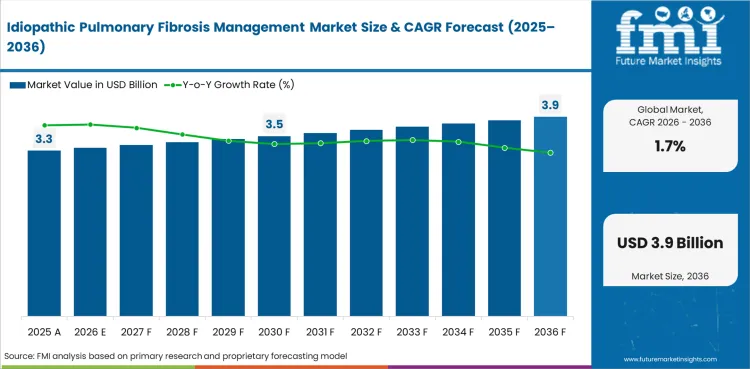
Idiopathic Pulmonary Fibrosis Management Market Size, Market Forecast and Outlook By FMI
Summary of the Idiopathic Pulmonary Fibrosis Management Market
- Demand and Growth Drivers
- Established antifibrotic standard of care sustains treatment demand.
- Diagnostic improvement enables earlier treatment initiation.
- Combination therapy investigation may expand per-patient expenditure.
- Product and Segment View
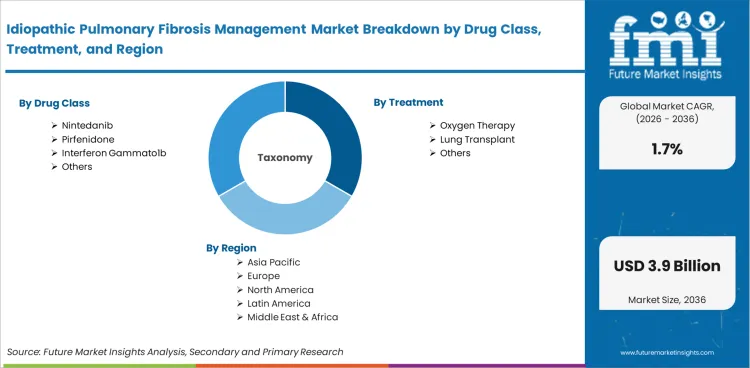
- Nintedanib is expected to lead with 39.6% of drug class demand in 2026.
- Oxygen therapy is expected to represent 57.3% of treatment demand.
- Oral administration is expected to account for 74.8% of route demand.
- Geography and Competitive Outlook
- The EU (1.8%) and Japan (1.7%) are expected to lead growth.
- The USA (1.6%) maintains demand through the largest diagnosed population.
- Companies with established antifibrotic products are expected to maintain the duopoly.
- Analyst Opinion
- The IPF market is a mature therapeutic category with established antifibrotic duopoly.
- Nintedanib and pirfenidone represent the treatment standard.
- The market grows modestly, reflecting stable incidence and established treatments.
- Companies advancing combination or novel mechanism drugs may disrupt the paradigm.
Idiopathic Pulmonary Fibrosis Management Market Definition
The idiopathic pulmonary fibrosis management market covers antifibrotic pharmaceutical treatments and supportive care for managing IPF, a chronic progressive lung disease of unknown cause.
Idiopathic Pulmonary Fibrosis Management Market Inclusions
Market scope covers all commercially available IPF management products categorized by drug class (nintedanib, pirfenidone, supportive therapies), treatment (oxygen therapy, pulmonary rehabilitation, lung transplantation, antifibrotic medication), route of administration (oral, inhaled, intravenous), and distribution channel (hospital pharmacies, retail pharmacies, specialty pharmacies). Revenue coverage spans 2026 to 2036.
Idiopathic Pulmonary Fibrosis Management Market Exclusions
The scope does not include treatments for other ILD subtypes, general respiratory support equipment, or lung transplantation surgical costs.
Idiopathic Pulmonary Fibrosis Management Market Research Methodology
- Primary Research: FMI analysts conducted interviews with pulmonologists, IPF centre directors, and specialty pharmacy managers.
- Desk Research: Combined data from IPF patient registries, pharmaceutical prescription databases, and respiratory disease treatment records.
- Market Sizing and Forecasting: Bottom-up sizing across drug class, treatment modality, and distribution channel segments with regional IPF diagnosis rate curves.
- Data Validation: Cross-checked quarterly against patient registry data, pharmaceutical sales volumes, and respiratory clinic utilization.
Why is the Idiopathic Pulmonary Fibrosis Management Market Growing?
- Established antifibrotic therapies nintedanib and pirfenidone have become standard of care, with clinical evidence demonstrating slowed disease progression.
- Improving diagnostic capabilities including high-resolution CT scanning are increasing confirmed IPF diagnosis rates.
- Growing clinical trial activity investigating combination approaches is expanding the treatment pipeline.
Demand reflects the chronic progressive nature of IPF affecting an estimated 13 to 20 per 100,000 individuals, with median survival of 3 to 5 years from diagnosis.
Adoption of nintedanib leads at 39.6% driven by INPULSIS trial evidence demonstrating reduced annual FVC decline rate.
Pricing reflects the specialty pharmaceutical positioning of antifibrotic agents, with chronic treatment generating sustained pharmaceutical expenditure.
Market Segmentation Analysis
- Nintedanib accounts for 39.6% of the drug class segment.
- Oxygen Therapy represents 57.3% of the treatment segment.
- Oral leads the route of administration segment with 74.8% share.
The IPF management market is segmented by drug class, treatment modality, route of administration, and distribution channel. Demand reflects the chronic progressive disease course, established antifibrotic duopoly, and comprehensive supportive care requirements.
Insights into the Nintedanib Drug Class Segment
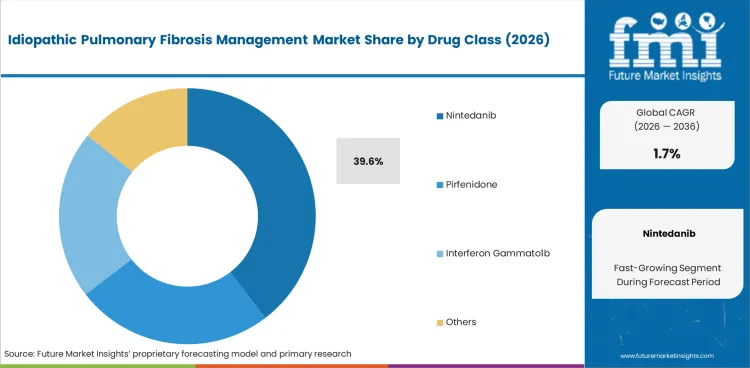
Nintedanib leads at 39.6% due to clinical evidence from INPULSIS trials demonstrating significant FVC decline rate reduction. Its triple tyrosine kinase inhibitor mechanism provides a distinct pharmacological approach.
Insights into the Oxygen Therapy Treatment Segment
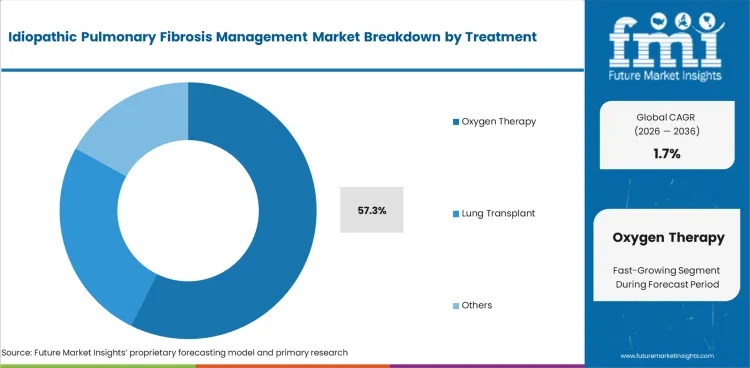
Oxygen therapy represents 57.3% of treatment demand, reflecting the progressive hypoxemia that develops in IPF patients requiring supplemental oxygen in later disease stages.
Idiopathic Pulmonary Fibrosis Management Market Drivers, Restraints, and Opportunities
- Regulatory and structural demand drivers are creating sustained procurement patterns.
- Cost and access constraints create segmentation in adoption rates.
- Technology and product development are expanding the addressable market.
The idiopathic pulmonary fibrosis management market is shaped by regulatory, technology, and procurement dynamics.
Antifibrotic Standard of Care
Demand is shaped by nintedanib and pirfenidone as standard treatment, with guidelines recommending initiation at diagnosis.
Diagnostic Rate Improvement
Growth reflects improving IPF diagnosis through HRCT and multidisciplinary assessment.
Combination Therapy Investigation
Adoption of combination approaches is being explored in clinical trials.
Mature Market Growth
The market grows modestly reflecting the established treatment landscape and stable disease incidence.
Analysis of Idiopathic Pulmonary Fibrosis Management Market By Key Countries
.webp)
| Country |
CAGR |
| EU |
1.8% |
| Japan |
1.7% |
| USA |
1.6% |
| South Korea |
1.6% |
| UK |
1.5% |
Source: FMI analysis based on primary research and proprietary forecasting model
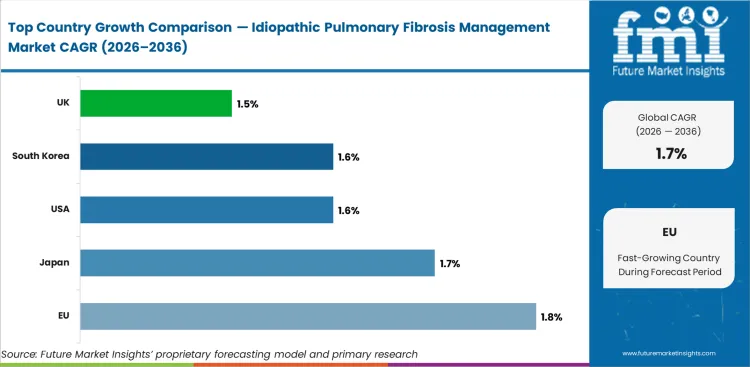
- EU leads with 1.8% CAGR.
- Japan at 1.7% reflects expanding capabilities.
- USA (1.6%) and South Korea (1.6%) maintain steady growth.
The global idiopathic pulmonary fibrosis management market is expected to grow at 1.7% per year from 2026 to 2036.
Demand Outlook for Idiopathic Pulmonary Fibrosis Management Market in the European Union
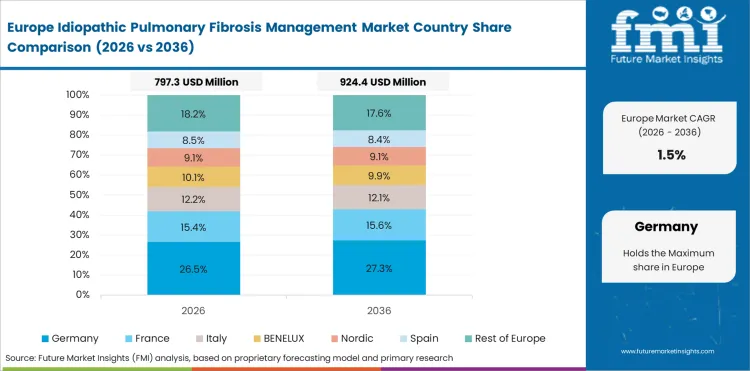
The EU is expected to grow at 1.8% through 2036, supported by established IPF treatment centres and EMA-approved therapies.
- IPF centres provide specialist prescribing.
- EMA-approved therapies ensure availability.
- Clinical research explores combination approaches.
Future Outlook for Idiopathic Pulmonary Fibrosis Management Market in Japan
Japan is expected to grow at 1.7% through 2036, driven by aging population prevalence and pirfenidone development heritage.
- Aging population increases IPF prevalence.
- Respiratory infrastructure supports treatment.
- Pirfenidone heritage provides clinical expertise.
Opportunity Analysis of Idiopathic Pulmonary Fibrosis Management Market in the United States
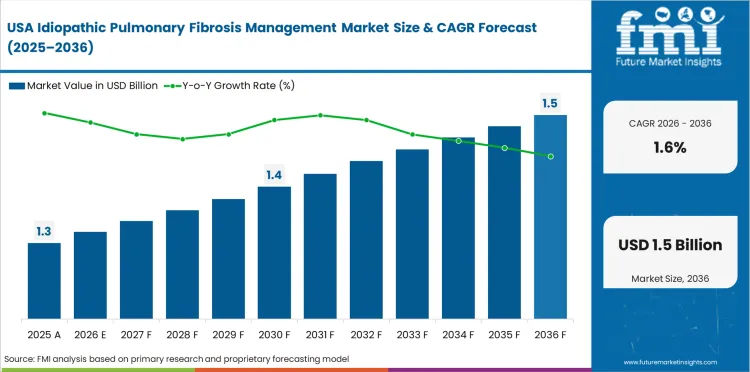
The USA is expected to grow at 1.6% through 2036, supported by the largest diagnosed IPF population.
- Largest IPF population sustains demand.
- FDA-approved therapies provide options.
- Specialist networks deliver prescribing.
In-depth Analysis of Idiopathic Pulmonary Fibrosis Management Market in South Korea
South Korea is expected to grow at 1.5% through 2036, supported by respiratory medicine advancement.
- Respiratory medicine provides specialist treatment.
- Antifibrotic access is established.
- Clinical trials advance understanding.
Sales Analysis of Idiopathic Pulmonary Fibrosis Management Market in the United Kingdom
The UK is expected to grow at 1.4% through 2036, supported by NICE-approved antifibrotic therapies.
- NICE approvals guide treatment access.
- NHS respiratory services provide care.
- IPF centres coordinate management.
Competitive Landscape and Strategic Positioning
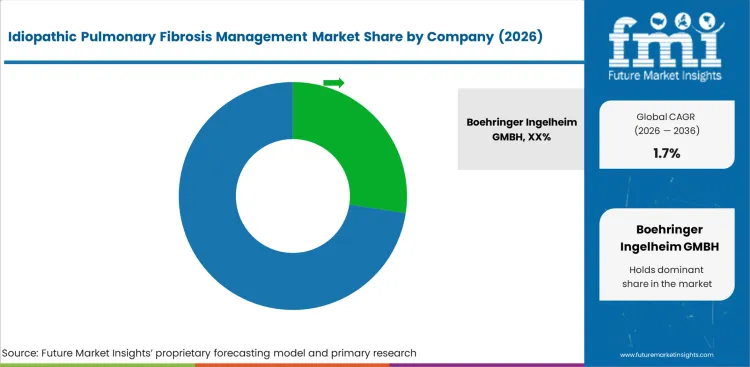
- Boehringer Ingelheim GMBH leads with an estimated 27.4% market share.
- The competitive landscape includes global and specialized players.
- Entry barriers include regulatory, relationship, and scale factors.
Boehringer Ingelheim GMBH leads through Ofev (nintedanib), the most widely prescribed antifibrotic for IPF.
Biogen maintains a position through pirfenidone (Esbriet), the first approved antifibrotic.
Novartis AG and MediciNova Inc. compete on respiratory pipeline programs.
Entry barriers include the established duopoly and high clinical trial costs.
Key Companies in the Idiopathic Pulmonary Fibrosis Management Market
Key global companies leading the idiopathic pulmonary fibrosis management market include:
- Boehringer Ingelheim GMBH (Germany) and Biogen (USA) hold dominant positions through the two approved antifibrotics.
- Novartis AG (Switzerland) and MediciNova Inc. (USA) compete on respiratory pipeline programs.
- Bristol-Myers Squibb (USA), FibroGen (USA), and Galapagos NV (Belgium) represent emerging pipeline positions.
Competitive Benchmarking: Idiopathic Pulmonary Fibrosis Management Market
| Company |
IPF Drug Portfolio |
Clinical Evidence |
Specialist Relationships |
Geographic Reach |
| Boehringer Ingelheim GMBH |
High |
Strong |
Strong |
Global |
| Biogen |
High |
Strong |
Strong |
Global |
| Novartis AG |
Low |
Moderate |
Strong |
Global |
| MediciNova Inc. |
Low |
Low |
Low |
N. America |
| Bristol-Myers Squibb |
Low |
Moderate |
Strong |
Global |
| FibroGen |
Low |
Moderate |
Moderate |
N. America, Global |
Source: Future Market Insights competitive analysis, 2026.
Key Developments in Idiopathic Pulmonary Fibrosis Management Market
- In 2025, Boehringer Ingelheim continued to support long-term clinical evidence generation for nintedanib in idiopathic pulmonary fibrosis, reinforcing its role in slowing forced vital capacity (FVC) decline in patients with progressive fibrotic lung disease.
- In 2025, FibroGen Inc. continued to advance research in antifibrotic therapies targeting novel pathways implicated in progressive pulmonary fibrosis, reflecting ongoing efforts to address unmet needs in idiopathic pulmonary fibrosis and related interstitial lung diseases.
Key Players in the Idiopathic Pulmonary Fibrosis Management Market
Major Global Players
- Boehringer Ingelheim GMBH
- Biogen
- Novartis AG
- Medicinova Inc.
- Bristol-Myers Squibb Company
- FibroGen Inc.
- Genentech (Roche)
Emerging Players/Startups
- FibroGen
- Galapagos NV
- Pliant Therapeutics
- Blade Therapeutics
- Samumed
Report Scope and Coverage
| Parameter |
Details |
| Quantitative Units |
USD 3.31 billion to USD 3.92 billion, at a CAGR of 1.7% |
| Market Definition |
The idiopathic pulmonary fibrosis management market covers antifibrotic pharmaceutical treatments and supportive care for managing IPF, a chronic progressive lung disease of unknown cause. |
| Regions Covered |
North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered |
USA, UK, France, Germany, Italy, South Korea, Japan, China, India, 30 plus countries |
| Key Companies Profiled |
Boehringer Ingelheim GMBH, Biogen, Novartis AG, Medicinova Inc., Bristol-Myers Squibb Company, FibroGen Inc., Genentech (Roche), Gilead Sciences, United Therapeutics, Veracyte Inc. |
| Forecast Period |
2026 to 2036 |
| Approach |
Hybrid bottom-up and top-down methodology starting with verified transaction data, projecting adoption velocity across segments and regions. |
Market Segmentation Analysis
Idiopathic Pulmonary Fibrosis Management Market Market Segmented by Drug Class:
- Nintedanib
- Pirfenidone
- Interferon Gammato1b
- Others
Idiopathic Pulmonary Fibrosis Management Market Market Segmented by Treatment:
- Oxygen Therapy
- Lung Transplant
- Others
Idiopathic Pulmonary Fibrosis Management Market Market Segmented by Route of Administration:
Idiopathic Pulmonary Fibrosis Management Market Market Segmented by Distribution Channel:
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
Idiopathic Pulmonary Fibrosis Management Market Market by Region:
- North America
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Research Sources and Bibliography
- 1. American Thoracic Society. (2025). ATS/ERS/JRS/ALAT Guidelines: IPF. ATS.
- 2. European Respiratory Society. (2024). ERS Statement on IPF. ERS.
- 3. USA Food and Drug Administration. (2025). FDA Approved Antifibrotics for IPF. FDA.
- 4. National Institute for Health and Care Excellence. (2024). NICE Appraisals: Nintedanib and Pirfenidone. NICE.
- 5. World Health Organization. (2024). WHO Classification of ILD. WHO.
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with publication dates, URLs, and supporting data for all cited works.
This Report Answers
- Estimating the market size and revenue from 2026 to 2036.
- Segmentation by drug class, treatment, route of administration, and distribution channel.
- Regional and country-level insights across more than 30 markets.
- Antifibrotic clinical evidence and pipeline analysis.
- Competitive landscape assessment.
- Investment opportunity identification.
- Treatment protocol and prescribing pattern analysis.
- Data delivery in PDF and Excel formats.
Frequently Asked Questions
What is the global market demand for Idiopathic Pulmonary Fibrosis Management in 2026?
In 2026, the global market is expected to be worth USD 3.31 billion.
How big will the Idiopathic Pulmonary Fibrosis Management Market be in 2036?
By 2036, the market is expected to be worth USD 3.92 billion.
How much is demand expected to grow between 2026 and 2036?
Between 2026 and 2036, demand is expected to grow at a CAGR of 1.7%.
Which Drug Class segment is expected to lead in 2026?
Nintedanib is expected to account for 39.6% of the segment in 2026.
What is causing demand to rise in EU?
EU is expected to grow at 1.8% through 2036.
What is causing demand to rise in Japan?
Japan is expected to grow at 1.7% through 2036.
What does this report mean by Idiopathic Pulmonary Fibrosis Management Market definition?
The idiopathic pulmonary fibrosis management market covers antifibrotic pharmaceutical treatments and supportive care for managing IPF, a chronic progressive lung disease of unknown cause..
How does FMI make the forecast?
Forecasting uses a hybrid bottom-up and top-down methodology starting with verified transaction data, projecting adoption velocity across segments and regions.



.webp)





