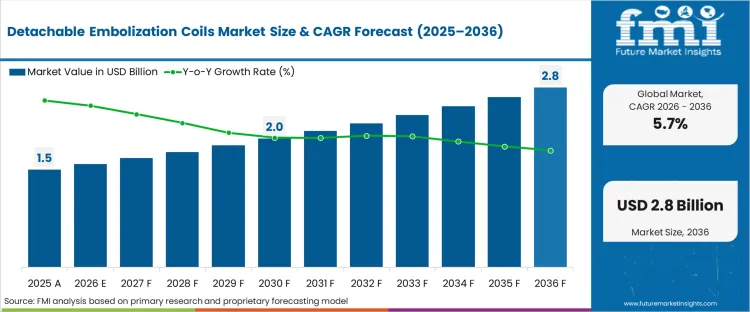
The Detachable Embolization Coils Market surpassed a value of USD 1.5 billion in 2025. The industry is set to cross USD 1.6 billion in 2026 at a CAGR of 5.7% during the forecast period. Demand outlook propels the valuation to USD 2.8 billion through 2036 as minimally invasive endovascular coiling displaces high-risk surgical clipping in progressively smaller and more anatomically complex neurovascular structures.
Hospital procurement departments are currently being forced to consolidate their neurovascular device portfolios, shifting from fragmented vendor sourcing toward integrated access-and-delivery platforms. This is not a clinical mandate but an economic one, driven by the need to standardize deployment mechanics across expanding interventional radiology teams. Buyers who delay this standardization face higher inventory carrying costs and increased procedural friction as newer, bioactive coated coils require specific microcatheter compatibilities. The structural transition is further complicated by physician preference, where tactile familiarity often overrides procurement's push for vendor consolidation.
Before growth in complex peripheral applications becomes self-reinforcing, the industry must cross a specific operational gate: the widespread adoption of automated or pressure-regulated detachment systems. Once interventionalists no longer rely on manual thermal or mechanical break points, the consistency of deployment improves, making the procedure viable for lower-volume ambulatory surgical centers rather than just specialized tertiary hubs. The market stabilizes around predictable per-procedure costs once this threshold is passed.
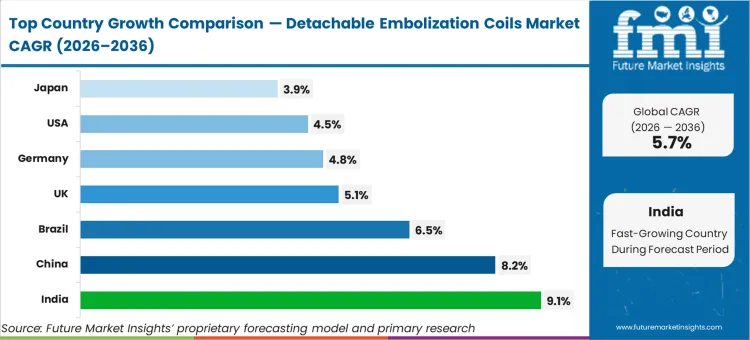
China is expected to advance at 8.2%, followed by India at 9.1%, Brazil at 6.5%, the United Kingdom at 5.1%, Germany at 4.8%, the United States at 4.5%, and Japan at 3.9%. This geographic divergence reflects where countries sit on the infrastructure curve; markets recording the highest rates are currently equipping newly established regional stroke centers, whereas slower-growth regions are primarily replacing legacy inventory within a mature installed base.
The detachable embolization coils market encompasses implantable wire devices designed to be navigated through a microcatheter and intentionally released into a blood vessel or aneurysm to induce thrombosis and occlude blood flow. The functional boundary of this market specifically requires a controlled detachment mechanism, whether thermal, mechanical, or electrolytic, distinguishing these devices from pushable or free-flow embolic agents.
Scope includes bare platinum coils, alloyed coils, hydrogel-coated variants, and bioactive modified coils utilized across neurovascular and peripheral interventions. The tracking covers the coil itself along with its integrated delivery pusher wire, provided they function as a single pre-packaged unit requiring a specific detachment controller. Embedded technologies such as electrolytic detachment systems are factored into the overall component valuation when sold alongside the primary coil inventory.
The market excludes liquid embolics, particulate agents, microspheres, and flow diverter stents. Flow diverters are excluded because they reconstruct the parent artery rather than filling the aneurysmal sac, representing a fundamentally different therapeutic mechanism and procurement category. Pushable coils lacking a specific, user-controlled detachment interface are similarly excluded due to their divergent risk profile and pricing structure.
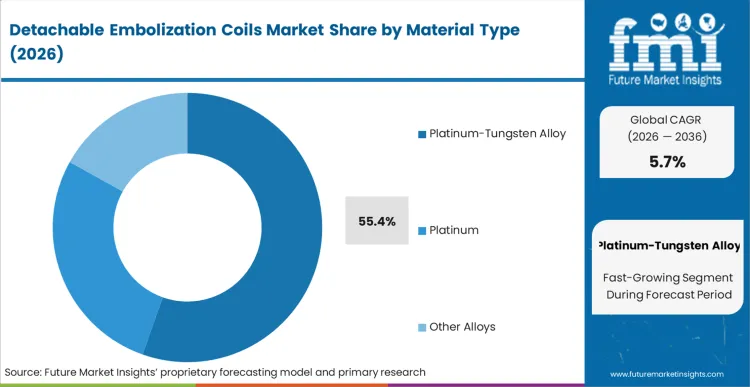
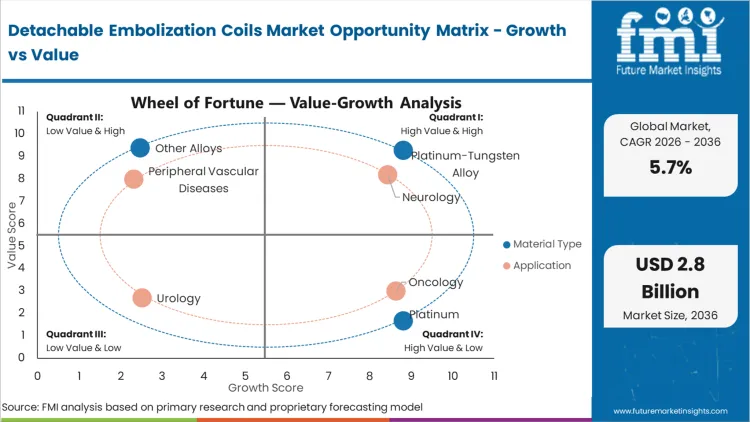
Legacy bare platinum configurations are currently failing to deliver the necessary volumetric packing density for wide-necked aneurysms, prompting the shift toward specialized alloys. Based on FMI's assessment, Platinum-Tungsten Alloy holds 55.4% share, addressing this exact mechanical deficit by providing the stiffness required to anchor the framing coil while maintaining radiopacity. Interventionalists initiating a coiling procedure must balance flexibility for navigation against the radial force needed for stabilization. The addition of tungsten fundamentally changes this operational calculation, allowing for a tighter packing matrix without the coil mass prolapsing into the parent vessel. Hospitals that continue to over-index on bare platinum for complex morphologies often face higher rates of late-stage coil compaction, forcing costly and risky retreatment procedures.
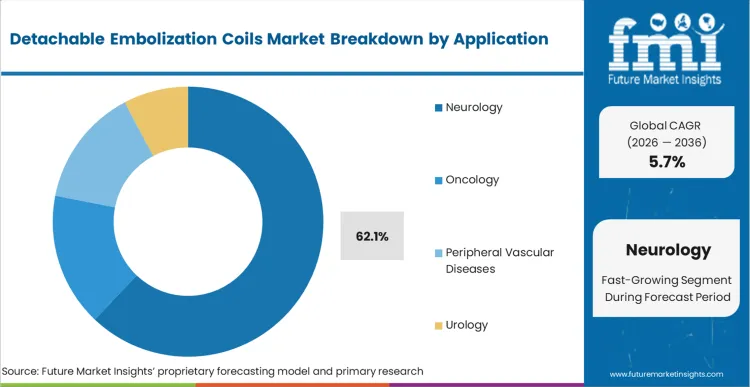
Neurointerventional surgery directors are currently facing a specific choice regarding how they equip their biplane suites for acute stroke and aneurysm management. Neurology holds 62.1% of the market share, driven entirely by the zero-tolerance threshold for off-target embolization in the cerebral vasculature. If a coil prematurely detaches or migrates during a peripheral tumor embolization, the consequence is localized ischemia; if it migrates during an intracranial procedure, the consequence is catastrophic neurological deficit. Therefore, the decision pattern in neurology strictly favors systems with instantaneous, predictable detachment mechanisms over those optimizing purely for cost. Facilities attempting to substitute less sophisticated, slow-detaching peripheral vascular devices into neuro-applications experience extended fluoroscopy times and elevated procedural risk profiles.
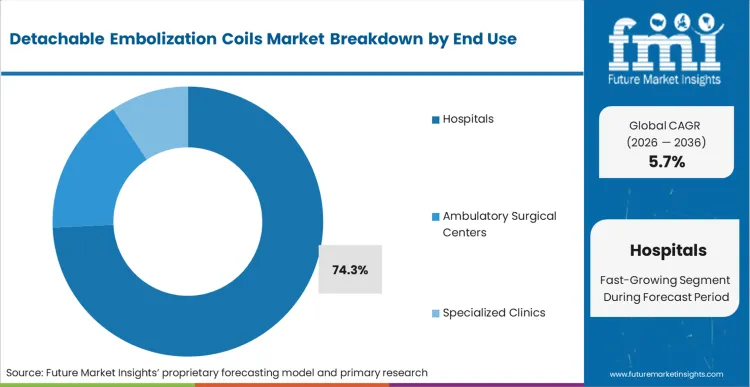
The reason Hospitals maintain a 74.3% share is tied directly to the capital infrastructure required to safely execute complex endovascular repairs. In FMI's view, the capacity to absorb the upfront cost of advanced biplane angiography systems and maintain a 24-hour on-call neurointerventional team restricts high-volume coiling to tertiary centers. Ambulatory surgical centers are beginning to absorb simpler peripheral embolizations, but they cannot economically justify the massive inventory of coil sizes, often hundreds of SKUs, required to be ready for unpredictable anatomical variations. Hospital procurement networks utilize their scale to negotiate consignment agreements, ensuring comprehensive sizing availability without tying up operational capital. Clinics attempting to manage this inventory on a direct-purchase basis find their margins entirely consumed by expired or unused niche coil sizes.
The necessity to shift complex vascular interventions from open surgical theaters to endovascular suites forces hospital administrators to upgrade their interventional radiology capabilities. This structural pressure requires procurement directors to establish dedicated endovascular inventory channels that prioritize high-reliability detachment systems. The commercial stakes are tied directly to patient throughput; biplane angiography suites represent one of the highest fixed-cost environments in a hospital. Any technology that reduces procedural time by ensuring predictable coil deployment directly increases the number of cases a facility can complete per day.
The primary structural friction in this market is the steep tactile learning curve associated with new deployment mechanisms. When a manufacturer introduces an upgraded electrolytic or mechanical detachment zone, it alters the physical feedback the physician feels through the pusher wire. This is not a temporary training issue; it is a structural barrier because highly experienced operators rely on muscle memory built over thousands of deployments to judge tension and prevent vessel rupture. While simulation training provides a partial solution, it cannot perfectly replicate the friction of tortuous in-vivo navigation, causing many senior interventionalists to resist migrating away from their established legacy systems.
Opportunities in the Detachable Embolization Coils Market
Based on the regional analysis, the Detachable Embolization Coils Market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania, and Middle East & Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 9.1% |
| China | 8.2% |
| Brazil | 6.5% |
| United Kingdom | 5.1% |
| Germany | 4.8% |
| United States | 4.5% |
| Japan | 3.9% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
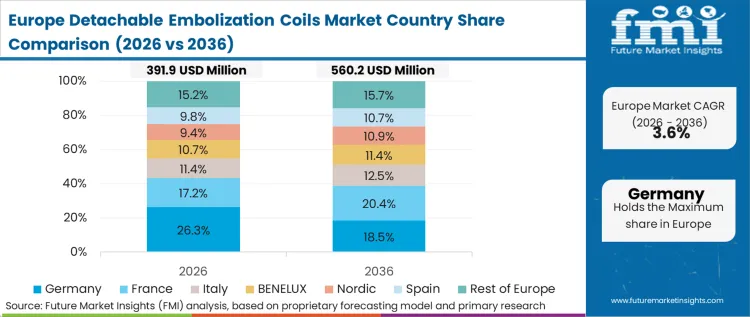
Vendors must prove that higher-priced bioactive or complex-alloy coils reduce downstream retreatment costs across a population level before gaining listing on approved national formularies, as per FMI's projection. Europe's trajectory is defined by its highly structured, centralized public procurement frameworks, fundamentally altering how vendors approach market access compared to fragmented private systems. Once approved, however, the adoption is rapid and uniform across the network. National health services do not evaluate coil technologies on an individual hospital basis, but rather conduct rigorous, multi-year health technology assessments to determine systemic cost-effectiveness.
FMI's report includes data on France, Italy, Spain, and the Nordics. The common structural pattern across these additional markets is the increasing reliance on pan-European clinical registries to validate post-market performance, dictating renewal contracts.
FMI analysts opine that the expansion of catheterization labs into tier-2 cities is the primary growth engine, but these new labs require coils with highly forgiving deployment characteristics as the physician skill base matures. Vendors cannot simply export Western consignment models here; instead, they must adapt to localized distribution networks and varying levels of physician endovascular training. Infrastructure readiness shapes the adoption curve across Asia Pacific, creating a stark divide between heavily concentrated metropolitan stroke centers and the rapidly expanding regional hospital networks.
FMI's report includes South Korea, Australia, and ASEAN nations. Across these territories, the development of localized clinical training fellowships is systematically lowering the barrier to entry for complex neurovascular procedures.
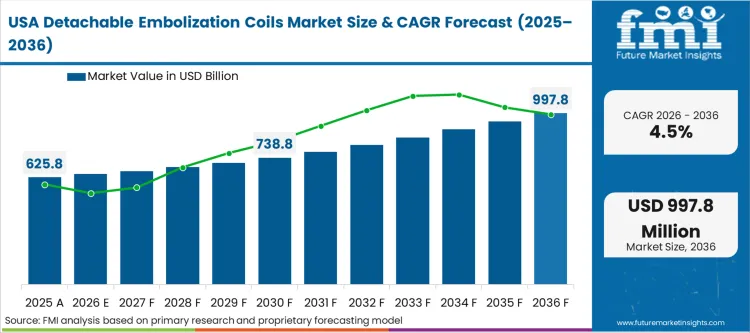
According to FMI's estimates, a manufacturer cannot succeed across the Americas with a single superior coil; they must offer a complete ecosystem of framing, filling, and finishing coils, alongside the proprietary microcatheters required to deliver them. These networks leverage their immense procedural volume to force vendors into portfolio-wide contracting, a trend spilling over into the privatized sectors of Latin America. The broader regional environment is driven by aggressive value analysis committee (VAC) oversight and the consolidation of independent hospitals into massive integrated delivery networks (IDNs).
FMI's report includes Canada and Mexico. The Canadian landscape mirrors the US in clinical sophistication but utilizes provincial tender processes that lengthen the procurement cycle and emphasize budget predictability over rapid technology turnover, a dynamic similarly observed in Latin America's heavily regulated public sectors.
The detachable embolization coils market operates under severe structural concentration, dictated by the immense capital required to achieve precision alloy manufacturing and the zero-tolerance regulatory burden of neurovascular approvals. Procurement directors at major stroke centers do not evaluate vendors on isolated technical specifications; they distinguish qualified partners from secondary suppliers based entirely on the ability to provide an uninterrupted, comprehensive sizing matrix. Dominant entities like Medtronic plc, Stryker Corporation, and Johnson & Johnson MedTech (CERENOVUS) maintain their primary formulary positions through highly integrated supply chain operations rather than sheer device engineering. These tier-1 suppliers guarantee that a hospital's consignment cabinet never lacks a critical finishing coil during an acute intervention, a logistical threshold that effectively locks out smaller manufacturers attempting to compete on unit price alone.
The defining structural advantage for incumbents lies within their established proprietary detachment generators, which are heavily embedded into the workflow of thousands of angiography suites. Terumo Corporation, Boston Scientific Corporation, and Penumbra, Inc. benefit directly from this installed base because interventional departments fiercely resist introducing multiple, incompatible detachment boxes into a crowded sterile field. Challengers such as MicroPort Scientific Corporation and BALT attempting to disrupt this stronghold must engineer mechanically agnostic detachment interfaces that bypass dense intellectual property thickets while guaranteeing zero-fail deployment across various neurovascular embolization devices. Replicating the physical coil is no longer the primary barrier; the actual hurdle is designing a universal deployment capability that integrates flawlessly into a hospital's existing capital equipment footprint without requiring nurses to memorize new operational sequences.
Large integrated delivery networks actively resist proprietary lock-in by mandating that their interventional suites maintain at least two approved vendors on the active formulary, creating a deliberate buffer against supply chain disruptions. Hospital procurement committees are aggressively pushing for commoditization to control costs, directly opposing the dominant vendors who rely on closed ecosystems to maintain high margins. The concentration is slowly fragmenting at the edges as companies like Cook Medical LLC and Kaneka Corporation commercialize highly specialized peripheral and ultra-soft finishing coils that target specific anatomical niches the broader portfolios overlook. As buyers gain bargaining power through the availability of simplified, battery-operated mechanical detachment systems, they force legacy manufacturers into comprehensive bundled contracting, shifting the competitive focus from individual coil superiority to total departmental pricing agreements.
| Metric | Value |
|---|---|
| Quantitative Units | USD 1.6 billion to USD 2.8 billion, at a CAGR of 5.7% |
| Market Definition | Implantable wire devices requiring a controlled detachment mechanism, navigated via microcatheter to purposefully occlude vascular structures. |
| Material Type Segmentation | Platinum, Platinum-Tungsten Alloy, Other Alloys |
| Application Segmentation | Neurology, Oncology, Peripheral Vascular Diseases, Urology |
| End User Segmentation | Hospitals, Ambulatory Surgical Centers, Specialized Clinics |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | India, China, Brazil, United Kingdom, Germany, United States, Japan, and 40 plus countries |
| Key Companies Profiled | Medtronic plc, Stryker Corporation, Terumo Corporation, Penumbra, Inc., Boston Scientific Corporation, MicroPort Scientific Corporation, Cook Medical LLC, Johnson & Johnson MedTech (CERENOVUS) |
| Forecast Period | 2026 to 2036 |
| Approach | The baseline value derives from a bottom-up aggregation of detachable embolization coil procedure volumes, applying region-specific adoption curves across neurovascular and peripheral embolization interventions to project future market expansion |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the Detachable Embolization Coils Market in 2026?How do proprietary generator boxes influence competition?
The market is expected to cross USD 1.6 billion in 2026. This valuation reflects the steady capital deployment by tertiary centers to maintain comprehensive neurovascular inventories, establishing a high revenue baseline even before accounting for procedural growth.
What will it be valued at by 2036?
The valuation reaches USD 2.8 billion by 2036. This cumulative buildup signals that complex endovascular coiling is firmly transitioning from an emergent, specialized intervention into standard prophylactic practice for unruptured aneurysms.
What CAGR is projected?
A CAGR of 5.7% is projected through the forecast period. This rate is heavily anchored by the physical limits of how quickly hospitals can train new interventional teams, serving as a realistic governor on otherwise rapidly expanding clinical demand.
Which Material Type segment leads?
Platinum-Tungsten Alloy holds 55.4% of the market share. The inclusion of tungsten provides the critical radial stiffness necessary to create a stable frame within irregular aneurysms, solving the compaction issues that historically plagued softer bare platinum configurations.
Which Application segment leads?
Neurology leads with 62.1% share. The absolute zero-tolerance for premature deployment in the cerebral vasculature forces procurement teams to invest heavily in the most precise, premium-priced detachment systems available, driving the bulk of category revenue.
Which End User segment leads?
Hospitals dominate with 74.3% share. The financial ability to host advanced biplane angiography systems and absorb the inventory carrying costs of massive coil sizing matrixes keeps complex interventions centralized in these high-acuity environments.
What drives rapid growth?
Expanding screening protocols identify unruptured aneurysms much earlier in the patient lifecycle. This forces neurointerventional departments to treat prophylactically, increasing overall procedural volumes and necessitating wider inventory stockpiles.
What is the primary restraint?
The steep tactile learning curve of new detachment systems slows adoption. Senior interventionalists resist migrating away from legacy coils because their muscle memory, critical for preventing vessel rupture, is tuned to the physical friction of their established platforms.
Which country grows fastest?
India advances at 9.1%, significantly faster than China's 8.2%. While China is restructuring its pricing through national tenders, India is actively building out first-time private catheterization infrastructure in tier-2 cities, creating massive initial outfitting volume.
How do European procurement frameworks impact vendors?
National health services in Europe rely on multi-year health technology assessments. Vendors must prove that premium bioactive coils reduce long-term retreatment rates before they can access the formulary, but once approved, adoption scales immediately across the network.
What changes when automated detachment systems become standard?
The reliance on manual physical breaks or complex electrolytic timing is removed. This operational shift lowers the deployment difficulty, allowing lower-volume ambulatory centers to safely adopt peripheral coiling procedures.
How do proprietary generator boxes influence competition?
Hospitals rarely want multiple, incompatible detachment generators cluttering the sterile field. Incumbents leverage this by locking facilities into their specific hardware ecosystems, making it highly difficult for challengers to introduce standalone coil lines.
Why is Volume-Based Procurement (VBP) critical in China?
VBP mandates shift the purchasing power entirely from physician preference to state-level administrative pricing. Vendors who win these tenders capture guaranteed massive volume but must survive on radically compressed unit margins.
What role do consignment models play in hospital purchasing?
Consignment shifts the financial burden of holding hundreds of coil SKUs from the hospital back to the vendor. Procurement directors favor manufacturers who utilize automated tracking cabinets, as it eliminates internal inventory management waste.
Why are flow diverters excluded from this analysis?
Flow diverters reconstruct the parent artery rather than filling the aneurysmal sac. They represent a distinct therapeutic approach with a different procurement cycle, regulatory pathway, and unit cost structure compared to intrasaccular coiling.
What makes the United Kingdom's NHS unique in this market?
NHS supply chain managers evaluate the total episodic cost of care rather than individual device pricing. They will approve higher-cost coil systems only if clinical data proves the devices reduce time spent in the angiography suite.
How does tactile feedback dictate physician preference?
Physicians use the friction transmitted through the pusher wire to confirm structural stability before detachment. Coils that lack the proper alloy stiffness fail to provide this necessary physical verification, leading to clinical rejection regardless of cost.
What causes premature detachment during navigation?
Tortuous vascular anatomy places extreme shear stress on the junction between the microcatheter and the coil. Systems without reinforced detachment zones risk breaking early, which is why neuro-specific coils command a massive premium over peripheral variants.
How is the shift to ambulatory surgical centers constrained?
ASCs operate on thinner margins and cannot afford to stock coils that expire unused. Until vendors develop highly consolidated, universal sizing kits, ASCs will struggle to economically support unpredictable endovascular interventions.
What is the strategic value of bioactive coatings?
Coils coated with hydrogels or specific polymers swell upon contact with blood, mechanically sealing microscopic gaps in the aneurysm neck. This accelerates the healing process and significantly reduces the statistical likelihood of requiring a second intervention.
How does Japan's regulatory environment shape adoption?
The PMDA enforces extremely conservative clinical validation protocols. This leads to a slower market entry for new coil technologies, but manufacturers who achieve approval benefit from intensely loyal hospital accounts that prioritize proven safety over price.
How do pricing structures differ between framing and finishing coils?
Framing coils require complex 3D shape retention and stiffer alloys to anchor the mass, commanding a premium. Finishing coils are ultra-soft and used purely for volumetric density, meaning hospitals often negotiate steeper volume discounts on these secondary units.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.