Single-Use Bioprocess Sensor Calibration and Validation Test Systems Market
The Single-Use Bioprocess Sensor Calibration And Validation Test Systems Market Is Segmented By Sensor Type (Ph Sensors, DO Sensors, Pressure Sensors, Conductivity Sensors, Flow Sensors, Biomass Sensors), Test Type (Calibration Systems, Validation Rigs, Simulator Modules, Reader Units, Software Suites), Workflow (Upstream, Downstream, Media Prep, Buffer Prep, Fill-Finish), End User (Biopharma Manufacturers, CDMOs, Equipment OEMs, QC Labs, Academic Centers), Deployment (At-Line, In-Line, Off-Line, Portable, Integrated), And Region. Forecast For 2026 To 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Single-Use Bioprocess Sensor Calibration and Validation Test Systems Market Size, Market Forecast and Outlook By FMI
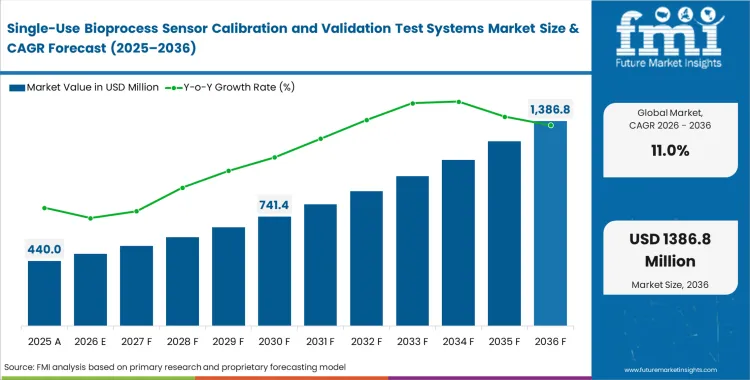
The single-use bioprocess sensor calibration and validation test systems market crossed a valuation of USD 400 million in 2025, with increasing sales across the globe are poised to scale USD 440 million in 2026 at a CAGR of 11.00% during this forecast period. The constantly expanding industry revenue carries overall opportunity to USD 1,250 million through 2036 as regulatory agencies shift from accepting post-batch statistical quality control to demanding pre-use parametric verification for disposable fluid paths.
Summary of Single-Use Bioprocess Sensor Calibration and Validation Test Systems Market
- The market is forecast to reach USD 1,250 million by 2036.
- The market is expected to grow at a CAGR of 11.0% from 2026 to 2036.
- The market was estimated at USD 400 million in 2025.
- The forecast period represents an incremental opportunity of USD 810 million.
- Regulatory shift toward pre-use parametric verification and rapid adoption of single-use bioprocessing systems are the primary demand drivers.
- Increasing deployment of real-time monitoring and digital factory integration is accelerating demand for automated calibration and validation systems.
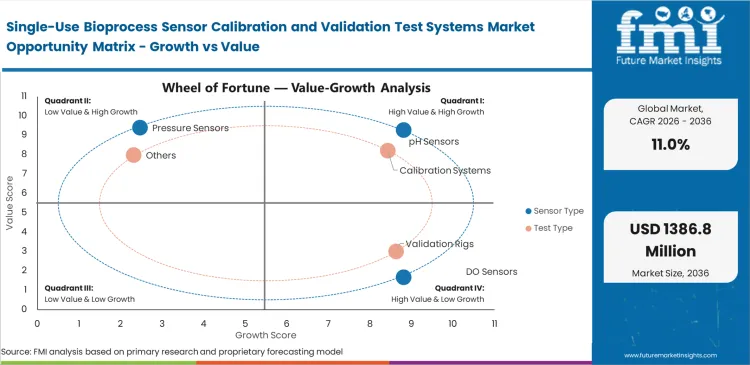
- Calibration systems lead the segment with a 38.0% share, while pH sensors dominate sensor focus with a 29.0% share.
- China, India, and the United States are key growth markets, supported by expansion of biopharmaceutical manufacturing capacity.
- Thermo Fisher Scientific, Hamilton Company, Cytiva, PreSens Precision Sensing, Mettler-Toledo, Broadley-James, and Sartorius are among the key players in the market.
Quality assurance leaders are running out of time to update their compliance methods. Handing a European inspector a paper certificate from the plastic supplier simply is not enough to pass GMP validation for single-use sensors anymore. Plant managers must now physically test and calibrate these sensors right on the production floor before starting any cell cultures. Contract manufacturers that put off buying this testing equipment end up scrambling to investigate costly failures after a slightly inaccurate new sensor ruins an entire high-value production run. Plastic single-use bioprocessing probes and sensors naturally fluctuate more than traditional stainless-steel equipment. This reality makes continuous disposable bioprocess sensor validation a mandatory cost of doing business, rather than just an optional safety net.
Installing automated bioprocess validation test rigs that connect directly to a factory's digital records changes the entire workflow. Pre-run testing times drop from three hours per single-use bioreactor down to under twenty minutes. This direct digital connection stops operators from making manual logging mistakes. It turns heavy single-use sensor qualification biopharma requirements into a quick setup chore instead of a massive operational bottleneck.
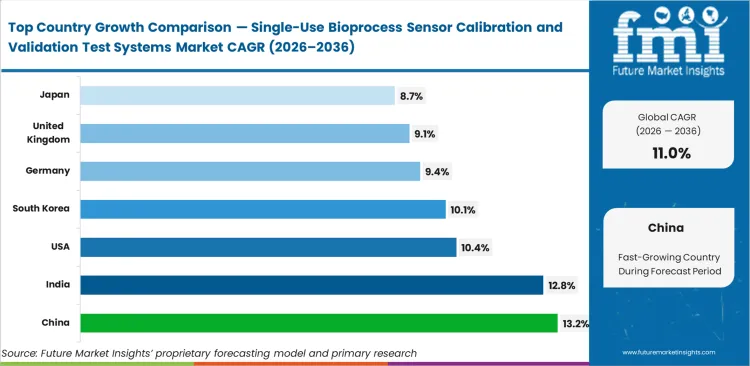
Germany is expected to advance at a 9.4% CAGR while United Kingdom demand likely to register at 9.1% amid regional pushes for closed-loop continuous processing. Japan projected to follow at an anticipated 8.7% growth. China poised to lead the regional adoption at an estimated 13.2% as local biologic manufacturing shifts aggressively from legacy stainless facilities toward entirely disposable infrastructure. India set to track closely at a predicted 12.8% on the back of surging biosimilar export volumes requiring stringent biopharma sensor calibration documentation. South Korea is seemingly ready to showcase a scale of 10.1% compound growth as domestic contract manufacturers secure large-scale commercial supply agreements requiring dedicated single-use bioprocess sensor calibration systems. United States expansion expected to run at 10.4% as early-generation disposable facilities retrofit automated reader units into existing suites. Structural divergence separates high-growth Asian hubs building greenfield disposable capacity from Western regions retrofitting complex calibration rigs into established quality control ecosystems.
Segmental Analysis
Single-Use Bioprocess Sensor Calibration and Validation Test Systems Market Analysis by Sensor Type
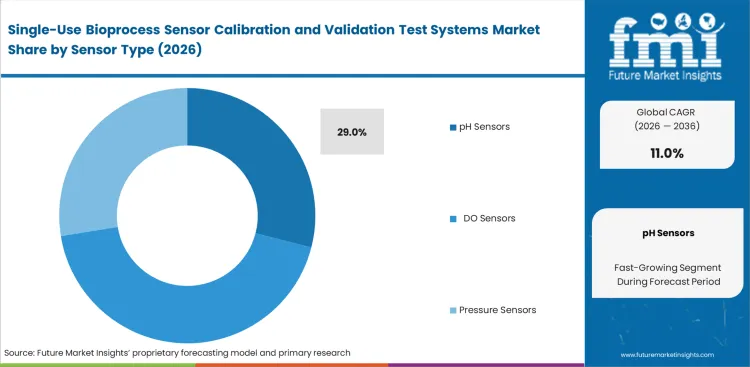
Maintaining exact acidity levels in mammalian cell cultures causes ongoing operational friction because optical dyes break down over time. Bioreactor operators understand that radiation sterilization changes the baseline accuracy of optical patches before they even reach the facility floor. The pH Sensors segment is anticipated to capture 29.0% of the market in 2026 as plant managers enforce mandatory pre-run calibration checks to avoid costly batch failures. Ignoring this benchtop step leaves downstream processing teams dealing with ruined protein structures. Many validation engineers miss how often standard bioprocess validation equipment struggles to correct for temperature shifts during long production runs. Companies that skip these verification routines end up throwing away highly valuable biological products due to unrecorded media changes.
- Calibration-Curve Correction: Optical dye degradation post-sterilization requires immediate mathematical offset calculation. Upstream managers gain reliable baseline readings while avoiding disastrous metabolic shifts during exponential growth phases.
- Temperature-Drift Compensation: Extended cultivation duration exposes patches to physical stress. Bioreactor operators must apply algorithmic correction factors to prevent false acidic readings from triggering unnecessary base additions.
- Post-Run Verification: Quality assurance teams extract used patches to confirm zero drift occurred during production. Regulatory affairs directors face immediate audit citations if final documentation lacks post-use stability proof.
Single-Use Bioprocess Sensor Calibration and Validation Test Systems Market Analysis by Test Type
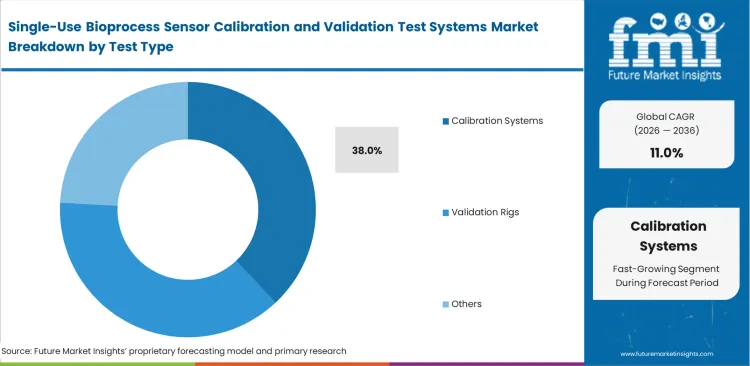
Market models indicate the calibration systems category is expected to hold a 38.0% share in 2026, driven by facilities abandoning slow manual buffer checks. Hand-verifying sensors wastes valuable cleanroom hours and exposes operations to manual logbook mistakes. Plants shifting toward automated closed system bioprocessing buy specialized benchtop readers to run verification sequences without human input. Validation teams simply connect un-inoculated bags to these consoles and let the pre-programmed tests run. High-end simulator modules sometimes complicate this by outputting raw hexadecimal data, forcing IT departments to buy extra middleware to sync results with electronic batch records. Factories avoiding the cost of these automated setups face constant labor bottlenecks during suite changeovers.
- Zero-Point Alignment: Automated gas-mixing stations flow precise nitrogen-oxygen blends over dry sensors. Validation engineers establish true baseline values before media fill, ensuring accurate single-use dissolved oxygen sensor validation.
- Linearity Confirmation: Benchtop modules test sensor responses across full expected operational ranges. Quality managers avoid discovering non-linear signal responses mid-way through critical high-density cell cultures.
- Data-Integrity Lock: Modern rigs automatically push calibration results directly to facility servers without human intervention. Compliance officers achieve 21 CFR Part 11 requirements while eliminating manual logbook entry deviations.
Single-Use Bioprocess Sensor Calibration and Validation Test Systems Market Analysis by Workflow
Primary fermentation requires perfect environmental control to keep sensitive cell cultures alive. Process development scientists know that even a tiny miscalculation in dissolved oxygen creates toxic lactate buildup, ruining the entire cell line. Securing reliable validation on upstream bioprocessing equipment guarantees maximum antibody yields before the product moves to filtration. Downstream processing sees plenty of sensor failures from high-pressure viral filters, but upstream workflows command the largest validation budgets because the biological material is at its most vulnerable state. In 2026, the upstream segment is poised to account for 68.0% of market share as a direct result of this extreme biological risk. Companies that underinvest in these early-stage validation protocols consistently face weaker product quality across different batches.
Media-Equilibration Checks: Pre-warmed media creates temporary sensor reading fluctuations. Process engineers use portable reader units to distinguish false signal noise from genuine environmental baseline shifts.
- Inoculation-Readiness Gates: Software systems refuse releasing bioreactors for cell transfer until all connected patches pass automated stability tests. Production supervisors avoid wasting high-value seed trains on unverified fluid paths.
- Perfusion-Loop Monitoring: Continuous cell culture requires weeks of uninterrupted measurement. Upstream managers face complete run failure if they neglect rigorous pre-use drift modeling on external circulation loop sensors.
Single-Use Bioprocess Sensor Calibration and Validation Test Systems Market Analysis by End User
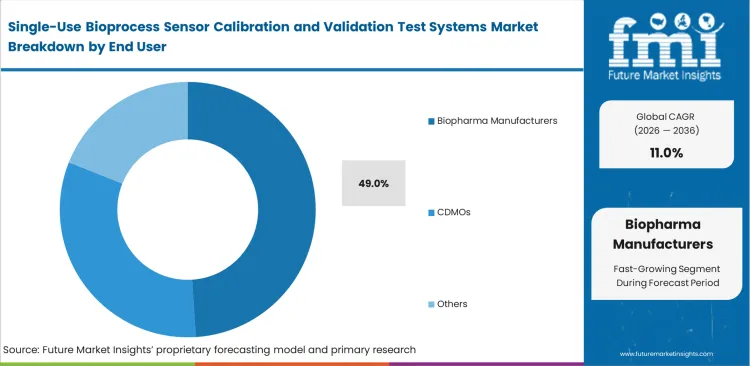
Building large new disposable manufacturing facilities requires identical testing equipment across different geographic locations. Internal manufacturing groups look for hardware that supports centralized data management and fleet-wide software updates. The Biopharma Manufacturers segment is forecast to command 49.0% share in 2026 as these global producers connect their validation tools directly into master execution systems. Contract manufacturers actually buy more validation rigs per bioprocessing systems suite because their rapid client changeovers demand faster testing speeds. Mid-sized drug developers that try to patch together custom calibration rigs from basic laboratory parts fail compliance audits when inspectors review their data security controls.
- Fleet-Standardization Rollout: Global therapeutic producers purchase matching calibration consoles for sites across three continents. Manufacturing heads ensure process development data from one site transfers perfectly to commercial scale elsewhere.
- Multi-Product Agility: Contract organizations switch between mammalian and microbial projects weekly. Suite managers depend on modular validation rigs to swap measurement parameters without completely rebuilding testing infrastructure.
- Compliance-Data Archiving: Large internal quality teams require ten-year retention of original calibration curves. Regulatory directors avoid warning letters by implementing validation systems with native encrypted cloud backup features.
Single-Use Bioprocess Sensor Calibration and Validation Test Systems Market Analysis by Deployment
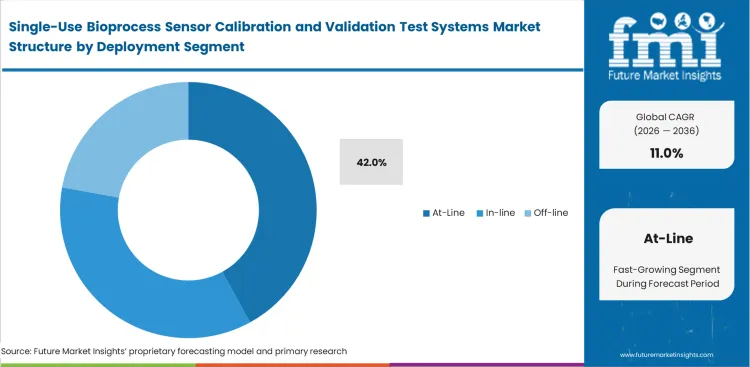
Facilities are expected to see at-line deployment account for 42.0% share in 2026 because physical proximity to the sterile boundary heavily reduces contamination risks. Moving a sensor-equipped bag outside a classified cleanroom for testing introduces unacceptable exposure hazards when bringing it back to the production floor. Operations managers place portable testing modules right next to the bioreactors to run quick transmitter verifications. In-line calibration sounds better in theory, but the tight physical space inside a disposable bag manifold makes bulkier at-line measurement technology in downstream processing setups much more practical for complex assemblies. Companies that insist on using separate, off-line calibration labs deal with broken optical connections when bags get bumped or dropped during transport.
- Sterile-Envelope Preservation: Testing equipment sits inches from designated production skids. Contamination-control specialists eliminate risks associated with moving bulky plastic assemblies through multiple airlock doors post-calibration.
- Rapid-Turnaround Verification: Floor operators hook up bags directly beside active processing lines. Shift supervisors reduce idle equipment time by validating next-batch sensors while current batches finish final transfer steps.
- Vibration-Shock Reduction: Transporting calibrated optical patches risks micro-fractures in sensitive fiber-optic connections. Facilities engineers prevent mechanical damage by bringing reader hardware to bioreactors rather than moving bags to remote labs.
Single-Use Bioprocess Sensor Calibration and Validation Test Systems Market Drivers, Restraints, and Opportunities
New rules from the European Medicines Agency (EMA Annex 1) require physical proof that a sterile boundary is secure before any manufacturing starts. Plant directors risk immediate shutdowns if they cannot show real-time calibration logs for every disposable probe attached to their fluid lines. The old method of handing an auditor a paper certificate from the plastic supplier simply fails today's inspections. Plant managers must install automated testing units across their production floors right now to meet these standards. Waiting to buy this equipment puts entire product lines at risk of costly compliance delays. As a multi-million dollar batch depends entirely on getting an accurate dissolved oxygen reading, buying on-site testing hardware has shifted from an optional upgrade to a basic cost of staying in business.
Strict equipment compatibility remains a significant barrier in this market. Sensors and testing units from different brands often operate on proprietary digital architectures, which limits interoperability on the same production floor. A facility using a Sartorius bioreactor bag usually needs a Sartorius-compatible testing unit for verification, and the same pattern applies to Cytiva and other major suppliers. That restricts buyer flexibility when sourcing routine consumables and validation equipment. In the absence of a common communication standard, facilities that want backup supply options often need duplicate testing hardware across platforms. Independent software layers may offer a partial workaround, yet deployment usually depends on extensive custom coding that remains difficult for many mid-sized drug manufacturers to justify.
- Universal Reader Integration: Building testing consoles that can read sensors from any brand changes the buying power dynamic. Supply chain heads gain serious negotiating leverage when they are no longer forced to buy their plastic bags from the exact same company that built their testing machine.
- Predictive Performance Tracking: Early-stage testing units catch tiny voltage drops before a sensor ever touches a live batch. Quality teams can throw away a faulty optical patch early in the process instead of losing a high-value cell culture two weeks later due to hidden sensor drift. This proactive approach saves massive amounts of expensive bioprocessing supplies from being wasted.
- High-Speed Flow Adaptation: Making testing units that work accurately under fast liquid flow opens up a new buying group in downstream processing. Plant managers need these specialized tools to get true pressure readings, keeping expensive viral filters safe from sudden bursting during rapid fluid transfers in closed system bioprocessing environments.
Regional Analysis
The transition toward fully disposable manufacturing suites drives distinct regional purchasing patterns, with emerging biopharma hubs building from scratch while established markets retrofit existing facilities. This divergence shapes how quickly different geographic zones adopt automated sensor validation systems. Based on regional analysis, the market is segmented into North America, Europe, Asia Pacific, and other regions across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 13.2% |
| India | 12.8% |
| United States | 10.4% |
| South Korea | 10.1% |
| Germany | 9.4% |
| United Kingdom | 9.1% |
| Japan | 8.7% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia Pacific Single-Use Bioprocess Sensor Calibration and Validation Test Systems Market Analysis
Heavy construction of commercial contract manufacturing hubs reshapes equipment buying across Asia Pacific. Local drug developers skip older stainless-steel setups and move directly into disposable architectures. This creates an immediate need for automated validation rigs that handle frequent product changes. Local equipment suppliers establish operations near major biopharma parks, bringing down baseline hardware costs and pushing Western suppliers to localize their software interfaces to remain competitive.
- China: Government-backed biologic parks require domestic sourcing for essential measurement infrastructure. Demand for single-use bioprocess sensor validation in China is anticipated to rise at a CAGR of 13.2% through 2036, as plant managers deploy large fleets of automated calibration rigs to standardize production across decentralized contract manufacturing networks. Domestic suppliers capture significant business by matching the validation speeds of Western continuous flow bioreactors.
- India: The country is set to record a CAGR of 12.8% in the sector during the assessment period due to biosimilar export targets forcing local facilities to match strict US FDA data integrity expectations. Quality directors install closed-loop simulator modules to prove zero human interference happens during pre-use sensor checks. Flawless audit trails help these facilities secure lucrative Western bioprocess containers and fluid transfer solutions supply contracts.
- South Korea: Massive investments in commercial-scale biomanufacturing push South Korean facilities to adopt high-velocity suite turnaround practices. Operations supervisors rely on at-line verification hardware to minimize idle time between large batch changeovers. South Korea is forecast to record steady growth in single-use sensor verification systems at a CAGR of 10.1% from 2026 to 2036 as fast qualification cycles remain a structural requirement for these sites to maintain global competitiveness.
- Japan: Strict domestic quality frameworks force Japanese therapeutic developers to implement double-redundant sensor verification steps. Validation engineers construct detailed offline testing procedures for every incoming disposable lot, pushing Japan as projected to witness 8.7% CAGR in the industry through 2036. Organizations achieve virtually zero in-process sensor failures but sacrifice suite turnaround speed to hit that quality metric.
FMI reports, Asia Pacific serves as the global proving ground for high-velocity disposable manufacturing. By skipping legacy stainless infrastructure entirely, local contract manufacturers rely heavily on automated pre-use verification, forcing Western suppliers to localize pricing and software to compete with emerging regional equipment hubs. The report also includes Southeast Asia and Australasia. Emerging biotechnology hubs in Singapore actively pilot universal reader platforms designed to break restrictive vendor lock-in dynamics.
North America Single-Use Bioprocess Sensor Calibration and Validation Test Systems Market Analysis
Advanced cell and gene therapy platforms in North America require a different approach to sterile boundary verification. Developers work with very small fluid volumes, where standard macro-sensor calibration can consume too much valuable product. Miniaturized testing modules are needed to validate micro-bioreactor arrays without creating large dead-volume losses. Heavy use of cell culture sampling devices also increases preference for reader units that offer fast stabilization times and built-in cloud compliance integration.
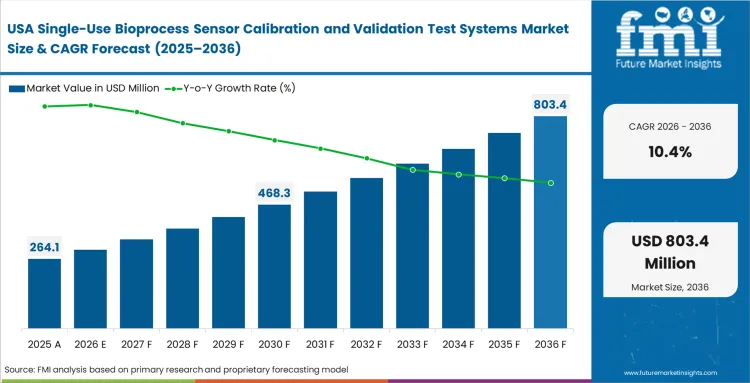
- United States: The United States sector is expected to register a CAGR of 10.4% through 2036, as cell therapy commercialization forces a shift from batch-level to patient-level validation architectures. Manufacturing directors place dedicated benchtop calibration modules inside individual isolation workstations to prevent cross-contamination risks. Facilities scaling autologous cell therapy manufacturing find that decentralized validation hardware, operating alongside United States upstream bioprocessing equipment and United States bioprocess containers and fluid transfer solutions, acts as a non-negotiable operational requirement.
FMI analyses, North American demand is defined by the shift toward decentralized cell and gene therapies. Handling micro-fluidic volumes requires exceptionally fast, miniaturized validation hardware that sits directly beside patient isolation workstations, embedding strict quality control directly into the most fragile production steps. Report includes Canada and Mexico. Canadian research hubs actively prototype next-generation calibration algorithms tailored for continuous bioprocessing environments.
Europe Single-Use Bioprocess Sensor Calibration and Validation Test Systems Market Analysis
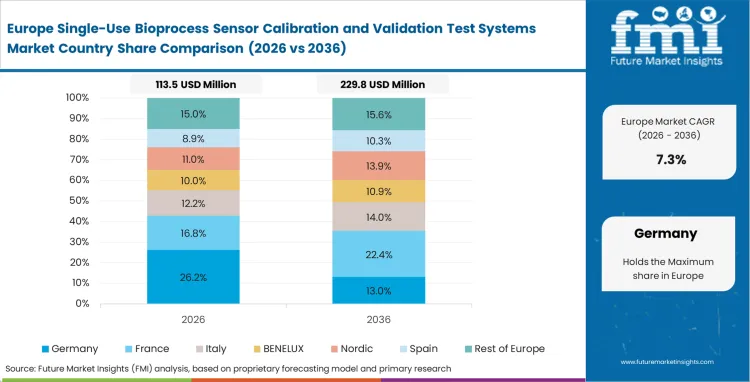
European Medicines Agency Annex 1 revisions fundamentally change how facilities verify sterile boundary integrity. Quality directors across the region aggressively decommission manual testing workflows in favor of fully automated, cryptographically secure validation rigs. Compliance officers mandate equipment that tracks specific gamma-irradiation lot numbers directly against individual sensor drift profiles. A strong regional preference for continuous processing models forces suppliers to engineer rigs that verify bioprocess technology sensors under constant flow conditions rather than static baseline states.
- Germany: Deep engineering expertise within domestic equipment manufacturers gives German facilities access to highly sophisticated multi-parameter simulator modules. Facility managers integrate these complex fusion bag testing units directly into central distributed control systems, leading Germany anticipated to see disposable sensor validation grow at a CAGR of 9.4% over the forecast period. Extensive digital integration allows pre-run qualification sequences to happen completely hands-free.
- United Kingdom: The rapid expansion of viral vector manufacturing capacity creates urgent demand for pressure-sensor validation tools in the United Kingdom. Downstream managers deploy specialized calibration hardware to protect fragile filtration setups from sudden rupture during high-velocity transfers through bioreactor single use protein a chromatography media. Resulting with the market adoption in the United Kingdom expected to grow at a CAGR of 9.1% during the study period. Specialized validation suites built adjacent to primary production floors are becoming the standard architectural layout across these sites.
FMI estimates, European facilities are rapidly automating their testing workflows to meet strict EMA Annex 1 data integrity mandates. By integrating cryptographically secure validation modules directly into central control networks, the region leads the global transition toward completely hands-free, continuous flow calibration. The report includes France, Italy, and Switzerland. Nordic bioclusters pioneer shared-access calibration data networks, allowing regional contract manufacturers to pool anonymized sensor failure statistics.
Competitive Aligners for Market Players
Equipment compatibility shapes how top suppliers maintain their market share. Sartorius and Thermo Fisher Scientific build testing units that only recognize their own disposable bags and sensors. Purchasing managers looking for less expensive alternative sensors quickly find that their current validation rigs block unrecognized connections. This setup effectively stops real price competition for daily manufacturing supplies. Smaller competitors like PreSens Precision Sensing approach this differently by offering open-architecture readers, including real-time bioprocess Raman analyzer platforms. These open systems appeal directly to mid-sized contract manufacturers that need more choices in their supply chain.
Historical performance data gives established companies a major advantage. Cytiva and Mettler-Toledo hold years of private records showing exactly how different sensors react after radiation exposure. New competitors can manufacture accurate testing machines, but they lack the long-term data needed to predict how sensor materials behave under stress. Plant managers operating portable bioprocessing bioreactors with continuous bioprocess fermentation monitoring stick to proven software to protect their production runs. Holding onto this specialized information lets the biggest suppliers keep their software prices high, even as the physical testing machines become easier to produce.
Major contract manufacturers are pushing back against restricted equipment by asking for universal industry standards. Supply chain leaders want to avoid buying multiple testing machines just to read different sensor brands. Companies like Broadley-James and Hamilton Company win these accounts by building equipment that connects easily with standard factory control networks. Independent drug developers now regularly choose their validation hardware based on whether it can read multiple brands of bioprocessing supplies. This buying pattern points to a gradual move away from facilities relying on just one equipment supplier.
Key Players in Single-Use Bioprocess Sensor Calibration and Validation Test Systems Market
- Sartorius
- Thermo Fisher Scientific
- Hamilton Company
- Cytiva
- PreSens Precision Sensing
- Mettler-Toledo
- Broadley-James
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 440 million to USD 1,250 million, at a CAGR of 11.00% |
| Market Definition | Equipment and software ecosystems designed to confirm measurement accuracy of disposable fluid-path sensors before committing active biological ingredients to sterile production sequences. |
| Segmentation | By Sensor Type, By Test Type, By Workflow, By End User, By Deployment, and By Region |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | United States, Canada, Brazil, Mexico, Germany, United Kingdom, France, Italy, Spain, China, Japan, South Korea, India, ASEAN, GCC, South Africa |
| Key Companies Profiled | Sartorius, Thermo Fisher Scientific, Hamilton Company, Cytiva, PreSens Precision Sensing, Mettler-Toledo, Broadley-James |
| Forecast Period | 2026 to 2036 |
| Approach | Annual replacement rates for benchtop reader modules across commercial-scale contract manufacturing sites. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Segments
Sensor Type
- pH Sensors
- DO Sensors
- Pressure Sensors
- Conductivity Sensors
- Flow Sensors
- Biomass Sensors
Test Type
- Calibration Systems
- Validation Rigs
- Simulator Modules
- Reader Units
- Software Suites
Workflow
- Upstream
- Downstream
- Media Prep
- Buffer Prep
- Fill-Finish
End User
- Biopharma Manufacturers
- CDMOs
- Equipment OEMs
- QC Labs
- Academic Centers
Deployment
- At-line
- In-line
- Off-line
- Portable
- Integrated
Regions:
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Bibliography
- Fratz-Berilla, E. J., Kohnhorst, C., Trunfio, N., Bush, X., Gyorgypal, A., & Agarabi, C. (2024). Evaluation of single-use optical and electrochemical pH sensors in upstream bioprocessing. Heliyon, 10(3), e25512.
- Ludolph, N., Haller, J., & Prediger, A. (2025). Measurement uncertainty analysis of single-use flow sensors. Frontiers in Bioengineering and Biotechnology, 13, 1455336.
- Cui, G., Wu, R., Cao, L., Abedin, S., Goel, K., Yoon, S., & Wang, X. (2025). Optical fiber pH and dissolved oxygen sensors for bioreactor monitoring: A review. Sensors, 26(1), 10.
- Isoko, K., Cordiner, J. L., Kis, Z., & Moghadam, P. Z. (2024). Bioprocessing 4.0: A pragmatic review and future perspectives. Digital Discovery, 3, 1662-1681.
- Wang, T., Cauchon, N. S., Kirwan, J. P., Joubert, M. K., Algorri, M., Bell, B., Soto, R. J., & Semin, D. J. (2025). Advancing the implementation of innovative analytical technologies in pharmaceutical manufacturing—Some regulatory considerations. Journal of Pharmaceutical Sciences, 114(2), 816-828.
- National Institute of Standards and Technology. (2024). Advancing cell-free manufacturing: Challenges in scale-up and automation workshop report (NIST SP 1500-26).
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Regulatory compliance shifts forcing biopharma manufacturers to adopt active pre-use verification protocols.
- Financial impacts of undetected sensor drift during high-titer continuous perfusion workflows.
- IT integration challenges surrounding proprietary hex-data streams from automated simulator modules.
- Contamination risks associated with transporting unwieldy disposable manifolds to off-line calibration laboratories.
- Vendor lock-in dynamics restricting procurement flexibility across global contract manufacturing sites.
- Rapid deployment of high-throughput validation rigs within emerging Asian biologic parks.
- Engineering constraints delaying truly continuous in-line flow sensor calibration.
- Decentralized validation strategies required for autologous cell therapy processing workstations.
Frequently Asked Questions
What are single-use bioprocess sensor calibration and validation test systems?
These systems comprise physical hardware, software suites, and reference standards engineered to verify accuracy, linearity, and zero-point stability of disposable measurement probes prior to or during sterile biomanufacturing runs, preventing costly biological material from entering unverified fluid paths.
Why do disposable bioprocess sensors need validation if they are factory calibrated?
Shipping conditions, temperature changes, and gamma-dose variations routinely induce baseline drift in sensitive optical patches post-sterilization, making pre-use verification directly on the shop floor a mandatory insurance policy against ruined batches.
Can single-use pH and DO sensors be recalibrated before use?
Operators conduct mathematical offset calculations using automated gas-mixing stations or dedicated benchtop consoles to correct optical dye degradation post-sterilization, providing a reliable baseline reading for mammalian cell culture environments.
What is the difference between verification, calibration, and validation in this market?
Verification confirms a sensor responds within acceptable ranges, calibration actively applies a mathematical offset to correct detected drift, and validation ensures the entire measurement ecosystem meets strict GMP data integrity requirements across a commercial manufacturing workflow.
Which companies supply test systems for single-use bioprocess sensors?
Major suppliers commanding this sector include Sartorius, Thermo Fisher Scientific, and Cytiva deploying proprietary closed digital ecosystems, while specialized challengers like PreSens Precision Sensing and Broadley-James offer alternative open-architecture reader platforms.
Why does upstream processing account for most demand?
Metabolic sensitivity peaks during primary fermentation phases, where process development scientists recognize even slight dissolved oxygen miscalibrations trigger toxic lactate accumulation, making accurate baseline readings critical for securing maximum antibody titer yields.
What factors drive adoption in China and India?
China sees massive deployment of automated calibration rigs to standardize production across greenfield contract manufacturing networks, while India requires stringent biopharma sensor calibration documentation to align biosimilar export volumes with strict US FDA expectations.
How do measurement uncertainty concerns affect buying decisions?
As minor hydrogen-ion shifts can devastate batch viability, procurement directors invest heavily in closed-loop simulator modules to prove zero human interference occurs during pre-use checks, effectively eliminating manual logbook entry deviations.
What documentation is needed for GMP qualification?
Regulatory affairs directors must produce active calibration logs, pre-use stability proofs, and linearity confirmation data for every disposable probe attached to fluid paths, which modern rigs automatically push directly to facility servers for 21 CFR Part 11 compliance.
How should CDMOs compare vendors in this category?
Contract organizations must evaluate vendor lock-in dynamics, prioritizing modular validation rigs and direct integration with agnostic distributed control systems to swap measurement parameters between vastly different biologic client projects without completely rebuilding testing infrastructure.
What value does Single-Use Bioprocess Sensor Calibration and Validation Test Systems Market reach by 2036?
Sales hit USD 1,250 million in 2036, reflecting industry-wide shifts toward fully disposable manufacturing suites requiring extensive pre-use verification infrastructure as facility managers refuse risking high-titer batches on uncalibrated equipment.
What was Single-Use Bioprocess Sensor Calibration and Validation Test Systems Market worth in 2025?
Total revenue stood at USD 400 million in 2025, driven by procurement focusing on standalone benchtop testing modules before European Medicines Agency regulatory updates made integrated validation systems an absolute operational requirement.
What compound annual growth rate dictates this expansion?
Expansion proceeds at 11.00% through 2036, driven by strict regulatory enforcement regarding sterile boundary integrity that forces manufacturing heads to purchase automated verification rigs to replace outdated paper-based certificate logging procedures entirely.
Why do pH sensors command dominant segment share?
As hydrogen-ion concentration directly impacts mammalian cell viability and gamma irradiation alters optical pH patch curves, bioreactor operators must conduct rigorous pre-inoculation mathematical offset adjustments to avoid destroying multi-million-dollar therapeutic batches.
What structural mechanism keeps Calibration Systems leading Test Type?
Validation engineers purchase automated rigs to eliminate manual buffer verification routines that waste hours of valuable cleanroom operator time, ensuring compliance officers achieve data integrity requirements reliably without paper-based transcription errors.
What operational reality pushes Biopharma Manufacturers to dominate end user share?
Massive greenfield facility expansions require standardized equipment fleets across multiple geographic sites, prompting manufacturing IT directors to mandate equipment capable of integrating natively with global manufacturing execution systems for perfect process development data transfer.
How does At-line deployment minimize operational risk?
Operations managers station portable testing modules immediately adjacent to bioreactor skids to protect sensitive fiber-optic connections from mechanical shock during transport, preventing the unacceptable contamination risks of taking sensor-equipped bags completely outside classified suites.
How do proprietary digital handshakes restrict procurement?
Strict vendor lock-in frustrates procurement directors attempting to build flexible multi-source supply chains because purchasing specific bioreactor bags requires matching proprietary testing modules, forcing facilities to purchase redundant calibration hardware if they dual-source consumable supplies.
Why do Downstream processing directors face unique pressure calibration challenges?
As viral clearance filters risk sudden rupture during transfers, engineers deploy specialized calibration hardware built specifically for high-velocity liquid environments to secure accurate pressure baseline readings that protect fragile downstream assemblies from unexpected failure.
How does Cell Therapy commercialization impact validation hardware design?
Autologous treatments handle incredibly small fluid volumes where standard macro-sensor calibration destroys too much product, forcing upstream managers to demand miniaturized testing modules capable of validating micro-bioreactor arrays without massive dead-volume losses.
What predictive capability do Quality managers expect from next-generation rigs?
Machine learning algorithms flag slight voltage irregularities during initial benchtop tests, allowing quality managers to preemptively scrap defective supplies before they cause severe late-stage perfusion culture failures and ruin active pharmaceutical ingredients.
How do massive internal quality teams handle long-term compliance archiving?
Regulatory directors implement validation systems with native encrypted cloud backup features, ensuring ten-year retention of original calibration curves so auditors can verify batch integrity long after physical disposable components enter incinerators.
Why do Process engineers use portable reader units during media equilibration?
Process engineers use portable units to distinguish false signal noise caused by pre-warmed media from genuine environmental baseline shifts, preventing control software from triggering unnecessary base additions during critical cell expansion phases.
What physical constraint limits true In-line continuous calibration adoption?
As physical space inside heavily populated disposable bag manifolds remains severely restricted, bulkier measurement rigs prove impractical for complex multi-sensor assemblies, keeping facilities heavily reliant on At-line configurations to preserve sterile envelopes without crowding skid footprints.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Sensor Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Sensor Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Sensor Type , 2026 to 2036
- pH Sensors
- DO Sensors
- Pressure Sensors
- pH Sensors
- Y to o to Y Growth Trend Analysis By Sensor Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Sensor Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Test Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Test Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Test Type, 2026 to 2036
- Calibration Systems
- Validation Rigs
- Others
- Calibration Systems
- Y to o to Y Growth Trend Analysis By Test Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Test Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Workflow
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Workflow, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Workflow, 2026 to 2036
- Upstream
- Downstream
- Media Prep
- Upstream
- Y to o to Y Growth Trend Analysis By Workflow, 2021 to 2025
- Absolute $ Opportunity Analysis By Workflow, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Biopharma Manufacturers
- CDMOs
- Others
- Biopharma Manufacturers
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Deployment
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Deployment, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Deployment, 2026 to 2036
- At-Line
- In-line
- Off-line
- At-Line
- Y to o to Y Growth Trend Analysis By Deployment, 2021 to 2025
- Absolute $ Opportunity Analysis By Deployment, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Sensor Type
- By Test Type
- By Workflow
- By End User
- By Deployment
- Competition Analysis
- Competition Deep Dive
- Sartorius
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Thermo Fisher Scientific
- Hamilton Company
- Cytiva
- PreSens Precision Sensing
- Sartorius
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Sensor Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Test Type, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Workflow, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Sensor Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Test Type, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Workflow, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Sensor Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Test Type, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Workflow, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Sensor Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Test Type, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Workflow, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Sensor Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Test Type, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Workflow, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Sensor Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Test Type, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Workflow, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Sensor Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Test Type, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Workflow, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Sensor Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Test Type, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Workflow, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Deployment, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Sensor Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Sensor Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Sensor Type
- Figure 6: Global Market Value Share and BPS Analysis by Test Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Test Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Test Type
- Figure 9: Global Market Value Share and BPS Analysis by Workflow, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Workflow, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Workflow
- Figure 12: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End User
- Figure 15: Global Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Deployment
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Sensor Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Sensor Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Sensor Type
- Figure 32: North America Market Value Share and BPS Analysis by Test Type, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Test Type, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Test Type
- Figure 35: North America Market Value Share and BPS Analysis by Workflow, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Workflow, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Workflow
- Figure 38: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by End User
- Figure 41: North America Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Deployment
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Sensor Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Sensor Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Sensor Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Test Type, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Test Type, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Test Type
- Figure 51: Latin America Market Value Share and BPS Analysis by Workflow, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Workflow, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Workflow
- Figure 54: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by End User
- Figure 57: Latin America Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Deployment
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Sensor Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Sensor Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Sensor Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Test Type, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Test Type, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Test Type
- Figure 67: Western Europe Market Value Share and BPS Analysis by Workflow, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Workflow, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Workflow
- Figure 70: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by End User
- Figure 73: Western Europe Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Deployment
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Sensor Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Sensor Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Sensor Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Test Type, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Test Type, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Test Type
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Workflow, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Workflow, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Workflow
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by End User
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Deployment
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Sensor Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Sensor Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Sensor Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Test Type, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Test Type, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Test Type
- Figure 99: East Asia Market Value Share and BPS Analysis by Workflow, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Workflow, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Workflow
- Figure 102: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by End User
- Figure 105: East Asia Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Deployment
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Sensor Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Sensor Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Sensor Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Test Type, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Test Type, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Test Type
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Workflow, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Workflow, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Workflow
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Deployment
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Sensor Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Sensor Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Sensor Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Test Type, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Test Type, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Test Type
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Workflow, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Workflow, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Workflow
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Deployment
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

