Intraosseous Devices Market
Intraosseous Devices Market Analysis - Trends & Forecast 2025 to 2035
Historical Data Covered: 2015 to 2023 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2026 to 2035
Intraosseous Devices Industry Outlook from 2025 to 2035
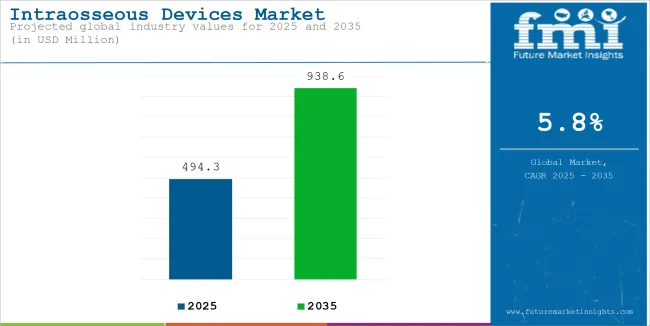
The global sales of intraosseous devices are estimated to be worth USD 494.3 million in 2025 and are anticipated to reach a value of USD 938.6 million by 2035. Sales are projected to rise at a CAGR of 5.8% over the forecast period between 2025 and 2035. The revenue generated by intraosseous devices in 2024 was USD 467.2 million.
The IO devices market has registered significant growth owing to the increased incidence of traumatic injuries and medical emergencies where quick vascular access is necessary. Trauma is the largest cause of mortality worldwide, accounting for over 4.4 million deaths every year from injuries.
In these critical scenarios, it becomes very difficult to get IV access due to blood loss or collapsed veins. IO devices provide the most life-saving alternative by allowing access to the vascular system through the bone marrow, which ensures prompt administration of drugs and fluid.
Their practical value in high-pressure emergency situations has rendered them indispensable in both pre-hospital care and the emergency department in hospitals where speed and reliability are the most important.
Global Intraosseous Devices Industry Analysis
| Attributes | Key Insights |
|---|---|
| Historical Size, 2024 | USD 467.2 million |
| Estimated Size, 2025 | USD 494.3 million |
| Projected Size, 2035 | USD 938.6 million |
| Value-based CAGR (2025 to 2035) | 5.8% |
Another significant driver in the market is the increasing adoption of IO devices in military and tactical medical applications. In battlefield settings, rapid intervention is often necessary for wounded soldiers.
Powered drill and impact-driven IO devices have gained preference due to their speed, reliability, and ease of use under stressful conditions. These technologies ensure faster fluid administration during critical moments, improving survival rates in combat zones.
Technological advancements have further strengthened market growth. Manufacturers are now developing smart IO devices with real-time pressure monitoring, LED guidance systems, and enhanced safety mechanisms.
These innovations help reduce procedural errors, particularly in chaotic trauma scenarios, making powered and automatic IO systems safer and more effective.
Additionally, rising collaborations between medical device companies and defense forces have become a key trend driving market expansion. Many defense agencies are forming strategic partnerships for the procurement of advanced IO devices, especially impact-driven and powered drill models suited for field trauma care.
This surge in tactical medical preparedness has further propelled the market, positioning IO devices as critical tools in emergency and battlefield medical interventions.
Semi Annual Market Update
A comparative analysis of fluctuations in compound annual growth rate (CAGR) for the intraosseous devices industry outlook between 2024 and 2025 on a six-month basis is shown below.
By this examination, major variations in the performance of these markets are brought to light, and trends of revenue generation are captured hence offering stakeholders useful ideas on how to carry on with the market’s growth path in any other given year. January through June covers the first part of the year called half1 (H1), while half2 (H2) represents July to December.
The table below compares the compound annual growth rate (CAGR) for the global intraosseous devices industry analysis from 2024 to 2025 during the first half of the year.
This overview highlights key changes and trends in revenue growth, offering valuable insights into market dynamics. H1 covers January to June, while H2 spans July to December.
In the first half (H1) of the decade from 2024 to 2034, the business is predicted to surge at a CAGR of 6.5%, followed by a slightly lower growth rate of 6.2% in the second half (H2) of the same decade.
| Particular | Value CAGR |
|---|---|
| H1 | 6.5% (2024 to 2034) |
| H2 | 6.2% (2024 to 2034) |
| H1 | 5.8% (2025 to 2035) |
| H2 | 5.3% (2025 to 2035) |
Moving into the subsequent period, from H1 2025 to H2 2035, the CAGR is projected to decrease slightly to 5.8% in the first half and projected to lower at 5.3% in the second half. In the first half (H1) the market witnessed a decrease of 70 BPS while in the second half (H2), the market witnessed a decrease of 90 BPS.
Key Industry Highlights
Rising Prevalence of Cardiovascular Events Driving the Market
The growing incidence of cardiovascular events has significantly improved the demand for intraosseous devices. In the United States alone, there are near about 356,000 cases of out-of-hospital sudden cardiac arrest in a year with rapid intervention only increasing the rate of survival chances.
In emergencies of cardiac problems, immediate access to the vessels for the distribution of drugs may be necessary where delays in saving medications can severely bring down the possibilities of survival.
The speed and reliability offered by automatic and powered drill IO devices make them popular in such cases, especially where IV access fails because of collapsed veins. Many paramedics and emergency medical teams rely on this kind of access in high-pressure situations.
Their ability to provide immediate vascular access within seconds makes them indispensable in cardiac emergencies. The market is gaining pace with time due to the increased focus on developing upgraded pre-hospital cardiac care protocols, integrated with an increased number of ALS systems, and now IO devices have become a common management process for cardiac arrest. Therefore, demand for vascular access devices that are fast, effective, and easy to use will continue to grow.
Rising Adoption in Pre-Hospital Emergency Services Boosting the Market
The rise in the application of intraosseous (IO) devices in pre-hospital emergency services has become the main growth driver for the market. EMS services are a key component of trauma care, and rapid vascular access can be the difference between life and death.
IO devices, particularly automatic and powered drill models, have become standard tools in ambulances and paramedic units due to their ability to establish immediate vascular access when traditional IV methods fail. Their use is particularly vital in high-stress situations such as cardiac arrests, trauma, and severe shock where vein access can be challenging.
EMS systems worldwide are increasingly equipping ambulances with powered IO devices, such as the EZ-IO®, for faster drug administration and fluid resuscitation in critical cases. Additionally, the trend of expanding advanced life support (ALS) ambulances has further fueled the demand for efficient vascular access tools like IO devices.
The rapid insertion capabilities of powered drill IO devices ensure faster patient stabilization, reducing mortality rates during pre-hospital care. As EMS systems continue to modernize and focus on equipping first responders with advanced medical technologies, the market for intraosseous devices is expected to witness sustained growth.
Increased Focus on Pediatric-Friendly IO Devices Expanding the Market
The increased emphasis on pediatric-friendly intraosseous (IO) devices has thrown open new growth avenues in the market. Pediatric cases often suffer from accessibility issues of vascular access, especially neonates and infants due to fragile and smaller veins. IV access becomes extremely challenging in critical situations like cardiac arrest, dehydration, or trauma in pediatric patients. IO access is generally safer and quicker in such circumstances.
Addressing this need, manufacturers are developing pediatric IO devices with ultra-fine needles, adjustable pressure systems, and depth control mechanisms to penetrate the bone with minimal damage. The Teleflex EZ-IO Pediatric System is one such device which can be used intraosseous access for small bone sizes with a reduced risk of complications.
More and more hospitals and pediatric care units are now integrating these devices as standard equipment for neonatal and pediatric emergency protocols. The training programs also include handling pediatric IO devices to prepare healthcare professionals for critical pediatric situations.
The rising focus on reducing pediatric mortality rates and improving emergency care outcomes will drive demand for dedicated pediatric IO devices, making this an important segment in the intraosseous devices market.
Limited Skillset and Training Requirements Restricting Market Expansion
While intraosseous (IO) devices are highly effective in emergency care, their adoption is often restricted by the specialized training required for proper use. Devices such as powered drills and impact-driven IO tools demand precise handling to avoid complications like bone fractures, tissue damage, or improper needle placement.
Without adequate training, there is a heightened risk of insertion errors, which could lead to compartment syndrome, bone marrow leakage, or infection.
This challenge is particularly evident in regions with limited medical infrastructure, where access to specialized training programs is scarce. For example, low-resource settings often face higher procedural error rates due to a lack of experienced emergency responders familiar with advanced IO device technologies.
Manual IO devices, though simpler, still require proper technique to ensure effective fluid delivery. Furthermore, continuous education and skill retention can be challenging for healthcare systems with high staff turnover.
To address these barriers, market players are increasingly focusing on developing user-friendly IO devices with visual or audio guidance for proper placement, as well as expanding virtual training modules to improve global proficiency. However, until consistent training standards are widely established, skillset limitations remain a significant restraint to the broader market adoption of IO devices.
2020 to 2024 Global Intraosseous Devices Industry Outlook Compared to Demand Forecast from 2025 to 2035
The global intraosseous devices industry recorded a CAGR of 3.6% during the historical period between 2020 and 2024. The growth of the intraosseous devices industry was positive as it reached a value of USD 467.2 million in 2024 from USD 419.1 million in 2020.
The market for intraosseous (IO) devices has historically grown as a result of improved trauma care recommendations and an increase in medical situations.
Adoption of IO devices, especially in emergency rooms and ICUs, increased. During this period, the focus shifted from traditional manual IO devices to automatic and powered drill systems due to their faster deployment and reduced dependency on operator skill.
The demand for pediatric-specific IO devices also increased, driven by a greater focus on neonatal and pediatric emergency care. Additionally, the defense sector became a key consumer, with several global military forces expanding their use of impact-driven and powered drill IO devices for battlefield trauma management.
Technological advancements also marked this period, including the development of smart IO devices with real-time pressure monitoring and guided needle insertion, improving procedural safety. Geographically, North America dominated the market due to advanced healthcare infrastructure and increasing trauma cases, while Asia-Pacific began showing higher growth potential due to expanding pre-hospital care systems.
The future of the intraosseous devices market is expected to be shaped by further technological innovations and expanded applications. Smart IO devices equipped with real-time feedback sensors, pressure monitoring, and automated needle depth adjustments are projected to gain traction, minimizing user error and enhancing patient safety.
Another key trend will be the growing integration of IO devices in pre-hospital emergency services worldwide, driven by improved EMS training and protocols emphasizing faster trauma response. Pediatric and neonatal IO devices will likely become more specialized, with adjustable needle sizes and improved pressure control features to reduce bone damage.
Additionally, emerging markets in Latin America, Africa, and Southeast Asia will see increased adoption due to growing healthcare investments and awareness about advanced trauma care. Military procurement for impact-driven and powered drill IO devices will also continue to rise, further expanding the market.
Market Concentration
Tier 1 companies hold a dominant 41.2% share of the global market. These industry leaders have significant market influence, robust revenue streams, and a broad global footprint.
Their diverse product portfolios cover various types of IO devices, including manual, automatic, impact-driven, and powered drill devices. These companies often set industry standards through strategic acquisitions, collaborations, and regulatory approvals.
By driving innovation and advancing the technology behind IO devices, Tier 1 companies play a central role in shaping market trends. Prominent Tier 1 players include Cook Medical Inc., Teleflex Incorporated, Becton, Dickinson and Company (BD), and Cardinal Health, Inc.
Tier 2 companies account for 26.5% of the global IO devices market, maintaining a strong presence in specific segments or regions. These companies are focused on specialized or cost-effective solutions, often targeting mid-sized hospitals, emergency medical services (EMS), and outpatient care centers. They emphasize portable, user-friendly, and niche IO devices, such as manual or affordable automatic models that cater to local market needs.
With an emphasis on providing efficient vascular access in less resource-intensive settings, Tier 2 companies offer practical solutions that meet the unique demands of smaller healthcare providers. Key players in this category include Pyng Medical Corp., PerSys Medical, Aero Healthcare, and Vidacare Corporation.
Country-wise Insights
The section below covers the industry analysis for intraosseous devices sales for different countries. Market demand analysis on key countries in several regions of the globe, including North America, Latin America, East Asia, South Asia & Pacific, Western Europe, Eastern Europe, and Middle East & Africa is provided.
The United States is anticipated to remain at the forefront in North America, with a CAGR of 1.8% through 2035. In South Asia & Pacific, India is projected to witness the highest CAGR in the market of 5.8% by 2035.
| Countries | Value CAGR (2025 to 2035) |
|---|---|
| United States | 1.8% |
| Germany | 1.6% |
| Italy | 2.0% |
| UK | 3.7% |
| China | 5.4% |
| India | 5.8% |
High Trauma and Cardiac Arrest Cases Drive the Intraosseous Devices Market in the United States
The United States dominates the global market with a high share in 2024. The United States is expected to exhibit a CAGR of 1.8% throughout the forecast period (2025 to 2035).
The United States dominates the intraosseous devices market, mainly because of the high incidence of trauma cases and sudden cardiac arrests that require prompt vascular access solutions.
With nearly 356,000 out-of-hospital cardiac arrests occurring every year, IO devices have become crucial tools for paramedics and first responders. A growing need in the use of ALS ambulances equipped with powered drill IO devices contributes to market demand.
This makes the USA also highly exposed to traumatic cases with an annual average of 3 million reported non-fatal injuries that demand efficient emergency vascular access tools. The rising standards of prehospital care protocols are also a part of increasing requirements, like American Heart Association-adopted ones.
IO devices then become a preferable choice in all difficult IV access cases. Strong collaborations between medical device manufacturers and emergency service providers, along with the country's focus on rapid intervention tools, have further solidified its market leadership.
Rising Geriatric Population and Chronic Disease Management Drives the Intraosseous Devices Market in Germany
In 2024, Germany held a dominant revenue in the Western Europe market and is expected to grow with a CAGR of 1.6%.
Germany's leadership in the intraosseous devices market is primarily driven by its aging population and the rising incidence of chronic illnesses requiring emergency care. With over 21% of the population aged 65 and older, Germany faces increasing cases of age-related conditions such as heart failure, leading to frequent medical emergencies where rapid vascular access is critical.
IO devices have become essential in geriatric care due to the challenges posed by collapsed veins in older patients, making traditional IV access difficult. Furthermore, Germany’s well-established emergency medical services (EMS) network, including specialized geriatric ambulances, often incorporates IO devices for efficient drug delivery in critical care situations.
The country’s emphasis on clinical training for paramedics and hospital staff has also supported the broader adoption of IO devices, ensuring effective use across various healthcare settings.
The German healthcare system also benefits from widespread collaborative research that focuses on the efficiency of emergency procedures, further increasing the reliance on IO devices, particularly in stroke and cardiac arrest situations, improving survival rates and recovery.
Expanding Military Medical Procurement Fuels the Intraosseous Devices Market in China
China occupies a leading value share in East Asia market in 2024 and is expected to grow with a CAGR of 5.4% during the forecasted period.
China's dominance in the intraosseous devices market is significantly influenced by expanding military medical procurement and rising demand for battlefield trauma care equipment.
The Chinese military has increasingly prioritized modernizing its combat medical response infrastructure, including the procurement of powered drill and impact-driven IO devices for rapid fluid administration in trauma cases.
This trend is further supported by the People's Liberation Army's (PLA) focus on tactical medical readiness, ensuring that field medics are equipped with advanced tools for immediate care. Additionally, China has seen a rise in mass casualty incidents related to industrial accidents and natural disasters, amplifying the need for reliable vascular access tools during emergency response efforts.
The presence of domestic manufacturers producing cost-effective IO devices has also made these tools more accessible across both military and civilian healthcare sectors, strengthening China’s market position.
Additionally, China's expanding population and urbanization are leading to higher emergency call volumes, further increasing the need for efficient trauma care solutions like IO devices, fueling market growth in both civilian and military sectors.
Category-wise Insights
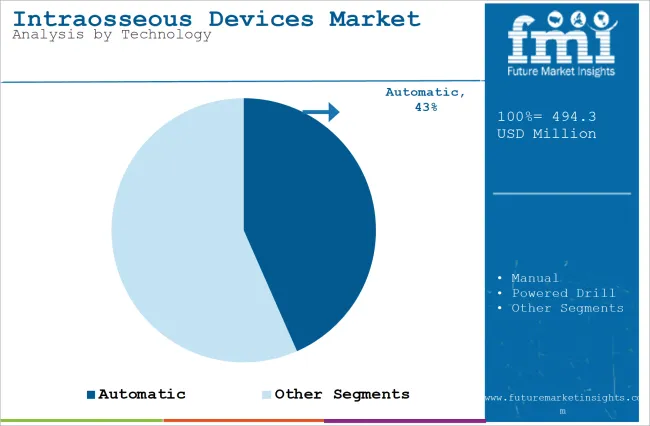
The section contains information about the leading segments in the industry. Based on technology, automatic segment are expected to account 43.4% of the global share in 2025.
Automatic IO Devices Set to Lead the Market Due to Speed and Accuracy
| Technology | Automatic |
|---|---|
| Value Share (2025) | 43.4% |
Automatic intraosseous (IO) devices are likely to lead the market because of their speed, accuracy, and ease of use. Unlike manual devices, which require a lot of skill and experience, automatic devices allow for rapid insertion, reducing the time spent on vascular access, especially in critical emergency situations. This is crucial in trauma cases, where every second counts.
Automatic IO devices contain built-in depth control mechanisms to automatically control insertion depth, avoiding bone damage or complications from unsafe needle placement. Moreover, the devices have been designed keeping in mind usability, which applies even to the well-trained healthcare professional and less-qualified first respondent.
This particular aspect is essential in prehospital care settings, such as ambulance services, because it requires the rapid decision-making under pressure by a paramedic. Automatic IO devices are preferred especially in time-dependent situations because they are efficient and reliable.
Hospitals Lead the Market Due to High Demand for Emergency and Critical Care Services
| By End User | Hospital |
|---|---|
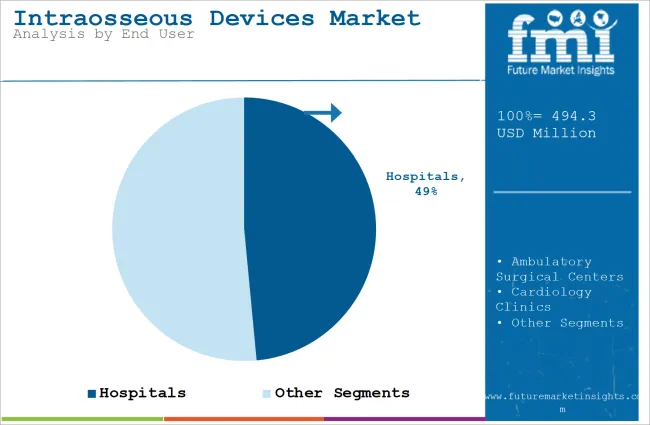
| Value Share (2025) | 48.5% |
Hospitals will account for 48.5% of the end user segment in 2025, and exhibit the highest CAGR in the forecast period.
Due to the large volumes of emergency and critical care cases, hospitals are likely to dominate the intraosseous (IO) devices market. Since hospitals are generally the first place where trauma patients, cardiac arrests, and other medical emergencies go, they will need reliable and efficient methods of vascular access in case traditional intravenous (IV) lines are not feasible.
IO devices are essential in critical care units, emergency rooms, and intensive care units (ICUs) for immediate medication and fluid delivery. Hospitals are increasingly adopting automatic and powered drill IO devices, which offer rapid and accurate insertion, reducing procedure time in high-pressure environments.
Furthermore, the growing need for quick responses in trauma situations, stroke management, and pediatric care has solidified the hospital's role as a key player in the IO devices market. With a continuous focus on improving patient outcomes and reducing mortality, hospitals are the largest consumers of IO devices.
Competitive Landscape
Key players in the intraosseous devices market are continuously driving product innovation and launches to stay ahead of the competition and meet the evolving needs of healthcare providers.
These companies are focusing on developing advanced, highly efficient IO devices, expanding their product portfolios to include cutting-edge solutions across various modalities such as manual, automatic, impact-driven, and powered drill systems.
This ongoing innovation helps them cater to the diverse demands of medical professionals, emergency services, and hospitals, solidifying their position in the global IO devices landscape.
Recent Industry Developments in Intraosseous Devices Market
- On June, 2023, Teleflex Incorporated, a prominent global provider of medical devices for critical care and surgery, has announced that its Arrow EZ-IO Needle is the first and only intraosseous (IO) needle to receive 510(k) clearance from the USA Food and Drug Administration (FDA) for MR Conditional labelling. The EZ-IO Needle, a key part of the Arrow EZ-IO Intraosseous Vascular Access System, features a patented diamond tip that ensures quick, precise, and stable insertion.
- On March, 2020, SAM Medical, a leader in trauma care medical devices, has unveiled the SAM IO Intraosseous Access System, designed for use by healthcare providers in both pre-hospital and hospital settings across civilian and military sectors.
Key Players of Intraosseous Devices Market
- Pyng Medical Corp.
- PerSys Medical
- Aero Healthcare
- Vidacare Corporation
- Carefusion Inc.
- Cook Medical Inc.
- Teleflex Incorporated
- Becton, Dickinson and Company (BD)
- Argon Medical Devices, Inc.
- SAM Medical
- BIOPSYBELL S.R.L.
- Cardinal Health, Inc.
- Medax SRL Unipersonale
- StarFish Medical
Key Segments of Intraosseous Devices Market
By Technology:
In terms of technology, the industry is divided into manual, automatic, impact-driven, powered drill.
By Route of Administration:
In terms of route of administration, the industry is divided into sternum, distal and proximal tibia, distal femur, calcaneus, head of humerus.
By End User:
In terms of end user, the industry is segregated into hospitals, ambulatory surgical centers and cardiology clinics
By Region:
Key countries of North America, Latin America, East Asia, South Asia & Pacific, Western Europe, Eastern Europe, and Middle East and Africa (MEA) have been covered in the report.
Frequently Asked Questions
What is the future of global intraosseous devices industry?
The global intraosseous devices industry is projected to witness CAGR of 5.8% between 2025 and 2035.
What was the worth of the global intraosseous devices industry in 2024?
The global intraosseous devices industry stood at USD 467.2 million in 2024.
What will the worth of global intraosseous devices industry by 2035 end?
The global intraosseous devices industry is anticipated to reach USD 938.6 million by 2035 end.
What is the expected CAGR for China during forecast period?
China is expected to show a CAGR of 5.4% in the assessment period.
Who are the key manufacturer of global intraosseous devices industry?
The key players operating in the global intraosseous devices industry include Pyng Medical Corp., PerSys Medical, Aero Healthcare, Vidacare Corporation, Carefusion Inc., Cook Medical Inc., Teleflex Incorporated, Becton, Dickinson and Company (BD), Argon Medical Devices, Inc., SAM Medical, BIOPSYBELL S.R.L., Cardinal Health, Inc., Medax SRL Unipersonale and StarFish Medical.
Table of Content
- Executive Summary
- Industry Introduction, including Taxonomy and Market Definition
- Market Trends and Success Factors, including Macro-Economic Factors, Market Dynamics, and Recent Industry Developments
- Global Market Demand (Value in USD) and Volume (Absolute Units) Analysis 2020 to 2024 and Forecast 2025 to 2035, including Historical Analysis and Future Projections
- Global Market - Pricing Analysis
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035
- By Technology
- By Route of Administration
- By End User
- By Region
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Technology
- Manual
- Automatic
- Impact-Driven
- Powered Drill
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Route of Administration
- Sternum
- Distal and Proximal Tibia
- Distal Femur
- Calcaneus
- Head of Humerus
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By End User
- Hospitals
- Ambulatory Surgical Centers
- Cardiology Clinics
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- North America
- Latin America
- Western Europe
- Eastern Europe
- South Asia & Pacific
- East Asia
- Middle East & Africa
- North America Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Latin America Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Western Europe Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Eastern Europe Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- South Asia & Pacific Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- East Asia Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Middle East and Africa Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Sales Forecast 2025 to 2035 By Technology, By Route of Administration, and by End User for 30 Countries
- Competition Outlook, including Market Structure Analysis, Company Share Analysis by Key Players, and Competition Dashboard
- Company Profile
- Pyng Medical Corp.
- PerSys Medical
- Aero Healthcare
- Vidacare Corporation
- Carefusion Inc.
- Cook Medical Inc.
- Teleflex Incorporated
- Becton, Dickinson and Company (BD)
- Argon Medical Devices, Inc.
- SAM Medical
- BIOPSYBELL S.R.L.
- Cardinal Health, Inc.
- Medax SRL Unipersonale
- StarFish Medical
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

