Intraosseous Infusion Devices Market
The Intraosseous Infusion Devices Market Is Segmented By Product (B.I.G, FAST1, EZ-IO, FASTR, IO Needles, Others), Route Of Administration (Sternum, Distal And Proximal Tibia, Distal Femur, Calcaneus, Head Of Humerus), Technology (Automatic, Manual), Patient Group (Adults, Pediatrics, Neonates, Geriatrics), Application (Cardiac Arrest, Trauma Management, Shock Treatment, Severe Dehydration, Difficult IV Access), End User (EMS, Hospitals, Ambulatory Surgical Centers, Cardiac Care Centers), And Region. Forecast from 2026 to 2036.
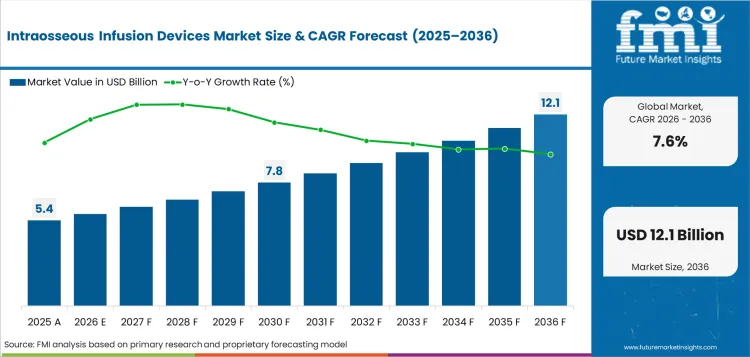
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Intraosseous Infusion Devices Market Size, Market Forecast and Outlook By FMI
Summary of the Intraosseous Infusion Devices Market
- Demand and Growth Drivers
- Emergency teams use intraosseous access after failed peripheral IV placement since fluid and drug delivery cannot wait during arrest or shock.
- Ambulance services and trauma centers keep stocked devices because response protocols require rapid vascular access readiness.
- Training programs raise device adoption as clinicians need repeat practice before urgent sternal or tibial access use.
- Product and Segment View
- Automatic systems lead technology demand since powered insertion supports more predictable placement during rushed emergency care.
- Adults lead patient group demand as cardiac arrest and trauma cases account for a large share of IO use.
- Sternal access is projected to support fast landmark identification in adult resuscitation care.
- Geography and Competitive Outlook
- India and South Korea is anticipated to record the highest growth outlook as trauma response upgrades and simulation training expand clinical familiarity.
- United States holds an established revenue base since emergency departments and EMS programs already use IO access protocols.
- Teleflex leads through Arrow EZ-IO access. Smiths Medical and BD compete through vascular access portfolios and hospital relationships.
- Analyst Opinion
- Sabyasachi Ghosh, Principal Analyst for Healthcare at FMI, observes that “IO device adoption is aligned to emergency timing and clinician confidence. Hospitals and EMS units require devices that can be placed quickly after peripheral IV access fails. Suppliers with training depth and reliable device availability hold a stronger position in trauma and resuscitation pathways. Growth through 2036 is likely to come from emergency system upgrades in India and wider protocol use across advanced hospital networks.”
- Intraosseous Infusion Devices Market Value Analysis
- Emergency response budgets keep device ordering steady because IO access is part of resuscitation readiness.
- Revenue gains come from automatic systems and single-use needle sets used in prehospital and hospital emergency care.
- System training affects replacement demand since stocked devices must match the models used in clinician education.
- Hospital and EMS purchasing cycles favor devices with proven access speed and accessory availability.
Intraosseous Infusion Devices Market Definition
The intraosseous infusion devices sector covers powered and manual access systems used to deliver fluids or medications through bone marrow access after peripheral IV placement fails.
Intraosseous Infusion Devices Market Inclusions
The study includes global sizing and forecast from 2026 to 2036 by product, route of administration, technology, patient group, application, end user, and geography.
Intraosseous Infusion Devices Market Exclusions
General IV catheters, infusion pumps, vascular closure devices, and surgical access tools stay outside this study unless they are sold for intraosseous access use.
Intraosseous Infusion Devices Market Research Methodology
- Primary Research: Involved review of product pages and public materials from suppliers active in intraosseous access.
- Desk Research: Combined emergency care references and published market pages for vascular access and emergency service adjacency.
- Market Sizing and Forecasting: Aligned device use with emergency readiness and protocol-based access needs.
- Data Validation: Checked segment leadership and country outlook against source figures and clinical use patterns.
Why is the Intraosseous Infusion Devices Market Growing?
- Emergency teams require rapid vascular access after failed IV attempts in cardiac arrest and severe shock.
- Training adoption across EMS and hospitals raises device familiarity before high-acuity use.
- Trauma care expansion increases device stocking across ambulances and emergency departments.
Hospitals and EMS teams sustain demand because intraosseous (IO) access provides a critical backup route when peripheral veins cannot be reached, a scenario that is common during cardiac arrest and severe trauma. Cardiac arrest care increasingly adopts IO devices as resuscitation teams require immediate medication delivery without delays caused by failed IV attempts. Automatic systems remain preferred in many emergency settings because powered insertion reduces hand‑force variation and supports faster access under time pressure.
Trauma care further accelerates demand as blood loss and circulatory collapse make venous access unreliable, particularly in prehospital environments. At the same time, expanded emergency training programs improve clinician confidence in sternal and tibial access, reinforcing routine device stocking. These factors link market growth directly to rising emergency case volume and broader upgrades in EMS and hospital critical care readiness.
Market Segmentation Analysis
- Automatic systems are estimated to account for 51.0% share in 2026 as powered insertion reduces force variation during field and emergency department use.
- Adults are estimated to represent 39.0% share in 2026 because sternal and tibial access are used often in adult cardiac arrest and trauma care.
- Cardiac arrest is estimated to contribute 34.0% share in 2026 since resuscitation protocols require fast drug access after failed IV placement.
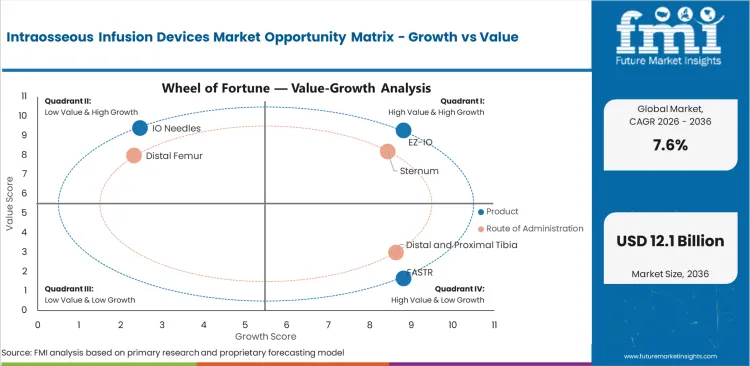
- Sternum is expected to make up 30.9% share in 2026 because adult landmark recognition is faster during urgent resuscitation.
Segmentation outlook divides market by tracks device type, access route, insertion technology, patient group, clinical use case, and care site. Product choice reflects training familiarity and response pressure within local protocols. Stocked access systems must match the equipment used in clinician practice.
Intraosseous Infusion Devices Market Analysis by Product
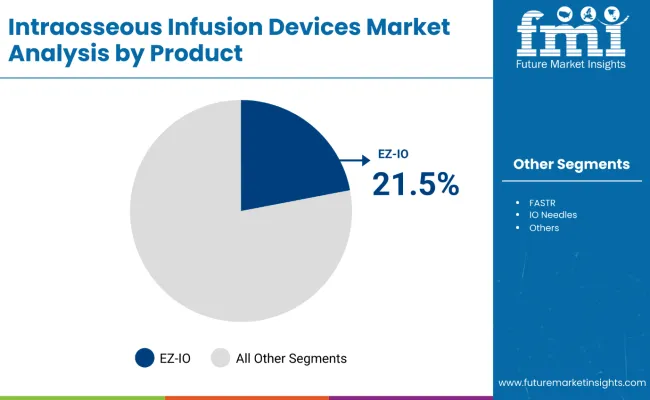
- EZ-IO is projected to contribute 21.5% share in 2026 as powered placement supports repeatable insertion during emergency access attempts.
- Clinical teams prefer the platform since established training materials and accessories help reduce hesitation during urgent use.
Insights into the IO Needles Segment
- IO needles support steady demand because single-use access kits require compatible needle lengths for adult and pediatric care.
- Stocked needle sets help hospitals and ambulance units maintain readiness across tibial, humeral, and sternal access pathways.
Intraosseous Infusion Devices Market Analysis by Route of Administration
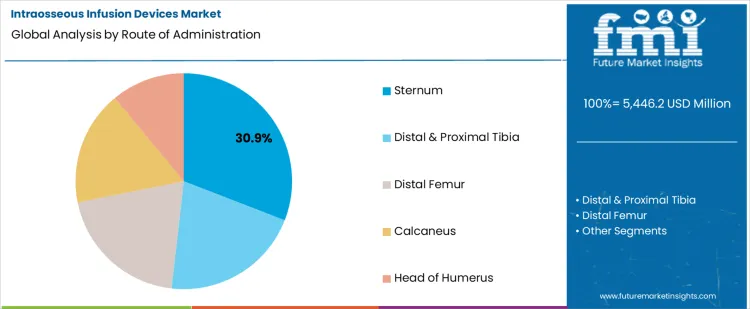
- Sternum is expected to account for 30.9% share in 2026 as fast adult landmark access supports resuscitation and severe trauma care.
- Adult emergency protocols support sternal use because central circulation access helps medication delivery during urgent intervention.
Insights into the Distal and Proximal Tibia Segment
- Tibial access segment garners a stable place in training since it is commonly taught across adult and pediatric emergency programs.
- Field clinicians use tibial landmarks in mixed patient groups as placement can be taught clearly during simulation practice.
Intraosseous Infusion Devices Market Analysis by Technology
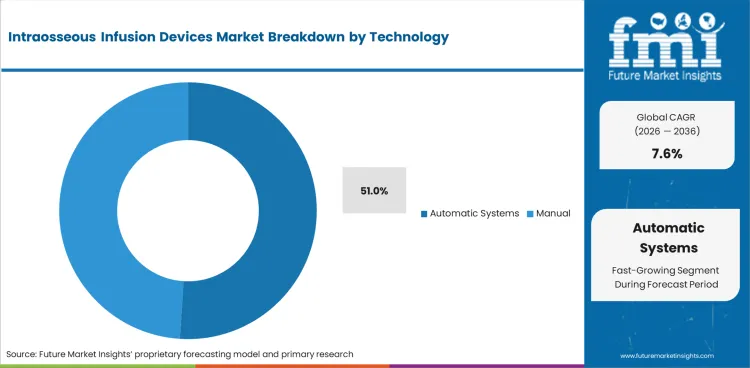
- Automatic systems are estimated to represent 51.0% share in 2026 as powered insertion helps reduce placement variation during urgent access attempts.
- Powered systems suit EMS and emergency department workflows and help clinicians apply a repeatable motion under pressure.
Insights into the Manual Segment
- Manual devices retain use in price-sensitive settings since they are simpler and need fewer powered components.
- StatPearls describes manual IO devices as inexpensive and reliable. These devices require more skill and time during emergencies.
Intraosseous Infusion Devices Market Analysis by Patient Group
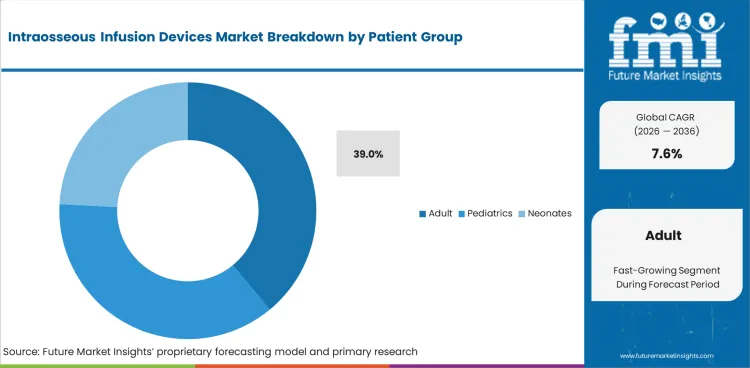
- By patient group, adult segment is estimated to account for 39.0% share in 2026 as cardiac arrest and trauma response protocols create frequent access needs.
- Adult care pathways use sternal and tibial routes more often since field teams manage arrest, shock, and severe injury before full hospital access.
Insights into the Pediatric Segment
- Pediatric use advances through training programs because difficult IV placement can delay medication in small children.
- Device sets with shorter needle lengths improve pediatric readiness since neonatal and child access needs differ from adult use.
Intraosseous Infusion Devices Market Analysis by Application
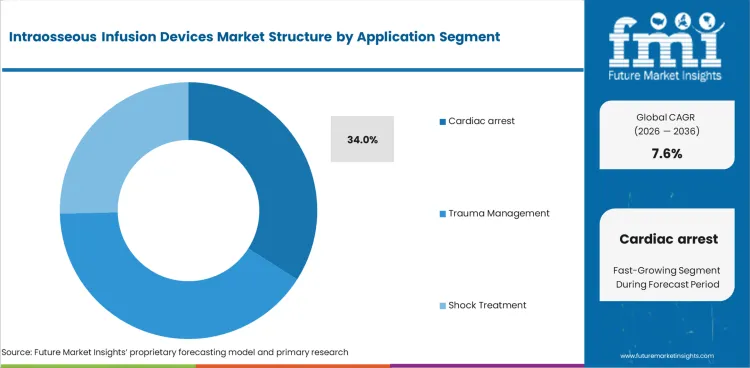
- Cardiac arrest is estimated to contribute 34.0% share of application demand in 2026 as drug delivery cannot pause after failed IV placement.
- Resuscitation teams use IO access since arrest care requires a fast route for medication and fluid administration.
Insights into the Trauma Management Segment
- Trauma management demand rises as ambulance and emergency teams need access options during blood loss and unstable circulation.
- IO access aligns with demand for rapid infusers in the USA because both device categories serve emergency fluid replacement and trauma stabilization.
Intraosseous Infusion Devices Market Analysis by End User
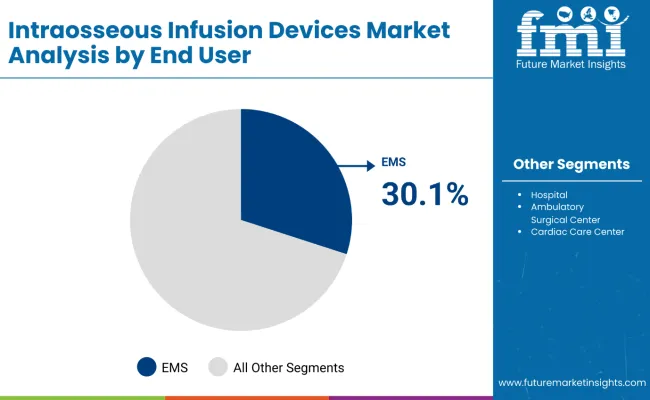
- EMS is projected to account for 30.1% share of end user demand in 2026 as ambulance teams need immediate access during field resuscitation.
- Prehospital use supports recurring device stocking because teams must carry access tools before patient transfer.
Insights into the Hospitals Segment
- Hospitals maintain a large demand base led by emergency departments and critical care units managing failed IV access inside structured clinical settings.
- Hospital ordering links IO devices with emergency carts and critical care inventory planning.
Intraosseous Infusion Devices Market Drivers Restraints and Opportunities
- Emergency access needs are expected to sustain device demand across EMS units and emergency departments through 2036.
- Training gaps is anticipated to slow adoption because clinicians require repeated practice before using IO devices in high-pressure care.
- Automatic insertion systems are expected to create clearer supplier value when devices are paired with education and stocked needle kits.
Intraosseous infusion device demand is projected to rise as cardiac arrest and trauma care place more pressure on fast vascular access. Hospitals and EMS units are expected to keep IO devices in emergency kits because failed IV access delays medication and fluid delivery. Adoption will be stronger in systems that invest in emergency training and standard device stocking.
Rapid Emergency Access Supports Demand
Cardiac arrest care will remain the strongest clinical driver as response teams need quick drug access after failed peripheral IV attempts. EMS units are expected to increase device readiness as advanced life support capacity expands across more services. Automatic systems will gain preference because powered insertion can improve placement confidence during urgent care.
Training Burden Slows Outcomes
Adoption may remain uneven in settings with limited simulation practice and lower emergency care budgets. Manual devices can reduce upfront cost but they need stronger placement skill during rushed use. Hospitals with limited training time may delay wider device use outside emergency and critical care units.
Automatic Systems Open Upside
Powered IO systems are expected to gain share as hospitals and ambulance services look for faster access with lower placement variation. Needle kits and training programs will increase repeat ordering across emergency networks. Suppliers that provide simple education and dependable device availability can capture stronger demand from trauma and cardiac care teams.
Regional Analysis
.webp)
| Country | CAGR 2026 to 2036 |
|---|---|
| India | 8.7% |
| South Korea | 8.2% |
| Germany | 8.0% |
| France | 7.8% |
| China | 7.6% |
Source: Future Market Insights analysis based on country CAGR set disclosed in the source report.
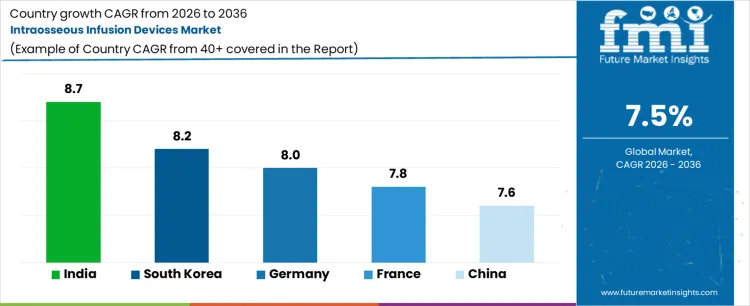
Analysis of Intraosseous Infusion Devices Market by Key Countries
- India is projected to post 8.7% CAGR through 2036 as ambulance network expansion and trauma center investment raise rapid access device stocking.
- South Korea is expected to advance at 8.2% CAGR through 2036 as simulation-based emergency training increases clinician familiarity with IO placement.
- Germany is forecast to record 8.0% CAGR over the projection period as prehospital emergency integration keeps device adoption with protocol-led care.
Country growth differs by emergency infrastructure and training depth. India and South Korea rise faster because device readiness is moving deeper into ambulance and hospital systems. Germany and France advance through structured emergency response models
Demand Outlook for Intraosseous Infusion Devices Market in India
The market in India is expected to record 8.7% CAGR through 2036 as road trauma volume and emergency department expansion increase IO device readiness. Ambulance networks and private hospitals are adding advanced life support capacity. High patient volume makes cost-optimized devices important for wider clinical use.
- Trauma care expansion increases device stocking across public and private emergency units.
- ALS training improves use confidence among doctors, paramedics, and critical care nurses.
- Large hospitals require reliable access tools because delayed IV placement slows early stabilization.
Demand Outlook for Intraosseous Infusion Devices Market in South Korea
Industry in South Korea is projected to rise at 8.2% CAGR through 2036 since protocol compliance and simulation training increase device familiarity among emergency teams. Hospitals use structured response pathways that support repeatable IO placement. Aging demographics add more acute care episodes that need fast access.
- Simulation centers improve confidence before clinicians use IO devices in live emergencies.
- Hospital networks support faster protocol adoption through coordinated emergency practice.
- Response time discipline keeps IO systems integrated in standard emergency kits.
Demand Outlook for Intraosseous Infusion Devices Market in Germany
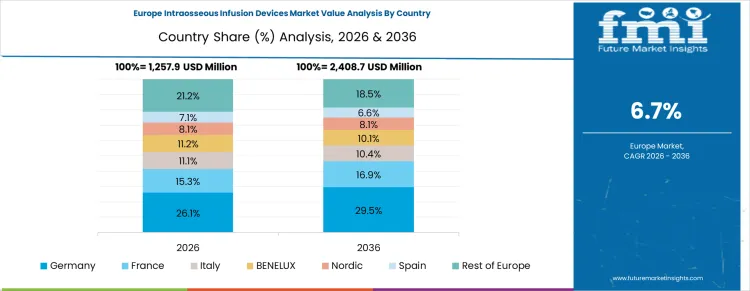
Germany is forecast to expand at 8.0% CAGR through 2036 as mature ambulance systems and trauma care capacity support field-based IO use. Public healthcare purchasing favors documented reliability and clinician training. Device adoption grows through replacement and standardization across emergency teams.
- Prehospital service integration supports recurring demand from ambulance systems.
- Trauma center throughput keeps rapid access devices in emergency inventories.
- Standardized training helps clinicians use the same device platform across care sites.
Sales Analysis of Intraosseous Infusion Devices Market in France
France is expected to record 7.8% CAGR through 2036 as national emergency response structures support rapid vascular access in shock and resuscitation care. Hospital-based critical care capacity adds demand for stocked IO devices. Regional emergency services maintain device use through protocol-led training.
- Structured emergency response systems keep IO access inside urgent care pathways.
- Hospital critical care investment supports device use after failed peripheral IV access.
- Public healthcare equipment planning increases access to standard emergency devices.
Opportunity Analysis of Intraosseous Infusion Devices Market in China
China is projected to post 7.6% CAGR through 2036 since trauma center development and emergency department expansion raise procedural adoption. Large hospital networks increase training reach among emergency clinicians. Domestic device availability helps extend access beyond top-tier urban hospitals.
- Trauma center growth raises demand for rapid access systems in acute care.
- Urban emergency departments increase IO use as clinician training expands.
- Domestic manufacturing improves supply reach for tertiary and secondary hospitals.
Competitive Position and Company Strategy
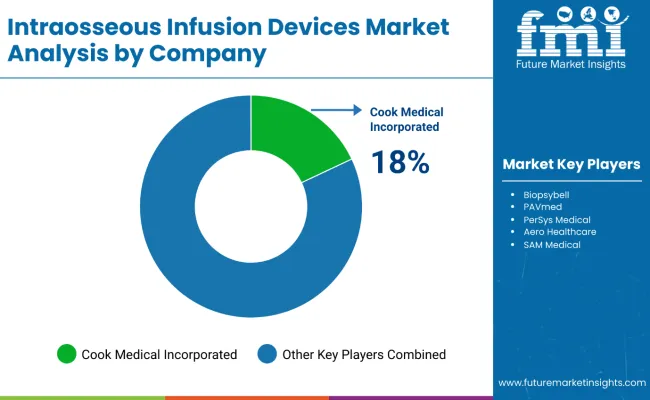
- Competition is based on insertion reliability and training depth. Compatibility with emergency drug and fluid delivery sets shapes hospital selection.
- Teleflex holds the clearest product position through Arrow EZ-IO and related clinical education materials.
- Smiths Medical and BD compete through vascular access reach. ICU Medical and Zhejiang Shanshan Broadmed rely on hospital supply relationships.
Competition in intraosseous infusion devices centers on clinician confidence. Teleflex offers Arrow EZ-IO recognition and a defined emergency medicine product page. The system is presented for difficult vascular access cases that are emergent, urgent, or medically necessary.
Hospital systems compare suppliers on device familiarity and training support. Manual systems can reduce acquisition cost. They require greater operator skill during emergencies. Automatic systems cost more. They reduce hand-force variation during urgent placement. Adjacent vascular access portfolios influence supplier reach since hospitals often buy access products through established device suppliers.
Competitive Benchmarking: Intraosseous Infusion Devices Market
| Company | IO Portfolio Depth | Training Support | Hospital Access | Global Reach |
|---|---|---|---|---|
| Teleflex | High | High | High | High |
| Smiths Medical | Medium | Medium | High | High |
| Becton Dickinson | Medium | Medium | High | High |
| ICU Medical | Medium | Medium | Medium | High |
| Zhejiang Shanshan Broadmed | Medium | Low | Medium | Medium |
Source: Future Market Insights competitive interpretation based on disclosed company set and product positioning.
Key Developments in Intraosseous Infusion Devices Market
- In May 2024, Teleflex launched the Arrow™ EZ-IO™ Intraosseous Access Procedure Tray for sterile-field IO access in hospital settings.
- In December 2024, a pediatric retrospective study of Arrow EZ-IO reported 96.2% access and infusion success with no serious adverse events.
- In May 2025, StatPearls updated its intraosseous vascular access review and classified IO devices as manual, powered, and spring-loaded systems.
- In October 2025, AHA published updated CPR and ECC guidance that keeps IO access as a reasonable alternative when IV access is delayed or not feasible.
Key Companies in the Intraosseous Infusion Devices Market
Major Global Companies
- Teleflex
- Smiths Medical
- Becton Dickinson
- ICU Medical
- Zhejiang Shanshan Broadmed
Other Companies
- Cook Medical
- Medline Industries
- PerSys Medical
- Piper Access
- Pyng Medical
Report Scope and Coverage
| Item | Value |
|---|---|
| Market value in 2025 | USD 5.1 billion |
| Market value in 2026 | USD 5.4 billion |
| Market value in 2036 | USD 11.2 billion |
| CAGR from 2026 to 2036 | 7.6% |
| Quantitative units | USD billion |
| Market definition | Devices used for intraosseous access in emergency and critical care |
| By product | B.I.G; FAST1; EZ-IO; FASTR; IO Needles; Others |
| By route of administration | Sternum; Distal and Proximal Tibia; Distal Femur; Calcaneus; Head of Humerus |
| By technology | Automatic; Manual |
| By patient group | Adults; Pediatrics; Neonates; Geriatrics |
| By application | Cardiac Arrest; Trauma Management; Shock Treatment; Severe Dehydration; Difficult IV Access |
| By end user | EMS; Hospitals; Ambulatory Surgical Centers; Cardiac Care Centers |
| Regions covered | Asia Pacific; Europe; North America; Latin America; Middle East and Africa |
| Countries covered | India; South Korea; Germany; France; China; United States; Brazil |
| Key companies profiled | Teleflex; Smiths Medical; Becton Dickinson; ICU Medical; Zhejiang Shanshan Broadmed |
| Forecast period | 2026 to 2036 |
| Approach | FMI public treatment model aligned with emergency access protocol and company evidence |
Source: Future Market Insights analysis, 2026.
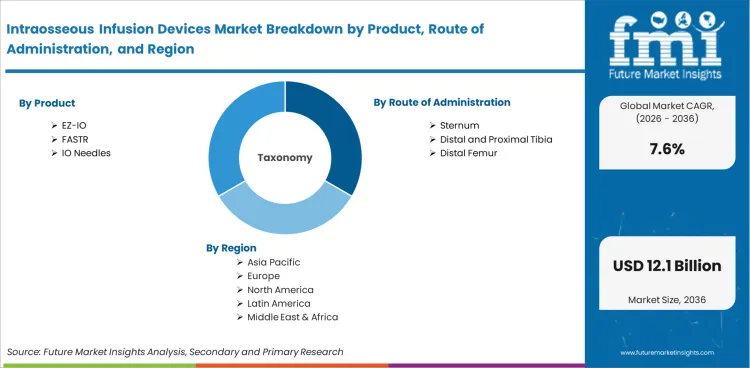
Intraosseous Infusion Devices Market Breakdown by Product, Route of Administration, Technology, Patient Group, Application, End User, and Region
Intraosseous Infusion Devices Market Segmented by Product
- B.I.G
- FAST1
- EZ-IO
- FASTR
- IO Needles
- Others
Intraosseous Infusion Devices Market Segmented by Route of Administration
- Sternum
- Distal and Proximal Tibia
- Distal Femur
- Calcaneus
- Head of Humerus
Intraosseous Infusion Devices Market Segmented by Technology
- Automatic
- Manual
Intraosseous Infusion Devices Market Segmented by Patient Group
- Adults
- Pediatrics
- Neonates
- Geriatrics
Intraosseous Infusion Devices Market Segmented by Application
- Cardiac Arrest
- Trauma Management
- Shock Treatment
- Severe Dehydration
- Difficult IV Access
Intraosseous Infusion Devices Market Segmented by End User
- EMS
- Hospitals
- Ambulatory Surgical Centers
- Cardiac Care Centers
Intraosseous Infusion Devices Market Segmented by Region
- North America
- United States
- Canada
- Latin America
- Brazil
- Mexico
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia and New Zealand
- Middle East and Africa
- Saudi Arabia
- Other GCC Countries
- South Africa
Research Sources and Bibliography
- American Heart Association. (2025). 2025 American Heart Association guidelines for CPR and ECC.
- Dornhofer, P., McMahon, K., & Kellar, J. Z. (2025). Intraosseous vascular access. In StatPearls. StatPearls Publishing.
- Ko, Y.-C., Lin, H.-Y., Huang, E. P.-C., Lee, A.-F., Hsieh, M.-J., Yang, C.-W., Lee, B.-C., Wang, Y.-C., Yang, W.-S., Chien, Y.-C., Sun, J.-T., Ma, M. H.-M., & Chiang, W.-C. (2024). Intraosseous versus intravenous vascular access in upper extremity among adults with out-of-hospital cardiac arrest. BMJ, 386, e079878.
- Kokori, E., Al-Hashemi, N., Aldeen, Z. S., Patel, R., Aderinto, N., Olatunji, G., Ojo, I. S., Abraham, I. C., & Shaka, H. (2025). Intraosseous vs. intravenous access in out-of-hospital cardiac arrest. International Journal of Emergency Medicine, 18, Article 131.
- Philbeck, T. E., McDonald, J. G., Carew, M., & Ross, C. (2024). Retrospective observational study of safety, performance, and duration of use of battery-powered intraosseous access device in pediatric patients. SAGE Open Medicine, 12, 20503121241306959.
- Rijnhout, T. W. H., Kieft, M., Klein, W. M., & Tan, E. C. T. H. (2024). Effectiveness of intraosseous access during resuscitation. BMC Emergency Medicine, 24, Article 117.
- Suman, S., Mishra, P. R., Mishra, P., & Pandey, S. (2025). Comparative safety and efficacy of four intraosseous devices for vascular access in the emergency department. The Journal of Emergency Medicine.
- Teleflex Incorporated. (2024, May 9). Teleflex expands intraosseous vascular access portfolio with new Arrow EZ-IO intraosseous access procedure tray.
The bibliography supports reader reference and direct source checking.
This Report Answers
- How large the intraosseous infusion devices sector is in 2026 and how far it can rise by 2036?
- Which product, route, and technology hold the strongest projected share positions in 2026?
- Why do automatic systems hold the clearest lead in current device structure?
- Why do India and South Korea lead the published country growth table through 2036?
- How do EMS units shape early access needs during cardiac arrest and trauma care?
- Which supplier capabilities help device companies defend their position in powered IO access and clinical training?
- What emergency care factors and vascular access pathways support the forecast method used in this study?
Frequently Asked Questions
How big is the intraosseous infusion devices market in 2026?
The intraosseous infusion devices market is valued at USD 5.4 billion in 2026 under the FMI forecast model.
What will be the size of the intraosseous infusion devices market in 2036?
The intraosseous infusion devices market is projected to reach USD 11.2 billion by 2036 under the FMI forecast model.
What CAGR is expected for the intraosseous infusion devices market?
The intraosseous infusion devices market is expected to expand at a 7.6% CAGR from 2026 to 2036 under the FMI model.
Which route leads the intraosseous infusion devices market?
Sternum leads route of administration with 30.9% share in 2026 because adult emergency access often needs fast landmark recognition.
Which technology leads the intraosseous infusion devices market?
Automatic systems lead technology demand with 51.0% share in 2026 as powered insertion supports more consistent emergency placement.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2026 to 2036
- EZ-IO
- FASTR
- IO Needles
- EZ-IO
- Y to o to Y Growth Trend Analysis By Product , 2021 to 2025
- Absolute $ Opportunity Analysis By Product , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Route of Administration
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Route of Administration, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Route of Administration, 2026 to 2036
- Sternum
- Distal and Proximal Tibia
- Distal Femur
- Sternum
- Y to o to Y Growth Trend Analysis By Route of Administration, 2021 to 2025
- Absolute $ Opportunity Analysis By Route of Administration, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- Automatic Systems
- Manual
- Automatic Systems
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Patient Group
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Patient Group, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Patient Group, 2026 to 2036
- Adult
- Pediatrics
- Neonates
- Adult
- Y to o to Y Growth Trend Analysis By Patient Group, 2021 to 2025
- Absolute $ Opportunity Analysis By Patient Group, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Cardiac arrest
- Trauma Management
- Shock Treatment
- Cardiac arrest
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- EMS
- Hospitals
- Ambulatory Surgical Centers
- EMS
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Route of Administration
- By Technology
- By Patient Group
- By Application
- By End User
- Competition Analysis
- Competition Deep Dive
- Teleflex
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Smiths Medical
- Becton Dickinson
- ICU Medical
- Zhejiang Shanshan Broadmed
- Cook Medical
- Medline Industries
- Teleflex
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Patient Group, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Patient Group, 2021 to 2036
- Table 13: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 14: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 19: Latin America Market Value (USD Million) Forecast by Patient Group, 2021 to 2036
- Table 20: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 21: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 25: Western Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 26: Western Europe Market Value (USD Million) Forecast by Patient Group, 2021 to 2036
- Table 27: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 28: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 31: Eastern Europe Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 32: Eastern Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 33: Eastern Europe Market Value (USD Million) Forecast by Patient Group, 2021 to 2036
- Table 34: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 35: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: East Asia Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 38: East Asia Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 39: East Asia Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 40: East Asia Market Value (USD Million) Forecast by Patient Group, 2021 to 2036
- Table 41: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 42: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 43: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: South Asia and Pacific Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 45: South Asia and Pacific Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 46: South Asia and Pacific Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 47: South Asia and Pacific Market Value (USD Million) Forecast by Patient Group, 2021 to 2036
- Table 48: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 49: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 50: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 51: Middle East & Africa Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 52: Middle East & Africa Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 53: Middle East & Africa Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 54: Middle East & Africa Market Value (USD Million) Forecast by Patient Group, 2021 to 2036
- Table 55: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 56: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Route of Administration
- Figure 9: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Technology
- Figure 12: Global Market Value Share and BPS Analysis by Patient Group, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Patient Group, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Patient Group
- Figure 15: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Application
- Figure 18: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by End User
- Figure 21: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 22: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 23: Global Market Attractiveness Analysis by Region
- Figure 24: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 29: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 30: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 31: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 32: North America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Product
- Figure 35: North America Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Route of Administration
- Figure 38: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Technology
- Figure 41: North America Market Value Share and BPS Analysis by Patient Group, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Patient Group, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Patient Group
- Figure 44: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 46: North America Market Attractiveness Analysis by Application
- Figure 47: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 48: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 49: North America Market Attractiveness Analysis by End User
- Figure 50: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 51: Latin America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Product
- Figure 54: Latin America Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Route of Administration
- Figure 57: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Technology
- Figure 60: Latin America Market Value Share and BPS Analysis by Patient Group, 2026 and 2036
- Figure 61: Latin America Market Y-o-Y Growth Comparison by Patient Group, 2026-2036
- Figure 62: Latin America Market Attractiveness Analysis by Patient Group
- Figure 63: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 64: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 65: Latin America Market Attractiveness Analysis by Application
- Figure 66: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 67: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 68: Latin America Market Attractiveness Analysis by End User
- Figure 69: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 70: Western Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Product
- Figure 73: Western Europe Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Route of Administration
- Figure 76: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 77: Western Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 78: Western Europe Market Attractiveness Analysis by Technology
- Figure 79: Western Europe Market Value Share and BPS Analysis by Patient Group, 2026 and 2036
- Figure 80: Western Europe Market Y-o-Y Growth Comparison by Patient Group, 2026-2036
- Figure 81: Western Europe Market Attractiveness Analysis by Patient Group
- Figure 82: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 83: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 84: Western Europe Market Attractiveness Analysis by Application
- Figure 85: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 86: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 87: Western Europe Market Attractiveness Analysis by End User
- Figure 88: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Product
- Figure 92: Eastern Europe Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 93: Eastern Europe Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 94: Eastern Europe Market Attractiveness Analysis by Route of Administration
- Figure 95: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 96: Eastern Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 97: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 98: Eastern Europe Market Value Share and BPS Analysis by Patient Group, 2026 and 2036
- Figure 99: Eastern Europe Market Y-o-Y Growth Comparison by Patient Group, 2026-2036
- Figure 100: Eastern Europe Market Attractiveness Analysis by Patient Group
- Figure 101: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 102: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 103: Eastern Europe Market Attractiveness Analysis by Application
- Figure 104: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 105: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 106: Eastern Europe Market Attractiveness Analysis by End User
- Figure 107: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 108: East Asia Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 109: East Asia Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 110: East Asia Market Attractiveness Analysis by Product
- Figure 111: East Asia Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 112: East Asia Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 113: East Asia Market Attractiveness Analysis by Route of Administration
- Figure 114: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 115: East Asia Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 116: East Asia Market Attractiveness Analysis by Technology
- Figure 117: East Asia Market Value Share and BPS Analysis by Patient Group, 2026 and 2036
- Figure 118: East Asia Market Y-o-Y Growth Comparison by Patient Group, 2026-2036
- Figure 119: East Asia Market Attractiveness Analysis by Patient Group
- Figure 120: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 121: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 122: East Asia Market Attractiveness Analysis by Application
- Figure 123: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 124: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 125: East Asia Market Attractiveness Analysis by End User
- Figure 126: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 127: South Asia and Pacific Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 128: South Asia and Pacific Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 129: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 130: South Asia and Pacific Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 131: South Asia and Pacific Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 132: South Asia and Pacific Market Attractiveness Analysis by Route of Administration
- Figure 133: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 134: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 135: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 136: South Asia and Pacific Market Value Share and BPS Analysis by Patient Group, 2026 and 2036
- Figure 137: South Asia and Pacific Market Y-o-Y Growth Comparison by Patient Group, 2026-2036
- Figure 138: South Asia and Pacific Market Attractiveness Analysis by Patient Group
- Figure 139: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 140: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 141: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 142: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 143: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 144: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 145: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 146: Middle East & Africa Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 147: Middle East & Africa Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 148: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 149: Middle East & Africa Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 150: Middle East & Africa Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 151: Middle East & Africa Market Attractiveness Analysis by Route of Administration
- Figure 152: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 153: Middle East & Africa Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 154: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 155: Middle East & Africa Market Value Share and BPS Analysis by Patient Group, 2026 and 2036
- Figure 156: Middle East & Africa Market Y-o-Y Growth Comparison by Patient Group, 2026-2036
- Figure 157: Middle East & Africa Market Attractiveness Analysis by Patient Group
- Figure 158: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 159: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 160: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 161: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 162: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 163: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 164: Global Market - Tier Structure Analysis
- Figure 165: Global Market - Company Share Analysis

