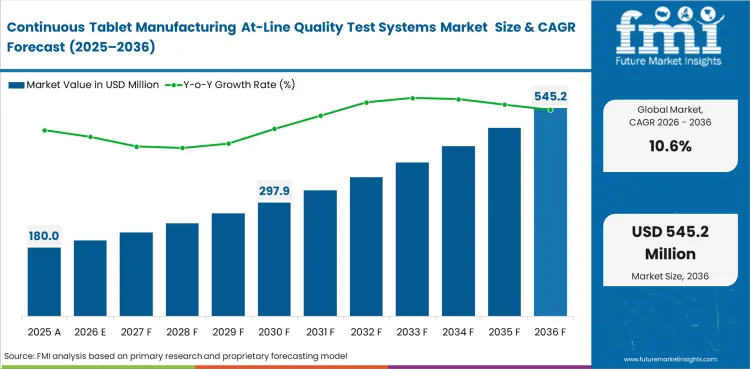
Continuous tablet manufacturing at-line quality test systems market crossed a valuation of USD 162.7 million in 2025 and is projected to reach USD 180 million in 2026. By 2036, the market is forecast to expand to USD 493 million at a 10.6% CAGR. Based on Future Market Insights analysis, the market is expected to create an incremental opportunity of USD 313 million during 2026 to 2036. Demand for continuous tablet manufacturing at-line quality test systems is rising because manufacturers need to detect variation earlier, keep tablet quality closer to production, and reply less on delayed laboratory results that come after the run.
| Parameter | Details |
| Market value (2026) | USD 180 million |
| Forecast value (2036) | USD 493 million |
| CAGR (2026 to 2036) | 10.6% |
| Estimated market value (2025) | USD 162.7 million |
| Incremental opportunity | USD 313 million |
| Leading test technology | NIR Spectroscopy (26.4% of test technology segment) |
| Leading quality attribute | Content Uniformity (23.8% of quality attribute segment) |
| Leading line type | Direct Compression (53.6% of line type segment) |
| Leading manufacturing stage | Compression (29.4% of manufacturing stage segment) |
| Leading system configuration | Standalone Modules (35.1% of system configuration segment) |
| Leading automation level | Fully Automated (48.7% of automation level segment) |
| Leading end user | Drug Manufacturers (43.6% of end user segment) |
| Leading throughput | Mid Throughput (38.2% of throughput segment) |
| Key players | Thermo Fisher Scientific, Bruker Corporation, SOTAX Group, Fette Compacting, GEA Group, Endress+Hauser, Sensum |
Source: Future Market Insights, 2026.
At-line measurements are becoming more important in tablet manufacturing because plant operations need faster visibility into variation during production. Early data helps teams fix problems before they affect more tablets. Equipment choice depends on measurement speed, stable readings and well-documented records. Adoption improves when manufacturers define how each reading supports routine process control and exception review.
India is expected to rise at 11.8% CAGR by 2036 because oral solid dosage expansion and line modernization are creating more room for first-time deployment. China is projected to grow at 11.2% CAGR as manufacturers continue adding capacity and upgrading line-side process control. Demand in the United States is anticipated to expand at 10.9% CAGR through 2036, supported by a large installed manufacturing base that can absorb added control systems without a full process reset. Germany is likely to register 10.1% CAGR, while Belgium is set to rise at 9.8% CAGR and Switzerland at 9.6% CAGR as established production environments continue adopting tighter control tools. Japan is expected to expand at 8.7% CAGR through 2036.
The continuous tablet manufacturing at-line quality test systems market includes instruments, modules, and software-linked testing units used near or within tablet production lines to assess critical quality attributes during continuous manufacturing. Scope remains limited to systems that support process control, better process visibility, exception review, and faster release readiness, which makes them functionally distinct from broader oral solid dosage pharmaceutical formulation activities that shape tablet design but do not themselves provide live line-side quality evidence.
The market includes spectroscopic, optical, imaging, and physical-testing systems used across blending, feed, compression, post-compression, and coating checkpoints in continuous tablet production. It also covers modular testers, integrated line units, and connected data-capture tools that support repeatable review during manufacturing, reflecting the same need for structured measurement and documented output seen in parts of the healthcare analytical testing services market, though here the role is tied directly to live production control rather than external laboratory support.
The scope excludes general offline laboratory analyzers, packaging counters, broad automation platforms that do not test tablet attributes directly, and downstream handling units used only for counting or secondary movement. Equipment used only after manufacturing is complete falls outside this market because it does not guide tablet quality decisions during production, unlike systems closer in purpose to process instrumentation equipment, where measurement has operational value only when it can influence real-time process understanding.
Demand for continuous tablet manufacturing at-line quality test systems is rising because tablet producers need quality evidence closer to the process. In continuous manufacturing, a small disturbance can spread quickly if it is not detected and contained in time. At-line testing matters because it gives earlier visibility, faster response, and better control over material at risk.
Content uniformity remains the leading quality checkpoint because release credibility starts with proof that tablet composition stays within acceptable limits during production. Manufacturers are giving more importance to systems that support repeatable checks without interrupting line flow, especially where quality teams need data that can feed directly into routine review and exception handling.
Expansion in oral solid dosage manufacturing is also supporting market growth, particularly in countries where new line installations and modernization programs are increasing first-time deployment. Manufacturers are assessing these systems as part of a broader control strategy that can reduce review delays, improve deviation tracing, and strengthen process confidence without forcing a full reset of manufacturing practice. Demand is becoming more relevant in routine production settings.
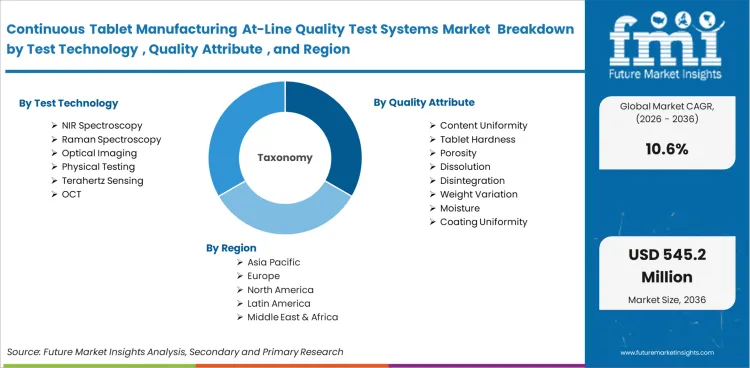
The continuous tablet manufacturing at-line quality test systems market is segmented by test technology, quality attribute, line type, manufacturing stage, system configuration, automation level, end user, and throughput. By Test technology, the market is divided into NIR spectroscopy, Raman spectroscopy, optical imaging, physical testing, terahertz sensing, and OCT. In terms of quality attribute, the market is classified into content uniformity, tablet hardness, porosity, dissolution, disintegration, weight variation, moisture, and coating uniformity. Based on line type, the market is segmented into direct compression, wet granulation, and dry granulation. By manufacturing stage, the market is divided into compression, blending, feed frame, post-compression, and coating. In terms of system configuration, the market is classified into standalone modules, integrated modules, skid systems, and portable units. Based on automation level, the market is segmented into fully automated, semi-automated, and manual assisted systems. By end user, the market is divided into drug manufacturers, CDMOs, technology centers, and academic labs. In terms of throughput, the market is classified into mid throughput, low throughput, high throughput, and pilot flexible systems.
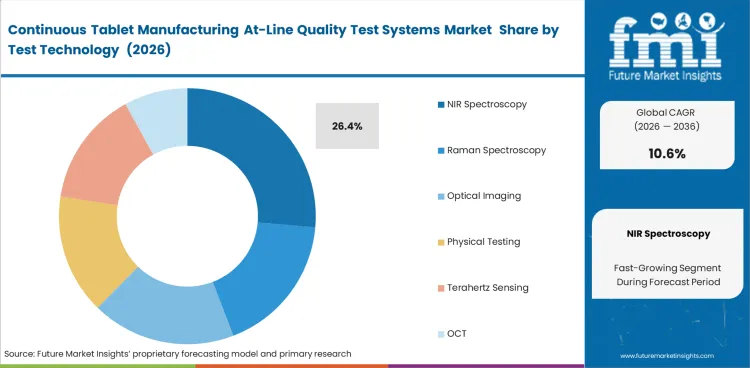
NIR Spectroscopy leads this segment because tablet manufacturing needs fast readings that fit routine review more easily. NIR Spectroscopy account for 26.4% share in 2026, supported by its ability to stay close to the process, limits sample handling, and supports repeat checks without interrupting production flow. Familiarity with IR spectroscopy and dual-wavelength Raman tools also helps to support adoption.
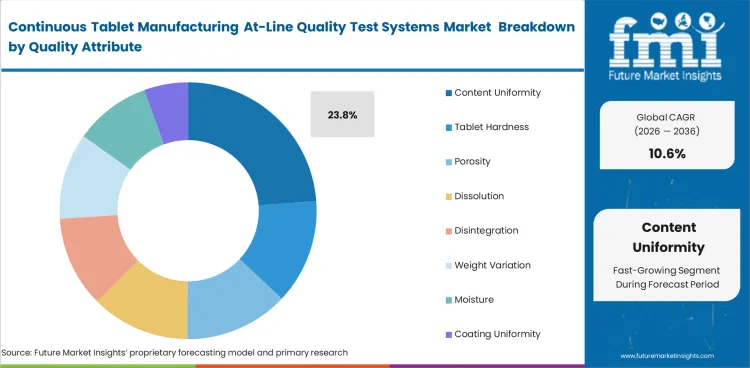
Content Uniformity leads this market because release decisions depend on stable tablet composition during the run. Content Uniformity is projected to hold 23.8% share in 2026 because many manufacturers keep at-line control focused on assay consistency until live production readings become more reliable. The relevance of healthcare analytical testing and pharmaceutical dissolution testing supports the same need.
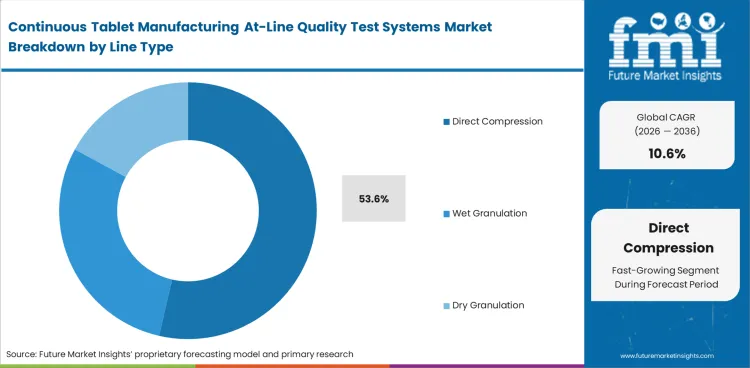
Direct Compression leads because continuous tablet manufacturing depends on testing points that fit one connected production flow. Direct Compression is likely to account for 53.6% share in 2026 because fewer unit operations make powder behavior, compression response, and finished tablet output easier to assess in sequence. Wet and dry granulation lines support at-line systems too, though added steps make data handoff and corrective action harder to standardize. The relevance of pharmaceutical continuous manufacturing equipment and oral solid dosage formulations supports this lead.
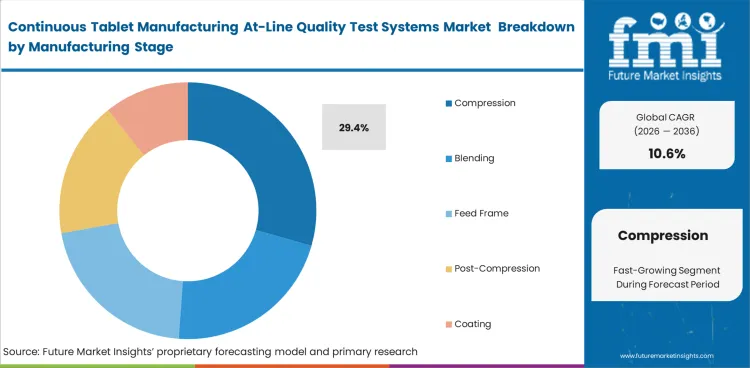
Compression is expected to account for 29.4% share in 2026 because weight consistency, hardness response, and visible defects become more important once the tablet is formed. Blending and feed stages remain relevant, though many manufacturers direct spending toward points where deviations have a clearer link to tablet acceptability. The relevance of tablet presses and process instrumentation equipment supports this lead because compression-stage decisions often combine machine behavior with attribute confirmation. Plants that invest too little at this stage may collect upstream signals without a clear view of finished-tablet impact.
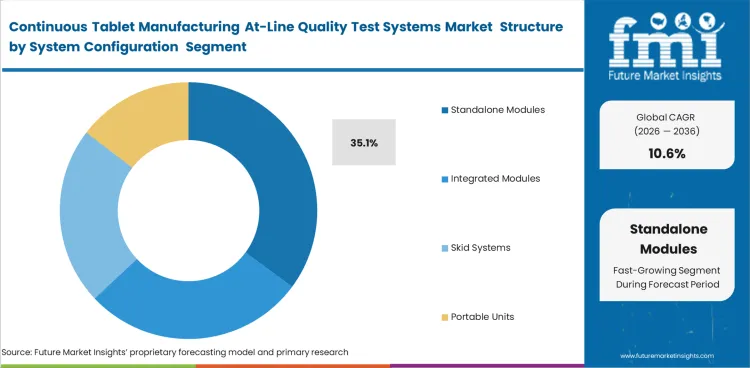
Standalone Modules is expected to account for 35.1% share in 2026 because phased adoption is easier when manufacturers are still deciding how data should be used, how alarms should be handled, and how validation requirements should be defined. Integrated modules gain become more important once programs are more established. Early spending often stays with equipment that can be introduced with less disruption to existing operations.
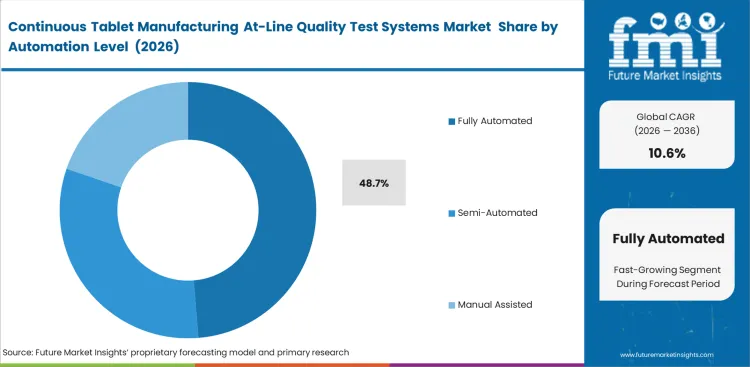
Manual review can support line operation, through it does not deliver the consistency continuous operations require. Fully Automated systems are anticipated to emerge with 48.7% market share in 2026 because repeatability depends on sampling, reading, alarm routing, and response with minimal operator variability. Semi-automated options fit smaller programs or development-heavy settings. Similar buying logic appears in adjacent process liquid analyzers and process spectroscopy categories.
The continuous tablet manufacturing at-line quality test systems market is expanding steadily as manufacturers place more weight on earlier quality visibility, tighter process control, and faster review readiness during live production. Adoption depends on whether measurement output can support repeatable operating action, fit validation logic, and hold up in routine quality review. Growth remains strong, though adoption is still shaped by qualification burden, integration complexity, and the practical challenge of turning line-side readings into defensible production decisions.
Demand is rising because continuous manufacturing leaves less time to detect problems after output has already moved through the line. Plants need at-line systems that show where variation begins, how long it lasts, and whether corrective action can still protect acceptable material. This is increasing the relevance of line-side quality checks in blending, compression, and post-compression stages, where earlier signals can reduce review delay and improve deviation tracing. Commercial value comes from helping manufacturers respond before a disturbance spreads across production.
Wider uptake remains constrained by the effort required to qualify each system within existing production and review structures. Manufacturers need more than accurate readings. They need clear rules for exception handling, alarm routing, operator response, and documentation fit before at-line output becomes part of normal plant control. This slows adoption, especially in compliance-heavy environments where quality teams are cautious about adding new control points that may increase validation work or create ambiguity during review.
Based on the regional analysis, Continuous Tablet Manufacturing At-Line Quality Test Systems Market is segmented into North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, and Middle East & Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 11.8% |
| China | 11.2% |
| United States | 10.9% |
| Germany | 10.1% |
| Belgium | 9.8% |
| Switzerland | 9.6% |
| Japan | 8.7% |
Source: Future Market Insights analysis, based on proprietary forecasting model and primary research
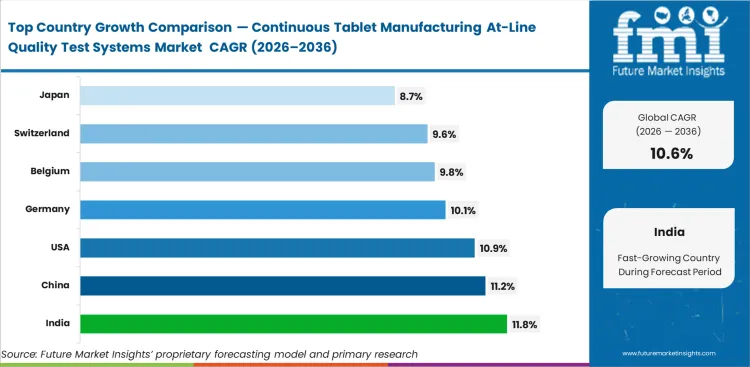
India is projected to expand at an 11.8% CAGR through 2036. Strong oral solid dosage manufacturing depth, ongoing capacity addition, and line modernization are supporting first-time deployment of at-line systems. Manufacturers want better live-run visibility without dependence on delayed laboratory confirmation. Earlier adoption can reduce review drag and improve how quickly deviations are traced to the source, which makes India the strongest growth market among the countries tracked.
China is expected to grow at a CAGR of 11.2% by 2036. Large-scale manufacturing and continued modernization support demand, through long-term adoption depends on how smoothly at-line systems fit high-throughput operations. Manufacturers want tools that improve control without increasing operator burden or slowing daily execution. Suppliers with stronger software handoff, cleaner integration, and practical plant support are better placed to expand in this market.
The United States is projected to record 10.9% CAGR from 2026 to 2036. A large installed manufacturing base gives the market greater capacity to absorb additional control systems without requiring a full process reset. Adoption is supported by familiarity with advanced manufacturing methods, though acceptance still moves through careful internal review. Plants gain value from at-line systems when they reduce review delay and improve operating confidence in routine production.
Germany is projected to expand at 10.1% CAGR through 2036. The market is shaped by a strong pharmaceutical equipment base and clear preference for repeatable operating fit. Manufacturers evaluate at-line systems on signal quality, service depth, and installation support. Vendors that can support disciplined rollout under compliance-heavy production conditions are better placed in Germany.
Belgium is expected to grow at a CAGR of 9.8% from 2026 to 2036. A concentrated pharmaceutical production base and strong process discipline support demand for at-line systems that shorten review cycles without weakening documentation quality. Adoption depends more on keeping quality decisions close to production while maintaining confidence during review.
Switzerland is projected to register a 9.6% CAGR through 2036. Demand comes from high-value manufacturing conditions where data credibility matters as much as instrument speed. Plants invest in dependable control, though they move carefully when new systems may complicate validated routines. Vendor selection depends on technical clarity, service quality, and long-use operating fit.
Japan is expected to expand at 8.7% CAGR through 2036. Plants move carefully because new control points must improve execution without increasing operating burden. Qualification discipline and use-case clarity matter strongly in system selection. This keeps adoption measured and supports stronger preference for long-term consistency than for rapid rollout.
The continuous tablet manufacturing at-line quality test systems market includes analytical instrument suppliers, tablet manufacturing technology specialists, and process-control participants serving pharmaceutical production environments that require repeatable quality evidence close to live operations. Competition depends on more than measurement capability. Manufacturers compare installation fit, service support, software handoff, validation readiness, and familiarity with oral solid dosage manufacturing before systems are accepted for routine use.
Thermo Fisher Scientific and Bruker Corporation remain well positioned because established analytical capability, technical credibility, and broader pharmaceutical reach support buyer confidence in regulated environments. SOTAX Group, Fette Compacting, and GEA Group benefit from closer alignment with tablet production workflows, where system acceptance depends on how well measurement tools fit operating practice rather than on reading speed in isolation. Endress+Hauser and Sensum remain relevant where buyers prioritize process integration, focused plant support, or more specialized deployment needs within continuous manufacturing lines.
Barriers to entry are moderate at the basic instrument level and higher where suppliers need to support validated plant use. A vendor may offer technically sound measurement, yet broader commercial adoption depends on proving that the system can hold up under documentation review, routine sampling discipline, and repeat-use production conditions.
Key global companies leading the continuous tablet manufacturing at-line quality test systems market include:
| Company | Analytical Capability | Validation Support | Integration Fit | Service / Application Support |
|---|---|---|---|---|
| Thermo Fisher Scientific | High | High | Moderate | Strong |
| Bruker Corporation | High | High | Moderate | Strong |
| SOTAX Group | High | High | Strong | Strong |
| Fette Compacting | Medium | Medium | Strong | Strong |
| GEA Group | Medium | Medium | Strong | Strong |
| Endress+Hauser | Medium | Medium | Moderate | Moderate |
| Sensum | Medium | Medium | Moderate | Moderate |
Source: Future Market Insights competitive analysis, 2026.
Recent Developments in Continuous Tablet Manufacturing At-Line Quality Test Systems Market
Major Global Players
Emerging Players / Specialized Participants
| Metric | Value |
|---|---|
| Quantitative Units | USD 162.7 million (2025) / USD 180.0 million (2026) to USD 493.0 million (2036), at a CAGR of 10.6% |
| Market Definition | The continuous tablet manufacturing at-line quality test systems market covers line-side and line-integrated tools used to measure tablet quality during continuous production. Scope remains limited to systems that support live manufacturing control, faster review, and repeatable quality decisions. General offline laboratory instruments without a direct continuous-tableting role remain outside scope. |
| Segmentation |
|
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, Middle East & Africa |
| Countries Covered | India, China, United States, Germany, Belgium, Switzerland, Japan, and 40 plus countries |
| Key Companies Profiled | Thermo Fisher Scientific, Bruker Corporation, SOTAX Group, Fette Compacting, GEA Group, Endress+Hauser, Sensum |
| Forecast Period | 2026 to 2036 |
| Approach | FMI combined primary interviews with plant and quality personnel, desk review of continuous tablet manufacturing use cases, and a bottom-up view of likely module deployment by line. Baseline assumptions were tied to installed line activity, adoption depth by quality task, and system configuration choices. Forecasts were checked through supplier coverage, adjacent equipment categories, and recurring buyer behavior seen in oral solid dosage manufacturing. |
Source: Future Market Insights analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is Continuous Tablet Manufacturing At-Line Quality Test Systems Market in 2026?
Continuous Tablet Manufacturing At-Line Quality Test Systems Market is expected to reach USD 180 million in 2026, showing clear commercial relevance.
What will the market be valued at by 2036?
FMI estimates market valuation will reach USD 493 million by 2036, reflecting broader use of line side testing.
What CAGR is projected for the market during 2026 to 2036?
FMI projects a CAGR of 10.6% from 2026 to 2036, indicating steady expansion in this specialized category.
Which Test Technology segment leads the market?
NIR Spectroscopy leads Test Technology, with 26.4% share expected in 2026 because it supports fast, repeatable checks.
Which Quality Attribute segment leads the market?
Content Uniformity leads the category, with 23.8% share expected in 2026, as assay consistency remains central.
Which Line Type segment leads the market?
Direct Compression leads Line Type, with 53.6% share expected in 2026 due to cleaner process flow.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.