In vitro Fertilization Monitoring System Market
In vitro Fertilization Monitoring System Market
Historical Data Covered: 2008 to 2016 | Base Year: 2017 | Estimated Year: 2018 | Forecast Period: 2019 to 2028
 Market Outlook
In latest decades, in vitro fertilization has become the fastest growing technology as infertility has become a major issue worldwide. According to the Centers for Disease Control and Prevention data for 2011- 2013 in the U.S. reported that about 7.5 million women had decreased the ability to get pregnant or carry a baby to long term.
In vitro fertilization monitoring (IVF) system also called time-lapse monitoring system is a big turn up and gives a ray of hope for women who have experienced repeated IVF failures. An IVF monitoring system gives information about the development of the embryos, and allows the embryologists to categorize the embryos, which leads to a successful pregnancy.
The in vitro fertilization monitoring systems have cameras present in the incubators that depict the development without the need of removing them from their optimal conditions. The pictures are collected and then can be depicted in the form of a film.
This technology helps in embryo selection by deselecting those with a lower implantation possibility. Also, in vitro fertilization monitoring technique increases the live birth rates and reduces the number of cycles. Earlier, the embryos were removed from the incubator to assess the cell division and its morphology at least once a day and were put under the microscope for examining the developmental progress, which affected the optimal culturing conditions.
With the advancement in monitoring, the embryologists identify the patterns of development or the abnormalities in embryos growth that gives an indication of the future development of the embryo. In vitro fertilization monitoring systems provide optimal incubator conditions for the developing embryo without removing it and gives detailed analysis allowing to pick out the embryos with the highest potential for pregnancy.
Advancements Influencing the Development of In Vitro Fertilization Monitoring System Market
In vitro fertilization is the fusion of eggs and sperms fertilized in culture mediums or test tubes resulting in formation of embryos, which are later implanted in the uterus. With the IVF monitoring system, it can be determined if the embryos will reach the blastocyst (16 celled) stage or not. Also, embryologists can analyze and review the number of cells, their growth and shape, level of fragmentation and thickness of the outer shell and later decide if it is suitable for the transfer.
IVF monitoring systems lower the possibilities of miscarriages. The monitoring system is designed to identify the potential embryos for treatment, while earlier the embryos were cultured in petri-dishes and were kept in the incubators within optimal conditions such as CO2, temperature and humidity, etc. Also the embryos were examined by removing them from incubators which hampered their development.
In vitro fertilization Monitoring System Assessment by End user
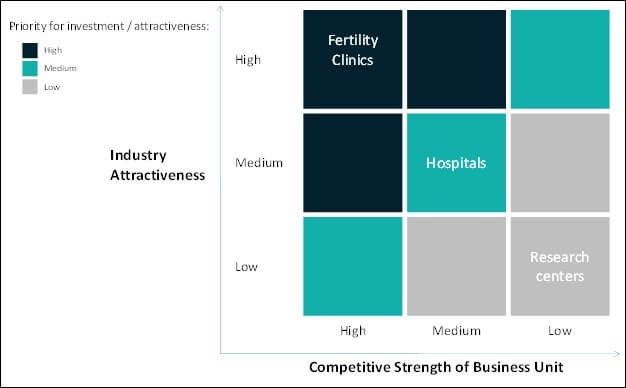
Factors Driving the In Vitro Fertilization Monitoring System Market
The in-vitro fertilization monitoring market is expected to rise globally as the technique offers improved implantation rates, environmental settings with adjustable CO2 & O2 levels and controlled temperatures with multiple image acquisitions. The increasing rates of pregnancy loss and miscarriages are also driving the growth of the market.
For instance, the launch of in vitro fertilization monitoring system from Vitrolife AB in 2012 has created more opportunities in the market. According to United Nations World Fertility Patterns 2015 nearly 50 per cent of the world’s population lives in countries with low levels of fertility. With in-vitro fertilization monitoring systems there would be a continuous monitoring of embryos and hence a higher success rate which would bring smiles on the faces of infertile couples. The system gives a detailed developmental information of the embryo, but does not guarantee a successful implant or pregnancy.
Competition Outlook
North America is expected to dominate the in vitro fertilization monitoring market globally, followed by Europe, owing to the high prevalence of infertility. The market in Asia Pacific region is expected to grow at a higher rate due to the evolution of higher infertility rates, and poor lifestyle conditions.
However, the market for in vitro fertilization monitoring systems in regions such as Latin America, and Middle East & Africa is estimated to show a constant growth due to the less technological advancements.
Competition Outlook
Examples of some of the key participants in the global in vitro fertilization monitoring system are Esco Micro Pte. Ltd. , Vitrolife, Parallabs Ltd. Thermofisher Scientific Inc, Genea Biomedx, Merck KGaA,, Ata International group Inc., Astec Co Ltd. , IVF Tech ApS and IKS International, among others. The companies are mainly focused on R&D to strengthen core competencies of their product portfolio.
Small-Scale Manufacturers/Providers:-
- Parallabs Ltd
- Genea Biomedx
- IVF Tech ApS
- IKS International among others
Medium-Scale Manufacturers/Providers:-
- Astec Co Ltd
- Merck KGaA
- ATA INTERNATIONAL GROUP INC
Large-Scale Manufacturers/Providers:-
- Thermofisher Scientific Inc
- Esco Micro Pte. Ltd
- Vitrolife
Key Data Points Covered in Report
The report covers exhaustive analysis on
- IVF Monitoring System Market by product type, end user and region
- Regional level market analysis of North America, Latin America, Europe, Asia Pacific, Middle East & Africa by product type, end use segments and country
- IVF Monitoring System Market Dynamics & Opportunity Assessment
- Historical Actual Market Size, 2013 - 2017
- IVF Monitoring System Market Size & Forecast 2018 - 2028
- IVF Monitoring System Market Current Trends/Issues/Challenges
- Competition & Companies involved
Research Methodology
FMI utilizes a triangulation methodology that is primarily based on experimental techniques such as patient-level data, number of procedures and capital equipment install base to obtain precise market estimations and insights on various medical devices and medical technology.
Bottom-up approach is always used to obtain insightful data for the specific country/regions. The country-specific data is again analyzed to derive data at a global level. This methodology ensures high quality and accuracy of information.
Secondary research is used at the initial phase to identify the feasibility of the target products/technology categories and its respective segments, product offerings, usage pattern as per disease indications, product installed base in target healthcare facilities, life span of a device, reimbursement scenario, adoption rate and future impact of new technologies.
Each piece of information is eventually analyzed during the entire research project, which builds a strong base for the primary research information.
Primary research participants include demand-side users such as key opinion leaders, physicians, surgeons, and supply-side providers of medical devices who provide valuable insights on trends, key treatment patterns, adoption rate, and purchasing pattern, technological development of medical devices, patient education, effectiveness of manufacturers and important strategies, pricing and competitive dynamics.
Quantitative and qualitative assessment of basic factors driving demand, economic factors/cycles and growth rates and strategies utilized by key players in the market is analyzed in detail while forecasting, in order to project Year-on-Year growth rates. These Y-o-Y growth projections are checked and aligned as per industry/product lifecycle and further utilized to develop market numbers at a holistic level.
Market Segmentation
By Product Type
- Embryoscope
- Primo Vision
- EEVA (Early Embryo Viability Assessment)
By End user
- Hospitals
- Fertility Clinics
- Research centers
By Region
- North America
- Latin America
- Europe
- Asia Pacific
- Middle East & Africa
Table of Content
- Executive Summary - In vitro Fertilization Monitoring System Market
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Inclusion and Exclusion
- Value Added Insights
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Supply Side Participants and their Roles
- Producers
- Mid-Level Participants (Traders/ Agents/ Brokers)
- Wholesalers and Distributors
- Value Added and Value Created at Node in the Supply Chain
- List of Raw Material Suppliers
- List of Existing and Potential Buyers
- Supply Side Participants and their Roles
- Pricing Analysis of In vitro Fertilization Monitoring System Market
- Pricing Analysis By Region
- Pricing Analysis By Country
- Pricing Analysis By End Use
- Pricing Analysis By Distributor
- Pricing Analysis By Application
- Value Chain Analysis
- Profit Margin Analysis
- Wholesalers and Distributors
- Retailers
- Product Portfolio Analysis
- Regulatory Landscape
- By Key Regions
- By Key Countries
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Investment Feasibility Matrix
- PESTLE Analysis
- Political Factors
- Government stability and change
- Taxation policy and incentives
- Trade tariffs and import/export regulations
- Political risk and corruption levels
- Economic Factors
- GDP growth and economic cycles
- Inflation and interest rates
- Exchange rate fluctuations
- Unemployment and consumer spending power
- Social Factors
- Demographic trends (age, population growth)
- Cultural attitudes and lifestyle changes
- Education levels and workforce skills
- Health consciousness and social values
- Technological Factors
- Rate of technological innovation
- R&D activity and automation trends
- Digital infrastructure and connectivity
- Intellectual property protection
- Legal Factors
- Employment and labor laws
- Health & safety regulations
- Consumer protection laws
- Data privacy and antitrust legislation
- Environmental Factors
- Environmental regulations and compliance
- Climate change impacts and carbon footprint
- Resource availability and sustainability practices
- Waste management and recycling initiatives
- Political Factors
- Market Background and Dynamics
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- SWOT Analysis of Global In vitro Fertilization Monitoring System Market
- Strengths
- Weaknesses
- Opportunities
- Threats
- Market Dynamics
- Global In vitro Fertilization Monitoring System Market Analysis 2020-2024 and Forecast, 2025-2035
- Historical Market Size Value (USD Mn) & Volume (Units) Analysis, 2020-2024
- Current and Future Market Size Value (USD Mn) & Volume (Units) Projections, 2025-2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Revenue Forecast Scenario, 2020-2035
- Conservative Scenario
- Likely Scenario
- Optimistic Scenario
- Global In vitro Fertilization Monitoring System Market Analysis 2020-2024 and Forecast 2025-2035, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Mn) & Volume (Units) Analysis By Application, 2020-2024
- Current and Future Market Size Value (USD Mn) & Volume (Units) Analysis and Forecast By Application, 2025-2035
- Y-o-Y Growth Trend Analysis By Application, 2020-2024
- Absolute $ Opportunity Analysis By Application, 2025-2035
- Market Attractiveness Analysis By Application, 2025-2035
- Global In vitro Fertilization Monitoring System Market Analysis 2020-2024 and Forecast 2025-2035, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Mn) & Volume (Units) Analysis By End Use, 2020-2024
- Current and Future Market Size Value (USD Mn) & Volume (Units) Analysis and Forecast By End Use, 2025-2035
- Y-o-Y Growth Trend Analysis By End Use, 2020-2024
- Absolute $ Opportunity Analysis By End Use, 2025-2035
- Market Attractiveness Analysis By End Use, 2025-2035
- Global In vitro Fertilization Monitoring System Market Analysis 2020-2024 and Forecast 2025-2035, By Region
- Introduction
- Historical Market Size Value (USD Mn) & Volume (Units) Analysis By Region, 2020-2024
- Current Market Size Value (USD Mn) & Volume (Units) Analysis and Forecast By Region, 2025-2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific & Pacific
- MEA
- Market Attractiveness Analysis By Region
- North America In vitro Fertilization Monitoring System Market Analysis 2020-2024 and Forecast 2025-2035
- Introduction
- Historical Market Size Value (USD Mn) & Volume (Units) Trend Analysis By Market Taxonomy, 2020-2024
- Current and Future Market Size Value (USD Mn) & Volume (Units) Analysis and Forecast By Market Taxonomy, 2025-2035
- By Country
- USA
- Canada
- By Application
- By End Use
- Market Attractiveness Analysis
- By Country
- By Application
- By End Use
- Market Trends
- Key Market Participants - Intensity Mapping
- Drivers and Restraints - Impact Analysis
- Country Level Analysis & Forecast
- USA In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- Canada In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- Mexico In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- USA In vitro Fertilization Monitoring System Market
- Latin America In vitro Fertilization Monitoring System Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) & Volume (Units) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) & Volume (Units) Forecast By Market Taxonomy, 2025-2035
- By Country
- Brazil
- Argentina
- Chile
- Rest of Latin America
- By Application
- By End Use
- Market Attractiveness Analysis
- By Country
- By Application
- By End Use
- Market Trends
- Key Market Participants - Intensity Mapping
- Drivers and Restraints - Impact Analysis
- Country Level Analysis & Forecast
- Brazil In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- Argentina In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- Chile In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- Rest of Latin America In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- Brazil In vitro Fertilization Monitoring System Market
- Western Europe In vitro Fertilization Monitoring System Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) & Volume (Units) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) & Volume (Units) Forecast By Market Taxonomy, 2025-2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Application
- By End Use
- Market Attractiveness Analysis
- By Country
- By Application
- By End Use
- Key Takeaways
- Market Trends
- Key Market Participants - Intensity Mapping
- Drivers and Restraints - Impact Analysis
- Country Level Analysis & Forecast
- Germany In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- UK In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- Italy In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- Spain In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- France In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- Nordic In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- BENELUX In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- Rest of Western Europe In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- Germany In vitro Fertilization Monitoring System Market
- Eastern Europe In vitro Fertilization Monitoring System Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) & Volume (Units) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) & Volume (Units) Forecast By Market Taxonomy, 2025-2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Application
- By End Use
- Market Attractiveness Analysis
- By Country
- By Application
- By End Use
- Key Takeaways
- Market Trends
- Key Market Participants - Intensity Mapping
- Drivers and Restraints - Impact Analysis
- Country Level Analysis & Forecast
- Russia In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- Poland In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- Hungary In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- Balkan & Baltic In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- Rest of Eastern Europe In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- Russia In vitro Fertilization Monitoring System Market
- East Asia In vitro Fertilization Monitoring System Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) & Volume (Units) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) & Volume (Units) Forecast By Market Taxonomy, 2025-2035
- By Country
- China
- Japan
- South Korea
- By Application
- By End Use
- Market Attractiveness Analysis
- By Country
- By Application
- By End Use
- Key Takeaways
- Market Trends
- Key Market Participants - Intensity Mapping
- Drivers and Restraints - Impact Analysis
- Country Level Analysis & Forecast
- China In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- Japan In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- South Korea In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- China In vitro Fertilization Monitoring System Market
- South Asia and Pacific In vitro Fertilization Monitoring System Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) & Volume (Units) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) & Volume (Units) Forecast By Market Taxonomy, 2025-2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Application
- By End Use
- Market Attractiveness Analysis
- By Country
- By Application
- By End Use
- Key Takeaways
- Market Trends
- Key Market Participants - Intensity Mapping
- Drivers and Restraints - Impact Analysis
- Country Level Analysis & Forecast
- India In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- ASEAN In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- Australia & New Zealand In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- Rest of South Asia and Pacific In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- India In vitro Fertilization Monitoring System Market
- MEA In vitro Fertilization Monitoring System Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) & Volume (Units) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) & Volume (Units) Forecast By Market Taxonomy, 2025-2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Application
- By End Use
- Market Attractiveness Analysis
- By Country
- By Application
- By End Use
- Key Takeaways
- Market Trends
- Key Market Participants - Intensity Mapping
- Drivers and Restraints - Impact Analysis
- Country Level Analysis & Forecast
- Kingdom of Saudi Arabia In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- Other GCC Countries In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- Turkiye In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- South Africa In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- South Africa In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- Rest of MEA In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- Rest of MEA In vitro Fertilization Monitoring System Market
- Introduction
- Market Analysis and Forecast by Market Taxonomy
- By Application
- By End Use
- Kingdom of Saudi Arabia In vitro Fertilization Monitoring System Market
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Region
- By Application
- By End Use
- Competition Analysis
- Competition Deep Dive
- Global Key Players
- Key Players by Region
- Key Players by Country
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global In vitro Fertilization Monitoring System Market Value (US$ Mn) Forecast by Region, 2020-2035
- Table 2: Global In vitro Fertilization Monitoring System Market Volume (Units) Forecast by Region, 2020-2035
- Table 3: Global In vitro Fertilization Monitoring System Market Value (US$ Mn) Forecast by Application, 2020-2035
- Table 4: Global In vitro Fertilization Monitoring System Market Volume (Units) Forecast by Application, 2020-2035
- Table 5: Global In vitro Fertilization Monitoring System Market Value (US$ Mn) Forecast by End Use, 2020-2035
- Table 6: Global In vitro Fertilization Monitoring System Market Volume (Units) Forecast by End Use, 2020-2035
- Table 7: North America In vitro Fertilization Monitoring System Market Value (US$ Mn) Forecast by Country, 2020-2035
- Table 8: North America In vitro Fertilization Monitoring System Market Volume (Units) Forecast by Country, 2020-2035
- Table 9: North America In vitro Fertilization Monitoring System Market Value (US$ Mn) Forecast by Application, 2020-2035
- Table 10: North America In vitro Fertilization Monitoring System Market Volume (Units) Forecast by Application, 2020-2035
- Table 11: North America In vitro Fertilization Monitoring System Market Value (US$ Mn) Forecast by End Use, 2020-2035
- Table 12: North America In vitro Fertilization Monitoring System Market Volume (Units) Forecast by End Use, 2020-2035
- Table 13: Latin America In vitro Fertilization Monitoring System Market Value (US$ Mn) Forecast by Country, 2020-2035
- Table 14: Latin America In vitro Fertilization Monitoring System Market Volume (Units) Forecast by Country, 2020-2035
- Table 15: Latin America In vitro Fertilization Monitoring System Market Value (US$ Mn) Forecast by Application, 2020-2035
- Table 16: Latin America In vitro Fertilization Monitoring System Market Volume (Units) Forecast by Application, 2020-2035
- Table 17: Latin America In vitro Fertilization Monitoring System Market Value (US$ Mn) Forecast by End Use, 2020-2035
- Table 18: Latin America In vitro Fertilization Monitoring System Market Volume (Units) Forecast by End Use, 2020-2035
- Table 19: Europe In vitro Fertilization Monitoring System Market Value (US$ Mn) Forecast by Country, 2020-2035
- Table 20: Europe In vitro Fertilization Monitoring System Market Volume (Units) Forecast by Country, 2020-2035
- Table 21: Europe In vitro Fertilization Monitoring System Market Value (US$ Mn) Forecast by Application, 2020-2035
- Table 22: Europe In vitro Fertilization Monitoring System Market Volume (Units) Forecast by Application, 2020-2035
- Table 23: Europe In vitro Fertilization Monitoring System Market Value (US$ Mn) Forecast by End Use, 2020-2035
- Table 24: Europe In vitro Fertilization Monitoring System Market Volume (Units) Forecast by End Use, 2020-2035
- Table 25: East Asia In vitro Fertilization Monitoring System Market Value (US$ Mn) Forecast by Country, 2020-2035
- Table 26: East Asia In vitro Fertilization Monitoring System Market Volume (Units) Forecast by Country, 2020-2035
- Table 27: East Asia In vitro Fertilization Monitoring System Market Value (US$ Mn) Forecast by Application, 2020-2035
- Table 28: East Asia In vitro Fertilization Monitoring System Market Volume (Units) Forecast by Application, 2020-2035
- Table 29: East Asia In vitro Fertilization Monitoring System Market Value (US$ Mn) Forecast by End Use, 2020-2035
- Table 30: East Asia In vitro Fertilization Monitoring System Market Volume (Units) Forecast by End Use, 2020-2035
- Table 31: South Asia In vitro Fertilization Monitoring System Market Value (US$ Mn) Forecast by Country, 2020-2035
- Table 32: South Asia In vitro Fertilization Monitoring System Market Volume (Units) Forecast by Country, 2020-2035
- Table 33: South Asia In vitro Fertilization Monitoring System Market Value (US$ Mn) Forecast by Application, 2020-2035
- Table 34: South Asia In vitro Fertilization Monitoring System Market Volume (Units) Forecast by Application, 2020-2035
- Table 35: South Asia In vitro Fertilization Monitoring System Market Value (US$ Mn) Forecast by End Use, 2020-2035
- Table 36: South Asia In vitro Fertilization Monitoring System Market Volume (Units) Forecast by End Use, 2020-2035
- Table 37: Oceania In vitro Fertilization Monitoring System Market Value (US$ Mn) Forecast by Country, 2020-2035
- Table 38: Oceania In vitro Fertilization Monitoring System Market Volume (Units) Forecast by Country, 2020-2035
- Table 39: Oceania In vitro Fertilization Monitoring System Market Value (US$ Mn) Forecast by Application, 2020-2035
- Table 40: Oceania In vitro Fertilization Monitoring System Market Volume (Units) Forecast by Application, 2020-2035
- Table 41: Oceania In vitro Fertilization Monitoring System Market Value (US$ Mn) Forecast by End Use, 2020-2035
- Table 42: Oceania In vitro Fertilization Monitoring System Market Volume (Units) Forecast by End Use, 2020-2035
- Table 43: MEA In vitro Fertilization Monitoring System Market Value (US$ Mn) Forecast by Country, 2020-2035
- Table 44: MEA In vitro Fertilization Monitoring System Market Volume (Units) Forecast by Country, 2020-2035
- Table 45: MEA In vitro Fertilization Monitoring System Market Value (US$ Mn) Forecast by Application, 2020-2035
- Table 46: MEA In vitro Fertilization Monitoring System Market Volume (Units) Forecast by Application, 2020-2035
- Table 47: MEA In vitro Fertilization Monitoring System Market Value (US$ Mn) Forecast by End Use, 2020-2035
- Table 48: MEA In vitro Fertilization Monitoring System Market Volume (Units) Forecast by End Use, 2020-2035
List of Figures
- Figure 1: Global In vitro Fertilization Monitoring System Market Value (US$ Mn) by Application, 2020-2035
- Figure 2: Global In vitro Fertilization Monitoring System Market Value (US$ Mn) by End Use, 2020-2035
- Figure 3: Global In vitro Fertilization Monitoring System Market Value (US$ Mn) by Region, 2020-2035
- Figure 4: Global In vitro Fertilization Monitoring System Market Value (US$ Mn) Analysis by Region, 2020-2035
- Figure 5: Global In vitro Fertilization Monitoring System Market Volume (Units) Analysis by Region, 2020-2035
- Figure 6: Global In vitro Fertilization Monitoring System Market Value Share (%) and BPS Analysis by Region, 2020-2035
- Figure 7: Global In vitro Fertilization Monitoring System Market Y-o-Y Growth (%) Projections by Region, 2020-2035
- Figure 8: Global In vitro Fertilization Monitoring System Market Value (US$ Mn) Analysis by Application, 2020-2035
- Figure 9: Global In vitro Fertilization Monitoring System Market Volume (Units) Analysis by Application, 2020-2035
- Figure 10: Global In vitro Fertilization Monitoring System Market Value Share (%) and BPS Analysis by Application, 2020-2035
- Figure 11: Global In vitro Fertilization Monitoring System Market Y-o-Y Growth (%) Projections by Application, 2020-2035
- Figure 12: Global In vitro Fertilization Monitoring System Market Value (US$ Mn) Analysis by End Use, 2020-2035
- Figure 13: Global In vitro Fertilization Monitoring System Market Volume (Units) Analysis by End Use, 2020-2035
- Figure 14: Global In vitro Fertilization Monitoring System Market Value Share (%) and BPS Analysis by End Use, 2020-2035
- Figure 15: Global In vitro Fertilization Monitoring System Market Y-o-Y Growth (%) Projections by End Use, 2020-2035
- Figure 16: Global In vitro Fertilization Monitoring System Market Attractiveness by Application, 2020-2035
- Figure 17: Global In vitro Fertilization Monitoring System Market Attractiveness by End Use, 2020-2035
- Figure 18: Global In vitro Fertilization Monitoring System Market Attractiveness by Region, 2020-2035
- Figure 19: North America In vitro Fertilization Monitoring System Market Value (US$ Mn) by Application, 2020-2035
- Figure 20: North America In vitro Fertilization Monitoring System Market Value (US$ Mn) by End Use, 2020-2035
- Figure 21: North America In vitro Fertilization Monitoring System Market Value (US$ Mn) by Country, 2020-2035
- Figure 22: North America In vitro Fertilization Monitoring System Market Value (US$ Mn) Analysis by Country, 2020-2035
- Figure 23: North America In vitro Fertilization Monitoring System Market Volume (Units) Analysis by Country, 2020-2035
- Figure 24: North America In vitro Fertilization Monitoring System Market Value Share (%) and BPS Analysis by Country, 2020-2035
- Figure 25: North America In vitro Fertilization Monitoring System Market Y-o-Y Growth (%) Projections by Country, 2020-2035
- Figure 26: North America In vitro Fertilization Monitoring System Market Value (US$ Mn) Analysis by Application, 2020-2035
- Figure 27: North America In vitro Fertilization Monitoring System Market Volume (Units) Analysis by Application, 2020-2035
- Figure 28: North America In vitro Fertilization Monitoring System Market Value Share (%) and BPS Analysis by Application, 2020-2035
- Figure 29: North America In vitro Fertilization Monitoring System Market Y-o-Y Growth (%) Projections by Application, 2020-2035
- Figure 30: North America In vitro Fertilization Monitoring System Market Value (US$ Mn) Analysis by End Use, 2020-2035
- Figure 31: North America In vitro Fertilization Monitoring System Market Volume (Units) Analysis by End Use, 2020-2035
- Figure 32: North America In vitro Fertilization Monitoring System Market Value Share (%) and BPS Analysis by End Use, 2020-2035
- Figure 33: North America In vitro Fertilization Monitoring System Market Y-o-Y Growth (%) Projections by End Use, 2020-2035
- Figure 34: North America In vitro Fertilization Monitoring System Market Attractiveness by Application, 2020-2035
- Figure 35: North America In vitro Fertilization Monitoring System Market Attractiveness by End Use, 2020-2035
- Figure 36: North America In vitro Fertilization Monitoring System Market Attractiveness by Country, 2020-2035
- Figure 37: Latin America In vitro Fertilization Monitoring System Market Value (US$ Mn) by Application, 2020-2035
- Figure 38: Latin America In vitro Fertilization Monitoring System Market Value (US$ Mn) by End Use, 2020-2035
- Figure 39: Latin America In vitro Fertilization Monitoring System Market Value (US$ Mn) by Country, 2020-2035
- Figure 40: Latin America In vitro Fertilization Monitoring System Market Value (US$ Mn) Analysis by Country, 2020-2035
- Figure 41: Latin America In vitro Fertilization Monitoring System Market Volume (Units) Analysis by Country, 2020-2035
- Figure 42: Latin America In vitro Fertilization Monitoring System Market Value Share (%) and BPS Analysis by Country, 2020-2035
- Figure 43: Latin America In vitro Fertilization Monitoring System Market Y-o-Y Growth (%) Projections by Country, 2020-2035
- Figure 44: Latin America In vitro Fertilization Monitoring System Market Value (US$ Mn) Analysis by Application, 2020-2035
- Figure 45: Latin America In vitro Fertilization Monitoring System Market Volume (Units) Analysis by Application, 2020-2035
- Figure 46: Latin America In vitro Fertilization Monitoring System Market Value Share (%) and BPS Analysis by Application, 2020-2035
- Figure 47: Latin America In vitro Fertilization Monitoring System Market Y-o-Y Growth (%) Projections by Application, 2020-2035
- Figure 48: Latin America In vitro Fertilization Monitoring System Market Value (US$ Mn) Analysis by End Use, 2020-2035
- Figure 49: Latin America In vitro Fertilization Monitoring System Market Volume (Units) Analysis by End Use, 2020-2035
- Figure 50: Latin America In vitro Fertilization Monitoring System Market Value Share (%) and BPS Analysis by End Use, 2020-2035
- Figure 51: Latin America In vitro Fertilization Monitoring System Market Y-o-Y Growth (%) Projections by End Use, 2020-2035
- Figure 52: Latin America In vitro Fertilization Monitoring System Market Attractiveness by Application, 2020-2035
- Figure 53: Latin America In vitro Fertilization Monitoring System Market Attractiveness by End Use, 2020-2035
- Figure 54: Latin America In vitro Fertilization Monitoring System Market Attractiveness by Country, 2020-2035
- Figure 55: Europe In vitro Fertilization Monitoring System Market Value (US$ Mn) by Application, 2020-2035
- Figure 56: Europe In vitro Fertilization Monitoring System Market Value (US$ Mn) by End Use, 2020-2035
- Figure 57: Europe In vitro Fertilization Monitoring System Market Value (US$ Mn) by Country, 2020-2035
- Figure 58: Europe In vitro Fertilization Monitoring System Market Value (US$ Mn) Analysis by Country, 2020-2035
- Figure 59: Europe In vitro Fertilization Monitoring System Market Volume (Units) Analysis by Country, 2020-2035
- Figure 60: Europe In vitro Fertilization Monitoring System Market Value Share (%) and BPS Analysis by Country, 2020-2035
- Figure 61: Europe In vitro Fertilization Monitoring System Market Y-o-Y Growth (%) Projections by Country, 2020-2035
- Figure 62: Europe In vitro Fertilization Monitoring System Market Value (US$ Mn) Analysis by Application, 2020-2035
- Figure 63: Europe In vitro Fertilization Monitoring System Market Volume (Units) Analysis by Application, 2020-2035
- Figure 64: Europe In vitro Fertilization Monitoring System Market Value Share (%) and BPS Analysis by Application, 2020-2035
- Figure 65: Europe In vitro Fertilization Monitoring System Market Y-o-Y Growth (%) Projections by Application, 2020-2035
- Figure 66: Europe In vitro Fertilization Monitoring System Market Value (US$ Mn) Analysis by End Use, 2020-2035
- Figure 67: Europe In vitro Fertilization Monitoring System Market Volume (Units) Analysis by End Use, 2020-2035
- Figure 68: Europe In vitro Fertilization Monitoring System Market Value Share (%) and BPS Analysis by End Use, 2020-2035
- Figure 69: Europe In vitro Fertilization Monitoring System Market Y-o-Y Growth (%) Projections by End Use, 2020-2035
- Figure 70: Europe In vitro Fertilization Monitoring System Market Attractiveness by Application, 2020-2035
- Figure 71: Europe In vitro Fertilization Monitoring System Market Attractiveness by End Use, 2020-2035
- Figure 72: Europe In vitro Fertilization Monitoring System Market Attractiveness by Country, 2020-2035
- Figure 73: East Asia In vitro Fertilization Monitoring System Market Value (US$ Mn) by Application, 2020-2035
- Figure 74: East Asia In vitro Fertilization Monitoring System Market Value (US$ Mn) by End Use, 2020-2035
- Figure 75: East Asia In vitro Fertilization Monitoring System Market Value (US$ Mn) by Country, 2020-2035
- Figure 76: East Asia In vitro Fertilization Monitoring System Market Value (US$ Mn) Analysis by Country, 2020-2035
- Figure 77: East Asia In vitro Fertilization Monitoring System Market Volume (Units) Analysis by Country, 2020-2035
- Figure 78: East Asia In vitro Fertilization Monitoring System Market Value Share (%) and BPS Analysis by Country, 2020-2035
- Figure 79: East Asia In vitro Fertilization Monitoring System Market Y-o-Y Growth (%) Projections by Country, 2020-2035
- Figure 80: East Asia In vitro Fertilization Monitoring System Market Value (US$ Mn) Analysis by Application, 2020-2035
- Figure 81: East Asia In vitro Fertilization Monitoring System Market Volume (Units) Analysis by Application, 2020-2035
- Figure 82: East Asia In vitro Fertilization Monitoring System Market Value Share (%) and BPS Analysis by Application, 2020-2035
- Figure 83: East Asia In vitro Fertilization Monitoring System Market Y-o-Y Growth (%) Projections by Application, 2020-2035
- Figure 84: East Asia In vitro Fertilization Monitoring System Market Value (US$ Mn) Analysis by End Use, 2020-2035
- Figure 85: East Asia In vitro Fertilization Monitoring System Market Volume (Units) Analysis by End Use, 2020-2035
- Figure 86: East Asia In vitro Fertilization Monitoring System Market Value Share (%) and BPS Analysis by End Use, 2020-2035
- Figure 87: East Asia In vitro Fertilization Monitoring System Market Y-o-Y Growth (%) Projections by End Use, 2020-2035
- Figure 88: East Asia In vitro Fertilization Monitoring System Market Attractiveness by Application, 2020-2035
- Figure 89: East Asia In vitro Fertilization Monitoring System Market Attractiveness by End Use, 2020-2035
- Figure 90: East Asia In vitro Fertilization Monitoring System Market Attractiveness by Country, 2020-2035
- Figure 91: South Asia In vitro Fertilization Monitoring System Market Value (US$ Mn) by Application, 2020-2035
- Figure 92: South Asia In vitro Fertilization Monitoring System Market Value (US$ Mn) by End Use, 2020-2035
- Figure 93: South Asia In vitro Fertilization Monitoring System Market Value (US$ Mn) by Country, 2020-2035
- Figure 94: South Asia In vitro Fertilization Monitoring System Market Value (US$ Mn) Analysis by Country, 2020-2035
- Figure 95: South Asia In vitro Fertilization Monitoring System Market Volume (Units) Analysis by Country, 2020-2035
- Figure 96: South Asia In vitro Fertilization Monitoring System Market Value Share (%) and BPS Analysis by Country, 2020-2035
- Figure 97: South Asia In vitro Fertilization Monitoring System Market Y-o-Y Growth (%) Projections by Country, 2020-2035
- Figure 98: South Asia In vitro Fertilization Monitoring System Market Value (US$ Mn) Analysis by Application, 2020-2035
- Figure 99: South Asia In vitro Fertilization Monitoring System Market Volume (Units) Analysis by Application, 2020-2035
- Figure 100: South Asia In vitro Fertilization Monitoring System Market Value Share (%) and BPS Analysis by Application, 2020-2035
- Figure 101: South Asia In vitro Fertilization Monitoring System Market Y-o-Y Growth (%) Projections by Application, 2020-2035
- Figure 102: South Asia In vitro Fertilization Monitoring System Market Value (US$ Mn) Analysis by End Use, 2020-2035
- Figure 103: South Asia In vitro Fertilization Monitoring System Market Volume (Units) Analysis by End Use, 2020-2035
- Figure 104: South Asia In vitro Fertilization Monitoring System Market Value Share (%) and BPS Analysis by End Use, 2020-2035
- Figure 105: South Asia In vitro Fertilization Monitoring System Market Y-o-Y Growth (%) Projections by End Use, 2020-2035
- Figure 106: South Asia In vitro Fertilization Monitoring System Market Attractiveness by Application, 2020-2035
- Figure 107: South Asia In vitro Fertilization Monitoring System Market Attractiveness by End Use, 2020-2035
- Figure 108: South Asia In vitro Fertilization Monitoring System Market Attractiveness by Country, 2020-2035
- Figure 109: Oceania In vitro Fertilization Monitoring System Market Value (US$ Mn) by Application, 2020-2035
- Figure 110: Oceania In vitro Fertilization Monitoring System Market Value (US$ Mn) by End Use, 2020-2035
- Figure 111: Oceania In vitro Fertilization Monitoring System Market Value (US$ Mn) by Country, 2020-2035
- Figure 112: Oceania In vitro Fertilization Monitoring System Market Value (US$ Mn) Analysis by Country, 2020-2035
- Figure 113: Oceania In vitro Fertilization Monitoring System Market Volume (Units) Analysis by Country, 2020-2035
- Figure 114: Oceania In vitro Fertilization Monitoring System Market Value Share (%) and BPS Analysis by Country, 2020-2035
- Figure 115: Oceania In vitro Fertilization Monitoring System Market Y-o-Y Growth (%) Projections by Country, 2020-2035
- Figure 116: Oceania In vitro Fertilization Monitoring System Market Value (US$ Mn) Analysis by Application, 2020-2035
- Figure 117: Oceania In vitro Fertilization Monitoring System Market Volume (Units) Analysis by Application, 2020-2035
- Figure 118: Oceania In vitro Fertilization Monitoring System Market Value Share (%) and BPS Analysis by Application, 2020-2035
- Figure 119: Oceania In vitro Fertilization Monitoring System Market Y-o-Y Growth (%) Projections by Application, 2020-2035
- Figure 120: Oceania In vitro Fertilization Monitoring System Market Value (US$ Mn) Analysis by End Use, 2020-2035
- Figure 121: Oceania In vitro Fertilization Monitoring System Market Volume (Units) Analysis by End Use, 2020-2035
- Figure 122: Oceania In vitro Fertilization Monitoring System Market Value Share (%) and BPS Analysis by End Use, 2020-2035
- Figure 123: Oceania In vitro Fertilization Monitoring System Market Y-o-Y Growth (%) Projections by End Use, 2020-2035
- Figure 124: Oceania In vitro Fertilization Monitoring System Market Attractiveness by Application, 2020-2035
- Figure 125: Oceania In vitro Fertilization Monitoring System Market Attractiveness by End Use, 2020-2035
- Figure 126: Oceania In vitro Fertilization Monitoring System Market Attractiveness by Country, 2020-2035
- Figure 127: MEA In vitro Fertilization Monitoring System Market Value (US$ Mn) by Application, 2020-2035
- Figure 128: MEA In vitro Fertilization Monitoring System Market Value (US$ Mn) by End Use, 2020-2035
- Figure 129: MEA In vitro Fertilization Monitoring System Market Value (US$ Mn) by Country, 2020-2035
- Figure 130: MEA In vitro Fertilization Monitoring System Market Value (US$ Mn) Analysis by Country, 2020-2035
- Figure 131: MEA In vitro Fertilization Monitoring System Market Volume (Units) Analysis by Country, 2020-2035
- Figure 132: MEA In vitro Fertilization Monitoring System Market Value Share (%) and BPS Analysis by Country, 2020-2035
- Figure 133: MEA In vitro Fertilization Monitoring System Market Y-o-Y Growth (%) Projections by Country, 2020-2035
- Figure 134: MEA In vitro Fertilization Monitoring System Market Value (US$ Mn) Analysis by Application, 2020-2035
- Figure 135: MEA In vitro Fertilization Monitoring System Market Volume (Units) Analysis by Application, 2020-2035
- Figure 136: MEA In vitro Fertilization Monitoring System Market Value Share (%) and BPS Analysis by Application, 2020-2035
- Figure 137: MEA In vitro Fertilization Monitoring System Market Y-o-Y Growth (%) Projections by Application, 2020-2035
- Figure 138: MEA In vitro Fertilization Monitoring System Market Value (US$ Mn) Analysis by End Use, 2020-2035
- Figure 139: MEA In vitro Fertilization Monitoring System Market Volume (Units) Analysis by End Use, 2020-2035
- Figure 140: MEA In vitro Fertilization Monitoring System Market Value Share (%) and BPS Analysis by End Use, 2020-2035
- Figure 141: MEA In vitro Fertilization Monitoring System Market Y-o-Y Growth (%) Projections by End Use, 2020-2035
- Figure 142: MEA In vitro Fertilization Monitoring System Market Attractiveness by Application, 2020-2035
- Figure 143: MEA In vitro Fertilization Monitoring System Market Attractiveness by End Use, 2020-2035
- Figure 144: MEA In vitro Fertilization Monitoring System Market Attractiveness by Country, 2020-2035
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

