In-vitro Human Models Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
In-vitro Human Models Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
In-vitro Human Models Market Forecast and Outlook 2026 to 2036
The in-vitro human models market is likely to value at USD 2,123.8 million in 2026 to reach USD 8,443.7 million by 2036, at a CAGR of 14.8%. Adoption upside is driven by pressure to reduce late-stage failure, shorten R&D timelines, and generate translational data that conventional animal models often fail to deliver. At the same time, execution risk persists due to variability in model performance, limited standardization, and lengthy internal validation cycles within large pharmaceutical organizations.
Economic sensitivity is moderate to high while leading pharma can absorb premium pricing, smaller biotechs and academic labs remain constrained by budget cycles and grant dependency. Regulatory acceptance is improving but remains inconsistent, creating adoption friction for formal safety submissions. Between 2026 and 2036, sustained value creation will depend less on technological novelty and more on proof of predictive relevance, workflow integration, and the ability of suppliers to support scalable, repeatable use across discovery portfolios rather than isolated pilot programs.
Quick Stats for In-vitro Human Models Market
- In-vitro Human Models Market Value (2026): USD 2,123.8 Million
- In-vitro Human Models Market Forecast Value (2036): USD 8,443.7 Million
- In-vitro Human Models Market Forecast CAGR (2026-2036): 14.8%
- Leading Product Type in In-vitro Human Models Market: Organ-on-Chip Systems (46% share)
- Key Growth Regions in In-vitro Human Models Market: North America, Europe, Asia Pacific, Latin America, Middle East & Africa
- Top Key Players in In-vitro Human Models Market: Emulate, CN Bio, TissUse, Mimetas, InSphero

In-vitro Human Models Market Key Takeaways
| Metric | Value |
|---|---|
| In-vitro Human Models Market Value (2026) | USD 2,123.8 Million |
| In-vitro Human Models Market Forecast Value (2036) | USD 8,443.7 Million |
| In-vitro Human Models Market Forecast CAGR (2026-2036) | 14.8% |
How Are Predictive Validity and R&D Efficiency Reprioritizing In-vitro Human Models?
The in-vitro human models market is accelerating as drug developers seek earlier, more reliable signals of human biology to reduce late-stage attrition. Advanced 2D/3D cultures, organoids, and organ-on-chip systems are being adopted to interrogate human-specific toxicity, efficacy, and mechanism of action before costly in-vivo or clinical work. For suppliers, value creation increasingly hinges on demonstrable predictive validity how well model outputs correlate with human outcomes rather than model novelty alone.
Commercial adoption is shaped by integration and throughput. Scaling programs often starts with 3D cell culture setups that can be standardized across sites before moving into higher-complexity microphysiological formats. Pharma and biotech teams favor platforms that fit existing discovery workflows, accept standard assay formats, and scale across programs. Models that support multiplexed readouts, automation, and compatibility with imaging and omics pipelines are gaining preference. Adoption accelerates when automated culture systems reduce manual variability and keep handling steps consistent across high-volume discovery workflows. Vendors offering standardized protocols, quality controls, and application-ready kits shorten internal validation cycles and improve uptake across multi-site R&D organizations and CROs. Procurement also tightens around cell culture media consistency, since media composition and lot control can materially shift phenotype stability and assay readouts.
Regulatory engagement and partnership models are further strengthening demand. As agencies encourage more human-relevant data packages, companies are investing in in-vitro models to support IND-enabling studies and risk assessments. Co-development agreements, service-plus-platform offerings, and data-sharing partnerships are becoming common, aligning incentives around decision impact rather than unit sales. For providers, competitive advantage increasingly depends on proof of relevance, ease of deployment, and the ability to support customers from pilot validation through portfolio-wide adoption positioning in-vitro human models as a core productivity lever in modern drug development.
How Is the In-Vitro Human Models Market Structured Across Key Segments?
The in-vitro human models market is segmented by product and application, reflecting how human-relevant platforms are adopted across preclinical and translational research workflows. By product, organ-on-chip systems lead the market, as they replicate human organ functions with higher physiological accuracy compared to conventional cell cultures. Other products include 3D tissue models and stem-cell derived models, which support broader biological complexity and disease relevance. Work on 3D bioprinted human tissues is expanding where labs want closer anatomical fidelity for screening and response mapping beyond conventional 3D cultures. By application, drug discovery represents the largest demand area, while toxicity testing and disease modeling continue to gain traction as alternatives to animal-based studies across pharmaceutical and academic research environments.
Why Do Organ-On-Chip Systems Lead Product Adoption in the In-Vitro Human Models Market?

Organ-on-chip systems account for 46% share of the in-vitro human models market because they closely mimic human tissue interfaces, fluid flow, and mechanical forces. These systems enable researchers to study drug response, metabolism, and toxicity under conditions that resemble human physiology. Pharmaceutical companies adopt organ-on-chip platforms to improve target validation and reduce late-stage drug failure. Integration with microfluidics and real-time sensing supports high-quality, reproducible data. Research institutions value their ability to model complex organ interactions. For technology providers, organ-on-chip systems offer premium pricing and long-term collaboration opportunities, explaining their leading position.
Why Does Drug Discovery Dominate Application Demand In The In-Vitro Human Models Market?

Drug discovery holds 54% share of the in-vitro human models market because early-stage screening and validation require predictive human-relevant data. In-vitro models help identify efficacy signals and safety risks before costly clinical development. Pharmaceutical and biotechnology companies use these models to prioritize compounds, optimize lead candidates, and reduce dependence on animal testing. High attrition rates in drug pipelines increase demand for more accurate preclinical tools. Continuous growth in small-molecule and biologics research sustains application volume. For model suppliers, drug discovery programs offer repeat usage and integration across multiple pipeline stages, reinforcing dominance of this application segment.
What are The Key Dynamics of the In-Vitro Human Models Market?
The in-vitro human models market is shaped by rising demand from pharmaceutical, biotechnology, and academic research sectors for predictive, human-relevant test systems that reduce reliance on traditional animal models. In-vitro human models-including organoids, tissue chips, 3D cell cultures, and engineered microphysiological systems-are used for drug discovery, toxicity screening, disease modeling, and mechanistic studies. Many teams still use cytotoxicity assays as a fast gating layer before moving candidates into higher-cost organ-level systems. For platform providers and material suppliers, the ability to deliver reproducible biological responses, scalability for high-throughput screening, and interoperability with automation and imaging systems are key factors influencing adoption. Where budgets allow, high throughput screening is paired with standardized plates, automation, and analytics to reduce cycle time per candidate series. Regulatory interest in alternative testing strategies and pressure to improve translational success strengthen the value proposition of human in-vitro models.
How are Innovation and Technology Trends Shaping Adoption in This Market?
Innovation and technology trends are rapidly shaping the in-vitro human models market through advancements in tissue engineering, microfluidics, and biofabrication. Adoption of 3D bioprinting is rising where teams need repeatable tissue constructs for model-building, assay development, and longer-run phenotype stability checks. Integration of microphysiological systems and organ-on-chip platforms enables dynamic fluid flow, tissue-tissue interfaces, and multi-organ connectivity to mimic in vivo human physiology. Stem-cell-derived organoids provide patient-specific disease models that support precision medicine research. For engineered tissues that must hold structure over time, scaffold technology choices increasingly influence reproducibility and readout consistency across programs. Automated imaging, high-content analysis, and AI-driven data interpretation enhance throughput and predictive power. Image-based decision workflows are tightening the link between phenotype and endpoint selection, which is why high content screening is being pulled closer to early model validation. For suppliers, delivering modular platforms that integrate with robotic liquid handling and analytics pipelines reduces barriers for adoption in centralized laboratories, CROs, and pharmaceutical R&D units.
Why do Standardization, Validation, and Regulatory Challenges Restrain Market Growth?
Standardization, validation, and regulatory challenges restrain growth of the in-vitro human models market because diverse model formats and biological variability create hurdles for reproducibility and cross-study comparability. Lack of universally accepted performance benchmarks and reference standards complicates qualification by pharmaceutical and toxicology labs that require confidence in predictive validity. Regulatory agencies are still evolving frameworks for accepting non-animal predictive models for safety assessment, which can delay integration into formal submission packages. These constraints increase qualification timelines, raise development costs, and temper adoption among conservative research organizations that prioritize validated, regulatory-accepted methods.
What is the Country-Level Overview for the In-vitro Human Models Market?
The in-vitro human models market is expanding as pharmaceutical companies, biotechnology firms, and research institutions increasingly adopt human-relevant testing platforms for drug discovery, toxicology, and disease modeling. Technologies such as organoids, organ-on-chip systems, and advanced 3D cell cultures are gaining traction due to their ability to improve translational accuracy and reduce late-stage development risk. Growth varies by country based on R&D investment intensity, regulatory acceptance of alternative models, availability of skilled researchers, and integration into preclinical workflows. Mature research markets emphasize validation, reproducibility, and scientific rigor, while emerging regions focus on expanding access, affordability, and research capacity. Market value is driven by demand for predictive models, collaboration between academia and industry, and increasing global research outsourcing.
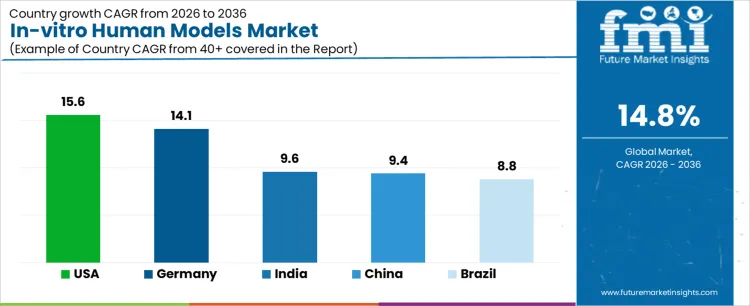
| Country | CAGR (%) |
|---|---|
| United States | 15.6 |
| Germany | 14.1 |
| India | 9.6 |
| China | 9.4 |
| Brazil | 8.8 |
How is the United States Leading Adoption of In-vitro Human Models?
The in-vitro human models market in the United States is expanding at a CAGR of 15.6% during 2026-2036, driven by strong pharmaceutical R&D spending and advanced biomedical research infrastructure. USA companies widely adopt organoids, organ-on-chip systems, and 3D cell culture models to improve translational accuracy and reduce late-stage drug failure. Adoption is led by large pharmaceutical firms, biotech startups, and academic research centers working in close collaboration. Buyers emphasize physiological relevance, reproducibility, and compatibility with high-throughput screening workflows.
Regulatory engagement and early validation for drug discovery and toxicology studies play an important role in purchasing decisions. Procurement cycles prioritize scientific validation, vendor credibility, and integration with existing laboratory platforms. Market value is driven by demand for predictive preclinical models across oncology, neurology, and rare disease research. The United States represents an innovation-driven market where growth is shaped by research intensity, technology maturity, and strong funding ecosystems.
Why Is Brazil Gradually Expanding Use of In-vitro Human Models?
Brazil’s in-vitro human models market is growing at a CAGR of 8.8% during 2026-2036, supported by increasing research activity in academic institutions and public health laboratories. Adoption is primarily focused on disease modeling, toxicology studies, and early-stage drug screening rather than full-scale pharmaceutical development. Buyers prioritize affordability, ease of use, and access to technical training. Partnerships with international suppliers and research collaborations help accelerate technology transfer. Procurement decisions are often influenced by grant funding, institutional budgets, and distributor support. Use of advanced models such as organoids is increasing gradually, particularly in cancer and infectious disease research. Market value is driven by expanding research participation and growing awareness of human-relevant testing methods. Brazil represents an emerging market where growth depends on education, funding availability, and incremental integration of advanced in-vitro platforms into research workflows.
Why Is China Rapidly Scaling the In-vitro Human Models Market?
China’s in-vitro human models market is expanding at a CAGR of 9.4% during 2026-2036, driven by strong investment in life sciences and growing domestic drug development capabilities. Pharmaceutical companies and research institutes increasingly deploy organoids and microphysiological systems to accelerate preclinical research and reduce development timelines. Buyers focus on scalability, throughput capacity, and alignment with local research priorities. Government-backed research programs and infrastructure development support widespread adoption. Procurement favors suppliers with local manufacturing, strong technical support, and competitive pricing. Qualification cycles are relatively short, enabling rapid experimentation and deployment. Market value is driven by high research volumes, expanding biotech pipelines, and integration of advanced models into routine drug screening. China represents a scale-oriented market where success depends on capacity, localization, and alignment with national research objectives.
Why Is India emerging As a High-Growth Market for In-vitro Human Models?
India’s in-vitro human models market is projected to grow at a CAGR of 9.6% during 2026-2036, supported by expanding pharmaceutical R&D, contract research activity, and academic research output. Adoption is led by CROs, biotech startups, and research institutes seeking cost-effective alternatives to animal testing. Buyers prioritize affordability, flexibility, and ease of integration into existing laboratory setups. Use of 3D cell culture and organoid systems is increasing, particularly in oncology and toxicity studies. Procurement cycles are relatively short, enabling faster adoption when funding is available. Market value is driven by growing participation in global research programs and outsourcing projects. India represents a fast-growing market where scalability, cost efficiency, and training support shape adoption of advanced in-vitro platforms.
How Is Germany Advancing High-Precision In-vitro Human Model Adoption?
Germany’s in-vitro human models market is growing at a CAGR of 14.1% during 2026-2036, characterized by strong emphasis on scientific rigor and model validation. Research institutions and pharmaceutical companies adopt advanced in-vitro systems to improve predictability in drug discovery and toxicology. Buyers prioritize reproducibility, detailed characterization, and compliance with research standards. Adoption is methodical, with extensive pilot studies before full-scale deployment. Procurement decisions involve scientific committees and long evaluation timelines. Market value is driven by demand for high-precision models in oncology, neurology, and regulatory research. Germany represents a quality-driven market where growth is shaped by validation depth, regulatory alignment, and long-term research collaboration rather than rapid expansion.
What Defines Competition in the In-Vitro Human Models Market?
Competition in the in-vitro human models market is anchored in biological fidelity, scalability, and documented predictive performance that matter to drug developers, toxicologists, and regulatory scientists. Emulate positions its organ-on-a-chip platforms with official materials that highlight multicellular tissue architecture and dynamic fluid flow, offering physiologically relevant responses that align with human biology more closely than conventional 2D cultures. Product literature emphasizes modular systems that support liver, lung, gut, and other organ models with validated endpoints suitable for ADME/Tox screening.
CN Bio competes through microphysiological systems designed for liver and multiorgan interactions. Brochures detail controlled microenvironments with real-time functional readouts, backed by comparative studies showing enhanced prediction of human responses. Its platform appeal centers on integration with standard laboratory automation and imaging systems, facilitating adoption in medium-throughput research workflows. TissUse differentiates with multi-organ chip platforms presented in official documentation that underscores co-culture stability and inter-tissue communication, appealing to complex disease modeling and multi-organ toxicology assessments.
Emerging models emphasize 3D architecture and tissue complexity. Mimetas markets its microfluidic 3D culture systems with product guides highlighting high-throughput compatibility and consistent tissue formation across chips. InSphero offers scaffold-free spheroid and microtissue platforms documented for reproducible function in oncology, liver toxicity, and metabolism studies. Official materials stress functional endpoints, assay compatibility, and data reproducibility.
Others in the market include niche platform providers and specialized human model developers that supply disease-specific tissues, co-culture systems, or bespoke assay modules. Across all suppliers, competitive differentiation is driven by brochure-verified biological relevance, scalability, and integration with existing workflows rather than pricing alone.
Key Players in In-vitro Human Models Market
- Emulate
- CN Bio
- TissUse
- Mimetas
- InSphero
- Others
Scope of Report
| Attribute | Description |
|---|---|
| Quantitative Unit | USD Million |
| Products | Organ-on-Chip Systems, 3D Tissue Models, Stem-Cell Derived Models |
| Applications | Drug Discovery, Toxicity Testing, Disease Modeling |
| Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Rest of Asia Pacific, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, Rest of Europe, United States, Canada, Mexico, Brazil, Chile, Rest of Latin America, Kingdom of Saudi Arabia, Other GCC Countries, Turkey, South Africa, Other African Union, Rest of Middle East & Africa |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Key Companies Profiled | Emulate, CN Bio, TissUse GmbH, Mimetas B.V., InSphero AG, Others |
| Additional Attributes | Dollar sales by product type and application are evaluated across preclinical research and advanced biomedical testing workflows. The report includes country-level demand assessment, growth projections for 2026-2036, analysis of regulatory acceptance and animal testing replacement trends, evaluation of model complexity and scalability, competitive benchmarking of technology providers, and assessment of adoption across pharmaceutical, biotechnology, and academic research institutions. |
In-vitro Human Models Market by Key Segments
Products
- Organ-on-chip systems
- 3D tissue models
- Stem-cell derived models
Applications
- Drug discovery
- Toxicity testing
- Disease modeling
Region
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- U.S. Food and Drug Administration. (2024). Advancing alternatives to animal testing: FDA activities on microphysiological systems and in‑vitro models. FDA Scientific Publications.
- National Institutes of Health. (2023). Tissue chips in space and human microphysiological systems research program. NIH NCATS.
- OECD. (2024). Guidance document on the use of new approach methodologies for toxicology and chemical risk assessment. Organisation for Economic Co‑operation and Development.
Frequently Asked Questions
How big is the in-vitro human models market in 2026?
The global in-vitro human models market is estimated to be valued at USD 2,123.8 million in 2026.
What will be the size of in-vitro human models market in 2036?
The market size for the in-vitro human models market is projected to reach USD 8,443.7 million by 2036.
How much will be the in-vitro human models market growth between 2026 and 2036?
The in-vitro human models market is expected to grow at a 14.8% CAGR between 2026 and 2036.
What are the key product types in the in-vitro human models market?
The key product types in in-vitro human models market are organ-on-chip systems, 3d tissue models and stem-cell derived models.
Which applications segment to contribute significant share in the in-vitro human models market in 2026?
In terms of applications, drug discovery segment to command 54.0% share in the in-vitro human models market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Products
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Products , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Products , 2026 to 2036
- Organ-on-chip systems
- 3D tissue models
- Stem-cell derived models
- Organ-on-chip systems
- Y to o to Y Growth Trend Analysis By Products , 2021 to 2025
- Absolute $ Opportunity Analysis By Products , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Applications
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Applications, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Applications, 2026 to 2036
- Drug discovery
- Toxicity testing
- Disease modeling
- Drug discovery
- Y to o to Y Growth Trend Analysis By Applications, 2021 to 2025
- Absolute $ Opportunity Analysis By Applications, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Products
- By Applications
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Products
- By Applications
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Products
- By Applications
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Products
- By Applications
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Products
- By Applications
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Products
- By Applications
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Products
- By Applications
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Products
- By Applications
- Competition Analysis
- Competition Deep Dive
- Emulate
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- CN Bio
- TissUse
- Mimetas
- InSphero
- Others
- Emulate
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Applications, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 4: Global Market Y to o to Y Growth Comparison by Products , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Products
- Figure 6: Global Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 7: Global Market Y to o to Y Growth Comparison by Applications, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Applications
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y to o to Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 21: North America Market Y to o to Y Growth Comparison by Products , 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Products
- Figure 23: North America Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 24: North America Market Y to o to Y Growth Comparison by Applications, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Applications
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 28: Latin America Market Y to o to Y Growth Comparison by Products , 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Products
- Figure 30: Latin America Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 31: Latin America Market Y to o to Y Growth Comparison by Applications, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by Applications
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 35: Western Europe Market Y to o to Y Growth Comparison by Products , 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Products
- Figure 37: Western Europe Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 38: Western Europe Market Y to o to Y Growth Comparison by Applications, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by Applications
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 42: Eastern Europe Market Y to o to Y Growth Comparison by Products , 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Products
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 45: Eastern Europe Market Y to o to Y Growth Comparison by Applications, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Applications
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 49: East Asia Market Y to o to Y Growth Comparison by Products , 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Products
- Figure 51: East Asia Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 52: East Asia Market Y to o to Y Growth Comparison by Applications, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by Applications
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y to o to Y Growth Comparison by Products , 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Products
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y to o to Y Growth Comparison by Applications, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Applications
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 63: Middle East & Africa Market Y to o to Y Growth Comparison by Products , 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Products
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 66: Middle East & Africa Market Y to o to Y Growth Comparison by Applications, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Applications
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

