Digital Pathology Scanner Calibration and Image Quality Test Systems Market
The digital pathology scanner calibration and image quality test systems market Is Segmented By Component (Test Slides, Calibration Software, Validation Services, Display QA Tools, Maintenance Kits), Scanner Type (Brightfield, Fluorescence, Hybrid, High-Throughput, Compact), Quality Parameter (Image Sharpness, Color Fidelity, Focus Accuracy, Scan Uniformity, Display Luminance), End User (Hospitals, Reference Labs, Cancer Centers, Academic Labs, Biopharma Labs), Workflow Stage (Routine QA, Pre-Installation, Revalidation, Troubleshooting, AI Normalization), And Region. Forecast For 2026 To 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Digital Pathology Scanner Calibration and Image Quality Test Systems Market Size, Market Forecast and Outlook By FMI
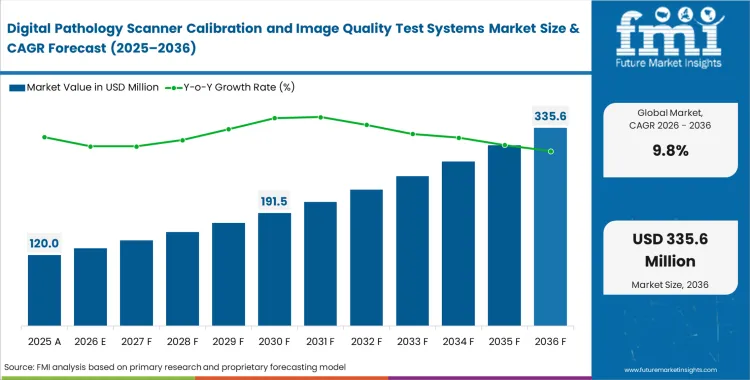
The digital pathology scanner calibration and image quality test systems market crossed a valuation of USD 109.3 million in 2025 with consistently progressing sales expected to surpass USD 120.0 million in 2026 at a CAGR of 9.8% during the forecast period. Demand outlook lifts total valuation to USD 305.6 million through 2036 as clinical laboratories implement mandatory pixel-level standardization protocols to prevent diagnostic algorithms from failing on uncalibrated whole slide imaging calibration systems.
Hospital laboratories are under heavy pressure to make sure every digital scanner across their network produces the exact same tissue colors. If they try to run new artificial intelligence tools on unchecked machines, the software silently misses critical disease patterns. This creates massive legal and malpractice risks for the healthcare provider. Waiting to buy proper quality control systems usually backfires. When inspectors audit the lab, staff often have to manually rescan thousands of slides just to prove the images are accurate. Building a reliable digital network simply cannot happen without strict image quality control.
Summary of Digital Pathology Scanner Calibration and Image Quality Test Systems Market
- Market Snapshot
- The digital pathology scanner calibration and image quality test systems market was valued at USD 109.3 million in 2025 and is set to climb to USD 305.6 million by 2036.
- Growth across this sector is projected at a 9.8% CAGR between 2026 and 2036, unlocking an additional USD 185.6 million in new revenue during this timeline.
- Buyers view this as a necessary insurance policy for their daily workflow, valuing exact color matches, sharp focus, and reliable screen performance far above the initial cost of the hardware itself.
- Sales naturally follow the adoption of primary whole-slide imaging systems used for standard preserved tissue samples, while faster, less standardized methods like frozen sections or fluid analysis typically bypass these strict calibration requirements.
- Demand and Growth Drivers
- Clinical laboratories must now prove their digital scanners perform identically to secure regulatory approval, turning image-quality testing from an optional check into a mandatory daily requirement.
- Recent studies prove that diagnostic algorithms perform much better across different hospital sites once physical scanners are mathematically color-matched, directly pushing IT officers to buy these tools.
- Lab workers face intense pressure to reduce the number of slides they have to rescan, forcing them to invest in quality assurance software that keeps machines running smoothly and prepares images for artificial intelligence, despite the ongoing maintenance expenses.
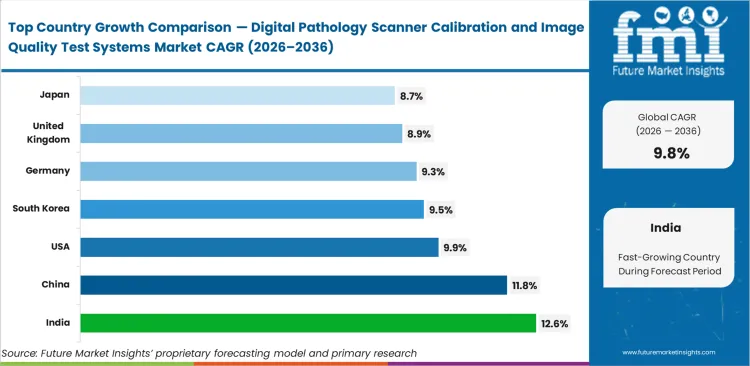
- National adoption rates vary based on local infrastructure build-outs. India is likely to lead global expansion at a 12.6% CAGR as it builds out remote diagnostic networks. China anticipated to follow closely behind, expanding at an 11.8% CAGR to handle massive screening volumes. The United States market poised to advance at a 9.9% CAGR through strict regulatory compliance, while South Korea set to post a 9.5% CAGR driven by early AI integration. Strict European standards likely to push Germany to a 9.3% CAGR, just ahead of the United Kingdom, which is estimated to tracks at an 8.9% CAGR. Japan forecasted to complete the major markets, set to scale at an 8.7% CAGR as aging populations drive steady testing demand.
- Budget constraints force many facilities to delay these purchases, treating calibration software and specialized monitors as secondary upgrades only added after the primary imaging hardware is fully paid for.
- Product and Segment View
- Available systems include physical glass test slides, specialized software, third-party validation services, monitor testing tools, and physical upkeep kits that guarantee diagnostic accuracy.
- These setups support a wide range of facilities, from massive reference laboratories processing thousands of daily samples to specialized biopharma research centers training proprietary deep learning models.
- Recurring demand for physical ground-truth testing helps the test slides component capture 34.0% share in 2026, serving as the foundation for daily scanner acceptance checks and cross-site alignment.
- Routine tissue staining dominates daily clinical workflows, pushing the brightfield segment to secure a massive 61.0% share in 2026 as hospitals prioritize their highest-volume diagnostic equipment.
- With a projected 27.0% share in 2026, the image sharpness segment ranks are projected as the top priority because focus failures and blurry scans cause the most immediate and visible disruptions to daily lab work.
- Managing decentralized fleets across multiple buildings allows hospitals anticipated to represent 38.0% share in 2026, driven by high clinical volumes and strict regulatory oversight.
- Daily testing mandates require continuous consumption of calibration materials, helping the routine QA segment likely to emerge with an estimated 41.0% share in 2026 as facilities test machines before every morning shift.
- Geography and Competitive Outlook
- Rapid hospital expansion pushes India, China, and South Korea forward as the fastest-growing territories, while the United States provides a stable, high-value foundation because its digital clinical networks are already deeply established.
- Major equipment manufacturers frequently bundle these testing tools directly with their primary machines to lock in buyers early. Independent software developers and specialized monitor companies push back by offering universal tools that prepare any brand of scanner for artificial intelligence integration, featuring key participants like Leica Biosystems, Hamamatsu, Philips, Roche Diagnostics, 3DHISTECH, and Barco.
- The competitive field for the key players is fragmented because these quality assurance layers are usually packaged inside larger equipment deals rather than sold on their own.
Strict government rules are the main reason hospitals are buying these systems right now. Regulators increasingly demand hard, mathematical proof of color accuracy before clearing new diagnostic software. Inspectors no longer accept a human technician just looking at a screen and guessing the color is right. As soon as these visual checks are rejected, lab officers are forced to buy universal validation tools that test any brand of scanner just to keep their facilities compliant and open.
Baseline divergence centers on whether a country relies on remote human interpretation or localized algorithmic screening with India forecasted to drive adoption at 12.6%, expanding telepathology networks across remote areas. China anticipated to post a 11.8% growth on the back of aggressive high-throughput digitization targets. United States facilities likely to record 9.9% expansion, focusing on FDA compliance for secondary consultation setups. South Kore projected to advance at 9.5%, while Germany expected to secure a 9.3% through strict European standard enforcement. United Kingdom and Japan poised to round out the major adopters at 8.9% and 8.7% respectively.
Segmental Analysis
Digital Pathology Scanner Calibration and Image Quality Test Systems Market Analysis by Component
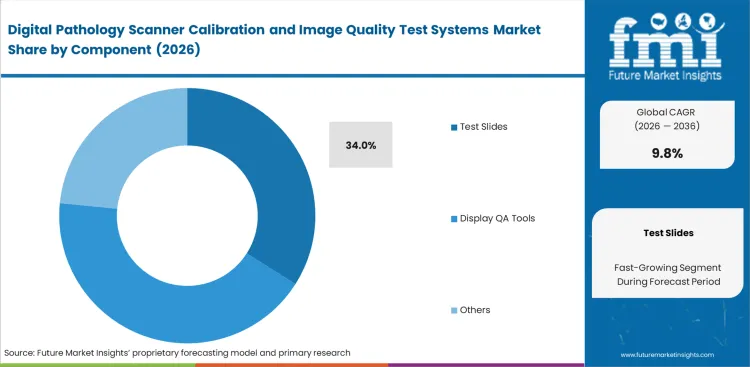
Laboratory quality officers rely on specialized glass targets to satisfy strict daily accreditation requirements. These physical digital pathology test slides establish an unalterable optical baseline. Software uses this physical baseline to identify subtle light source decay over time. FMI projects the test slides category to represent 34.0% of market share in 2026 as active diagnostic facilities continuously consume these items. Facilities relying solely on software without physical testing face severe operational penalties during routine regulatory audits. Integrating digital pathology displays further increases the need for constant physical-to-screen validation.
- Physical Ground Truth: Glass targets establish an unalterable optical baseline for scanner sensors. Laboratory QA officers secure daily operational compliance against CAP guidelines, avoiding sudden laboratory shutdown mandates.
- Automated Drift Detection: Calibration algorithms track color shifts across thousands of scanned pixels. Pathology IT officers reduce morning verification time, preventing workflow bottlenecks prior to peak scanning hours.
- Monitor Luminance Validation: External sensor tools verify medical-grade display brightness levels. Diagnostic pathologists maintain visual confidence in subtle tissue contrast, eliminating false negative cancer screening results.
Digital Pathology Scanner Calibration and Image Quality Test Systems Market Analysis by Scanner Type
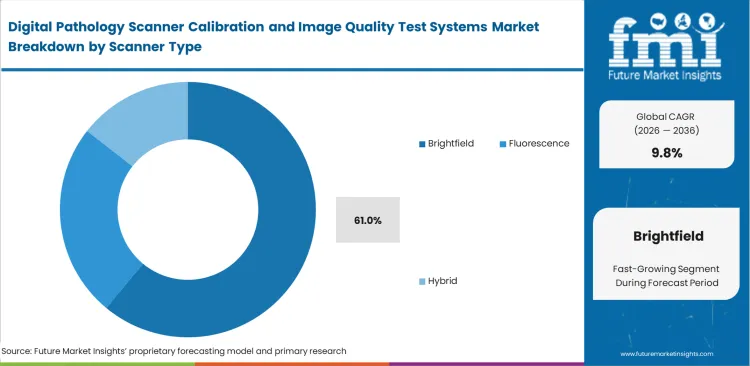
In 2026, the brightfield segment is poised to account for 61.0% of market share due to the massive volume of routine hematoxylin and eosin staining handled by core hospital laboratories. Sourcing officers naturally prioritize their validation budgets for these specific high-throughput units. Routine brightfield scanning forms the vulnerable backbone of daily clinical diagnosis. Algorithmic screening tools deployed across digital telepathology networks generate massive false-positive rates when they process uncalibrated brightfield data. Resolving this baseline focus degradation remains an immediate clinical priority to prevent system-wide diagnostic errors.
- Routine Stain Standardization: Algorithmic profiles correct daily variations in hematoxylin and eosin slide imaging. Buyers extend legacy scanner lifespans by mathematically correcting minor hardware degradation over time.
- Multichannel Alignment: Fluorescence targets verify exact spatial registration across multiple emission wavelengths. Research imaging officers ensure precise biomarker colocalization, preventing failed clinical trial data submissions.
- Throughput Optimization: High-speed calibration targets analyze focus mapping during continuous loading. Operations group sustain peak scanning rates without sacrificing critical optical sharpness parameters.
Digital Pathology Scanner Calibration and Image Quality Test Systems Market Analysis by Quality Parameter
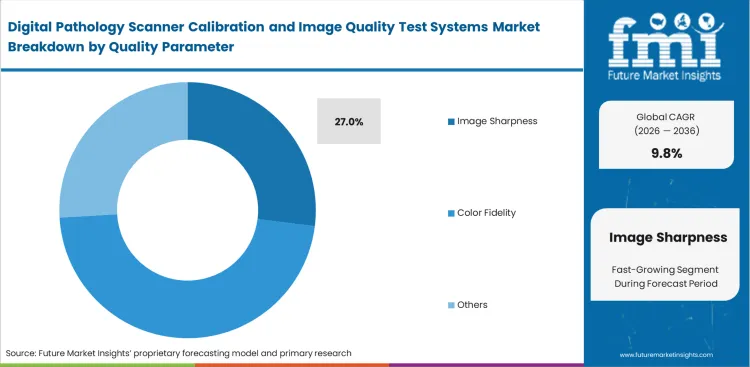
Vertical mapping errors ruin entire batches of digitized tissue samples. Technicians must rescan compromised slides manually. Pathology officers prioritize reliable pathology scanner focus tests to prevent these workflow delays caused by uneven tissue thickness. Autofocus mechanisms wear down mechanically much faster than LED light sources decay optically. This reality pushes facilities to prioritize focal adjustments, with the image sharpness segment projected to secure a 27.0% share in 2026 across global installations. Advanced quantitative pathology imaging software cannot calculate cellular metrics accurately without perfectly established focal planes.
- Focal Mapping Verification: Focus targets test the scanner's ability to track uneven tissue thickness. Pathology officers eliminate out-of-focus slide rejections, saving hours of manual rescanning labor.
- Illumination Consistency: Uniformity tools detect dust or sensor degradation across the imaging field. Laboratory technicians avoid artificial shading artifacts that confuse automated cell-counting algorithms.
- Color Profile Normalization: Standardized dye targets ensure exact hue reproduction across different hardware brands. Clinical AI leads deploy identical diagnostic models across heterogeneous hospital networks without retraining algorithms.
Digital Pathology Scanner Calibration and Image Quality Test Systems Market Analysis by End User
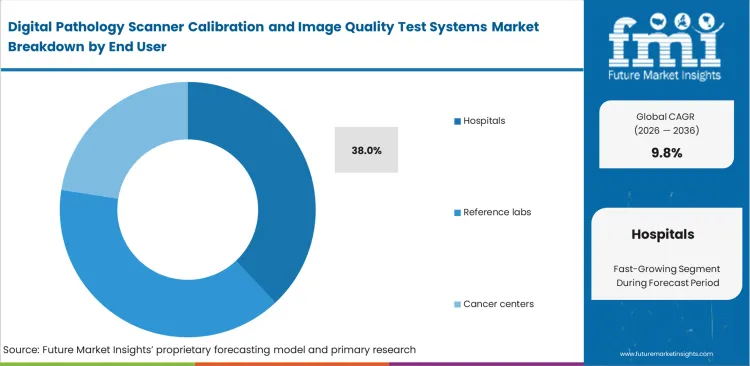
Network administration officers need identical digital outputs across multiple campus locations to allow central specialists to review slides safely. Hospitals purchase expensive quality assurance networks primarily to defend against malpractice audits rather than to accelerate diagnostic speed. Based on this need for strict fleet synchronization, hospitals are likely set to represent 38.0% of market share in 2026 as they deploy standardized digital pathology scanners across satellite labs. Failure to implement identical calibration standards forces central pathologists to reject external scans. This rejection destroys the economic advantage of consolidated diagnostic reviews and disrupts cellular pathology workflows.
- Fleet Synchronization: Cloud-based software tracks calibration data across multiple regional facilities. Network administration officers route digital slides to any available specialist without worrying about location-specific image variation.
- High-Volume Throughput Tracking: Scanner calibration for reference labs automates morning verification protocols using robotic slide handlers. Operations group process thousands of daily samples without losing production hours to manual system testing.
- Algorithm Validation Environments: Biopharma laboratories utilize strict color targets to train proprietary deep learning models. Research scientists guarantee regulatory bodies that clinical trial data relies on mathematically stable imaging platforms.
Digital Pathology Scanner Calibration and Image Quality Test Systems Market Analysis by Workflow Stage
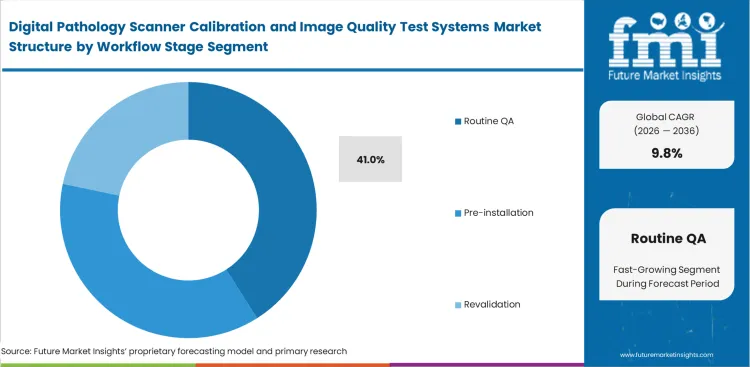
Quality assurance officers run standardized resolution targets every morning before loading active patient samples. This daily testing frequency ensures the routine QA segment is estimated to account for 41.0% share in 2026, consuming budget allocations much faster than one-time installation checks. Executing a strict digital pathology revalidation workflow proves daily compliance with clinical laboratory improvement amendments. Skipping morning calibration leaves diagnostic facilities legally vulnerable to false misdiagnosis claims if a scanner fails mid-shift. Managing legacy pathology devices demands even more intensive daily scrutiny to keep aging optical components within safe diagnostic tolerance limits.
- Daily Baseline Verification: Technicians run standardized resolution targets before loading patient samples. Quality assurance officers satisfy external regulatory auditors, preventing costly facility accreditation suspensions.
- Algorithm Prep Normalization: Software normalizes image color spaces before feeding data into deep learning models. Clinical AI leads drastically reduce model false-positive rates, accelerating automated triage workflows.
- Post-Repair Validation: A dedicated digital pathology quality control service verifies optical integrity following preventive maintenance. Laboratories confidently return hardware to clinical service without risking patient diagnostic accuracy.
Digital Pathology Scanner Calibration and Image Quality Test Systems Market Drivers, Restraints, and Opportunities
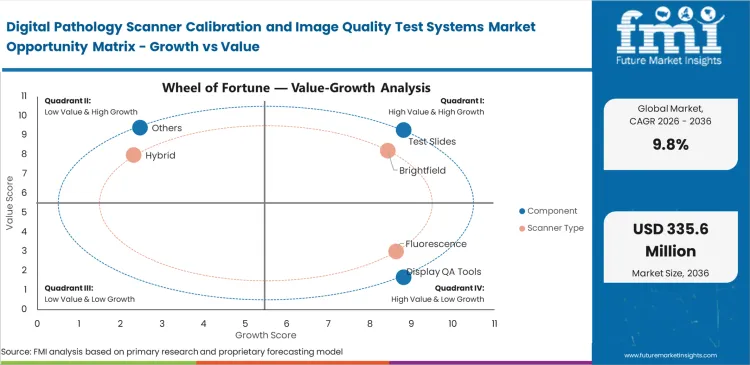
Diagnostic software simply cannot read uncalibrated slide images accurately. When hospital IT teams try to run new AI tools on unchecked scanners, the programs fail to recognize basic tissue patterns. Fixing these errors manually for every individual scanner costs too much time and money. Hospital officers now view objective validation tools as mandatory purchases rather than optional upgrades. Preparing equipment for algorithmic analysis directly accelerates new procurement contracts.
Taking scanners offline for daily testing creates a massive bottleneck for high-volume laboratories. A full morning calibration check pulls a machine out of service for up to twenty minutes exactly when technicians need to start processing overnight slide backlogs. Operations officers must constantly balance strict quality control requirements against the pressure to finish daily patient caseloads. Fast automated testing software helps reduce this delay. Technicians still have to load physical test slides manually, a step that actively pauses regular clinical work.
- Built-In Scanner Testing: Putting microscopic test targets permanently inside the machine removes the need for morning setup delays. Technicians can keep running patient slides all day while the equipment monitors its own optical accuracy.
- Fixing Old Slide Archives: New software programs can analyze and correct color errors in older digital files. Research teams can utilize years of archived anatomic pathology records to train modern diagnostic software.
- Universal Management Software: Hospital networks frequently operate multiple scanner brands across different buildings. Creating a single software platform that connects every machine lets IT officers standardize pathology devices across the entire hospital system from one central desk.
Regional Analysis
Regional adoption depends heavily on local healthcare infrastructure and direct regulatory pressures. Mature markets prioritize strict legal compliance frameworks, while developing economies scale remote diagnostic networks to bridge severe specialist shortages.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 12.6% |
| China | 11.8% |
| United States | 9.9% |
| South Korea | 9.5% |
| Germany | 9.3% |
| United Kingdom | 8.9% |
| Japan | 8.7% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Digital Pathology Scanner Calibration and Image Quality Test Systems Market Analysis
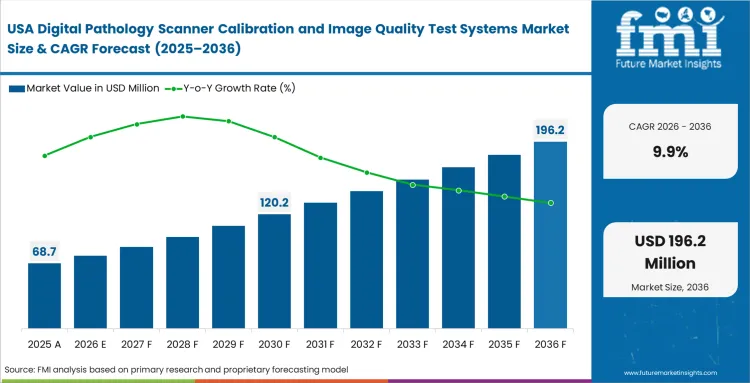
- United States: Strict clearance pathways for secondary diagnostic software compel American hospitals to prove their physical hardware remains optically stable. Network administrators secure continuous reimbursement for remote consultations by documenting this baseline fidelity, helping the United States market grow at a CAGR of 9.9% during the study period. Specialized domestic digital cytology networks benefit directly from these enforced localized compliance rules.
FMI notes, North American adoption remains tightly bound to legal defense and specific reimbursement criteria rather than pure clinical throughput expansion.
Europe Digital Pathology Scanner Calibration and Image Quality Test Systems Market Analysis
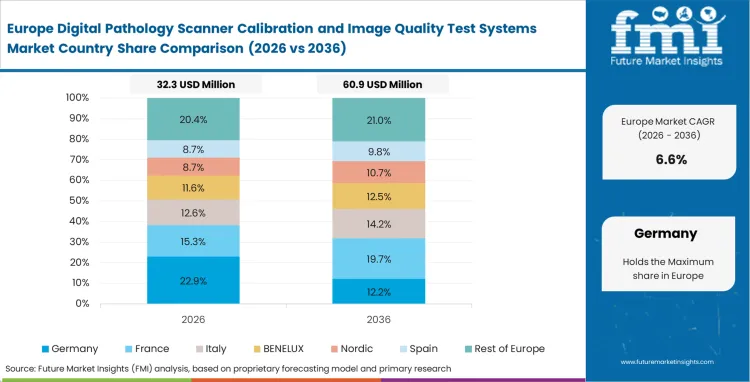
- Germany: Germany is projected to witness a 9.3% CAGR in this sector through 2036 as regional health authorities heavily penalize facilities that fail to update their legacy hardware validation protocols. Adherence to strict European in vitro diagnostic regulations forces quality assurance officers to purchase automated testing tools to avoid these severe operational disruptions.
- United Kingdom: National Health Service consolidation initiatives force distinct geographic trusts to establish identical optical baselines. Purchasing officers negotiate massive software licensing deals to synchronize image outputs across these regional networks, positioning the UK sector to register a CAGR of 8.9% from 2026 to 2036. Remote diagnostic networks require absolute mathematical image stability to function safely across trust borders.
Per FMI’s assessments, European procurement strategies clearly prioritize cross-border standardization and public health network synchronization over individual clinic upgrades.
Asia Pacific Digital Pathology Scanner Calibration and Image Quality Test Systems Market Analysis
- China: National mandates tied to hyper-scale laboratory environments are pushing adoption higher in China, where the market is expected to grow at a CAGR of 11.8% over the forecast period to handle very large daily scanning volumes. Robotic QA systems are becoming important in maintaining workflow continuity under heavy clinical throughput conditions.
- India: Large telepathology deployments across rural regions often operate without on-site optical technicians to manage equipment. That is increasing investment in remote calibration tools, with India projected to record a CAGR of 12.6% during the assessment period. Automated oversight plays a central role in linking rural patients with urban oncologists while supporting reliable slide review across distributed settings.
- Japan: Aging demographics are generating high daily cancer screening volumes that place growing pressure on traditional pathology workflows. Japan is likely to post a CAGR of 8.7% by 2036 as laboratories work to make better use of limited technician capacity. Automated daily verification sequences help reduce rescan bottlenecks when routine slide volumes remain consistently high.
- South Korea: Clinical AI developers are moving quickly to deploy models across highly connected metropolitan hospital networks. These systems require tightly standardized imaging output from base hardware, which is helping South Korea advance at a CAGR of 9.5% during the study period. Objective quality testing is increasingly treated as a baseline requirement before AI-enabled diagnostics can be used at scale
FMI analyses, Asia Pacific expansion relies entirely on scaling automated diagnostic infrastructure to compensate for concentrated populations and a lack of localized specialists.
Competitive Aligners for Market Players
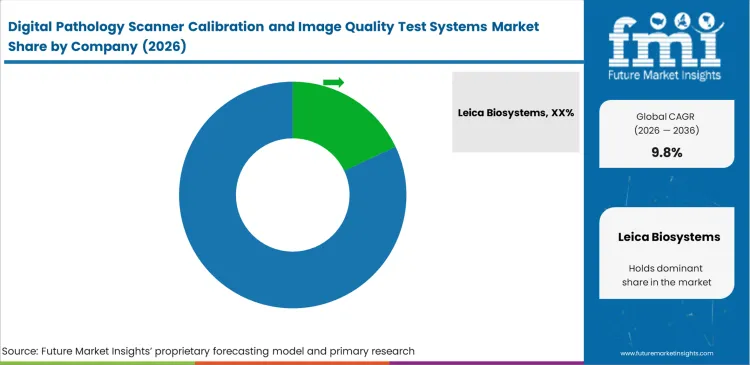
Leica Biosystems, Hamamatsu Photonics, and Roche Diagnostics essentially control the validation landscape by forcing buyers to use proprietary calibration tools alongside their primary scanners. Hospitals initially tolerate these closed ecosystems just to get their digital operations running without integration headaches. Independent software developers like 3DHISTECH disrupt this dynamic by offering validation platforms that operate across multiple machine brands. FMI notes that buying officers are actively rejecting single-vendor lock-in. They now specifically seek a versatile digital pathology QA vendor capable of normalizing optical data from any scanner in their network.
Raw optical data streams give incumbent manufacturers a massive structural edge over third-party challengers. Companies like Koninklijke Philips use their vast installed base to build highly accurate, device-specific degradation models that outside software simply cannot replicate. Independent developers face the grueling task of reverse-engineering undocumented digital slide formats to build clinical diagnostics integration libraries. This technical friction pushes laboratory buyers to find a reliable pathology scanner calibration supplier that can bridge vendor gaps without losing critical image metadata.
IT officers at large healthcare networks now write mandatory interoperability standards into all new procurement contracts. They deliberately source independent display QA tools from companies like Barco to verify image quality outside the primary scanner's proprietary ecosystem. Laboratories completely reject systems that rely on manual calibration checks. They demand the best digital pathology QA software that feeds compliance reports directly into central anatomic pathology information systems. If a software platform fails to automatically document performance across mixed scanner brands, hospital administrators immediately remove it from consideration.
Key Players in Digital Pathology Scanner Calibration and Image Quality Test Systems Market
- Leica Biosystems
- Hamamatsu Photonics
- Koninklijke Philips
- Roche Diagnostics
- 3DHISTECH
- Barco
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 120.0 million to USD 305.6 million, at a CAGR of 9.8% |
| Market Definition | Scanner image quality assurance pathology involves the physical targets, optical measurement software, and specialized protocols required to verify digital slide accuracy. These setups ensure diagnostic scanners capture physical tissue properties with mathematical consistency across varied lighting and focal conditions. |
| Segmentation | By component, scanner type, quality parameter, end user, and workflow stage |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Countries Covered | United States, Germany, United Kingdom, China, India, Japan, South Korea |
| Key Companies Profiled | Leica Biosystems, Hamamatsu Photonics, Koninklijke Philips, Roche Diagnostics, 3DHISTECH, Barco |
| Forecast Period | 2026 to 2036 |
| Approach | Annual scanner installation volumes cross-referenced with mandatory daily QA protocol adoption rates anchor the projection. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Segments
By Component
- Test slides
- Calibration software
- Validation services
- Display QA tools
- Maintenance kits
By Scanner Type
- Brightfield
- Fluorescence
- Hybrid
- High-throughput
- Compact
By Quality Parameter
- Image sharpness
- Color fidelity
- Focus accuracy
- Scan uniformity
- Display luminance
By End User
- Hospitals
- Reference labs
- Cancer centers
- Academic labs
- Biopharma labs
By Workflow Stage
- Routine QA
- Pre-installation
- Revalidation
- Troubleshooting
- AI normalization
Regions:
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Bibliography
- Food and Drug Administration. (2024). Use of whole slide imaging in nonclinical toxicology studies: Questions and answers. USA Department of Health and Human Services.
- Zhang, D. Y., Venkat, A., Khasawneh, H., Sali, R., Zhang, V., & Pei, Z. (2024). Implementation of digital pathology and artificial intelligence in routine pathology practice. Laboratory Investigation, 104(9), 102111.
- Kubota, A., Shibata, M., Kikuchi, S., & Yoneyama, T. (2024). Colour reproduction evaluation of whole-slide imaging scanners for digital pathology. Computer Methods in Biomechanics and Biomedical Engineering: Imaging & Visualization, 12(1), 2359396.
- Jurgas, A., Wodzinski, M., D’Amato, M., van der Laak, J., Atzori, M., & Müller, H. (2024). Improving quality control of whole slide images by explicit artifact augmentation. Scientific Reports, 14(1), 17847.
- Ji, X., Salmon, R., Mulliqi, N., Khan, U., Wang, Y., Blilie, A., Olsson, H., Pedersen, B. G., Sørensen, K. D., Ulhøi, B. P., Kjosavik, S. R., Janssen, E. A. M., Rantalainen, M., Egevad, L., Ruusuvuori, P., Eklund, M., & Kartasalo, K. (2025). Physical color calibration of digital pathology scanners for robust artificial intelligence-assisted cancer diagnosis. Modern Pathology, 38(5), 100715.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Routine QA protocols determining daily physical slide utilization rates.
- Pathology scanner calibration challenges shaping high-throughput scanner adoption cycles.
- Digital pathology scanner procurement checklist requirements pushing physical baseline target consumption.
- Hardware-agnostic software demand altering incumbent vendor lock-in strategies.
- Telepathology network expansion driving remote color calibration deployment.
- Algorithmic failure liability forcing hospital IT officers into rigorous validation.
- Brightfield scanner vulnerabilities necessitating specific vertical mapping tools.
- Pathology image quality standardization mandates synchronizing decentralized pathology facilities.
Frequently Asked Questions
What is the digital pathology scanner calibration market?
The sector provides physical artifacts and software to verify optical fidelity and ensure continuous image consistency across scanning hardware.
Why is color calibration important in digital pathology?
It prevents diagnostic algorithms from failing silently on uncalibrated images, ensuring mathematical equivalence across different scanning environments.
How large is the market in 2025 and 2036?
The market is valued at USD 109.3 million in 2025 and is expected to reach USD 305.6 million by 2036.
Which segment leads scanner QA spending?
Routine QA leads as daily guidelines require continuous system baseline verification before processing active clinical samples.
Which countries are adopting digital pathology QA fastest?
India and China lead global growth, driven by massive telepathology network expansions and high-throughput digitization targets respectively.
Who are the key companies in pathology scanner QA?
Leica Biosystems, Hamamatsu Photonics, Koninklijke Philips, Roche Diagnostics, 3DHISTECH, and Barco lead the sector globally.
How does scanner calibration affect pathology AI accuracy?
Deep learning models fail on uncalibrated images, making pathology AI model normalization calibration a strict prerequisite for software deployment.
What drives demand for pathology image quality test systems?
Mandatory pixel-level standardization protocols and malpractice liability risks compel immediate laboratory compliance and hardware procurement.
How often should pathology scanners be validated?
Accreditation bodies require daily routine verification using physical glass test slides and optical tracking software.
What are whole slide image quality metrics?
These essential metrics include image sharpness, color fidelity, focus accuracy, scan uniformity, and display luminance.
Which tools are used for pathology scanner QA?
Laboratories utilize physical glass test slides, specialized calibration software algorithms, and dedicated display QA monitors.
How do hospitals differ from reference labs in QA demand?
Hospitals require fleet synchronization across decentralized networks, while reference labs need high-volume automated throughput verification.
What limits rapid global implementation?
Comprehensive morning calibration sequences consume valuable scanner throughput capacity, creating operational workflow bottlenecks in busy clinics.
What is included in scanner validation and revalidation?
It includes optical hardware baseline tracking, color profile normalization, and post-repair operational integrity verification.
Which scanner type leads adoption?
Brightfield systems hold 61.0% share in 2026 due to dominant routine staining workflows requiring continuous cross-site diagnostic equivalence.
Why is image sharpness prioritized?
Mechanical autofocus degrades faster than LED lighting, causing immediate clinical workflow disruption if undetected before slide processing.
Which component holds the largest share?
Test slides account for 34.0% share in 2026, providing the only regulatory-recognized physical optical ground truth.
What opportunity exists in software development?
Creating hardware-agnostic management platforms helps standardize outputs across mixed vendor scanner fleets without demanding exclusive lock-in.
How do calibration systems impact liver cancer diagnostics?
Standardizing subtle tissue color rendering prevents false negative evaluations during critical primary tumor screenings and remote specialist reviews.
How do these systems assist lung cancer diagnostics?
Precise focus tracking ensures pathologists identify subtle malignant cellular changes without rescanning compromised tissue sections manually.
How will the sector evolve by 2036?
Calibration mechanisms will migrate from external workflows directly onto the scanner stage for continuous, real-time optical verification.
What separates pathology scanner QC slides vs software?
Slides provide regulatory physical ground truth, while software tracks algorithmic color shifts continuously to detect hidden mechanical drift.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Component
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Component , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Component , 2026 to 2036
- Test Slides
- Display QA Tools
- Others
- Test Slides
- Y to o to Y Growth Trend Analysis By Component , 2021 to 2025
- Absolute $ Opportunity Analysis By Component , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Scanner Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Scanner Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Scanner Type, 2026 to 2036
- Brightfield
- Fluorescence
- Hybrid
- Brightfield
- Y to o to Y Growth Trend Analysis By Scanner Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Scanner Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Quality Parameter
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Quality Parameter, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Quality Parameter, 2026 to 2036
- Image Sharpness
- Color Fidelity
- Others
- Image Sharpness
- Y to o to Y Growth Trend Analysis By Quality Parameter, 2021 to 2025
- Absolute $ Opportunity Analysis By Quality Parameter, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- Reference labs
- Cancer centers
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Workflow Stage
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Workflow Stage, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Workflow Stage, 2026 to 2036
- Routine QA
- Pre-installation
- Revalidation
- Routine QA
- Y to o to Y Growth Trend Analysis By Workflow Stage, 2021 to 2025
- Absolute $ Opportunity Analysis By Workflow Stage, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Component
- By Scanner Type
- By Quality Parameter
- By End User
- By Workflow Stage
- Competition Analysis
- Competition Deep Dive
- Leica Biosystems
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Hamamatsu Photonics
- Koninklijke Philips
- Roche Diagnostics
- 3DHISTECH
- Barco
- Leica Biosystems
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Scanner Type, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Quality Parameter, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Scanner Type, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Quality Parameter, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Scanner Type, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Quality Parameter, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Scanner Type, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Quality Parameter, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Scanner Type, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Quality Parameter, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Scanner Type, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Quality Parameter, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Scanner Type, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Quality Parameter, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Scanner Type, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Quality Parameter, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Component
- Figure 6: Global Market Value Share and BPS Analysis by Scanner Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Scanner Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Scanner Type
- Figure 9: Global Market Value Share and BPS Analysis by Quality Parameter, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Quality Parameter, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Quality Parameter
- Figure 12: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End User
- Figure 15: Global Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Workflow Stage
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Component
- Figure 32: North America Market Value Share and BPS Analysis by Scanner Type, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Scanner Type, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Scanner Type
- Figure 35: North America Market Value Share and BPS Analysis by Quality Parameter, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Quality Parameter, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Quality Parameter
- Figure 38: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by End User
- Figure 41: North America Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Workflow Stage
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Component
- Figure 48: Latin America Market Value Share and BPS Analysis by Scanner Type, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Scanner Type, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Scanner Type
- Figure 51: Latin America Market Value Share and BPS Analysis by Quality Parameter, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Quality Parameter, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Quality Parameter
- Figure 54: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by End User
- Figure 57: Latin America Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Workflow Stage
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Component
- Figure 64: Western Europe Market Value Share and BPS Analysis by Scanner Type, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Scanner Type, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Scanner Type
- Figure 67: Western Europe Market Value Share and BPS Analysis by Quality Parameter, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Quality Parameter, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Quality Parameter
- Figure 70: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by End User
- Figure 73: Western Europe Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Workflow Stage
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Component
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Scanner Type, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Scanner Type, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Scanner Type
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Quality Parameter, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Quality Parameter, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Quality Parameter
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by End User
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Workflow Stage
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Component
- Figure 96: East Asia Market Value Share and BPS Analysis by Scanner Type, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Scanner Type, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Scanner Type
- Figure 99: East Asia Market Value Share and BPS Analysis by Quality Parameter, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Quality Parameter, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Quality Parameter
- Figure 102: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by End User
- Figure 105: East Asia Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Workflow Stage
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Component
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Scanner Type, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Scanner Type, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Scanner Type
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Quality Parameter, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Quality Parameter, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Quality Parameter
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Workflow Stage
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Component
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Scanner Type, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Scanner Type, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Scanner Type
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Quality Parameter, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Quality Parameter, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Quality Parameter
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Workflow Stage
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

