Digital Immunoassays Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
Digital Immunoassays Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Digital Immunoassays Market Forecast and Outlook 2026 to 2036
In 2026, the digital immunoassays market stands at USD 2.4 billion and is projected to reach USD 7.1 billion by 2036, implying a CAGR of 11.4%. Use is concentrated in clinical diagnostics, hospitals, and reference laboratories that require high sensitivity, low detection limits, and consistent quantitative accuracy. Demand clusters around oncology, cardiology, infectious disease, and critical care testing, where clinical decisions depend on early and precise measurement. Buying decisions are led by clinical value and workflow fit rather than by instrument price alone. The product mix spans instrument platforms, consumables, and test cartridges, which anchors long term relationships once a system is installed.
Adoption varies widely by region because laboratory infrastructure, reimbursement policies, and routine test volumes differ in scale and stability. Large health systems with centralized labs standardize platforms to simplify validation, quality control, and staff training, which concentrates volumes among a smaller set of suppliers. Smaller hospitals and private labs enter selectively, often starting with limited menus tied to specific clinical pathways. Utilization rates and menu breadth matter more to economics than headline placements. The category grows through expansion of high value testing programs and deeper integration into care pathways, not through uniform replacement of conventional immunoassay methods.
Quick Stats for Digital Immunoassays Market
- Digital Immunoassays Market Value (2026): USD 2.4 billion
- Digital Immunoassays Market Forecast Value (2036): USD 7.1 billion
- Digital Immunoassays Market Forecast CAGR 2026 to 2036: 11.4%
- Leading Type in Digital Immunoassays Market: Protein biomarker assays
- Key Growth Regions in Digital Immunoassays Market: Asia Pacific, North America, Europe
- Top Players in Digital Immunoassays Market: Quanterix Corporation, Thermo Fisher Scientific, Bio Rad Laboratories, Danaher Corporation, PerkinElmer, Abbott Laboratories, Siemens Healthineers
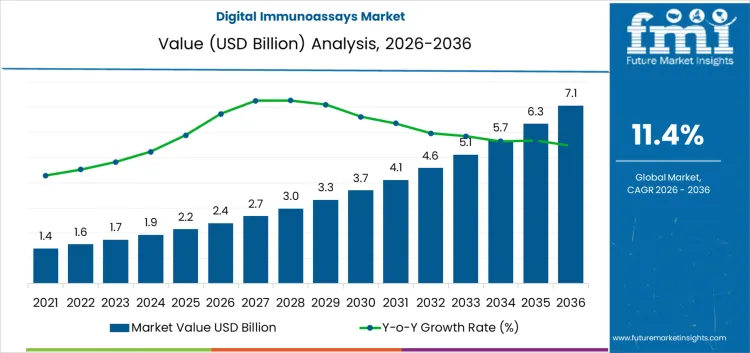
What is the Growth Forecast for Digital Immunoassays Market through 2036?
The digital immunoassays market is expanding as clinical diagnostics shifts from qualitative and semi-quantitative testing toward absolute quantification and ultra-low concentration detection. At an early stage, the category remains concentrated in research laboratories and advanced clinical reference centers. Momentum builds as early adopters in oncology, cardiology, and infectious disease monitoring begin standardizing digital readout platforms. In this phase, digital immunoassays move beyond pilot evaluations and start entering structured clinical use, especially in areas where ultra-high sensitivity, early detection, and frequent biomarker tracking are more important than test cost or maximum throughput. Growth is driven mainly by expanding clinical relevance and new use cases rather than replacement of conventional immunoassays.
As the installed base expands, adoption broadens across hospitals, while pharmaceutical and biotechnology companies increasingly rely on these platforms for therapy monitoring and clinical trial workflows. Demand gradually shifts from instrument placements toward higher utilization, supported by test menu expansion and increased testing intensity per system. Over time, the segment becomes defined by repeat consumable consumption, longitudinal patient monitoring, and protocol-led testing programs. Value creation is driven by deeper clinical integration and higher test frequency rather than pricing. Competitive strength rests on assay breadth, reproducibility, and strong clinical validation, as laboratories prioritize diagnostic confidence and seamless workflow integration over incremental gains in analytical sensitivity.
Digital Immunoassays Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 2.4 billion |
| Forecast Value (2036) | USD 7.1 billion |
| Forecast CAGR 2026 to 2036 | 11.4% |
How Is the Digital Immunoassays Market Advancing Diagnostic Accuracy and Speed?
Digital immunoassays are increasingly adopted to detect and quantify biomarkers with high sensitivity and precision in clinical diagnostics, pharmaceutical research, and point-of-care applications. Historically, conventional immunoassays relied on bulk detection methods, which limited sensitivity, throughput, and reproducibility. Modern digital immunoassay platforms use microfluidics, digital counting, and high-resolution detection to provide single-molecule sensitivity, faster assay times, and improved quantification accuracy. Clinical laboratories, diagnostic companies, and research institutions prioritize analytical sensitivity, reproducibility, and integration with laboratory information systems. Early adoption focused on specialized research labs, while current demand spans hospitals, diagnostic centers, and pharmaceutical development, driven by the need for early disease detection, personalized medicine, and high-throughput testing. Detection precision, assay reproducibility, and platform compatibility influence supplier selection.
Meeting the growing need for rapid, precise diagnostics is shaping market growth. Compared with conventional immunoassays, digital platforms emphasize higher sensitivity, lower sample volume requirements, and faster turnaround times for clinical decision-making. Cost structures depend on sensor technology, microfluidic design, and software integration, concentrating margins among suppliers capable of delivering reliable, high-performance systems. Healthcare providers and researchers adopt digital immunoassays to enhance diagnostic accuracy, accelerate drug development, and improve patient outcomes. By 2036, digital immunoassays are expected to become a standard tool in clinical and research settings, supporting precise biomarker detection, efficient workflows, and informed healthcare decisions.
What Factors Are Shaping the Demand for Digital Immunoassays, Segment wise by Assay Type and End User, in 2026?
The digital immunoassays market in 2026 is segmented by assay type and by end user setting. By assay approach, demand is divided into protein biomarker assays, nucleic acid digital immunoassays, single molecule counting assays, and digital ELISA platforms, each offering different balances between sensitivity, throughput, and workflow complexity. By end user, demand is organized around hospital and clinical laboratories, research institutes and biopharma companies, point of care testing centers, and diagnostic service providers, which differ in sample volume, regulatory burden, and tolerance for specialized instrumentation. These segments reflect how users match analytical performance needs with operational and economic constraints.
Why Do Protein Biomarker Assays Anchor Routine Clinical Use?
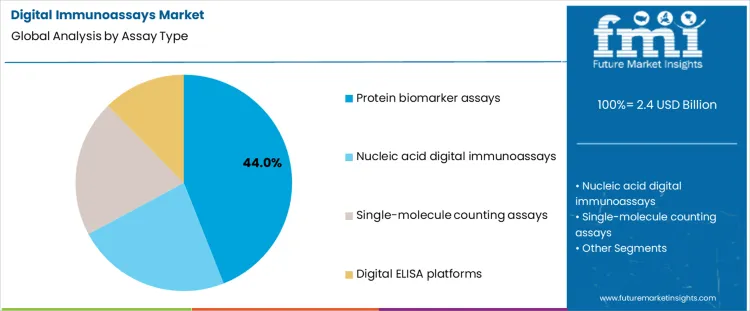
Protein biomarker assays account for about 44% of demand in 2026 because they align closely with established clinical testing needs. Many disease areas, including oncology, cardiology, and infectious disease management, rely on protein level measurements rather than on genetic markers. Digital formats improve sensitivity and quantification, but they fit into familiar laboratory workflows and reporting structures. Hospitals and large laboratories can integrate these assays into existing sample preparation and quality control processes with limited retraining. Test menus are also broad and continually expanding, which supports repeat purchasing of reagents and consumables. This combination of clinical relevance, workflow compatibility, and menu breadth keeps protein biomarker assays as the largest assay category.
Nucleic acid, single molecule, and platform focused approaches serve more specialized roles. Nucleic acid digital immunoassays overlap with molecular diagnostics and are used where combined protein and genetic information is required. Single molecule counting pushes sensitivity limits, but it is applied mainly in research or in very low abundance targets. Digital ELISA platforms represent the instrumentation layer rather than a specific test type and are adopted as part of broader platform decisions. These areas grow with innovation, yet they do not displace the broad clinical base of protein biomarker testing.
Why Do Hospital and Clinical Laboratories Dominate End User Demand?
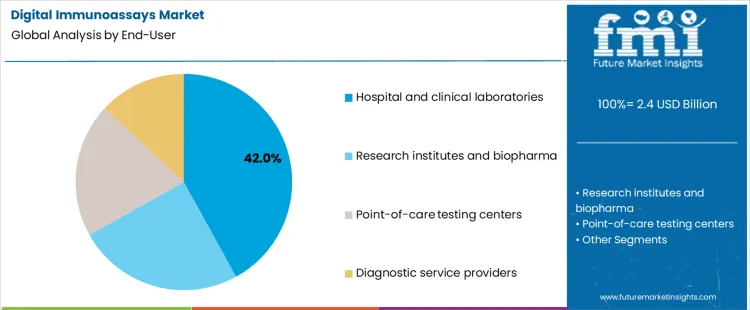
Hospital and clinical laboratories represent about 42% of demand in 2026 because they handle the largest share of routine diagnostic testing. These labs operate high throughput workflows and are under constant pressure to improve detection limits and reduce repeat testing. Digital immunoassays allow earlier detection and more precise monitoring without changing the basic structure of laboratory operations. Once a platform is validated, it can be rolled out across many test types, which increases utilization and justifies capital investment. The centralized role of these labs in patient care pathways ensures steady and predictable demand for both instruments and consumables.
Research institutes, biopharma, point of care centers, and service providers follow different adoption paths. Research groups focus on exploratory or low volume testing rather than routine diagnostics. Biopharma uses digital immunoassays mainly in development and trial settings. Point of care centers prioritize speed and simplicity, which limits use of complex digital platforms. Diagnostic service providers act as outsourced labs and adopt technologies selectively based on client demand. These segments are important for growth and innovation, but they do not match the volume concentration and routine usage found in hospital and clinical laboratory settings.
How Are the Key Dynamics Reshaping the Digital Immunoassays Market?
The category is being shaped by how laboratories and care pathways value earlier and more precise detection rather than by incremental analyzer upgrades. Digital immunoassays promise measurable gains in sensitivity and quantitative confidence, but adoption is tempered by validation burden, workflow disruption, and reimbursement alignment. At the same time, health systems are consolidating testing onto fewer, more capable platforms and extending menus around them. This creates an opportunity for digital formats to become anchor technologies once they are proven and standardized. The result is a market driven by clinical performance ceilings and platform governance, where scale comes from ecosystem position rather than one off assay wins.
Why Is Ultra High Sensitivity Driving Digital Immunoassays?
Demand is anchored in clinical questions that conventional assays struggle to answer. In cardiology, oncology, neurology, and infectious disease, earlier detection and finer quantification change triage, monitoring, and therapy decisions. Digital immunoassays reduce signal ambiguity at very low concentrations, enabling measurement where analog methods approach their limits. As care pathways move toward risk stratification and longitudinal monitoring, confidence at the low end of the curve becomes more valuable than throughput alone. Once a test proves clinical utility and is written into protocols, volumes scale quickly. Growth follows high impact indications and menu expansion on validated platforms.
What Is Slowing Wider Adoption of Digital Immunoassays?
The main barrier is system change, not assay performance. Introducing digital methods requires new instruments, training, quality controls, and LIS integration. Clinical acceptance depends on outcome data and comparability studies, which take time and funding. Labs also worry about throughput, consumable costs, and maintenance in high volume settings. Reimbursement policies often lag technical capability, weakening business cases. In regulated environments, any platform change triggers audits and documentation updates. These factors lengthen decision cycles and keep many sites in evaluation or limited rollout phases even when analytical benefits are clear.
How Are Platforms Changing the Digital Immunoassays Market?
The category is becoming platform governed. Health systems and reference labs increasingly standardize on a small number of analyzer families and expand test menus around them to maximize utilization and simplify oversight. Digital immunoassays that secure a place on these platforms scale rapidly across sites and indications. Suppliers now compete on menu breadth, automation, and service reliability, not only on sensitivity. Procurement shifts from single assay purchases to multi year platform commitments. Over time, success depends on ecosystem position and pace of clinically relevant menu expansion rather than isolated performance claims.
What is the Demand for Digital Immunoassays by Country?
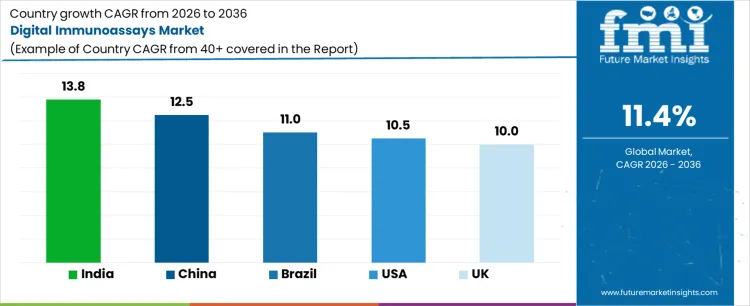
| Country | CAGR (%) |
|---|---|
| US | 10.5% |
| UK | 10.0% |
| China | 12.5% |
| India | 13.8% |
| Brazil | 11.0% |
Demand for digital immunoassays is rising as healthcare providers and diagnostic laboratories adopt advanced testing solutions for rapid, accurate, and high-throughput detection of biomarkers and diseases. India leads with a 13.8% CAGR, driven by growing diagnostic infrastructure, increasing prevalence of chronic and infectious diseases, and adoption of automated immunoassay platforms. China follows at 12.5%, supported by expanding clinical laboratories and investment in advanced diagnostic technologies. Brazil records 11.0% growth, shaped by modernization of healthcare facilities and adoption of digital testing methods. The US grows at 10.5%, influenced by integration of digital immunoassays in clinical workflows. The UK shows 10.0% CAGR, reflecting steady adoption in diagnostic laboratories and healthcare centers.
How is the United States experiencing growth in the digital immunoassays market?
United States is experiencing growth at a CAGR of 10.5%, driven by the large installed base of clinical laboratories, hospitals, and research facilities across California, Massachusetts, and New York. Replacement cycles for immunoassay equipment are predictable, supporting steady adoption of digital platforms. Industry concentration in healthcare networks and diagnostic companies anchors demand. Investments focus on accuracy, high-throughput capabilities, and integration with laboratory information systems. Growth reflects increasing demand for rapid, reliable diagnostics, streamlined workflows, and enhanced data connectivity in clinical testing environments.
- Clinical laboratories and hospitals drive adoption.
- Urban healthcare clusters concentrate demand.
- Accuracy and throughput guide investment.
- System integration enhances workflow efficiency.
How is the United Kingdom witnessing growth in the digital immunoassays market?
United Kingdom is witnessing growth at a CAGR of 10%, supported by labor cost structure and productivity pressure in healthcare facilities in London, Manchester, and Birmingham. Digital immunoassays reduce manual handling, improve turnaround time, and optimize workforce efficiency. Demand is concentrated in NHS hospitals, research centers, and diagnostic labs. Investments focus on automation, standardized workflows, and staff training. Growth reflects the push to improve lab productivity, reduce errors, and enhance patient care while managing personnel costs.
- NHS hospitals and research labs drive adoption.
- Urban healthcare hubs concentrate demand.
- Automation and workflow efficiency guide investment.
- Staff training enhances operational effectiveness.
How is China experiencing growth in the digital immunoassays market?
China is experiencing growth at a CAGR of 12.5%, fueled by industry concentration and anchor sectors in Beijing, Shanghai, and Guangzhou. Major hospitals, biotech companies, and research institutes form concentrated demand hubs for digital immunoassays. Investments prioritize high-throughput platforms, data integration, and compatibility with large-scale lab operations. Growth reflects rapid healthcare infrastructure expansion, increased clinical testing volume, and adoption of modern diagnostics in high-demand urban centers.
- Hospitals, biotech, and research institutes drive adoption.
- Urban centers concentrate demand.
- High-throughput and integration capabilities guide investment.
- Industry concentration accelerates adoption.
How is India witnessing growth in the digital immunoassays market?
India is experiencing growth at a CAGR of 13.8%, supported by exposure to export markets and global healthcare cycles in metropolitan hubs such as Mumbai, Delhi, and Bengaluru. Diagnostic companies catering to both domestic and international markets adopt digital immunoassays to meet international quality standards. Demand is concentrated in private hospital networks and high-volume diagnostic labs. Investments focus on accuracy, workflow automation, and regulatory compliance. Growth reflects increasing clinical testing needs, global collaboration, and rising adoption of internationally standardized diagnostic platforms.
- Private hospitals and high-volume labs drive adoption.
- Metropolitan hubs concentrate demand.
- Accuracy and compliance guide investment.
- Export exposure encourages adoption of digital platforms.
How is Brazil experiencing growth in the digital immunoassays market?
Brazil is experiencing growth at a CAGR of 11%, driven by sensitivity to downtime and operational risk in healthcare facilities in São Paulo, Rio de Janeiro, and Paraná. Digital immunoassays reduce errors, improve turnaround times, and enhance reliability in laboratories. Demand is concentrated in hospitals, private diagnostic chains, and research centers. Investments focus on robust systems, data connectivity, and preventive maintenance. Growth reflects the need for continuous diagnostic services, operational efficiency, and reliable testing capabilities in a healthcare environment sensitive to delays or equipment failures.
- Hospitals and diagnostic chains drive adoption.
- Urban centers concentrate demand.
- Reliability and connectivity guide investment.
- Operational risk sensitivity encourages adoption.
Digital Immunoassays Market What Distinguishes the Leading Technology Providers?
Competition in the digital immunoassays market is defined by assay sensitivity, instrument throughput, and integration with laboratory workflows. Quanterix Corporation supplies digital immunoassay platforms that use single-molecule detection to measure low-abundance proteins with high precision, supported by software for data analysis and reporting. Thermo Fisher Scientific provides digital immunoassay systems integrated with its broader portfolio of life science instruments and reagents, enabling standardized workflows in clinical and research settings. Bio-Rad Laboratories offers immunoassay instruments and kits that emphasize multiplexing capability and compatibility with existing laboratory automation. Danaher Corporation delivers solutions through multiple subsidiaries with varied assay formats and support services, targeting clinical diagnostics and biopharma research.
PerkinElmer supplies digital immunoassay platforms coupled with analytics software tailored for high-content screening and detailed biomarker profiling. Abbott Laboratories provides point-of-care and laboratory-based digital immunoassay systems designed for clinical decision support with emphasis on user interface and turnaround time. Siemens Healthineers offers integrated digital immunoassay solutions that align with hospital laboratory systems and quality control protocols. Other regional instrument manufacturers and specialized assay developers contribute niche products for specific analytes or research applications. Differentiation arises from detection limits, ease of use, throughput, reagent availability, interoperability with laboratory information systems, and regulatory compliance. Suppliers that combine validated performance data, robust service support, and scalable workflows maintain relevance as demand grows for precise, high-sensitivity immunoassay testing across clinical, biopharma, and research laboratories.
Key Players in the Digital Immunoassays Market
- Quanterix Corporation
- Thermo Fisher Scientific
- Bio-Rad Laboratories
- Danaher Corporation
- PerkinElmer
- Abbott Laboratories
- Siemens Healthineers
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD billion |
| Assay Type | Protein Biomarker Assays, Nucleic Acid Digital Immunoassays, Single-Molecule Counting Assays, Digital ELISA Platforms |
| End-User | Hospital and Clinical Laboratories, Research Institutes and Biopharma, Point-of-Care Testing Centers, Diagnostic Service Providers |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, United States, Canada, Mexico, Brazil, Chile, Saudi Arabia, Turkey, South Africa, and other regional markets |
| Key Companies Profiled | Quanterix Corporation, Thermo Fisher Scientific, Bio-Rad Laboratories, Danaher Corporation, PerkinElmer, Abbott Laboratories, Siemens Healthineers |
| Additional Attributes | Dollar sales by assay type and end-user; protein biomarker assays and hospital labs as leading segments; growth driven by ultra-high sensitivity testing, menu expansion, and platform standardization; economics shaped by consumables pull-through, clinical validation, and workflow integration rather than instrument pricing alone. |
Digital Immunoassays Market Segmentation
Assay Type:
- Protein biomarker assays
- Nucleic acid digital immunoassays
- Single-molecule counting assays
- Digital ELISA platforms
End-User:
- Hospital and clinical laboratories
- Research institutes and biopharma
- Point-of-care testing centers
- Diagnostic service providers
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- Medical Device Coordination Group. (2024). Guidance on safety reporting in performance studies under Regulation (EU) 2017/746 (IVDR) (MDCG 2024-4). European Commission.
- National Institutes of Health. (2024). Regulatory knowledge guide for in vitro diagnostics. National Institutes of Health.
- U.S. Food and Drug Administration. (2024, December 20). Overview of IVD regulation. U.S. Food and Drug Administration.
Frequently Asked Questions
How big is the digital immunoassays market in 2026?
The global digital immunoassays market is estimated to be valued at USD 2.4 billion in 2026.
What will be the size of digital immunoassays market in 2036?
The market size for the digital immunoassays market is projected to reach USD 7.1 billion by 2036.
How much will be the digital immunoassays market growth between 2026 and 2036?
The digital immunoassays market is expected to grow at a 11.4% CAGR between 2026 and 2036.
What are the key product types in the digital immunoassays market?
The key product types in digital immunoassays market are protein biomarker assays , nucleic acid digital immunoassays, single‑molecule counting assays and digital elisa platforms.
Which end‑user segment to contribute significant share in the digital immunoassays market in 2026?
In terms of end‑user, hospital and clinical laboratories segment to command 42.0% share in the digital immunoassays market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Assay Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Assay Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Assay Type, 2026 to 2036
- Protein biomarker assays
- Nucleic acid digital immunoassays
- Single‑molecule counting assays
- Digital ELISA platforms
- Protein biomarker assays
- Y to o to Y Growth Trend Analysis By Assay Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Assay Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End‑User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End‑User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End‑User, 2026 to 2036
- Hospital and clinical laboratories
- Research institutes and biopharma
- Point‑of‑care testing centers
- Diagnostic service providers
- Hospital and clinical laboratories
- Y to o to Y Growth Trend Analysis By End‑User, 2021 to 2025
- Absolute $ Opportunity Analysis By End‑User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Assay Type
- By End‑User
- By Country
- Market Attractiveness Analysis
- By Country
- By Assay Type
- By End‑User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Assay Type
- By End‑User
- By Country
- Market Attractiveness Analysis
- By Country
- By Assay Type
- By End‑User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Assay Type
- By End‑User
- By Country
- Market Attractiveness Analysis
- By Country
- By Assay Type
- By End‑User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Assay Type
- By End‑User
- By Country
- Market Attractiveness Analysis
- By Country
- By Assay Type
- By End‑User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Assay Type
- By End‑User
- By Country
- Market Attractiveness Analysis
- By Country
- By Assay Type
- By End‑User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Assay Type
- By End‑User
- By Country
- Market Attractiveness Analysis
- By Country
- By Assay Type
- By End‑User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Assay Type
- By End‑User
- By Country
- Market Attractiveness Analysis
- By Country
- By Assay Type
- By End‑User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Assay Type
- By End‑User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Assay Type
- By End‑User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Assay Type
- By End‑User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Assay Type
- By End‑User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Assay Type
- By End‑User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Assay Type
- By End‑User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Assay Type
- By End‑User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Assay Type
- By End‑User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Assay Type
- By End‑User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Assay Type
- By End‑User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Assay Type
- By End‑User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Assay Type
- By End‑User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Assay Type
- By End‑User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Assay Type
- By End‑User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Assay Type
- By End‑User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Assay Type
- By End‑User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Assay Type
- By End‑User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Assay Type
- By End‑User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Assay Type
- By End‑User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Assay Type
- By End‑User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Assay Type
- By End‑User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Assay Type
- By End‑User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Assay Type
- By End‑User
- Competition Analysis
- Competition Deep Dive
- Quanterix Corporation
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Thermo Fisher Scientific
- Bio‑Rad Laboratories
- Danaher Corporation
- PerkinElmer
- Abbott Laboratories
- Siemens Healthineers
- Quanterix Corporation
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Assay Type, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End‑User, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Assay Type, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by End‑User, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Assay Type, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by End‑User, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Assay Type, 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by End‑User, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Assay Type, 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by End‑User, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Assay Type, 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by End‑User, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Assay Type, 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by End‑User, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Assay Type, 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by End‑User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Assay Type, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Assay Type, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Assay Type
- Figure 6: Global Market Value Share and BPS Analysis by End‑User, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End‑User, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by End‑User
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Assay Type, 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Assay Type, 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Assay Type
- Figure 23: North America Market Value Share and BPS Analysis by End‑User, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by End‑User, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by End‑User
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Assay Type, 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Assay Type, 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Assay Type
- Figure 30: Latin America Market Value Share and BPS Analysis by End‑User, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by End‑User, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by End‑User
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Assay Type, 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Assay Type, 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Assay Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by End‑User, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by End‑User, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by End‑User
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Assay Type, 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Assay Type, 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Assay Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by End‑User, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by End‑User, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by End‑User
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Assay Type, 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Assay Type, 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Assay Type
- Figure 51: East Asia Market Value Share and BPS Analysis by End‑User, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by End‑User, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by End‑User
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Assay Type, 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Assay Type, 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Assay Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by End‑User, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by End‑User, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by End‑User
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Assay Type, 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Assay Type, 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Assay Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by End‑User, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by End‑User, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by End‑User
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

