Single Use Pump Market
Single Use Pump Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Single Use Pump Market Forecast and Outlook 2026 to 2036
The single use pump market increases from USD 1.6 billion in 2026 to USD 5.1 billion by 2036, reflecting a CAGR of 12.20%. Adoption remains concentrated in bioprocessing and sterile fluid handling environments where cleaning validation and cross contamination risk carry high operational cost. Regulatory oversight and inspection practice favor disposable fluid paths, which shifts procurement toward fully documented, pre-qualified assemblies. Compliance cost and batch release requirements influence vendor positioning, since suppliers must support material traceability, extractables data, and change control. Facilities with frequent product changeovers adopt these systems faster than plants optimized for long continuous production runs.
Over the period, growth varies by region based on the pace of biologics capacity expansion and the structure of contract manufacturing networks. Markets with dense clusters of CDMOs and vaccine producers show faster uptake, while others remain more conservative due to waste handling rules and supply chain dependency concerns. Import regulations and local quality audits shape supplier access and product mix. Purchasing decisions are often centralized at corporate level rather than at site level. The market expands through process standardization and regulatory alignment, not through uniform replacement of reusable pumping systems.
Quick Stats of the Single Use Pump Market
- Single Use Pump Market Value (2026): USD 1.6 billion
- Single Use Pump Forecast Value (2036): USD 5.1 billion
- Single Use Pump Forecast CAGR 2026 to 2036: 12.2%
- Leading Product in Single Use Pump Market: Peristaltic Pumps (34 % share)
- Leading End-Use Industry in Single Use Pump Market: Healthcare and Medical (41 % share)
- Key Growth Regions in Single Use Pump Market: India, China, Brazil, USA, UK
- Top Players in Single Use Pump Market: Sartorius, Watson Marlow Fluid Technology Group, Graco Inc., Colfax (WEMCO), Verder Liquids, IDEX Corporation, ProMinent Fluid Controls
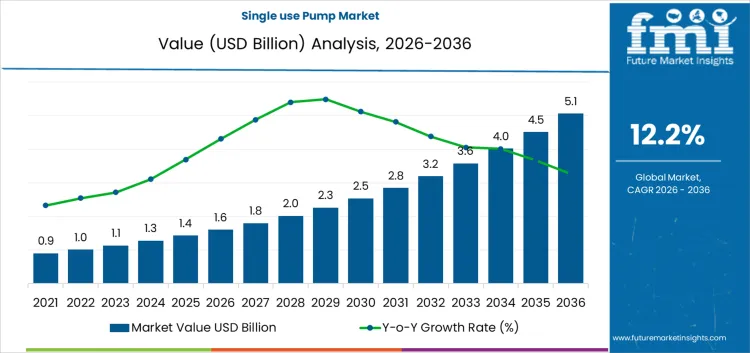
What is the Growth Forecast for Single Use Pump Market through 2036?
Between 2026 and 2031, the single use pump market is projected to expand from USD 1.6 billion along a trajectory consistent with a 12.2% CAGR, reflecting changes in how bioprocessing and sterile fluid handling are organized rather than simple capacity additions. During this phase, manufacturers continue to replace fixed stainless-steel transfer systems with disposable flow paths to reduce cleaning validation time, cross-contamination risk, and changeover downtime. Demand is driven by multiproduct facilities, clinical-scale production, and contract manufacturing environments where flexibility carries more value than asset utilization. Purchasing decisions are therefore made at the process design level, not at the component level. Pump selection emphasizes compatibility with single-use assemblies, predictable flow control, and reliability over long batch runs rather than peak performance specifications.
From 2031 to 2036, the market is expected to reach USD 5.1 billion, with growth shaped by the normalization of fully disposable upstream and downstream process chains. As biologics portfolios diversify and batch sizes fragment, facilities rely even more on modular, rapidly reconfigurable systems where pumps are treated as consumable process tools rather than capital equipment. This shifts spending toward recurring purchases tied to production cycles. Value creation comes from higher installation density and broader application coverage, not from price escalation. Supplier competitiveness depends on integration with tubing, connectors, and sensor ecosystems, as well as supply security and validation support, since production continuity becomes more critical than marginal efficiency gains.
Single Use Pump Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 1.6 billion |
| Forecast Value (2036) | USD 5.1 billion |
| Forecast CAGR 2026 to 2036 | 12.2% |
How Is the Single-Use Pump Market Supporting Sterile and Efficient Fluid Handling?
Single-use pumps are increasingly adopted to handle liquids in pharmaceutical, biotechnology, and food processing applications while minimizing contamination risks and cleaning requirements. Historically, fluid transfer relied on reusable pumps, which required extensive cleaning and sterilization between batches, increasing downtime and cross-contamination risk. Modern single-use pumps integrate disposable fluid paths, modular components, and sterile connectors to enable rapid setup, easy replacement, and compliance with cGMP standards. Biopharmaceutical manufacturers, contract development and manufacturing organizations (CDMOs), and food processors prioritize contamination control, process reliability, and regulatory compliance. Early adoption focused on high-value biologics and vaccine production, while current demand spans monoclonal antibody production, cell therapy, and sterile food processing, driven by scalability, regulatory pressure, and process efficiency. Flow rate consistency, material compatibility, and disposable component quality influence supplier selection.
Increasing demand for sterile manufacturing, accelerated production cycles, and regulatory compliance are shaping market growth. Compared with conventional reusable pumps, single-use systems emphasize contamination prevention, reduced cleaning validation, and operational flexibility. Cost structures depend on disposable component manufacturing, pump design, and quality assurance, concentrating margins among suppliers capable of delivering reliable, certified systems. Manufacturers adopt single-use pumps to reduce downtime, maintain sterility, and scale production efficiently. By 2036, single-use pumps are expected to become standard in biopharmaceutical and sterile production processes, supporting operational efficiency, contamination control, and regulatory compliance across high-value liquid handling applications.
What Factors Are Shaping the Demand for Single Use Pumps by Pump Architecture and End Use Sector in 2026?
The single use pump market in 2026 is segmented by product type and by end use industry. By pump architecture, demand is divided into peristaltic, diaphragm, syringe, and gear designs, each offering different flow control behavior, shear characteristics, and integration paths with disposable fluid paths. By end use, demand is organized around healthcare and medical, food and beverage, pharmaceuticals and biotechnology, and industrial or chemical processing, which differ in cleanliness standards, batch sizes, and validation burden. These segments reflect how operators manage contamination risk, changeover time, and documentation while selecting pumping solutions that must support frequent product switches without extensive cleaning or downtime.
Why Do Peristaltic Pumps Lead Adoption in Single Use Applications?
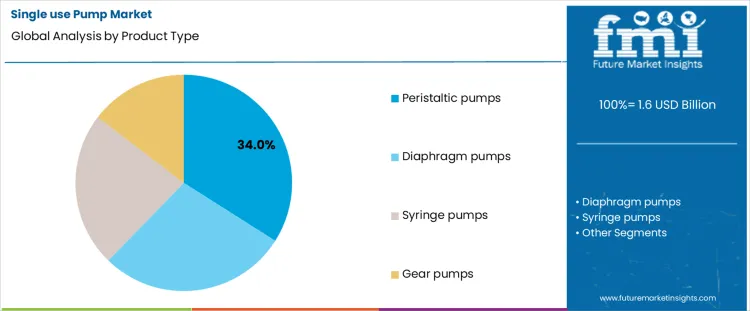
Peristaltic pumps account for about 34% of demand in 2026, reflecting their natural compatibility with disposable tubing sets. Fluid contacts only the tube, which eliminates internal wetted surfaces and simplifies validation between batches. This feature suits environments where rapid changeover and strict contamination control are required. The pumping action is gentle and predictable, which helps protect sensitive biological or formulated products from shear stress. Installation is straightforward because operators can replace tubing without disassembling the drive unit. Maintenance tasks are limited and do not interrupt production for cleaning. This combination of containment, operational simplicity, and process safety keeps peristaltic designs as the default choice in many single use process lines.
Diaphragm, syringe, and gear pumps address narrower operating windows. Diaphragm pumps handle higher pressures and certain dosing tasks, yet they introduce valves and chambers that complicate disposable integration. Syringe pumps provide precise metering in laboratory or filling steps, though their throughput is limited and their duty cycles are short. Gear pumps offer stable flow at higher pressures, yet their internal clearances and rotating parts make full disposability or low cost fluid paths harder to achieve. These designs are selected for specific process steps rather than as general purpose transfer tools. Their roles remain important, yet they do not match the broad applicability of peristaltic systems across single use production environments.
Why Does Healthcare and Medical Use Represent the Largest End Use Segment?
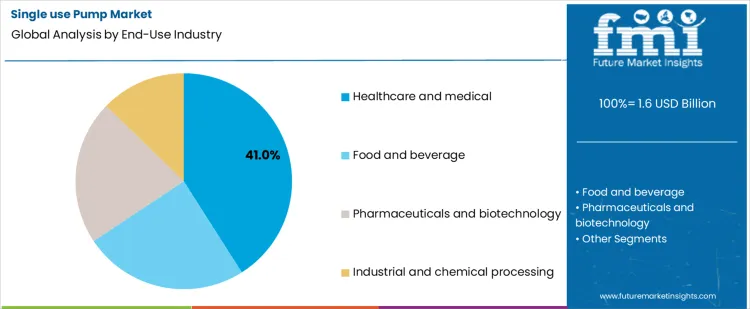
Healthcare and medical applications represent about 41% of demand in 2026 because clinical and hospital environments place high value on contamination avoidance and fast setup. Single use pumps are used in dialysis support, perfusion circuits, drug preparation, and various point of care fluid handling tasks where cleaning and revalidation are impractical. Patient safety protocols favor components that arrive sterile and are discarded after use. Procurement decisions in these settings also consider labor savings and reduced turnaround time between procedures. The steady flow of procedures and the diversity of fluids handled create consistent demand for disposable pumping solutions, which explains why healthcare and medical use anchors the largest share of the overall market.
Pharmaceutical, biotechnology, food, and industrial processing follow different adoption patterns. In bioprocessing, single use systems are common, yet pump selection is tied to specific unit operations and scale. Food and beverage uses disposables mainly for short runs, seasonal products, or allergen control, which limits volume. Industrial and chemical users adopt single use components only in specialized or hazardous handling scenarios. These sectors value disposability, yet their processes often tolerate cleaning in place or use fixed piping. As a result, their aggregate demand remains secondary to healthcare, where disposability is directly linked to patient safety and routine clinical workflows.
Why Are Workflow Simplification and Infection Control Policies Driving the Single Use Pump Market through 2036?
Demand is anchored in how hospitals manage risk and staff time. Single use pumps remove the need for cleaning, calibration, and maintenance, which reduces turnaround time between patients and limits cross contamination exposure. In high throughput areas such as oncology infusion, pain management, and ambulatory surgery, predictability and speed matter as much as unit cost. Many protocols now specify disposable delivery systems to standardize dosing and documentation. During staffing shortages, devices that reduce handling steps become more attractive. Once adopted into clinical pathways, these pumps are consumed per case, tying volume directly to procedure flow rather than to capital equipment replacement cycles.
What Unit Cost, Waste Burden, and Performance Limits Are Slowing Broader Substitution?
Constraints come from operating economics and technical boundaries. Disposable pumps carry higher per treatment cost than amortized reusable systems, which makes finance departments cautious in cost sensitive departments. Waste disposal and environmental compliance add indirect cost and administrative burden. Some therapies require variable flow rates, alarms, or integration with monitoring systems that simple mechanical pumps cannot provide. Clinicians also worry about flow accuracy over long infusions in certain use cases. As a result, hospitals selectively deploy single use pumps where simplicity is sufficient and keep reusable smart pumps for complex regimens, which limits the addressable share of total infusion volume.
How Are Care Setting Shifts and Packaging Strategies Changing the Role of These Devices?
The role of single use pumps is expanding beyond acute wards. Home infusion, day care oncology, and outpatient procedures favor devices that require minimal training and no return logistics. This pushes manufacturers to design pumps that are bundled with drug packaging or therapy kits. Some pharmaceutical partners view the pump as part of the product presentation rather than as separate equipment. Procurement is also shifting toward therapy based contracts instead of device line items. Over time, single use pumps become a delivery format embedded in treatment pathways, not just a substitute for traditional infusion hardware.
What is the Demand for Single-Use Pumps by Country?
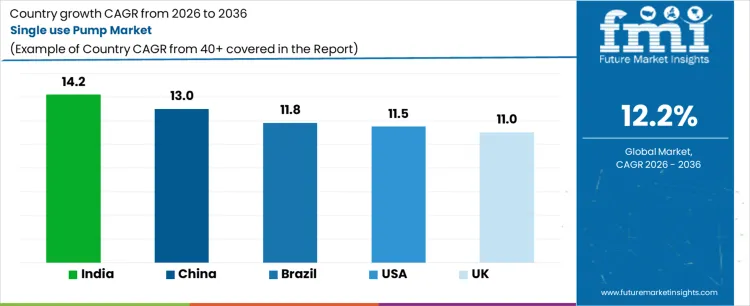
| Country | CAGR (%) |
|---|---|
| USA | 11.5% |
| UK | 11.0% |
| China | 13.0% |
| India | 14.2% |
| Brazil | 11.8% |
Demand for single-use pumps is rising as pharmaceutical, biotechnology, and healthcare manufacturers adopt disposable pumping systems to reduce contamination risk, simplify cleaning processes, and improve operational efficiency. India leads with a 14.2% CAGR, driven by rapid expansion of biopharmaceutical production, increasing adoption of disposable technologies, and rising demand for sterile manufacturing processes. China follows at 13.0%, supported by growing pharmaceutical manufacturing, biologics production, and adoption of single-use systems. Brazil records 11.8% growth, shaped by healthcare infrastructure expansion and process optimization initiatives. The USA grows at 11.5%, influenced by advanced biopharma manufacturing and disposable technology adoption. The UK shows 11.0% CAGR, reflecting steady integration of single-use pump systems in pharmaceutical and research applications.
How is the United States experiencing growth in the single use pump market?
United States is experiencing growth at a CAGR of 11.5%, driven by rising demand in biopharmaceutical, medical, and laboratory applications where sterility and contamination control are critical. Single use pumps are optimized for fluid transfer, chemical compatibility, and ease of integration with disposable bioprocess systems. Demand is concentrated in biotech clusters such as Boston, San Francisco, and San Diego, where contract manufacturing organizations and research laboratories are expanding. Investments focus on device reliability, chemical resistance, and compliance with FDA and USP standards. Growth reflects increasing biopharmaceutical production, adoption of disposable manufacturing technologies, and focus on operational efficiency in sterile processes.
- Biopharmaceutical production drives adoption.
- Pumps optimize sterility and chemical compatibility.
- Biotech clusters concentrate demand.
- Device reliability and regulatory compliance guide investment.
How is the United Kingdom witnessing growth in the single use pump market?
United Kingdom is witnessing growth at a CAGR of 11%, supported by expanding biopharmaceutical research, vaccine production, and sterile fluid handling in London, Cambridge, and Oxford. Single use pumps are optimized for ease of use, contamination prevention, and integration with disposable bioprocess systems. Demand is concentrated in contract manufacturing organizations, research laboratories, and pharmaceutical production centers. Investments prioritize chemical resistance, device reliability, and compliance with MHRA and European pharmaceutical standards. Growth reflects increasing adoption of disposable manufacturing processes, focus on sterile operations, and expansion of biotech and vaccine production facilities.
- Biopharmaceutical research drives adoption.
- Pumps enhance contamination control and integration.
- Biotech hubs concentrate demand.
- Chemical resistance and compliance guide investment.
How is China experiencing growth in the single use pump market?
China is experiencing growth at a CAGR of 13%, fueled by rapid expansion of vaccine manufacturing, biologics production, and biopharmaceutical R&D in cities such as Shanghai, Beijing, and Suzhou. Single use pumps are optimized for sterile fluid transfer, chemical compatibility, and high reliability in disposable production systems. Demand is concentrated in industrial biotech parks, pharmaceutical companies, and research facilities. Investments focus on system reliability, regulatory compliance, and material performance. Growth reflects government initiatives to boost domestic biologics production, adoption of disposable bioprocessing technologies, and increasing global export of vaccines and therapeutics.
- Vaccine and biologics production drives adoption.
- Pumps optimize sterile fluid transfer and reliability.
- Industrial biotech parks concentrate demand.
- Regulatory compliance and material performance guide investment.
How is India witnessing growth in the single use pump market?
India is witnessing growth at a CAGR of 14.2%, supported by the expansion of contract manufacturing, vaccine production, and biologics development in Hyderabad, Bengaluru, and Pune. Single use pumps are optimized for contamination-free fluid handling, chemical resistance, and integration with disposable bioprocess systems. Demand is concentrated in biopharmaceutical manufacturing hubs, R&D centers, and vaccine production facilities. Investments prioritize device reliability, ease of use, and compliance with national and international pharmaceutical regulations. Growth reflects rising domestic vaccine production, adoption of single use technologies, and modernization of biopharma facilities.
- Vaccine production and biologics manufacturing drive adoption.
- Pumps enhance sterility and chemical resistance.
- Biopharma hubs concentrate demand.
- Device reliability and regulatory compliance guide investment.
How is Brazil experiencing growth in the single use pump market?
Brazil is experiencing growth at a CAGR of 11.8%, fueled by expansion in biopharmaceutical manufacturing, vaccine production, and laboratory research in São Paulo, Rio de Janeiro, and Minas Gerais. Single use pumps are optimized for sterile fluid handling, chemical compatibility, and operational reliability in disposable systems. Demand is concentrated in industrial biotech facilities, research centers, and vaccine production units. Investments focus on device durability, ease of integration, and compliance with ANVISA and pharmaceutical standards. Growth reflects increasing domestic biologics manufacturing, adoption of disposable processing technologies, and modernization of sterile production infrastructure.
- Biopharmaceutical manufacturing drives adoption.
- Pumps optimize sterility and chemical compatibility.
- Industrial biotech and vaccine hubs concentrate demand.
- Device durability and regulatory compliance guide investment.
Who Competes in the Single Use Pump Market and What Defines Their Capabilities?
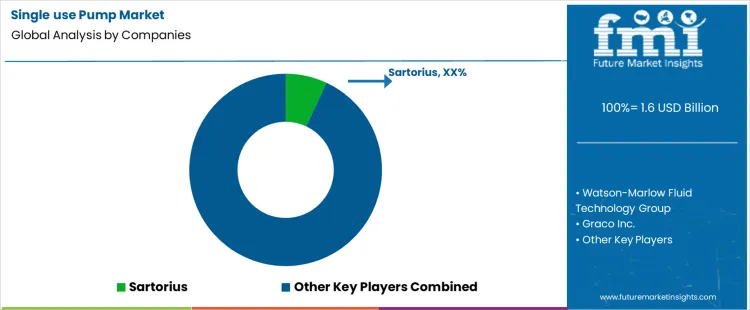
Competition in the single use pump market is shaped by flow reliability, sterile design, and compatibility with bioprocessing and fluid handling applications. Sartorius supplies single use pumps engineered for controlled fluid transfer in biopharmaceutical production, with emphasis on integrated sterile pathways and modular system connectivity. Watson Marlow Fluid Technology Group provides peristaltic single use pumps known for gentle fluid handling and straightforward tube replacement without product contact. Graco Inc. delivers diaphragm based and peristaltic options suited to aseptic manufacturing environments, with focus on cleanability and serviceability. Colfax’s WEMCO division offers robust pumping solutions adapted for high viscosity and abrasive media in disposable configurations. Verder Liquids supplies compact single use pumps tailored for laboratory and process scale operations requiring reliable, low contamination fluid transfer.
IDEX Corporation provides precision engineered single use pump modules with diverse head materials and sealing options to meet specific process requirements. ProMinent Fluid Controls offers diaphragm and metering single use pumps designed for accurate dosing in water treatment and process utilities, where sterility and repeatability are priorities. Other suppliers and regional manufacturers contribute disposable pump heads and assemblies that integrate with broader fluid systems. Market differentiation arises from ease of installation, sterile barrier performance, pump head materials, flow accuracy, and adaptability to automation and sensor feedback. Adoption reflects requirements in bioprocessing, pharmaceutical dosing, laboratory workflows, and any application where contamination avoidance and single use convenience reduce cleaning validation burden. Competitive positioning depends on product reliability, service support, and ability to document compatibility with industry standards for process safety and quality control.
Key Players in the Single Use Pump Market
- BD (Becton Dickinson)
- Cook Medical
- Teleflex (AngioDynamics)
- Boston Scientific
- Smith’s Medical
- Merit Medical Systems
- Medtronic
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD billion |
| Product Type | Peristaltic Pumps, Diaphragm Pumps, Syringe Pumps, Gear Pumps |
| End-Use Industry | Healthcare and Medical, Food and Beverage, Pharmaceuticals and Biotechnology, Industrial and Chemical Processing |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, United States, Canada, Mexico, Brazil, Chile, Saudi Arabia, Turkey, South Africa, and other regional markets |
| Key Companies Profiled | Sartorius, Watson Marlow Fluid Technology Group, Graco Inc., Colfax (WEMCO), Verder Liquids, IDEX Corporation, ProMinent Fluid Controls, and other regional manufacturers |
| Additional Attributes | Dollar sales by pump type and end-use industry, peristaltic pumps forming the largest product segment due to natural compatibility with disposable tubing sets, healthcare and medical representing the largest end-use segment because of contamination control and rapid setup requirements, market growth driven by the shift from reusable stainless steel systems to fully disposable fluid paths, strong influence of regulatory compliance, cleaning validation avoidance, and changeover time reduction on procurement decisions, adoption concentrated in biopharmaceutical manufacturing, CDMOs, clinical production, and sterile processing environments, purchasing centralized at corporate or process design level, demand tied to batch size fragmentation and multiproduct facilities, and competitive positioning based on system integration with tubing, connectors, sensors, documentation support, and supply security rather than peak performance specifications. |
Single Use Pump Market Segmentation
Product Type:
- Peristaltic pumps
- Diaphragm pumps
- Syringe pumps
- Gear pumps
End-Use Industry:
- Healthcare and medical
- Food and beverage
- Pharmaceuticals and biotechnology
- Industrial and chemical processing
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- World Health Organization. (2024). Medical devices: Key facts and regulatory overview.
- World Health Organization. (2023). WHO global model regulatory framework for medical devices including IVDs.
- USA Food and Drug Administration. (2024). Current good manufacturing practice (CGMP) for finished pharmaceuticals.
- USA Food and Drug Administration. (2023). Process validation: General principles and practices.
- National Institute of Biomedical Imaging and Bioengineering. (2025). Biomanufacturing and bioprocessing technologies.
Frequently Asked Questions
How big is the single use pump market in 2026?
The global single use pump market is estimated to be valued at USD 1.6 billion in 2026.
What will be the size of single use pump market in 2036?
The market size for the single use pump market is projected to reach USD 5.1 billion by 2036.
How much will be the single use pump market growth between 2026 and 2036?
The single use pump market is expected to grow at a 12.2% CAGR between 2026 and 2036.
What are the key product types in the single use pump market?
The key product types in single use pump market are peristaltic pumps, diaphragm pumps, syringe pumps and gear pumps.
Which end-use industry segment to contribute significant share in the single use pump market in 2026?
In terms of end-use industry, healthcare and medical segment to command 41.0% share in the single use pump market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type, 2026 to 2036
- Peristaltic pumps
- Diaphragm pumps
- Syringe pumps
- Gear pumps
- Peristaltic pumps
- Y to o to Y Growth Trend Analysis By Product Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End-Use Industry
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End-Use Industry, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End-Use Industry, 2026 to 2036
- Healthcare and medical
- Food and beverage
- Pharmaceuticals and biotechnology
- Industrial and chemical processing
- Healthcare and medical
- Y to o to Y Growth Trend Analysis By End-Use Industry, 2021 to 2025
- Absolute $ Opportunity Analysis By End-Use Industry, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By End-Use Industry
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End-Use Industry
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By End-Use Industry
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End-Use Industry
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By End-Use Industry
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End-Use Industry
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By End-Use Industry
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End-Use Industry
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By End-Use Industry
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End-Use Industry
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By End-Use Industry
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End-Use Industry
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By End-Use Industry
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End-Use Industry
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End-Use Industry
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End-Use Industry
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End-Use Industry
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End-Use Industry
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End-Use Industry
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End-Use Industry
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End-Use Industry
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End-Use Industry
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End-Use Industry
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End-Use Industry
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End-Use Industry
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End-Use Industry
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End-Use Industry
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End-Use Industry
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End-Use Industry
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End-Use Industry
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End-Use Industry
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End-Use Industry
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End-Use Industry
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End-Use Industry
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End-Use Industry
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End-Use Industry
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By End-Use Industry
- Competition Analysis
- Competition Deep Dive
- Sartorius
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Watson‑Marlow Fluid Technology Group
- Graco Inc.
- Colfax (WEMCO)
- Verder Liquids
- IDEX Corporation
- ProMinent Fluid Controls
- Sartorius
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End-Use Industry, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by End-Use Industry, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by End-Use Industry, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by End-Use Industry, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by End-Use Industry, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by End-Use Industry, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by End-Use Industry, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by End-Use Industry, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by End-Use Industry, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End-Use Industry, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by End-Use Industry
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Product Type
- Figure 23: North America Market Value Share and BPS Analysis by End-Use Industry, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by End-Use Industry, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by End-Use Industry
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Product Type
- Figure 30: Latin America Market Value Share and BPS Analysis by End-Use Industry, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by End-Use Industry, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by End-Use Industry
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by End-Use Industry, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by End-Use Industry, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by End-Use Industry
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by End-Use Industry, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by End-Use Industry, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by End-Use Industry
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Product Type
- Figure 51: East Asia Market Value Share and BPS Analysis by End-Use Industry, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by End-Use Industry, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by End-Use Industry
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by End-Use Industry, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by End-Use Industry, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by End-Use Industry
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by End-Use Industry, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by End-Use Industry, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by End-Use Industry
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

