Lateral Flow Assay Component Market
The lateral flow assay component market is segmented by Product (Membranes, Sample Pads, Conjugate Pads, Absorbent Pads, Laminates, Cassettes and Housings, Other Products), by Application (Clinical Diagnostics, Food Safety and Quality Testing, Veterinary Diagnostics, Drug Development and Quality Testing, Other Applications), and by Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Lateral Flow Assay Component Market Size, Market Forecast and Outlook By FMI
In 2025, the lateral flow assay component market was valued at USD 388.4 million. Based on Future Market Insights' analysis, demand for lateral flow assay components is estimated to grow to USD 404.3 million in 2026 and USD 580.5 million by 2036. FMI projects a CAGR of 4.1% during the forecast period.
An absolute dollar growth of USD 176.2 million between 2026 and 2036 reflects technological intensification in point-of-care diagnostics rather than volume-only test kit expansion. Growth depends on increased conversion from single-plex to multiplex lateral flow formats that demand engineered membranes with distinct capture lines, specialized conjugate pads with multiple dried reagent zones, and advanced laminates supporting complex fluid dynamics.
Summary of the Lateral Flow Assay Component Market
- Market Definition
- The market comprises specialized membranes, pads, laminates, and housings engineered for lateral flow test manufacturing across diagnostic and testing applications.
- Demand Drivers
- Expanding point-of-care testing adoption in infectious disease diagnosis and chronic disease management.
- Increasing food safety testing requirements and veterinary diagnostic utilization.
- Technological advancement enabling multiplex detection and quantitative readouts.
- Key Segments Analyzed
- Product: Membranes lead, accounting for 42.7% share in 2025 due to their critical role in determining test sensitivity and specificity.
- Application: Clinical Diagnostics dominates, driven by rapid test utilization in infectious disease, cardiac marker, and drug screening applications.
- Region: North America leads value, while Asia-Pacific drives volume through test kit manufacturing scale.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant for Healthcare at Future Market Insights, comments: "This updated report captures a market where component value is shifting from commodity nitrocellulose toward engineered materials enabling multiplex detection and quantitative results. Suppliers offering precision membranes with controlled pore size distribution and customized protein binding capacity are securing preferred vendor status with leading assay developers."
- Strategic Implications/Executive Takeaways
- Invest in membrane manufacturing capacity to address supply chain concentration risks and capture growing demand.
- Develop conjugate pad technologies supporting multiple dried reagent stabilization for multiplex assays.
- Pursue ISO 13485 certification to meet IVDR and FDA quality system requirements for component suppliers.
- Methodology
- Built on primary interviews with component manufacturers and assay developers.
- Benchmarked against lateral flow test production data and material consumption analysis.
- Validated using trade data and internal peer review as per FMI modeling standards.
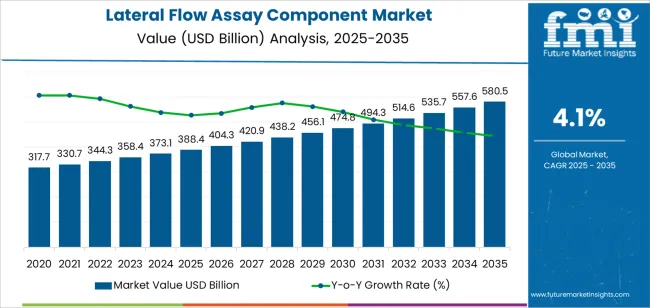
As per FMI, component manufacturers are capturing value not from incremental test strip volumes alone but from engineering complexity embedded in high-performance membranes with controlled pore sizes, streptavidin-coated conjugate pads, and precision-molded cassettes that define modern rapid test performance.
This performance reflects accelerating demand for high-quality components across clinical diagnostics, food safety testing, and veterinary applications. North America leads global expansion supported by advanced diagnostic manufacturing and rigorous quality standards. Europe follows driven by in vitro diagnostic regulation compliance and food safety testing requirements. Asia-Pacific maintains steady growth through rapid test adoption and manufacturing scale-up.
Lateral Flow Assay Component Market Definition
The lateral flow assay component market encompasses the global supply of specialized materials and consumables used in manufacturing lateral flow test strips. Core components include nitrocellulose membranes that immobilize capture reagents and control lines, sample pads that receive and filter patient specimens, conjugate pads containing dried detection reagents, absorbent pads that wick fluid and maintain flow, laminates that provide structural support, and cassettes or housings that protect strips and provide sample addition ports and result windows. These components determine test performance characteristics including sensitivity, specificity, speed, and stability.
Market Inclusions
The report includes global and regional market size estimates in value terms, with a forecast period from 2026 to 2036. It provides segmentation by Product, Application, and Region. The analysis incorporates manufacturing technology trends, material science innovations, regulatory alignment strategies, and performance benchmarking across component formats. Regional demand dynamics, country-level growth rates, and competitive positioning are also evaluated as per FMI methodology.
Market Exclusions
The scope excludes complete lateral flow test kits sold to end-users, assay development services, and reader instruments. It also excludes raw materials not specifically engineered for lateral flow applications such as generic adhesives or untreated papers. Custom components for research-only applications are not considered due to limited commercial scale.
Research Methodology
- Primary Research: Interviews were conducted with lateral flow component manufacturers, assay developers, diagnostic company procurement executives, and quality control specialists.
- Desk Research: Public company filings, IVD industry statistics, patent filings, technical literature, and regulatory guidance documents were reviewed.
- Market-Sizing and Forecasting: A hybrid top-down and bottom-up model was constructed, reconstructing demand from lateral flow test production volumes, component consumption per test, and replacement cycles across application segments.
- Data Validation and Update Cycle: Outputs were subjected to variance testing against production capacity data, trade flow benchmarking, and structured internal review prior to publication.
Lateral Flow Assay Component Market Drivers, Restraints, and Opportunities
Future Market Insights analysis indicates the market has historically existed as a specialized materials category serving diagnostic test manufacturers. Its baseline scale is anchored in infectious disease testing volumes, with respiratory pathogen testing demonstrating the highest utilization rates [1]. As per FMI, the market's valuation is increasingly influenced by multiplex assay adoption and quantitative lateral flow platforms requiring enhanced component performance.
FMI analysts observe a clear stratification between standard components optimized for single-plex qualitative tests and advanced materials engineered for multiplex detection and quantitative readouts. Standard components face pressure where price competition drives commoditization, while growth concentrates in specialized formats where performance characteristics justify premium pricing.
- Point-of-Care Testing Expansion: The global point-of-care diagnostics market expansion drives lateral flow component demand, with rapid tests enabling decentralized testing in physician offices, urgent care centers, and home settings [2]. The COVID-19 pandemic established widespread acceptance of lateral flow testing, creating sustained demand for components serving respiratory pathogen panels. Cardiac marker testing using lateral flow formats expands with aging populations requiring rapid rule-out in emergency settings [3].
- Multiplex Assay Development: Assay developers increasingly pursue multiplex formats detecting multiple pathogens or biomarkers from single specimens, requiring engineered membranes with distinct capture lines and conjugate pads supporting multiple dried reagents [4]. Respiratory panels detecting SARS-CoV-2, influenza A, influenza B, and respiratory syncytial virus simultaneously demonstrate multiplex feasibility. Gastrointestinal pathogen panels and sexually transmitted infection combinations expand multiplex applications [5].
- Quantitative Lateral Flow Platforms: Transition from qualitative visual read to quantitative digital readout creates demand for components with reduced variability and enhanced consistency [6]. Membranes with controlled pore size distribution and uniform protein binding capacity enable reproducible signal generation essential for quantitative interpretation. Reader-compatible cassettes with integrated calibration standards support instrumented result acquisition [7].
- Regulatory Scrutiny and Quality Requirements: The EU In Vitro Diagnostic Regulation imposes stricter requirements on component suppliers, with assay manufacturers requiring documentation of component traceability and performance consistency [8]. ISO 13485 certification for component manufacturers becomes increasingly necessary for supply chain participation. FDA's final rule on laboratory developed tests published in May 2024 will increase scrutiny of test components used in LDTs [9].
- Nitrocellulose Supply Chain Concentration: Nitrocellulose membrane manufacturing remains concentrated among few global suppliers, creating vulnerability to supply disruptions as demonstrated during pandemic demand surges [10]. Assay developers seek secondary qualified sources and invest in membrane characterization capabilities to mitigate single-source risk. Alternative membrane materials including polyethersulfone and nylon emerge for applications requiring distinct protein binding characteristics [11].
Segmental Analysis
Lateral Flow Assay Component Market Analysis by Product
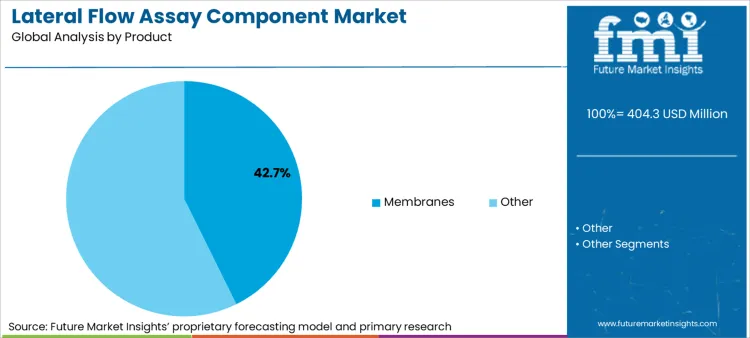
Based on FMI's lateral flow assay component market report, demand for membranes is estimated to lead the product segment, capturing 42.7% of the market share in 2025 and maintaining dominance through 2035. Nitrocellulose membranes serve as the critical component where capture antibodies are immobilized and test lines develop, directly determining assay sensitivity and specificity. The segment's leadership reflects the essential role of membrane selection in assay performance optimization. Growth is supported by increasing multiplex assay development requiring membranes with consistent flow rates and protein binding capacity across multiple capture lines. Membrane manufacturers differentiate through controlled pore size distribution, reduced lot-to-lot variability, and customized protein binding characteristics for specific analyte classes.
Sample pads and conjugate pads represent essential components for specimen processing and reagent delivery, with advanced pads supporting dried reagent stabilization for multiplex assays. Absorbent pads maintain consistent fluid wicking rates critical for reproducible results across test batches. Laminates provide structural support and backing for strip assembly, with pressure-sensitive adhesives requiring compatibility with membrane materials. Cassettes and housings protect test strips while providing sample addition ports and result windows, with precision molding ensuring consistent alignment for reader-based interpretation.
Lateral Flow Assay Component Market Analysis by Application
Clinical diagnostics generates maximum demand for lateral flow assay components, accounting for the largest share driven by infectious disease testing, cardiac marker assays, and drug screening applications. Infectious disease testing for respiratory pathogens, sexually transmitted infections, and tropical diseases represents the highest-volume application segment [12]. Cardiac marker testing for troponin, CK-MB, and myoglobin enables rapid rule-out in emergency settings. Drugs of abuse testing serves workplace screening, forensic applications, and pain management compliance monitoring.
Food safety and quality testing represents the second-largest application segment, with lateral flow formats detecting pathogens including Salmonella, Listeria, and E. coli in food production environments. Mycotoxin testing in grain and feed products utilizes lateral flow technology for rapid screening. Allergen detection tests support food manufacturer compliance with labeling requirements. Veterinary diagnostics applications include infectious disease testing in companion animals and livestock, with rapid test formats enabling point-of-care diagnosis in clinical settings.
Regional Analysis
The lateral flow assay component market is segmented geographically across North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa. Growth dynamics differ based on diagnostic manufacturing concentration, regulatory frameworks, and end-user testing volumes.
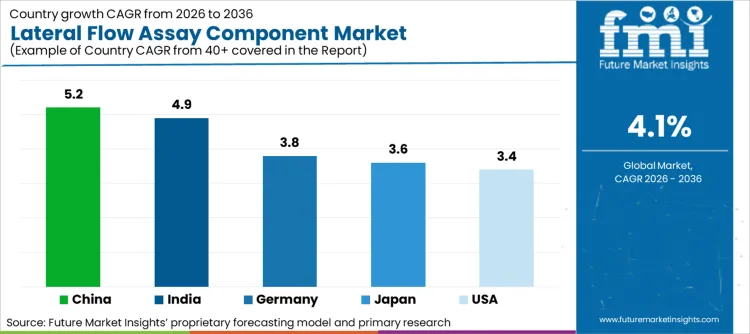
| Country | CAGR (2026-2036) |
|---|---|
| China | 5.2% |
| India | 4.9% |
| Germany | 3.8% |
| Japan | 3.6% |
| United States | 3.4% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Market Analysis
North America represents the largest value market for lateral flow assay components, driven by advanced diagnostic manufacturing, rigorous quality standards, and high testing volumes. The region's diagnostic companies serve global markets with products meeting FDA and international regulatory requirements, demanding components with documented performance and supply chain reliability.
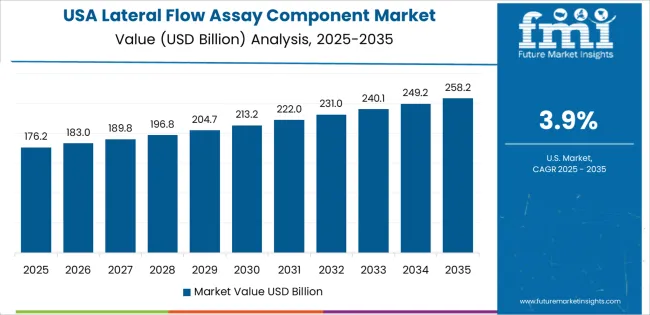
- United States: Demand for lateral flow assay components in the United States is projected to rise at 3.4% CAGR through 2036. The USA market benefits from concentration of leading diagnostic manufacturers including Abbott, QuidelOrtho, and Becton Dickinson with extensive lateral flow product portfolios [13]. FDA's final rule on laboratory developed tests published May 2024 will increase scrutiny of test components used in LDTs, potentially driving component qualification requirements [9].
The National Institutes of Health Rapid Acceleration of Diagnostics initiative continues funding for lateral flow test development, supporting component innovation. USA manufacturers maintain stringent vendor qualification programs requiring ISO 13485 certification and extensive lot-to-lot characterization data.
FMI's analysis of the lateral flow assay component market in North America consists of country-wise assessment including the United States and Canada. Readers can find regulatory guidance tracking, manufacturer concentration analysis, and component qualification requirements.
Europe Market Analysis
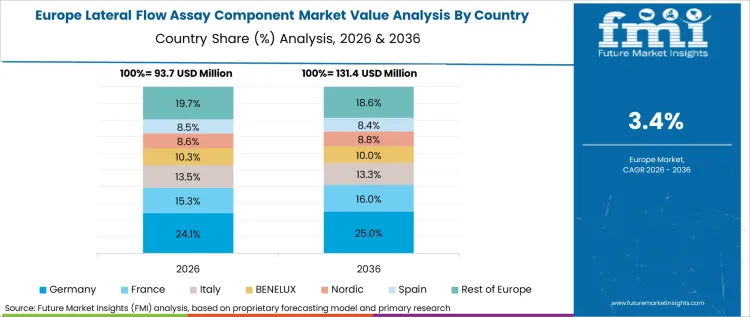
Europe represents a mature market characterized by IVDR compliance requirements, strong diagnostic manufacturing presence, and food safety testing utilization. The region's transition to In Vitro Diagnostic Regulation imposes stricter requirements on component suppliers, with assay developers requiring documented traceability and performance consistency.
- Germany: Demand for lateral flow assay components in Germany is projected to rise at 3.8% CAGR through 2036. Germany's strong diagnostic manufacturing base includes leading companies serving European and global markets. The country's central European location positions it as a distribution hub for component supply across the region.
German manufacturers emphasize quality systems and technical documentation meeting IVDR requirements, driving demand for components with comprehensive characterization data. The country's food safety testing infrastructure utilizes lateral flow formats for pathogen and allergen detection, supporting component demand from test kit manufacturers [8].
FMI's analysis of the lateral flow assay component market in Europe consists of country-wise assessment including Germany, United Kingdom, France, Italy, and Spain. Readers can find IVDR implementation tracking, manufacturer mapping, and quality system requirements.
Asia-Pacific Market Analysis
Asia-Pacific represents the fastest-growing regional market for lateral flow assay components, driven by test kit manufacturing scale, expanding domestic diagnostic production, and cost-competitive component sourcing.
- China: Demand for lateral flow assay components in China is projected to rise at 5.2% CAGR through 2036. China leads regional growth as the world's largest lateral flow test kit manufacturer, supplying products to domestic and export markets. The country's diagnostic industry benefits from government support for in vitro diagnostic development and manufacturing scale-up. Chinese manufacturers produce components for infectious disease tests, drugs of abuse screens, and pregnancy tests serving global demand.
The National Medical Products Administration regulatory framework for IVDs establishes quality requirements that component suppliers must meet for domestic market participation. Chinese companies including Wuxi Diagnostic and Beijing O&D Biotech manufacture lateral flow components serving both domestic and international customers [14].
FMI's analysis of the lateral flow assay component market in Asia-Pacific consists of country-wise assessment including China, India, Japan, and South Korea. Readers can find manufacturing capacity tracking, export flow analysis, and regulatory framework assessment.
- India: Demand for lateral flow assay components in India is projected to rise at 4.9% CAGR through 2036. India's diagnostic manufacturing sector expands under the government's Production Linked Incentive scheme for pharmaceuticals and medical devices. Indian manufacturers produce lateral flow tests for infectious diseases including malaria, dengue, and tuberculosis serving domestic and export markets.
The country's large generic pharmaceutical industry includes diagnostic subsidiaries requiring component supply. Indian companies including J. Mitra and Tulip Diagnostics manufacture lateral flow products requiring consistent component quality.
- Japan: Demand for lateral flow assay components in Japan is projected to rise at 3.6% CAGR through 2036. Japan's diagnostic industry maintains high quality standards with domestic manufacturers serving the country's advanced healthcare system. Japanese companies including Fujifilm and Sekisui Medical manufacture lateral flow products for infectious disease testing and cardiac markers.
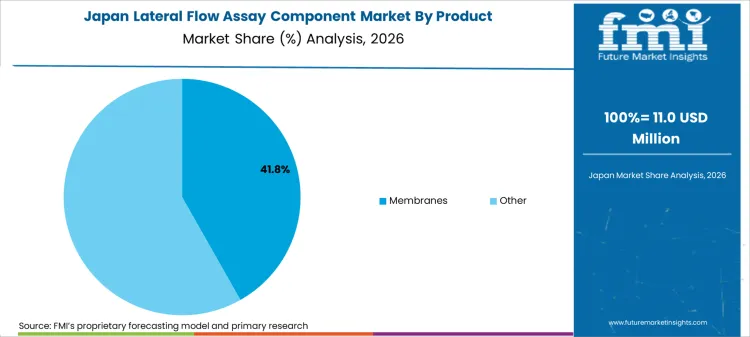
The country's aging population drives chronic disease monitoring applications including HbA1c testing using lateral flow formats. Japanese manufacturers emphasize precision and consistency in component selection, supporting demand for high-quality membranes and pads.
Competitive Landscape for Market Players
Specialized material science companies with deep expertise in membrane technology, filtration media, and diagnostic component manufacturing characterize the lateral flow assay component market. Leading firms are differentiating through product consistency, technical support for assay development, and supply chain reliability.
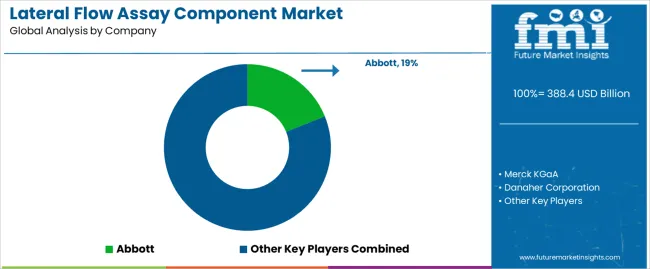
Abbott leverages vertical integration with internal component manufacturing supporting its extensive lateral flow product portfolio while also supplying select components to external customers. Merck KGaA provides membranes and reagents through its MilliporeSigma brand, with extensive technical documentation supporting regulatory submissions. Danaher Corporation's Cytiva business supplies membranes and components for diagnostic manufacturing.
Sartorius Stedim Biotech offers membranes and pads with comprehensive characterization data supporting IVDR compliance. Ahlstrom-Munksjö specializes in sample pads, conjugate pads, and wicking materials optimized for lateral flow applications. DCN Diagnostics provides custom component development and manufacturing services supporting assay developer scale-up. PerkinElmer Inc. supplies components and reagents through its diagnostics business, supporting infectious disease and food safety testing applications.
Buyer behavior in this market reflects dual sourcing strategies to mitigate supply chain risk and rigorous vendor qualification programs requiring extensive lot-to-lot characterization data. Framework agreements with diagnostic manufacturers often include multi-year volume commitments with quality agreements specifying acceptable variability limits and change notification requirements.
Recent Developments
- Merck KGaA - Membrane Capacity Expansion (2025): Merck expanded nitrocellulose membrane manufacturing capacity at its Ireland facility to address supply chain concentration concerns and meet growing demand.
- Sartorius Stedim Biotech - Quality Documentation Enhancement (2025): Sartorius enhanced its component technical documentation packages supporting customer IVDR compliance and regulatory submissions.
- DCN Diagnostics - Custom Component Development (2025): DCN Diagnostics expanded custom conjugate pad development services supporting multiplex assay stabilization requirements.
Key Players in the Lateral Flow Assay Component Market
- Abbott
- Merck KGaA
- Danaher Corporation
- Sartorius Stedim Biotech
- Advanced Microdevice Pvt. Ltd
- Ahlstrom-Munksjö
- Nupore Filtration
- DCN Diagnostics
- Ballya Bio
- PerkinElmer Inc.
Scope of Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 404.3 million (2026) to USD 580.5 million (2036), at a CAGR of 4.1% |
| Market Definition | The lateral flow assay component market comprises specialized membranes, pads, laminates, and housings for lateral flow test manufacturing. |
| Product Segmentation | Membranes, Sample Pads, Conjugate Pads, Absorbent Pads, Laminates, Cassettes and Housings, Other Products |
| Application Segmentation | Clinical Diagnostics, Food Safety and Quality Testing, Veterinary Diagnostics, Drug Development and Quality Testing, Other Applications |
| Region Segmentation | North America, Latin America, Europe, Asia-Pacific, Middle East & Africa |
| Key Countries Covered | United States, Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, China, India, Japan, South Korea, and 40+ countries |
| Key Companies Profiled | Abbott, Merck KGaA, Danaher Corporation, Sartorius Stedim Biotech, Advanced Microdevice Pvt. Ltd, Ahlstrom-Munksjö, Nupore Filtration, DCN Diagnostics, Ballya Bio, PerkinElmer Inc. |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up market modeling validated through primary interviews with component manufacturers and assay developers, supported by test production data and material consumption analysis |
Lateral Flow Assay Component Market Analysis by Segments
Product:
- Membranes
- Sample Pads
- Conjugate Pads
- Absorbent Pads
- Laminates
- Cassettes and Housings
- Other Products
Application:
- Clinical Diagnostics
- Food Safety and Quality Testing
- Veterinary Diagnostics
- Drug Development and Quality Testing
- Other Applications
Region:
- North America
- Latin America
- Europe
- Asia-Pacific
- Middle East & Africa
Bibliography
- Centers for Disease Control and Prevention. (2025). Rapid Diagnostic Tests for Infectious Diseases: Utilization and Impact. CDC.
- World Health Organization. (2025). Point-of-Care Diagnostics Landscape Report 2025. WHO Press.
- American Heart Association. (2024). Rapid Cardiac Marker Testing in Emergency Settings: Clinical Practice Guidelines. AHA Journals.
- Analytical Chemistry. (2025). Multiplex Lateral Flow Assay Development: Challenges and Opportunities. ACS Publications.
- Journal of Clinical Microbiology. (2024). Multiplex Respiratory Pathogen Panel Performance Characteristics. ASM.
- Biosensors and Bioelectronics. (2025). Quantitative Lateral Flow Platforms: Technology Review. Elsevier.
- Clinical Chemistry. (2024). Reader-Based Lateral Flow Interpretation: Analytical Performance. AACC.
- European Commission. (2025). In Vitro Diagnostic Regulation (EU) 2017/746 Implementation Guidance. Official Journal of the European Union.
- USA Food and Drug Administration. (2024). Final Rule: Laboratory Developed Tests. Federal Register.
- Nature Reviews Bioengineering. (2025). Nitrocellulose Membrane Supply Chain Resilience. Nature Publishing.
- Journal of Membrane Science. (2024). Alternative Membrane Materials for Lateral Flow Applications. Elsevier.
- The Lancet Infectious Diseases. (2025). Global Infectious Disease Testing Volumes and Trends. Elsevier.
- USA Food and Drug Administration. (2025). 510(k) Clearances for Lateral Flow Assays Database. FDA.
- National Medical Products Administration. (2025). China IVD Regulatory Framework and Component Requirements. NMPA.
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
This Report Addresses
- Market intelligence to enable structured strategic decision-making across component manufacturers, assay developers, diagnostic company executives, and healthcare investors.
- Market size estimation and 10-year revenue forecasts from 2026 to 2036, including validated CAGR and absolute dollar opportunity sizing.
- Growth opportunity mapping across Product, Application, and regional demand pockets, with emphasis on membrane technology advancement and multiplex assay adoption.
- Segment and regional forecasting covering membranes, pads, and housings, alongside application-specific demand trajectories across clinical diagnostics and food safety testing.
- Competition strategy assessment, including manufacturing capacity investment, quality system certification, and buyer leverage dynamics shaping supply agreements.
- Product and technology tracking focused on nitrocellulose membrane characterization, conjugate pad stabilization, and cassette design for reader compatibility.
- Regulatory policy analysis covering IVDR compliance requirements, FDA LDT final rule implications, and component qualification standards influencing supplier selection.
- Report delivery in PDF, Excel, and presentation-ready formats to support executive planning, capacity investment decisions, and competitive benchmarking.
Frequently Asked Questions
How large is the demand for lateral flow assay components in the global market in 2026?
Demand for lateral flow assay components is estimated to be valued at USD 404.3 million in 2026.
What will be the market size of lateral flow assay components in the global market by 2036?
Market size for lateral flow assay components is projected to reach USD 580.5 million by 2036.
What is the expected demand growth for lateral flow assay components between 2026 and 2036?
Demand for lateral flow assay components is expected to grow at a 4.1% CAGR between 2026 and 2036.
Which product segment is poised to lead global sales?
Membranes are estimated to lead the product segment, holding 42.7% share in 2025, reflecting their critical role in determining test sensitivity and specificity through capture line immobilization.
How significant is the role of clinical diagnostics in driving component demand?
Clinical diagnostics represents the dominant application segment, driven by infectious disease testing, cardiac marker assays, and drug screening applications requiring high-volume test production.
What is driving demand in North America?
Advanced diagnostic manufacturing concentration, rigorous quality standards, and high testing volumes are supporting North American demand growth.
What is the United States growth outlook in this report?
The United States is projected to expand at a 3.4% CAGR during 2026 to 2036.
What is driving demand in Europe?
IVDR compliance requirements, strong diagnostic manufacturing presence, and food safety testing utilization are supporting European market growth.
What is the Germany growth outlook in this report?
Germany is projected to expand at a 3.8% CAGR during 2026 to 2036, supported by diagnostic manufacturing base and central European distribution position.
Does the report cover China in its regional analysis?
Yes, China is included within Asia-Pacific and is a high-growth country market covered in the report.
What is the China growth outlook in this report?
China is projected to expand at a 5.2% CAGR during 2026 to 2036, supported by position as the world's largest lateral flow test kit manufacturer.
Does the report cover India in its regional analysis?
Yes, India is included within Asia-Pacific and is covered in the country-wise assessment.
What is the India growth outlook in this report?
India is projected to expand at a 4.9% CAGR during 2026 to 2036, supported by diagnostic manufacturing expansion under the Production Linked Incentive scheme.
Does the report cover Japan in its regional analysis?
Yes, Japan is included within Asia-Pacific under the regional scope of analysis.
What is the Japan growth outlook in this report?
Japan is projected to expand at a 3.6% CAGR during 2026 to 2036, supported by high-quality diagnostic manufacturing and aging population driving testing demand.
What is driving demand in Asia-Pacific?
Test kit manufacturing scale, expanding domestic diagnostic production, and cost-competitive component sourcing are supporting Asia-Pacific demand growth.
What defines a lateral flow assay component as part of this market?
A component is included if it is specifically engineered for lateral flow test manufacturing including membranes, pads, laminates, and housings used in commercial diagnostic production.
What is included in the scope of this lateral flow assay component market report?
Scope includes market sizing and forecasting for 2026 to 2036, segmentation by product, application, and region, along with competitive assessment and country-wise outlook.
What is excluded from the scope of this report?
The scope excludes complete lateral flow test kits, assay development services, reader instruments, and raw materials not specifically engineered for lateral flow applications.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2026 to 2036
- Membranes
- Membranes
- Y to o to Y Growth Trend Analysis By Product , 2021 to 2025
- Absolute $ Opportunity Analysis By Product , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- Competition Analysis
- Competition Deep Dive
- Abbott
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Merck KGaA
- Danaher Corporation
- Sartorius Stedim Biotech
- Advanced Microdevice Pvt. Ltd
- Ahlstrom-Munksjö
- Nupore Filtration
- DCN Diagnostics
- Abbott
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 3: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 5: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: Latin America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 7: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Western Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 9: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Eastern Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 11: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: East Asia Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 13: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: South Asia and Pacific Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 15: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 16: Middle East & Africa Market Value (USD Million) Forecast by Product , 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Region
- Figure 9: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 10: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 11: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 12: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 17: North America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 18: North America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 19: North America Market Attractiveness Analysis by Product
- Figure 20: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 21: Latin America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 22: Latin America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 23: Latin America Market Attractiveness Analysis by Product
- Figure 24: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 25: Western Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 26: Western Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 27: Western Europe Market Attractiveness Analysis by Product
- Figure 28: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: Eastern Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 30: Eastern Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 31: Eastern Europe Market Attractiveness Analysis by Product
- Figure 32: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: East Asia Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 34: East Asia Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 35: East Asia Market Attractiveness Analysis by Product
- Figure 36: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 37: South Asia and Pacific Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 38: South Asia and Pacific Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 39: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 40: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Middle East & Africa Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 42: Middle East & Africa Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 43: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 44: Global Market - Tier Structure Analysis
- Figure 45: Global Market - Company Share Analysis

