Healthcare and Laboratory Labels Market
The Healthcare and Laboratory Labels Market is Segmented by End User (Hospitals, Diagnostic Laboratories, Pharmaceutical Companies, Research Institutions), Label Technology (RFID Labels, Advanced 2D Barcodes, Smart Labeling Systems, 1D Barcodes, Intelligent Tags, Direct Part Marking), and Application Focus (Specimen Identification, Device Traceability, Medication Safety, Patient Safety Workflows, Sample Custody, Asset Tracking, Cold-Chain Labeling, Sterile Processing). Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Healthcare and Laboratory Labels Market Size, Market Forecast and Outlook By FMI
The healthcare and laboratory labels market is expected to expand from USD 8.6 billion in 2026 to USD 11.2 billion by 2036. The market is anticipated to register a 2.7% CAGR during the forecast period. Hospitals are likely to lead the end user segment with a 40.0% share in 2026.
Summary of the Healthcare and Laboratory Labels Market
- Demand and Growth Drivers
- Compliance-led identification is expected to keep label demand steady across hospitals and laboratory workflows.
- Laboratory automation is likely to increase use of RFID and advanced 2D codes for sample tracking.
- Sterilization and cryogenic storage requirements are anticipated to raise demand for durable label constructions.
- Product and Segment View
- Hospitals are expected to lead end user demand because labels support medication safety and specimen identification.
- Diagnostic laboratories are likely to increase label use as automated sample handling raises scan reliability requirements.
- RFID labels and smart labeling systems are anticipated to gain relevance where non-line-of-sight readability is needed.
- Geography and Competitive Outlook
- Japan is expected to lead country-level expansion as automation and identification discipline strengthen label requirements.
- India is likely to record strong expansion as diagnostics capacity and export-oriented pharmaceutical activity increase.
- Companies with stronger materials science and verification support are anticipated to hold better positions with regulated buyers.
- Analyst Opinion
- Ismail Sutaria, Principal Consultant for Packaging at FMI, suggests, “The healthcare and laboratory labels market is shifting toward labels that function as compliance and data-capture tools. Hospitals will remain the largest end user, while smart labels are likely to gain advantage as automation reduces tolerance for scan errors and manual verification.”
- Healthcare and Laboratory Labels Market Value Analysis
- The healthcare and laboratory labels market is moving from basic identification supply toward compliance-led workflow support.
- Industry value is likely to rise as hospitals use labels to reduce medication and specimen handling errors.
- Product uptake is expected to improve as RFID and advanced 2D codes support automated laboratory environments.
- Spending is anticipated to be supported by UDI rules, cold-chain labeling needs, and higher demand for durable adhesives.
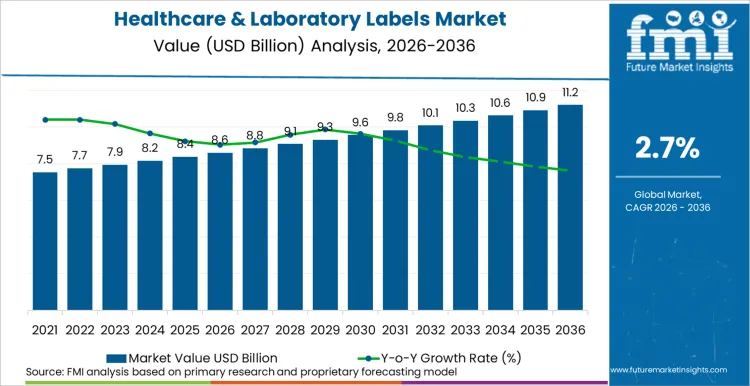
Healthcare & Laboratory Labels Key Takeaways
| Item | Metrics |
|---|---|
| Market Value (2026E) | USD 8.6 Billion |
| Projected Value (2036F) | USD 11.2 Billion |
| Forecast CAGR (2026-2036) | 2.7% |
Growth is best understood through compliance workload and automation density, not just hospital expansion. As therapy pipelines expand in rare and complex diseases, traceability expectations tend to harden across sample custody, cold-chain labelling, and device identification, reinforcing demand for higher-performance constructions such as those used alongside rare neurological disease treatment programmes.
Which Factors will Influence Course of the Healthcare & Laboratory Labels Market’s Expansion Trajectory?
Regulatory mandates are elevating label performance from a packaging detail to a compliance critical specification. FMI analysis indicates that UDI and related device identification rules raise the economic cost of label failure, forcing procurement teams to prioritize durability over unit price. Requirements tied to persistent identification are driving demand for face stocks and adhesives validated for abrasion resistance, chemical exposure, and repeated reprocessing cycles. The direct marking framework under 21 CFR 801.45 reinforces this shift by increasing remediation risk when identification degrades. As a result, labels are being specified to survive sterilisation, wipe downs, and long lifecycle use rather than short term handling. Compliance standards are therefore acting as a structural growth driver by resetting minimum performance thresholds across clinical and laboratory environments.
Why Is Lab Automation Making Non Line of Sight Readability a Procurement Requirement
Laboratory automation is changing how labels are read, verified, and valued. High throughput labs are increasingly built around robotics, conveyors, and enclosed systems where manual orientation is not feasible. FMI expects this architecture to tighten tolerance for print quality variation and accelerate adoption of RFID and advanced 2D codes that can be read without line of sight. Procurement decisions are shifting toward integrated smart labelling systems that combine tag design, printer calibration, and verification tools to ensure scan reliability at speed. Labels that fail under automated handling introduce workflow bottlenecks and data risk. Automation therefore increases the strategic value of smart labels as enablers of throughput, traceability, and error reduction rather than simple identification markers.
How Do Trade Policy and Input Volatility Slow Full Scale Smart Label Adoption
Trade policy uncertainty and input cost volatility continue to moderate the pace of smart label conversion. RFID economics remain sensitive to tag pricing, inlay availability, and substrate costs. FMI observes that when tariffs or supply disruptions raise unit costs, healthcare and laboratory buyers prioritize deployment in high risk workflows first rather than across entire fleets. This leads to phased adoption patterns where RFID is concentrated in specimen tracking and critical device identification, while 1D and 2D barcodes remain in lower risk applications. Hybrid labelling environments persist as cost management tools rather than technical limitations. Input volatility therefore slows market wide conversion but does not reverse the long term shift toward smart labelling in regulated settings.
How is the Market for Healthcare & Laboratory Labels Segmented?
The healthcare and laboratory labels market serves a diverse set of end users, including hospitals, diagnostic laboratories, pharmaceutical companies, and research institutions. Hospitals and diagnostic labs account for the largest share due to high volumes of specimen identification, patient safety requirements, and compliance driven labeling needs. Pharmaceutical companies use specialized labels to support drug development, manufacturing, and regulatory traceability across controlled environments. Research institutions represent a smaller but critical segment, relying on durable and data rich labels for sample tracking, long term storage, and experimental integrity across laboratory workflows.
Which dynamics sustain Hospitals as the leading end user?
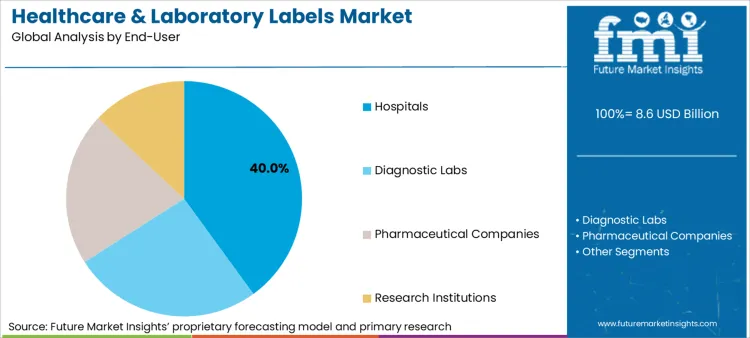
Hospitals account for a 40.0% share in 2026 as labelling is embedded across the highest concentration of risk-bearing touchpoints. These include bedside medication administration, specimen collection, sterile processing workflows, and repeated device reprocessing cycles. Each of these environments combines human interaction, regulatory oversight, and automation constraints, amplifying the operational cost of label failure. Hospital dominance is sustained by three structural factors. Labels must maintain legibility and adhesion through sterilisation, chemical exposure, and handling stress. Hospital workflows demand interoperability across printers, scanners, and clinical information systems, raising switching costs once standards are set. Misidentification events in hospitals generate disproportionate operational disruption, extending beyond rework into patient safety reviews and compliance escalation.
Which forces intensify demand from Diagnostic Labs?
Diagnostic labs are the most automation-sensitive end user. As specimen volumes scale, handling shifts from technician-driven steps to robotics and track systems. Demand is likely to concentrate in labels engineered for cold storage, centrifugal stress, and high-speed application. Where labs serve complex disease cohorts, identity and custody requirements tend to harden further, mirroring the operational discipline that supports neurology clinical trial execution and broader rare disease clinical trial expansion.
Which Procurement Patterns shape Pharmaceutical Companies and Research Institutions?
Pharmaceutical companies and research institutions behave differently: they buy labels as part of quality systems. Demand strengthens for constructions that support chain-of-identity, audit readiness, and multi-language compliance, especially where labelling must survive cold-chain handling and long storage windows. FMI expects incremental share gains in these end users where biologics and gene therapy pipelines expand, reinforcing the need for controlled labelling protocols similar to those used in orphan drug development ecosystems and nucleic acid and gene therapies in neuromuscular disorders.
What are the Key Trends in the Healthcare and Laboratory Labels Market?
FMI expects intelligent labels to move from limited pilots into formal policy driven adoption as operational scale increases. RFID and advanced 2D codes deliver the highest value where asset tracking, sample custody, and compliance reporting intersect across multi site networks. As healthcare systems and laboratory operators expand geographically, manual reconciliation becomes inefficient and error prone. Automated identification enables real time visibility, audit readiness, and standardized data capture across locations. Once these systems are embedded, reverting to manual or low capability labeling raises operational risk. Intelligent labels therefore evolve from optional efficiency tools into required infrastructure aligned with compliance, traceability, and enterprise wide data governance, driving sustained demand growth through 2036.
Why Is Materials Science Emerging as a Core Competitive Advantage
Materials science is becoming a decisive differentiator in healthcare and laboratory labels as performance thresholds tighten. FMI observes that buyers are actively reducing variability in adhesive chemistry, face stock stability, and print permanence because inconsistency creates scan failures and manual interventions downstream. Labels must withstand sterilisation cycles, chemical wipe downs, abrasion, and long term storage without degradation. Failure at any point disrupts automated workflows and compromises data integrity. Suppliers capable of engineering materials that deliver predictable performance across harsh conditions gain procurement preference. This focus shifts competition away from price and toward validated durability, positioning materials expertise as a long term competitive moat.
How Are Sustainability Requirements Reshaping Label Design Constraints
Sustainability expectations are extending beyond packaging into healthcare labeling systems. Supply chains are beginning to apply recyclability criteria without relaxing performance or readability standards. FMI notes that this creates a complex design constraint where materials must support recycling streams while maintaining compatibility with smart identification technologies.
Avery Dennison recognition for recyclable RFID labels compatible with PET recycling illustrates this direction. Demand is rising for constructions that do not automatically exclude products from recycling pathways. This shift favors suppliers that can integrate smart functionality with circular material compatibility.
How is the Healthcare & Laboratory Labels Landscape Profiled across Key Countries?
Against the global CAGR of 2.7%, all listed countries show above average expansion, indicating structurally stronger demand drivers at the national level. Japan leads at 4.5%, supported by advanced laboratory automation and strict identification discipline in clinical workflows. India follows closely at 4.4%, driven by rapid healthcare infrastructure buildout and expanding diagnostic capacity. China, at 4.1%, benefits from scale expansion in hospital networks and regulatory tightening around traceability. The United States, with 3.7%, reflects steady compliance led upgrades rather than volume growth. The United Kingdom, at 3.5%, shows moderate growth tied to digital traceability and procurement standardization.
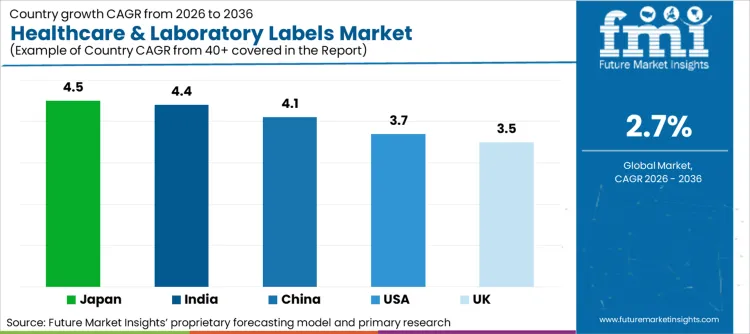
| Country | CAGR (2026-2036) |
|---|---|
| USA | 3.7% |
| UK | 3.5% |
| China | 4.1% |
| Japan | 4.5% |
| India | 4.4% |
Which factors keep the anchor for medical-grade labelling in USA?
Healthcare & laboratory labels demand in the USA is projected to grow at 3.7% CAGR through 2036. FMI views the USA as the market where compliance and scale meet. Large provider networks standardise label specifications across hundreds of sites, which raises the value of predictable performance, vendor qualification, and integrated verification. The UDI direct-marking framework under 21 CFR 801.45 reinforces the procurement bias toward permanence and traceability, pushing both label and device identigher reliability thresholds. The operational effect is a tightening of acceptable failure rates, opines FMI. When scan integrity is treated as a patient-safety control, labels become part of the clinical risk management system rather than a consumable line item. USA demand also benefits from the economics of automation. High labour costs increase the return on investment for reducing manual relabelling, rework, and specimen reconciliation. This creates a pull for smart labelling that integrates with hospital information systems and lab information systems, consistent with the blueprint’s shift toward intelligent labels.
Which realities sustain UK demand under structured compliance and central procurement?
Healthcare & laboratory labels demand in the UK is expected to progress at a 3.5% CAGR through 2036. FMI expects UK growth to be shaped by two forces: compliance readiness and procurement centralisation. NHS-aligned procurement behaviour tends to reward suppliers that can demonstrate reproducible performance, documentation discipline, and continuity of supply. From FMI’s perspective, UK demand will strengthen for labels engineered for hospital sterilisation environments and for laboratories that are increasing automation density. The blueprint highlights the role of central labelling registration requirements through EUDAMED-related deadlines affecting European market behaviour, with the UK specifically included in that regional framing. FMI also expects UK labs to segment label demand: routine workflows remain barcode-heavy, while higher-risk sample journeys adopt higher-spec constructions and verification. This mirrors the broader discipline required in complex therapy ecosystems, including those adjacent to neurodegenerative disease care pathways where diagnostic confidence and longitudinal tracking matter.
Which levers accelerate China’s shift toward pre-approval and scale-ready labelling?
Healthcare & laboratory labels demand in China is projected to rise at 4.1% CAGR through 2036, supported by scale and industriats growth to be driven by labelling protocols that tighten earlier in the product lifecycle, pushing manufacturers and labs to adopt scale-ready standards before commercial expansion. The blueprint notes NMPA Announcement No. 96 (2025) as a driver requiring specific labelling protocols for pre-approval commercial-scale batches to reduce supply chain gaps. FMI’s interpretation is that China is reducing the distance between regulatory submission labelling and commercial labelling. This increases demand for suppliers that can provide validated constructions, multilingual content control, and rapid iteration without requalification delays. China’s manufacturing depth also supports faster adoption of smart labelling components as local supply chains mature. FMI expects the strongest pull in high-throughput diagnostic environments and in hospital networks adopting asset tracking, where RFID reduces manual reconciliation.
Which mechanisms position Japan as a premium market for information-dense labels?
Healthcare & laboratory labels demand in Japan is estimated to expand at a 4.5% CAGR through 2036. FMI expects Japan to remain a premium market because compliance and consumer safety norms translate into information density and high expectations for label readability and permanence. The blueprint highlights a PMD Act update of September 2025 that MNGs on packaging, increasing demand for multi-layer booklet labels. FMI’s view is that the practical consequence is not just more text. It is tighter design tolerances, stronger adhesive performance, and consistent print quality under handling. This favours suppliers with precision converting, high-quality materials, and robust quality systems. Japan’s labelling demand is also reinforced by ageing demographics and high utilisation of diagnostic services, which increases the operational premium on error prevention and scan reliability.
Which structural advantages make India a high-growth market for smart labelling adoption?
Healthcare & laboratory labels demand in India is expected to grow at a 4.4% CAGR through 2036. FMI attributes India’s growth to expanding healthcare infrastructure, export-oriented pharma capacity, and a pragmatic shift toward automation in high-volume labs. Two signals stand out. The blueprint notes CDSCO system-auto-generated Neutral Codes (early 2026) to automate export-specific labelling for global markets. increase demand for standardised, audit-friendly label production, with stronger emphasis on data integrity and repeatability. The local build-out of RFID manufacturing supports faster smart label adoption. Avery Dennison’s Pune RFID inlay and label manufacturing facility is positioned to reduce lead times and improve responsiveness, strengthening the economics of scaling smart labelling beyond pilots. In FMI’s judgement, India’s adoption pattern will be staged: smart labels expand first in export-aligned pharma workflows and high-throughput diagnostics, then broaden into hospital asset tracking as system integration improves.
How Competitive will the Healthcare & Laboratory Labels Landscape be?
The competitive landscape is defined by portfolio breadth and qualification credibility. In FMI’s view, leadership is increasingly determined by three capabilities: materials performance in harsh conditions, integration with scanning and printing ecosystems, anlated documentation. Avery Dennison is strengthening its positioning around intelligent labels and local manufacturing. Its PET-recycling-stream compatible RFID label recognition demonstrates a strategy that treats sustainability as a design constraint rather than a marketing layer. The Pune RFID facility expands regional responsiveness, improving lead times and supporting faster iteration for India-specific operating conditions.
Brady is building a broader identification stack that extends beyond traditional labels into durable marking and specialty identification. The Mecco acquisition expands capabilities in direct part marking, supporting healthcare durable assets where permanence and harsh-environment resilience are non-negotiable. Zebra Technologies is positioned at the intersection of data capture and workflow digitisation. FMI expects Zebra’s advantage to show up where hospitals and labs treat labelling as part of a connected asset and data strategy, especially in RFID-enabled workflows. 3M, Sato Holdings, Labelmaster, and Lintec compete through combinations of materials science, printing systems, and healthcare-specific catalogues. FMI expects share gains for suppliers that deliver validated constructions across autoclave, cryogenic, and chemical exposure conditions, supported by verification and documentation. Where the customer environment becomes more regulated, the winner is typically the supplier that reduces qualification friction and scan failure risk, not the lowest unit cost.
Recent Development
- In June 2025, Avery Dennison introduced the first RFID label certified for recyclability (APR DfR), enabling PET-compatible smart labeling solutions. This innovation supports sustainable packaging and traceability across sectors, including healthcare and laboratory applications.
- In December 2025, SATO launched its CL4/6-SXR industrial label printers featuring AI-based predictive maintenance and enhanced print precision. The innovation improves operational efficiency, reduces downtime, and supports high-accuracy labeling in healthcare and laboratory environments.
Key Players in Healthcare & Laboratory Labels Market
- Avery Dennison
- Brady
- 3M
- Zebra Technologies
- Sato Holdings
- Labelmaster
- Lintec Corporation
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 8.6 billion |
| End User Category | Hospitals, Diagnostic Laboratories, Pharmaceutical Companies, Research Institutions |
| Label Technology | RFID Labels, Advanced 2D Barcodes, Smart Labeling Systems, 1D Barcodes, Intelligent Tags, Direct Part Marking |
| Material Type | Adhesive Systems (Autoclave-Resistant, Cryogenic-Compatible, Chemical-Resistant), Face Stocks, Recyclable Constructions, Multi-Layer Booklet Labels, PET-Compatible RFID Inlays |
| Application Focus | Specimen Identification, Device Traceability, Medication Safety, Patient Safety Workflows, Sample Custody, Asset Tracking, Cold-Chain Labeling, Sterile Processing |
| Performance Requirements | Sterilization Cycle Durability, Chemical Wipe-Down Resistance, Abrasion Resistance, Scan Integrity, Print Permanence, Non-Line-of-Sight Readability, Adhesion Under Extreme Conditions |
| Integration Systems | Automated Handling Equipment, Robotics and Conveyor Systems, Hospital Information Systems (HIS), Laboratory Information Systems (LIS), Data Capture Platforms, Printer and Scanner Ecosystems |
| Compliance Frameworks | UDI (Unique Device Identification), 21 CFR 801.45 Direct Marking Requirements, EUDAMED Registration, Chain-of-Identity Protocols, Multi-Language Compliance Standards |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, Middle East & Africa |
| Countries Covered | United States, United Kingdom, China, Japan, India, and 15+ countries |
| Key Companies Profiled | Avery Dennison, Brady Corporation, 3M, Zebra Technologies, Sato Holdings, Labelmaster, Lintec Corporation |
| Additional Attributes | Dollar sales by end user and technology type, regional demand trends, competitive landscape analysis, intelligent label adoption pathways, materials science differentiation, automation density impact, regulatory compliance drivers, sustainability and recyclability integration, error avoidance economics, verification and documentation systems, risk-bearing workflow positioning, lab throughput optimization, procurement standardization, and hybrid identification strategies |
Healthcare & Laboratory Labels Market by Segments
End User:
- Hospitals
- Diagnostic Labs
- Pharmaceutical Companies
- Research Institutions
Region:
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia & Pacific
- Middle East & Africa
Bibliography
- Brady Corporation. (2025, August 4). Brady Corporation acquires Mecco (press release).
- Avery Dennison. (2025, April 23). Avery Dennison opens first India-based RFID production facility (press release).
- Avery Dennison. (2025, June 25). Industry-first RFID label recognised for compatibility with PET recycling stream (press release). Regulations. (2025). 21 CFR § 801.45: Devices that must be directly marked with a unique device identifier.
- CCL Industries Inc. (2026, January 16). Press release listing: Agreement to acquire ALT Technologies for CCL Design.
- Zebra Technologies Corporation. (2025, August 5). Q2 2025 earnings call transcript (CEO remarks on connected solutions and RFID interest).
Frequently Asked Questions
How big is the global healthcare & laboratory labels market?
The global healthcare & laboratory labels market is estimated to be valued at USD 8.6 billion in 2026 and is projected to reach USD 11.2 billion by 2036. Market size reflects steady, compliance-driven demand rather than discretionary volume growth, with labels embedded across essential clinical and laboratory workflows.
What is the growth outlook for the healthcare & laboratory labels market over the next 10 years?
Between 2026 and 2036, the market is expected to expand at a 2.7% CAGR. Growth is shaped by tighter traceability requirements, rising laboratory automation, and increasing reliance on durable and information-dense labels in regulated healthcare environments.
Which end-use industries drive demand for healthcare & laboratory labels?
Hospitals account for the largest share due to high-risk labelling touchpoints such as medication administration, specimen handling, and device reprocessing.
What are the key regional differences in adoption across healthcare & laboratory labels?
USA leads due to stringent device identification and traceability requirements. Japan and India exhibit faster growth driven by regulatory updates and expanding diagnostic and pharmaceutical capacity, while the UK and China reflect structured compliance-led adoption tied to centralised healthcare systems and pre-approval labelling standards.
Who are the leading suppliers, and how is leadership defined in this market?
Market leadership is defined by the ability to deliver validated label performance, regulatory documentation support, and integration with printing, scanning, and data capture ecosystems. Companies such as Avery Dennison, Brady, 3M, Zebra Technologies, Sato Holdings, Labelmaster, and Lintec Corporation compete through materials science expertise, intelligent labelling capabilities, and proven reliability in harsh healthcare and laboratory environments.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End-User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End-User , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End-User , 2026 to 2036
- Hospitals
- Diagnostic Labs
- Pharaceutical Companies
- Research Institutions
- Hospitals
- Y to o to Y Growth Trend Analysis By End-User , 2021 to 2025
- Absolute $ Opportunity Analysis By End-User , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By End-User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By End-User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By End-User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By End-User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By End-User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By End-User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By End-User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By End-User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By End-User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By End-User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By End-User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By End-User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By End-User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By End-User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By End-User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By End-User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By End-User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By End-User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By End-User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By End-User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By End-User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By End-User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By End-User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By End-User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By End-User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By End-User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By End-User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By End-User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By End-User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By End-User
- Competition Analysis
- Competition Deep Dive
- Avery Dennison
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Brady
- 3M
- Zebra Technologies
- Sato Holdings
- Labelmaster
- Lintec Corporation
- Avery Dennison
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by End-User , 2021 to 2036
- Table 3: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by End-User , 2021 to 2036
- Table 5: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: Latin America Market Value (USD Million) Forecast by End-User , 2021 to 2036
- Table 7: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Western Europe Market Value (USD Million) Forecast by End-User , 2021 to 2036
- Table 9: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Eastern Europe Market Value (USD Million) Forecast by End-User , 2021 to 2036
- Table 11: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: East Asia Market Value (USD Million) Forecast by End-User , 2021 to 2036
- Table 13: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: South Asia and Pacific Market Value (USD Million) Forecast by End-User , 2021 to 2036
- Table 15: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 16: Middle East & Africa Market Value (USD Million) Forecast by End-User , 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by End-User , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by End-User , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by End-User
- Figure 6: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Region
- Figure 9: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 10: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 11: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 12: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 17: North America Market Value Share and BPS Analysis by End-User , 2026 and 2036
- Figure 18: North America Market Y-o-Y Growth Comparison by End-User , 2026-2036
- Figure 19: North America Market Attractiveness Analysis by End-User
- Figure 20: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 21: Latin America Market Value Share and BPS Analysis by End-User , 2026 and 2036
- Figure 22: Latin America Market Y-o-Y Growth Comparison by End-User , 2026-2036
- Figure 23: Latin America Market Attractiveness Analysis by End-User
- Figure 24: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 25: Western Europe Market Value Share and BPS Analysis by End-User , 2026 and 2036
- Figure 26: Western Europe Market Y-o-Y Growth Comparison by End-User , 2026-2036
- Figure 27: Western Europe Market Attractiveness Analysis by End-User
- Figure 28: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: Eastern Europe Market Value Share and BPS Analysis by End-User , 2026 and 2036
- Figure 30: Eastern Europe Market Y-o-Y Growth Comparison by End-User , 2026-2036
- Figure 31: Eastern Europe Market Attractiveness Analysis by End-User
- Figure 32: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: East Asia Market Value Share and BPS Analysis by End-User , 2026 and 2036
- Figure 34: East Asia Market Y-o-Y Growth Comparison by End-User , 2026-2036
- Figure 35: East Asia Market Attractiveness Analysis by End-User
- Figure 36: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 37: South Asia and Pacific Market Value Share and BPS Analysis by End-User , 2026 and 2036
- Figure 38: South Asia and Pacific Market Y-o-Y Growth Comparison by End-User , 2026-2036
- Figure 39: South Asia and Pacific Market Attractiveness Analysis by End-User
- Figure 40: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Middle East & Africa Market Value Share and BPS Analysis by End-User , 2026 and 2036
- Figure 42: Middle East & Africa Market Y-o-Y Growth Comparison by End-User , 2026-2036
- Figure 43: Middle East & Africa Market Attractiveness Analysis by End-User
- Figure 44: Global Market - Tier Structure Analysis
- Figure 45: Global Market - Company Share Analysis

