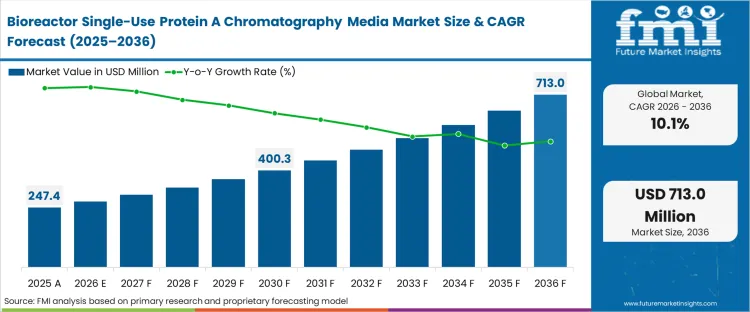
The bioreactor single-use protein A chromatography media market was valued at USD 247.4 million in 2025. The industry is expected to cross USD 272.4 million in 2026 at a protein A media CAGR of 10.10% during the forecast period. Continued investment drives the valuation upward to USD 713.0 million through 2036 as high-titer continuous upstream processes force downstream purification trains to abandon rigid stainless-steel cleaning validation cycles in favour of campaign-matched disposable protein A chromatography media.
Downstream process engineers are being forced to align their capture phase directly with upstream productivity. The decision is no longer about maximizing the lifetime cycles of a protein a resins bed, but about matching the media’s functional lifecycle to a specific clinical or commercial campaign. Delaying this transition leaves biomanufacturers with mismatched process tempos, where upstream bioreactors sit idle waiting for downstream changeover and cleaning-in-place validation. Integrating single-use antibody capture media market formats eliminates this bottleneck entirely, allowing facilities to increase their annual batch turnover without expanding their physical footprint.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 272.4 Million |
| Industry Value (2036) | USD 713.0 Million |
| CAGR (2026-2036) | 10.10% |
The critical threshold is the harmonization of titer capacities between the single-use bioreactors market limits and disposable column volumes. Once an operation matches the binding capacity of a pre-packed or flexible protein a resins share format to the exact yield of a 2,000 L run, the economic penalty of discarding the media vanishes. Contract manufacturing organizations typically trigger this change to guarantee multi-product facility turnaround times, turning a high-cost capital consumable into a predictable operating expense.
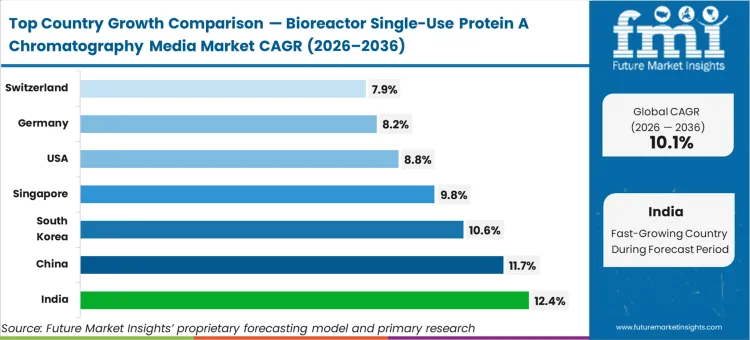
India is anticipated to expand at 12.4%, driven by aggressive clinical-stage CDMO capacity expansions prioritizing flexible processing over rigid infrastructure. China follows at 11.7% as regional biologics scale-up heavily favors protein A chromatography media for single-use bioreactors. South Korea is estimated to advance at 10.6% due to focused investments in modular manufacturing hubs. Singapore is poised to grow at 9.8%, reflecting its role as an agile biomanufacturing anchor in Asia. The United States is projected to record 8.8%, Germany 8.2%, and Switzerland 7.9%. The divergence between these Asian hubs and mature Western markets reflects a fundamental difference in installed base; newer facilities skip legacy stainless infrastructure entirely, embedding disposable capture protocols from day one.
To explain the bioreactor single-use protein A chromatography media market in simple terms, this market encompasses affinity chromatography materials specifically engineered, formatted, or utilized for limited-cycle or single-campaign capture of immunoglobulins. What is single-use protein A chromatography media operationally? It tracks media deployed explicitly to eliminate cleaning validation and cross-contamination risks between product changeovers, distinguishing it from traditional multi-cycle resin pools intended for years of continuous regeneration.
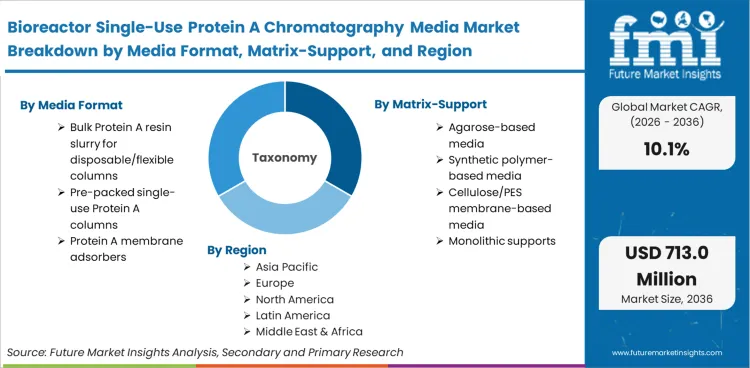
Included within scope are agarose, synthetic polymer, and membrane-based Protein A supports deployed as bulk slurries for flexible column packing, factory-sealed pre-packed columns, and membrane adsorbers designed for single-use or campaign-use. The scope incorporates media used across clinical and commercial scales, specifically within closed system bioprocessing environments where the downstream capture step is physically discarded after the defined batch or campaign is complete.
The market explicitly excludes reusable Protein A resins permanently installed in stainless steel column architectures intended for hundreds of cleaning and sanitisation cycles over multi-year lifespans. Also excluded are ion exchange, hydrophobic interaction, and mixed-mode resins, as well as the physical hardware of the chromatography skids themselves. These exclusions are maintained because the economics of permanent capital equipment and polishing resins do not share the campaign-specific, consumable-driven procurement dynamics that govern single-use affinity capture.
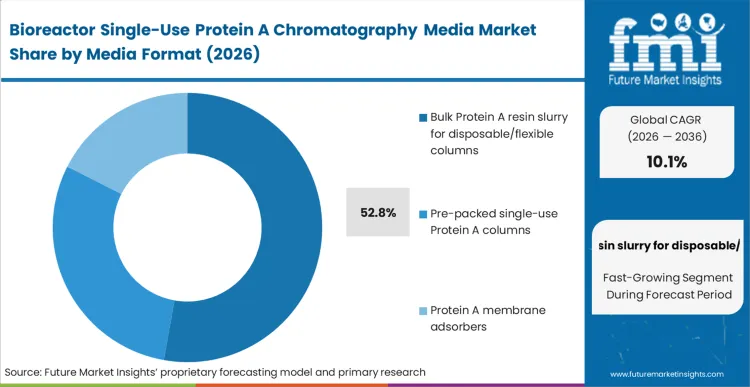
Pre-packed column architectures fundamentally alter how a downstream suite operates by moving the burden of bed integrity and qualification back to the vendor within the prepacked protein A columns market. While Bulk Protein A resin slurry for disposable/flexible columns holds a dominant 52.8% share, primarily due to the vast installed base of operators who still prefer to pack their own single-use hardware, pre-packed formats represent the actual trajectory of the market.
According to FMI's estimates, facilities evaluating prepacked protein A column vs bulk resin options at commercial scale are unwilling to tolerate the labor and failure risks of in-house packing. By procuring a closed, pre-validated capture step, facility managers isolate their operations from packing-induced channeling errors. CDMOs that delay the integration of pre-packed formats struggle to maintain competitive turnaround times, losing contracts to facilities that can guarantee faster product-to-product changeovers.
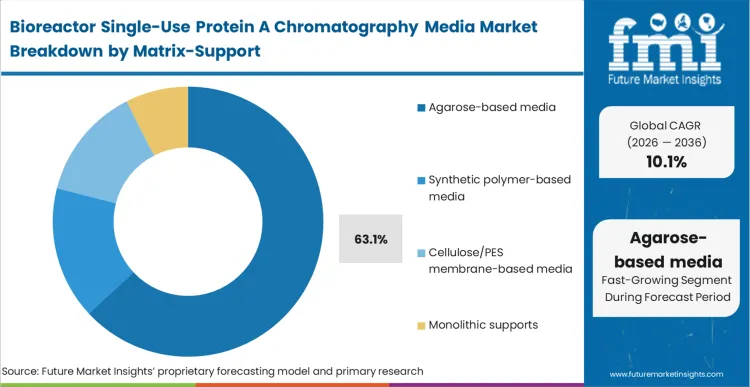
Agarose dominates this market not because it is the most advanced polymer, but because its regulatory pathway is virtually friction-free. The material captures a 63.1% share as regulatory affairs teams at major biopharmas continually default to agarose-based protein A resin for mAbs given its long history of successful BLA filings.
FMI analysts opine that the chemical stability and alkaline resistance of modern agarose allow it to survive aggressive sanitisation even within a limited-campaign lifecycle. Synthetic polymers are attempting to breach this stronghold by offering rigid architectures that do not compress at the high flow rates required by modern continuous processing. However, process development teams evaluating protein A membrane vs resin and synthetic alternatives must justify the qualification risk against the established baseline of agarose. Facilities that fail to evaluate synthetic alternatives risk hitting flow-rate bottlenecks as upstream titers intensify beyond the limits of soft gels.
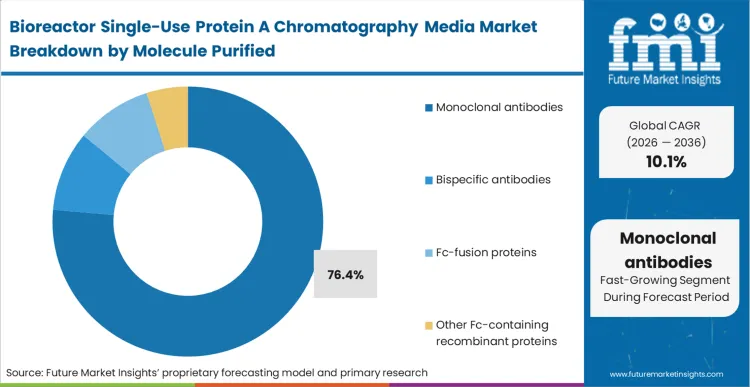
The absolute reliance of monoclonal antibody capture using single-use protein A media on the specific binding affinity of the staphylococcal ligand locks this segment into a 76.4% share of the market. This dynamic forces CDMOs to center their entire downstream platform architecture around this single purification step. As per FMI's projection, the requirement to remove host cell proteins and DNA in one highly efficient capture phase makes alternative separation techniques economically unviable for mAbs. When asking how does antibody pipeline growth affect protein A capture media demand, bispecifics and complex Fc-fusion proteins introduce severe binding complications, often requiring engineered or alkali-stabilized ligand variants to prevent product degradation during elution. Development teams attempting to push bispecifics through legacy mAb capture protocols face crippling aggregate formation and yield loss, directly impacting the commercial viability of their clinical pipelines.
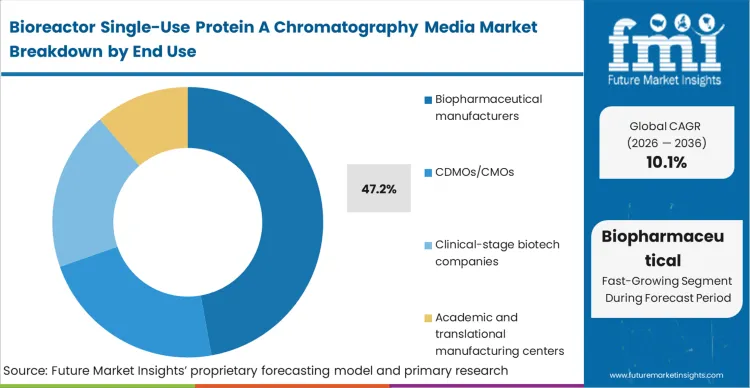
Contract manufacturing organizations (CDMOs/CMOs) are the fundamental engine driving the transition from stainless to disposable, even as biopharmaceutical manufacturing protein A media buyers hold the larger 47.2% share based on legacy volume. CDMOs sell agility; their business model collapses if a suite is offline for three weeks of cleaning validation between a client's Phase II breast cancer biologic and another client's autoimmune therapeutic.
Based on FMI's assessment, established biopharmaceutical manufacturers are slowly adopting this CDMO logic for their own multi-product regional hubs, realizing that capital-heavy, single-product stainless facilities are a liability in a fragmented blockbuster era. Manufacturing directors who cling to dedicated, multi-year resin lifespans in a multi-product environment face crippling overhead costs and uncompetitive suite utilization rates.

The physical geometry of modern single-use bioreactors dictates the downstream capture market, anchoring the 501-2,000 L single-use-linked campaigns segment to a 44.1% share. Because the limit of a disposable upstream bag is roughly 2,000 litres, the downstream capture column must be precisely sized to process this specific volume within a single operational change.
In FMI's view, scaling beyond this volume requires multiplexing bioreactors, which completely changes the cadence of the downstream suite. Facilities attempting to process multiple 2,000 L harvests through an undersized, multi-cycle column negate the time savings of single-use technology. Operations managers must perfectly match their pre-packed column volume to the upstream titre to ensure the entire harvest is captured continuously without product pooling or degradation.

The necessity to execute rapid product changeovers without cross-contamination risk forces CDMO facility directors to transition away from permanent stainless-steel capture columns toward flexible bioprocess chromatography media. As biologics pipelines fragment into targeted, smaller-volume therapeutics, the time spent on cleaning-in-place (CIP) and sterilization validation directly destroys the facility's profit margin. Multi-product suites can no longer afford the downtime associated with proving a reusable resin is free of a previous batch's active pharmaceutical ingredient. By discarding the capture media after a campaign, operators convert an unpredictable validation roadblock into a highly predictable consumable expense, driving the single-use downstream purification media market.
The severe supply chain consolidation among major chromatography resin manufacturers creates a bottleneck that threatens clinical timelines. Facility managers are painfully aware that transitioning to single-use, pre-packed columns locks them into proprietary hardware and specific single-use pump connections. When evaluating reusable protein A resin vs single-use protein A media, buyers realize that if a dominant supplier experiences a manufacturing delay or reprioritizes production for pandemic response, biomanufacturers lack the flexibility to simply pack a different resin into their existing hardware. While vendor-managed inventory programs and regional supply agreements attempt to buffer this risk, dual-sourcing pre-packed architectures remains technically difficult and regulatory intensive.
Regional analysis indicates that the Bioreactor Single-Use Protein A Chromatography Media Market is categorized into North America, Europe, Asia Pacific, and other regions covering over 40 countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 12.4% |
| China | 11.7% |
| South Korea | 10.6% |
| Singapore | 9.8% |
| United States | 8.8% |
| Germany | 8.2% |
| Switzerland | 7.9% |
The aggressive scale-up of biologics manufacturing across the Asia Pacific is entirely unencumbered by legacy stainless-steel infrastructure, allowing the region to adopt fully disposable architectures from inception. Rather than retrofitting old facilities, capital is flowing directly into modular, flexible plants designed specifically for high-turnover CDMO operations. FMI analysts opine that this advantage allows Asian manufacturers to execute tech transfers and initiate clinical runs in a fraction of the time required by mature Western sites. The procurement strategy here focuses on securing reliable, high-volume streams of protein purification resin to feed these newly minted suites.
FMI's report includes additional Southeast Asian and emerging Pacific markets not detailed above. Across these secondary hubs, the lack of deeply experienced downstream packing personnel structurally forces an reliance on pre-packed, plug-and-play chromatography formats to ensure process consistency.
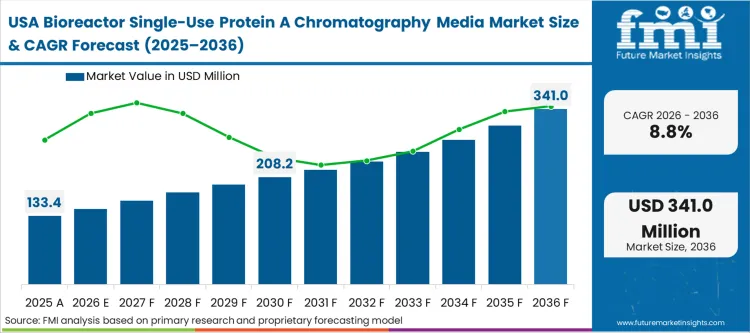
The North American market is defined by the tension between a massive, deeply entrenched stainless-steel installed base and the immediate need to process highly complex, low-volume advanced therapeutics. While mega-blockbuster mAbs still flow through permanent facilities, the sheer volume of novel bispecifics and fragmented clinical pipelines forces procurement teams to aggressively integrate single-use chromatography resin formats into new suite designs. As per FMI's projection, the regulatory comfort level with single-use technologies here is absolute, switching the buyer focus entirely toward supply chain resilience and multi-vendor qualification strategies.
FMI's report includes Canadian market dynamics. Canadian biomanufacturing investments are heavily skewed toward pandemic-readiness and sovereign production, structural priorities that mandate the rapid scalability only achievable through fully disposable process trains.
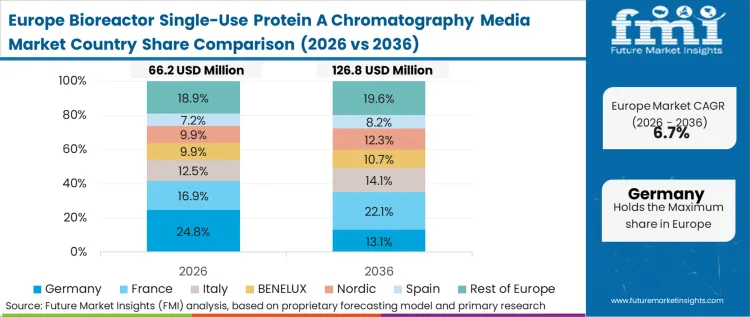
European biomanufacturing operates under the heaviest legacy infrastructure burden, yet it is leading the global industry toward continuous and intensified bioprocessing systems. The structural dynamic here involves retrofitting existing footprints with modular, single-use islands to handle the influx of complex Fc-fusion proteins that perform poorly on older legacy lines. According to FMI's estimates, European regulatory insistence on absolute viral clearance and cross-contamination prevention in multi-product facilities provides the final economic justification for abandoning reusable Protein A in flexible suites.
FMI's report includes the United Kingdom and broader EU member states. Across these markets, the regulatory phase-out of certain cleaning chemicals and the high cost of purified water generation structurally advantage facilities that eliminate the CIP requirement entirely through disposable capture.
The single-use protein A resin market is highly concentrated due to the extreme technical and regulatory barriers required to validate a primary capture ligand. Buyers evaluating Cytiva, Merck KGaA, and Repligen do not simply compare pricing; they evaluate the vendor’s absolute ability to guarantee supply continuity and the regulatory history of the specific ligand. The primary variable separating qualified protein A chromatography media suppliers from unqualified vendors is the existence of a comprehensive Drug Master File (DMF) and a proven track record of successful BLA inclusions.
Incumbents like Cytiva hold structural advantages through their deep integration into legacy BLA filings, creating a massive barrier to entry because switching a primary capture resin post-approval is economically disastrous for the buyer. Single-use protein A resin manufacturers challenging these incumbents must build not just equivalent binding capacities, but robust, localized packing infrastructure that can deliver preparative chromatography columns faster than the legacy players. Formulating a novel, high-performance synthetic matrix is irrelevant if the vendor cannot guarantee global, redundant supply of the pre-packed hardware.
Large CDMO buyers aggressively resist single-vendor lock-in by designing downstream suites capable of accepting hardware from multiple suppliers. When identifying which companies supply protein A media for disposable workflows, the structural tension between a CDMO’s need for generic, interchangeable preparative and process chromatography connections and the vendor’s incentive to enforce proprietary flow paths will define procurement strategies through 2036. The market will remain concentrated as the sheer capital required to maintain dual-continent resin manufacturing and packing facilities locks out smaller regional participants.
| Metric | Value |
|---|---|
| Quantitative Units | USD 272.4 Million to USD 713.0 Million, at a CAGR of 10.10% |
| Market Definition | This market tracks affinity capture media consumed in single-campaign or limited-cycle downstream bioprocessing. It is defined by an operational boundary: the deliberate discarding of high-cost Protein A materials after a specific batch to bypass the time and cost penalties of cleaning validation in multi-product facilities. |
| Media format Segmentation | Bulk Protein A resin slurry for disposable/flexible columns, Pre-packed single-use Protein A columns, Protein A membrane adsorbers |
| Matrix/support Segmentation | Agarose-based media, Synthetic polymer-based media, Cellulose/PES membrane-based media, Monolithic supports |
| Molecule purified Segmentation | Monoclonal antibodies, Bispecific antibodies, Fc-fusion proteins, Other Fc-containing recombinant proteins |
| End user Segmentation | Biopharmaceutical manufacturers, CDMOs/CMOs, Clinical-stage biotech companies, Academic and translational manufacturing centers |
| Regions Covered | North America, Europe, Asia Pacific, and other regions |
| Countries Covered | India, China, South Korea, Singapore, United States, Germany, Switzerland, and 40 plus countries |
| Key Companies Profiled | Cytiva (Danaher), Merck KGaA, Ecolab (Purolite), Repligen Corporation, Sartorius Stedim Biotech, Tosoh Bioscience, JSR Life Sciences |
| Forecast Period | 2026 to 2036 |
| Approach | Primary interviews were conducted with downstream process development scientists and bioprocess engineering directors. Baseline sizing was anchored to single-use bioreactor volume deployments and clinical pipeline milestones. Forecasts were cross-validated against regional CDMO capacity expansions and dominant vendor supply data. |
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
What is the forecast for single-use protein A media through 2036?
The market reaches USD 713.0 million by 2036. This expansion reflects the total architectural movement of new downstream facilities toward modular, campaign-specific purification trains capable of matching upstream single-use bioreactor limits.
Who are the leading suppliers of protein A capture media for single-use facilities?
The market is dominated by legacy vendors like Cytiva, Merck KGaA, and Sartorius, alongside specialty hardware providers like Repligen. These suppliers lead because they pair proven, BLA-validated agarose and synthetic resin chemistries with immense global column-packing infrastructure.
Compare protein A membrane adsorbers versus prepacked columns in single-use mAb purification?
Pre-packed columns offer identical binding capacities and scalability to traditional resin beds, making tech transfer straightforward. Membrane adsorbers bypass diffusion limits to achieve extreme flow rates, making them ideal for rapid cycling of very high-titer, low-volume clinical runs where cycle speed outweighs absolute binding capacity.
Why are prepacked protein A columns used with single-use bioreactors?
They structurally match the exact harvest volume of 2,000L upstream bags. Facility managers utilize them to bypass the massive labor and cross-contamination risks associated with packing and cleaning stainless-steel columns between batches.
How is protein A media used in single-use bioprocessing?
It is physically discarded after a predefined campaign rather than being subjected to weeks of chemical sanitisation. This transforms a high-cost capital asset into a consumable, drastically reducing the facility downtime between multi-product manufacturing runs.
What is single-use protein A chromatography media?
It refers to affinity capture formats such as pre-packed columns, flexible bulk slurries, or membrane adsorbers deployed specifically to eliminate cross-contamination and cleaning validation between biological product runs.
Which Media format segment leads?
Bulk Protein A resin slurry for disposable/flexible columns leads with 52.8% share. This format dominates because operations teams with deep in-house packing expertise prefer to maintain geometric control over their disposable column architectures while still avoiding permanent CIP validation.
Which Matrix/support segment leads?
Agarose-based media dominates with 63.1% share. Regulatory affairs groups default to this matrix because decades of successful BLA filings eliminate the intense scrutiny associated with qualifying novel synthetic polymers for primary capture.
Which Molecule purified segment leads?
Monoclonal antibodies account for 76.4% of the market. The sheer volumetric demand for these therapeutics, combined with their absolute reliance on the specific binding affinity of staphylococcal ligands, forces CDMOs to standardize their chromatography accessories and consumables around this purification step.
What drives rapid growth?
The severe economic penalty of idle suites drives the transition. Contract manufacturers cannot afford the two-to-three-week downtime required to validate equipment cleanliness between client campaigns, forcing the adoption of campaign-use disposable hardware.
What is the primary restraint?
The consolidation of global resin supply creates severe operational risk. Facility managers who adopt proprietary pre-packed columns lock themselves into specific bioprocessing supplies ecosystems, leaving them vulnerable to single-source manufacturing delays.
Which country grows fastest?
India leads at 12.4%, heavily outpacing the United States at 8.8%. India's massive influx of greenfield CDMO investments prioritizes flexible, multi-product suite designs that utilize disposable capture from day one, whereas the US market must carefully navigate retrofitting older, massive stainless-steel installations.
Why does China command an 11.7% growth rate?
Chinese biologics scale-up requires immediate processing capacity that bypasses lengthy hard-piping construction. The domestic market relies on single-use downstream architectures to match their rapid upstream bioreactor deployments.
How does South Korea utilize disposable Protein A formats?
South Korean mega-hubs utilize these formats as modular pilot lines. This allows them to quickly run complex clinical batches without risking contamination within their massive, permanent commercial stainless-steel infrastructure.
What structural barrier prevents synthetic polymers from overtaking agarose?
Synthetic matrices offer superior rigidity, but process development scientists are hesitant to absorb the massive qualification risk required to prove viral clearance and product safety profiles on a novel material when agarose already has guaranteed regulatory acceptance.
How do CDMOs handle bispecific antibody capture?
Bispecifics often require highly engineered, alkali-stable ligand variants to prevent aggregation during the low-pH elution phase, forcing procurement teams to source premium, specialized pre-packed formats rather than standard mAb resins.
What changes when a facility moves from 500 L to 2,000 L scale?
The 2,000 L scale represents the structural limit of standard single-use upstream bags. Downstream managers must procure pre-packed columns precisely sized to process this exact harvest volume continuously, establishing a hard geometric constraint on vendor supply.
Why do traditional biopharmaceutical manufacturers adopt CDMO strategies?
Established brand owners are facing fragmented clinical pipelines rather than single mega-blockbusters. They are adopting localized single-use capture models, including japan protein a resins, to increase the flexibility of their legacy multi-product manufacturing hubs.
How do pre-packed columns reduce validation burdens?
By offloading column packing and bed integrity testing entirely to the vendor, quality assurance teams can execute faster batch releases by relying on the supplier's provided performance certificates.
What role does Singapore play in the global supply chain?
Singapore acts as an agile, highly regulated biomanufacturing anchor. Because facility downtime in this premium-labor market is exceptionally costly, the high price of disposable pre-packed capture columns is economically justified by the increased suite utilization.
How do major players protect their market position?
Incumbents leverage deep integration into existing Drug Master Files (DMFs). Once a specific resin is written into a therapeutic's BLA, the massive regulatory cost of switching materials effectively locks out challenger products for the lifecycle of that drug.
Is protein A membrane chromatography replacing resin columns?
Membranes bypass the diffusion limits of standard porous beads, allowing process engineers to achieve extreme flow velocities. This is structurally necessary when processing very high-titer, low-volume clinical runs where cycle speed is the primary objective, though bulk resin currently maintains dominance by volume.
Why are downstream operations switching from volume to campaign-based procurement?
Clinical yields are highly variable. Instead of buying bulk resin intended to last for years, procurement directors now purchase pre-packed columns designed specifically to last only for the duration of a single Phase II campaign, hedging against clinical trial failures.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.