The single-use bioreactor bag integrity and leak test equipment market is projected to grow from USD 62 million in 2026 to USD 162.3 million by 2036 at a 10.1% CAGR. The market stood at USD 56.3 million in 2025, and the forecast implies an incremental opportunity of USD 100.3 million over the assessment period. Demand is growing as larger disposable bag formats become more common in biologics production, pushing suppliers and manufacturers towards repeatable leak-testing methods that support stronger release records and reduce uncertainty before use.
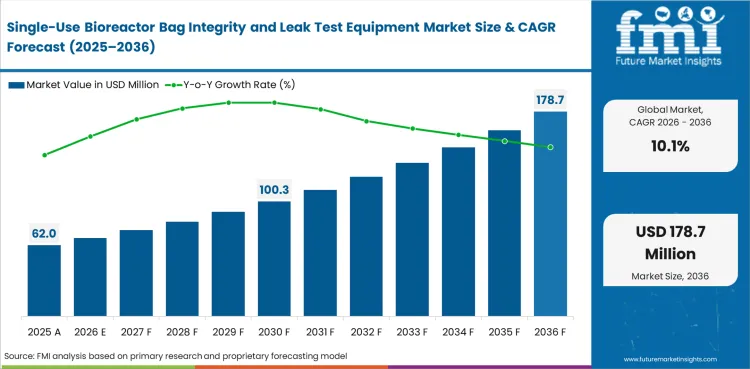
| Parameter | Details |
|---|---|
| Market value (2026) | USD 62 million |
| Forecast value (2036) | USD 162.3 million |
| CAGR (2026 to 2036) | 10.1% |
| Estimated market value (2025) | USD 56.3 million |
| Incremental opportunity | USD 100.3 million |
| Leading technology | Pressure decay testing (36% of technology segment) |
| Leading application | Supplier QA (34% of application segment) |
| Leading end use | Bag makers (37% of end-user segment) |
| Key players | Sartorius AG, ZELTWANGER Leaktesting & Automation GmbH, Cincinnati Test Systems, Pfeiffer Vacuum+Fab Solutions, Bonfiglioli Engineering |
Source: Future Market Insights, 2026.
Visual inspection is becoming less dependable in this market. Larger flexible bags, higher batch value, and tighter quality review are increasing demand for test systems that deliver stable readings and usable records. Adoption improves when bag suppliers and biologics manufacturers define clear leakage limits by bag type, because testing fits more naturally into release, incoming inspection, and validation workflows.
Across tracked countries, India is expected to grow at 11.6% CAGR through 2036, followed by China at 10.8%, Singapore at 10.4%, Ireland at 9.8%, United States at 9.4%, Germany at 8.7%, and Switzerland at 8.5%. Faster growth in India and China reflects new biologics capacity and rising single-use installation activity, while Singapore and Ireland remain strong because dense biomanufacturing clusters place greater operational value on bag reliability and documented integrity checks.
The single-use bioreactor bag integrity and leak test equipment market includes systems that detect leaks, verify seal quality, and confirm whether disposable bioreactor bags are suitable for sterile bioprocess use. Scope is restricted to test equipment, related fixtures, software, and associated testing functions rather than the bag assemblies themselves. This keeps the category separate from the broader single-use bioreactors market.
Scope includes pressure decay, helium-based, vacuum decay, tracer gas, force decay, and related integrity test systems used for single-use bioreactor bags, mixing bags, liners, and other flexible assemblies in upstream sterile processing. Bench units, semi-automatic stations, automated cells, fixtures, software, and validation-related accessories are included when they serve this application. Coverage also extends to equipment used for supplier release, incoming inspection, point-of-use verification, and related checks linked to bioprocess containers and fluid transfer solutions.
Revenue from single-use bags, single-use bioreactors, fluid-path consumables, and general sterile-transfer assemblies is excluded unless it is tied directly to test hardware sales. Generic package inspection systems for vials, syringes, blister packs, and broad pharma CCIT platforms are excluded when they are not designed for flexible bioprocess bags.
Demand is rising because disposable process assemblies are becoming more important in upstream biologics manufacturing, especially in mixing, holding, and reactor-linked applications. As larger 3D bags move deeper into commercial workflows, a leak creates more than a minor quality issue. It can interrupt batch preparation, delay line clearance, create deviation work, and complicate release review. This increases the value of equipment that deliver repeatable results under routine operating conditions, particularly in facilities expanding their use of upstream bioprocessing equipment.
Supplier-side testing is adding further support to market growth. Bag manufacturers need dependable defect screening before assemblies reach customer facilities, since weak release controls can create post-shipment disputes and increase incoming inspection burden. This is increasing adoption of pressure decay and other methods that fit practical release work without excessive setup difficulty. Many facilities are also moving from manual routines toward semi-automatic systems because they improve workflow control, reduce operator dependence, and generate records that are easier to review within broader bioprocess validation frameworks.
The single-use bioreactor bag integrity and leak test equipment market is segmented by Test Method, Deployment, Bag Format, Capacity Range, Automation, End User, and Region. By Test Method, the market is divided into Pressure Decay, Helium Vacuum, Vacuum Decay, Tracer Gas, and Force Decay. In terms of Deployment, the market is classified into Supplier QA, Point-of-Use, At-Line, In-Line, and Lab Testing. Based on Bag Format, the market is segmented into 3D Bags, 2D Bags, Bioreactor Liners, Mixing Bags, and Storage Bags. By Capacity Range, the market is divided into Up to 50 L, 51-200 L, 201-1000 L, 1001-2000 L, and Above 2000 L. In terms of Automation, the market is classified into Manual, Semi-Automatic, Automatic, and Robotic systems. Based on End User, the market is segmented into Bag Makers, Biopharma, CDMOs, and QC Labs.
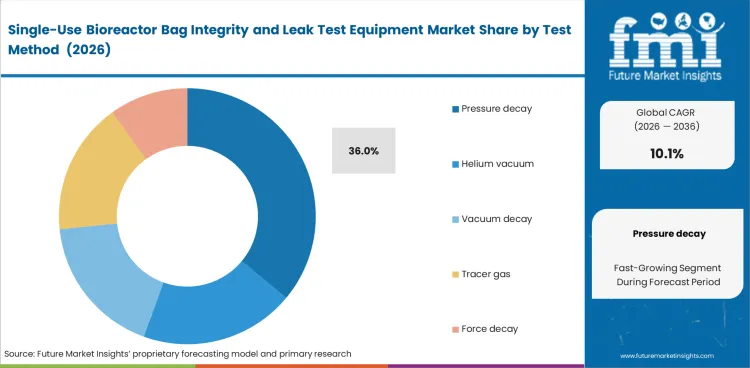
Pressure decay leads this segment because it offers a practical balance between repeatability, cycle time, and setup burden. In 2026, this segment is projected to account for 36% share. Helium-based and vacuum-based methods remain relevant in more demanding applications, though many bag manufacturers and biologics facilities prefer methods that integrate more easily into routine testing. In flexible films, stable and reviewable output often matters more than technical claims that are harder to apply consistently in day-to-day release work.
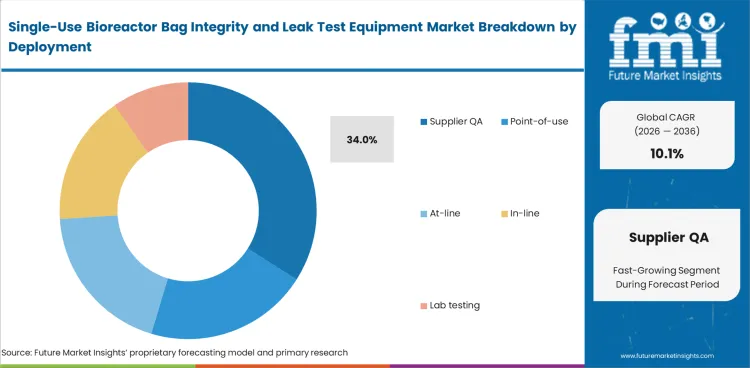
Supplier QA remains the leading deployment segment because defect checks begin before a bag is sterilized, packed, and shipped to the biologics site. The segment is expected to account for 34% share in 2026. Bag manufacturers rely on dedicated testing stations to identify manufacturing defects early, reduce downstream disputes, and support stronger release documentation. Point-of-use testing remains relevant in higher-risk workflows, but strong supplier-side assurance reduces the need for repeat checks later in the chain.
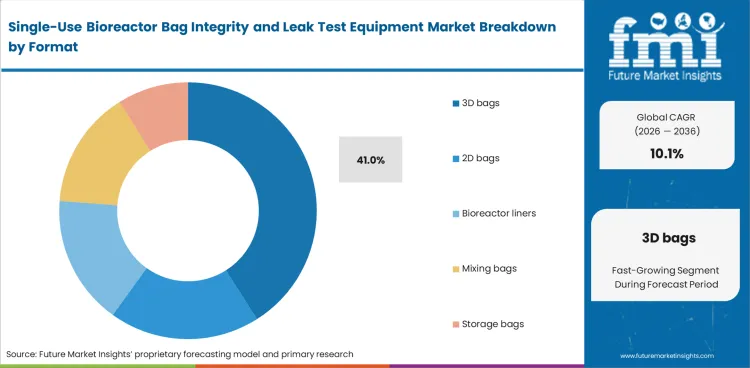
Bag geometry affects equipment performance directly, which keeps format as a central selection factor in this market. Larger flexible assemblies do not behave like flat pouches, and 3D bags create added testing difficulty because chamber fit, trapped volume, film movement, and handling variation can all influence the reading. 3D bags are projected to account for 41% share in 2026. Their wide use in mixing, holding, and reactor-linked applications keeps testing demand concentrated around this format.
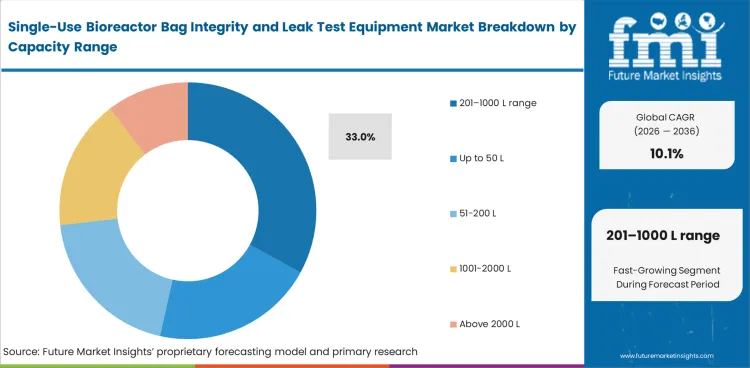
The 201-1000 L range sits near the center of many biologics operations, which keeps it important in this market. Users in this range balance higher bag value with manageable throughput and fixture complexity, which supports a projected 33% share in 2026. Smaller formats are easier to handle, though they do not always justify the same level of dedicated testing. Very large systems often require more specialized fixtures and method adjustments. This operating balance is similar to adoption patterns seen in portable bioprocessing bioreactors, where flexibility and practical scale fit remain important.
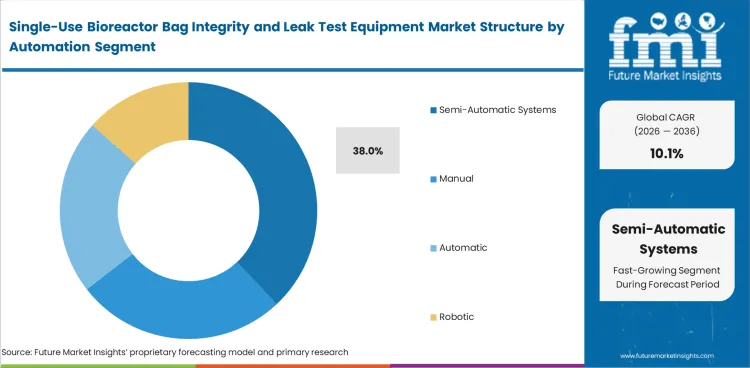
Semi-automatic systems are expected to account for 38% share in 2026 because they offer a practical middle ground between manual testing and full automation. Manual routines are still used, but repeatability, data quality, and operator dependence become harder to control as bag volumes rise. Semi-automatic platforms improve workflow discipline and electronic record capture without adding the higher cost, engineering support, and changeover burden often associated with fully automated systems. This preference is becoming more visible in facilities investing in adjacent quality tools, including single-use bioprocess sensor calibration and validation test systems.
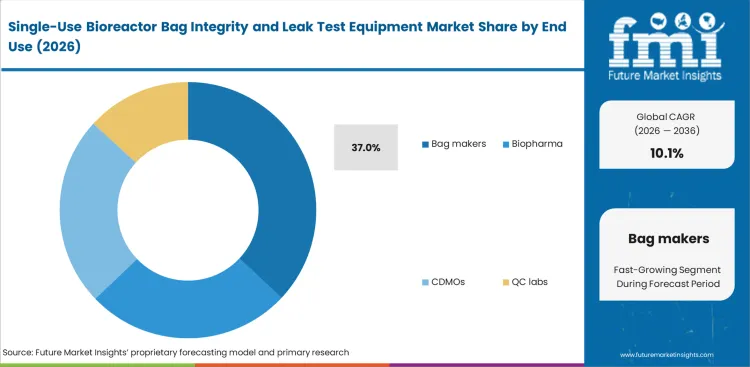
Bag makers remain the largest end-user group because defect screening begins before assemblies move into customer operations. The segment is projected to hold 37% share in 2026. Weak release testing at the supplier level can lead to shipment disputes, added incoming checks, and more review work once bags enter biologics sites. Biopharma companies and CDMOs represent notable demand as well, especially where internal quality rules require incoming verification. But supplier-side testing remains dominant because it addresses uncertainty earlier and with greater efficiency.
The single-use bioreactor bag integrity and leak test equipment market is expanding steadily with wider use of disposable process assemblies, growing dependence on larger 3D bag formats, and stronger demand for repeatable leak testing across supplier release and biologics site workflows. Equipment selection is moving beyond defect detection toward systems that support cleaner records, easier validation, and more dependable release decisions. Method transfer across bag families and setup variability in flexible assemblies still create challenges, though growth opportunities remain strong as suppliers and end users formalize bag assurance beyond visual checks.
Demand is supported by broader use of single-use bags across mixing, holding, and reactor-linked biologics operations, where a leak can disrupt preparation work, delay release, and trigger added review. The commercial effect becomes more serious as bag size increases because defect-related replacement, investigation, and handling carry greater process consequences. This increases the value of equipment that can deliver repeatable readings under daily operating conditions rather than only under controlled technical demonstrations.
Market growth remains constrained because flexible bag systems do not behave uniformly during testing. Chamber fit, trapped volume, film movement, connector placement, and handling setup can all influence result stability, which makes it harder to apply one method consistently across different bag formats. This increases validation effort and slows broader standardization, especially for users trying to scale test routines across multiple assemblies without adding review complexity.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 11.6% |
| China | 10.8% |
| Singapore | 10.4% |
| Ireland | 9.8% |
| United States | 9.4% |
| Germany | 8.7% |
| Switzerland | 8.5% |
Source: Future Market Insights, 2026.
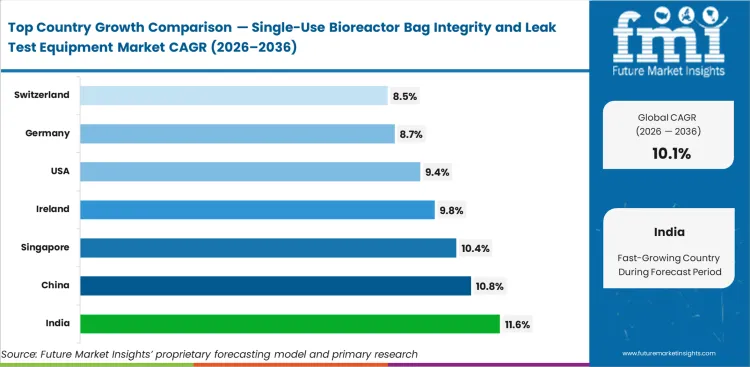
The United States is anticipated to grow at a CAGR of 9.4% through 2036, supported by a broad installed base of disposable process systems, strong commercial biologics production, and rising need for repeatable bag-release and incoming-check routines. Manufacturers in the country place greater value on equipment that can produce stable output and review-ready records without disrupting existing site procedures.
Ireland is expected to rise at a CAGR of 9.8% from 2026 to 2036, with demand supported by concentrated biologics activity, export-oriented sterile processing, and stronger operational focus on bag reliability. Compact and well-documented testing systems are gaining interest because they fit busy production environments where dependable release work carries direct commercial importance.
Germany is projected to grow at a CAGR of 8.7% by 2036 as disciplined biologics production, structured quality practices, and careful upgrade cycles sustain demand for integrity testing systems. Equipment decisions in the country are usually linked to method fit, software quality, and manageable validation effort rather than first-time adoption. This favors suppliers that support stable performance under existing site controls.
Switzerland is expected to grow at a CAGR of 8.5% through 2036, reflecting the country has high-value biopharma base and a disciplined quality environment. Adoption stays measured because many facilities operate under careful capital review. Integrity testing remains important where specialized assemblies and dependable output take priority over broader-volume release capacity.
China is projected to expand at a CAGR of 10.8% through 2036. Broader biologics investment, stronger local manufacturing depth, and wider use of single-use hardware across production sites support this growth. Plants are moving toward more formal integrity assurance routines as higher bag volumes make manual-only checks less reliable in daily operations.
India is likely to record a CAGR of 11.6% by 2036, supported by new biomanufacturing setups, wider single-use adoption, and stronger quality-system buildout across expanding facilities. Manufacturers prefers equipment that is easier to validate, works across multiple bag types, and fits semi-automatic workflows without adding excessive cost or review burden.
Singapore is set to witness at a CAGR of 10.4% through 2036, supported by dense high-value biomanufacturing activity and strong site discipline. Manufacturers prefer repeatable systems with efficient record handling because space, speed, and review efficiency carry more importance in compact facilities that run high-value production with limited tolerance for process disruption.
The single-use bioreactor bag integrity and leak test equipment market includes suppliers that combine leak-testing expertise with a practical understanding of flexible bioprocess bag behavior. Sartorius AG remains well positioned because of its close connection to single-use process environments, while ZELTWANGER Leaktesting & Automation, Cincinnati Test Systems, and Pfeiffer Vacuum+Fab Solutions compete through chamber design, controlled leak-detection capability, and support for repeatable test performance.
Bonfiglioli Engineering maintains relevance through its experience in inspection and validation-oriented testing environments. LACO Technologies and InterTech Development Company continue to serve specialized requirements where manufacturers need narrower bag-format support, targeted service, or simpler software handling. Manufacturers in this market place more weight on stable output during real bag handling, practical fixture adaptability, and review-ready records than on generic inspection claims.
Barriers to entry are moderate at the hardware level, though they rise when suppliers must prove method stability across multiple bag families, support validation-heavy workflows, and provide record traceability that fits formal release and incoming inspection routines. Vendors that reduce uncertainty during supplier QA and point-of-use assurance are better placed to strengthen their competitive position.
Key global companies leading the single-use bioreactor bag integrity and leak test equipment market include:
| Company | Bioprocess Bag Fit | Validation Support | Software / Traceability | Flexible-Bag Test Capability |
|---|---|---|---|---|
| Sartorius AG | High | High | Strong | High |
| ZELTWANGER Leaktesting & Automation GmbH | Medium | Medium | Strong | High |
| Cincinnati Test Systems | Medium | Medium | Moderate | High |
| Pfeiffer Vacuum+Fab Solutions | Medium | Medium | Moderate | High |
| Bonfiglioli Engineering | Medium | High | Moderate | Medium |
| LACO Technologies | Medium | Medium | Moderate | Medium |
| InterTech Development Company | Medium | Medium | Moderate | Medium |
Source: Future Market Insights competitive analysis, 2026.
Key Developments in Single-Use Bioreactor Bag Integrity and Leak Test Equipment Market
Major Global Players
Emerging Players / Specialized Challengers

| Metric | Value |
|---|---|
| Quantitative Units | USD 56.3 million (2025) / USD 62.0 million (2026) to USD 162.3 million (2036), at a CAGR of 10.1% |
| Market Definition | The single-use bioreactor bag integrity and leak test equipment market includes systems that detect leaks, verify seal quality, and confirm whether disposable bioreactor bags are suitable for sterile bioprocess use. Scope is restricted to test equipment, related fixtures, software, and associated testing functions rather than the bag assemblies themselves. |
| Segmentation |
|
| Regions Covered | North America, Europe, Asia Pacific |
| Countries Covered | United States, Germany, Ireland, Switzerland, China, India, Singapore, and 40 plus countries |
| Key Companies Profiled | Sartorius AG, ZELTWANGER Leaktesting & Automation GmbH, Cincinnati Test Systems, Pfeiffer Vacuum+Fab Solutions, Bonfiglioli Engineering, LACO Technologies, InterTech Development Company |
| Forecast Period | 2026 to 2036 |
| Approach | FMI combined primary interviews with bag makers, validation leads, and biologics site quality teams with desk review of public filings, guidance, association material, and method papers |
Source: Future Market Insights, 2026.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the single-use bioreactor bag integrity and leak test equipment market in 2026?
The market is projected to be anticipated at USD 62 million in 2026.
What will the market be worth by 2036?
FMI estimates the market will reach USD 162.3 million by 2036.
What CAGR is expected between 2026 and 2036?
The market is forecast to expand at a 10.1% CAGR during the period.
Which test method leads the market?
Pressure decay leads because it offers repeatable results and fits routine release workflows.
Which deployment setting remains dominant?
Supplier QA remains dominant because defect screening begins before shipment.
Which bag format leads adoption?
3D bags lead because larger flexible assemblies need more dependable integrity checks.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.