- Figure 51: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by End User
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Protein Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Protein Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Protein Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Protein Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Protein Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Protein Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

