Nasal Sprays Market
The Nasal Sprays Market Is Segmented By Product (Nasal Decongestant Sprays, Nasal Steroid Sprays, Nasal Antihistamine Sprays, Nasal Cromolyn Sodium Sprays, Nasal Anticholinergic Sprays, Nasal Triamcinolone Sprays, Others), Formulation (Aqueous-Based, Hydroalcoholic-Based, Non-Aqueous-Based Solutions, Suspensions, Emulsions, Dry Powder or Microparticles), Packaging (Pressurized Canisters, Pump Bottles), Dosage (Multi-Dose, Bi-Dose, Single-Unit Dose), Distribution Channel (Institutional Sales, Retail Sales), And Indication (Allergic Rhinitis, Nasal Congestion, Sinusitis, Nonallergic Rhinitis, Other Inflammatory Nasal Conditions). Forecast For 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Nasal Sprays Market Forecast and Outlook By FMI
Summary of the Nasal Sprays Market
- Demand and Growth Drivers
- Seasonal allergy burden and repeat congestion treatment support high refill activity in OTC nasal sprays.
- Aqueous spray systems stay commercially important as they pair familiar daily use with stable metered delivery.
- New approvals in emergency allergy care and intranasal therapy are expanding the addressable product bases beyond routine rhinitis relief.
- Product and segment view
- Decongestant sprays remain the leading product group as fast relief drives everyday pharmacy turnover.
- Aqueous formulations stay ahead because approved intranasal products still rely heavily on pump-ready liquid systems.
- Retail sales and multi-dose packs retain a large share due to repeat household use centering on stocked home-care products.
- Geography and competitive outlook
- India and China hold the strongest country growth outlook as pharmacy access and branded intranasal use are still expanding.
- Canada and France show steadier expansion since seasonal allergy treatment is well-established and pharmacy demand is steady.
- Competition centers on brand familiarity and channel depth, while new intranasal launches add value in specialty uses.
- Analyst opinion
- Sabyasachi Ghosh, Principal Consultant for Healthcare observe that, "Nasal spray demand is concentrated around products that solve everyday congestion and rhinitis symptoms without adding complexity for households or prescribers. Brands that keep strong shelf presence and dependable refill availability can still defend share in mature spray lines. Newer intranasal launches in allergy rescue and specialty care add value even though near-term revenue is influenced mainly from pump-based products used in repeat home care."
- Nasal Sprays Market value analysis
- Repeat purchase in allergy and congestion care keeps a large base of annual demand moving through retail pharmacies and drug stores.
- Value is still concentrated in easy-use spray formats as daily home treatment favors familiar metered delivery and refill continuity.
- Retail sales retain a large revenue block as most routine symptom relief purchases move through pharmacy-led self-care.
- Price discipline in mature OTC lines still limits faster value expansion in this market across the full forecast period.
Nasal Sprays Market Definition
The Nasal Sprays market covers intranasal pharmaceutical spray products used for local or systemic therapeutic delivery. It includes congestion relief sprays and allergy control sprays, along with intranasal therapies used in inflammatory nasal conditions or selected specialty indications.
Nasal Sprays Market Inclusions
Market scope includes finished nasal spray products sold through retail pharmacies and drug stores, along with hospital channels and specialty care settings. Counted products include decongestant and steroid sprays, while other covered sprays include antihistamine and cromolyn sodium lines and anticholinergic and triamcinolone sprays in liquid or dry intranasal formats.
Nasal Sprays Market Exclusions
The scope excludes oral allergy medicines and nebulized inhalation therapies, along with saline irrigation systems sold outside spray presentation. It also excludes injectable allergy therapies and nasal implants, along with respiratory devices that are not commercialized as nasal spray products.
Nasal Sprays Market Research Methodology
- Primary research: Checked physician use patterns and pharmacy movement, while also reviewing intranasal product access across prescription channels and OTC channels.
- Desk research: Surveyed regulatory guidance and scientific literature, along with official product information and company release activity dated through 2025.
- Market sizing and forecasting: Analysis were built from symptom prevalence and refill cycles, along with formulation mix and packaging norms across covered countries.
- Data validation: Used source-based product and formulation shares, while packaging and dosage splits were modelled from device use and channel and indication splits were modelled from refill behavior.
Why Is The Nasal Sprays Market Growing?
- Allergy and congestion treatment generate repeat spray purchases across pharmacy-led self-care in most covered countries.
- Aqueous pump systems sustain lead as approved products rely on simple daily-use delivery.
- Higher value intranasal launches are adding revenue while routine rhinitis and congestion products protect the core unit base.
- Retail availability keeps household adoption strong in markets that already use branded and generic spray lines.
Household treatment of congestion and nasal allergy treatment supports a durable refill cycle for nasal sprays. Fast local relief keeps these products close to routine home-care use, and that pattern protects volume in retail channels. Prescribers also continue to use intranasal steroids and antihistamine sprays for symptom control in recurrent nasal conditions.
Product mix is improving as intranasal delivery enters higher value uses such as allergy rescue and specialized prescription care. Launches from Milestone and Lupin support that view, and ARS Pharma adds proof thatnasal drug delivery can move into emergency allergy treatment. That expansion adds value to the route without cutting the steady unit base built by decongestant and rhinitis products.
Market Segmentation Analysis
- Decongestant sprays are expected to account for 46.1% of product demand in 2026 as fast relief products still dominate routine symptom-led purchases.
- Aqueous formulations are projected to contribute 54.1% of formulation demand in 2026 since most marketed nasal sprays rely on pump-ready liquid systems.
- Pump bottles are expected to represent 78.0% of packaging demand in 2026 due to non-propellant metered delivery defining the standard spray format.
- Multi-dose formats are expected to make up 74.0% of dosage demand in 2026 as allergy and rhinitis treatment favors repeat daily use.
The market is segmented by product, including nasal decongestant, steroid, antihistamine, cromolyn sodium, anticholinergic, triamcinolone, and other nasal sprays, with decongestants leading due to fast‑acting relief demand. It is further segmented by formulation, covering aqueous‑based, hydroalcoholic‑based, non‑aqueous solutions, suspensions, emulsions, and dry powder or microparticles, where aqueous systems remain dominant. By packaging and dosage, the market comprises pump bottles and pressurized canisters, along with multi‑dose, bi‑dose, and single‑unit dose formats, supported by repeat household use. The market is also segmented by distribution channel and indication, spanning retail and institutional sales and addressing allergic rhinitis, nasal congestion, sinusitis, nonallergic rhinitis, and other inflammatory nasal conditions.
Nasal Sprays Market analysis by Product
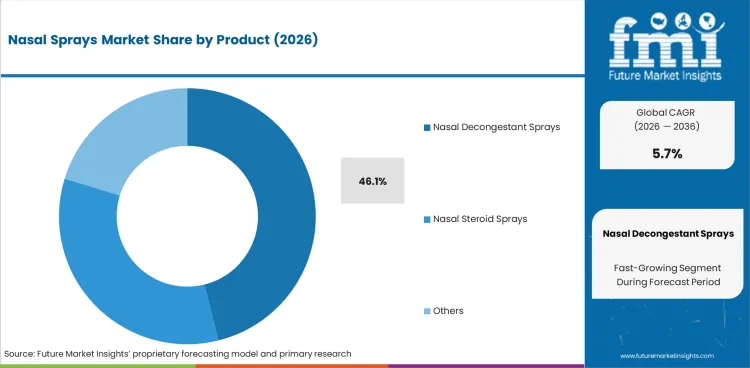
- Decongestant sprays are expected to account for 46.1% of product demand in 2026 due to nasal blockage driving fast relief purchases across common upper-airway complaints.
- OTC turnover supports this lead because decongestant products stay easy to recognize and easy to restock in routine pharmacy care.
Insights into the Nasal Steroid Sprays segment
- Nasal steroid sprays should advance at asteady paceas recurrent allergy control requires longer-duration symptom management.
- Prescriber use and repeat refill demand support this segment in chronic rhinitis treatment even without matching decongestant unit velocity.
Nasal Sprays Market analysis by Formulation
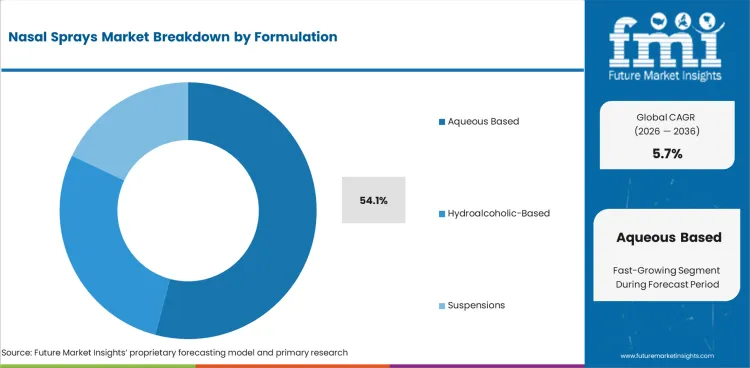
- Aqueous formulations are projected to contribute 54.1% of formulation demand in 2026 as most approved sprays still use pump-ready liquid systems.
- Formulation familiarity keeps this format central to steroid and antihistamine sprays, while drug formulation work also supports its long commercial lead.
Insights into the Dry Powder or Microparticles segment
- Dry powder or microparticle formats should expand from a smaller base as developers pursue shelf-life gains and specialized intranasal delivery.
- Commercial scale is still limited, yet this format keeps strategic interest in advanced nasal therapy programs with storage and formulation upside.
Nasal Sprays Market analysis by Packaging
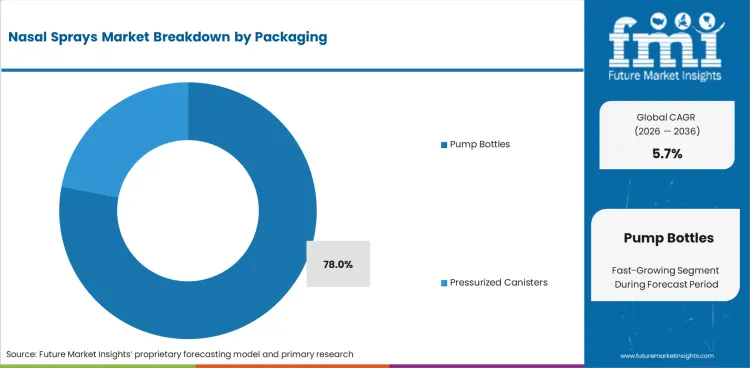
- Pump bottles are expected to account for 78.0% of packaging demand in 2026 as most marketed sprays still use standard metered pump hardware.
- Daily household use reinforces this lead, and alignment with aqueous spray systems keeps pump delivery firmly in the leading position across mainstream sprays.
Insights into the Pressurized Canisters segment
- Pressurized canisters retain a smaller place in nasal sprays because propellant-based delivery serves fewer mainstream product lines.
- Their role is more selective and remains tied to product designs that need a distinct spray profile and can align with electronic nasal spray devices in premium settings.
Nasal Sprays Market analysis by Dosage
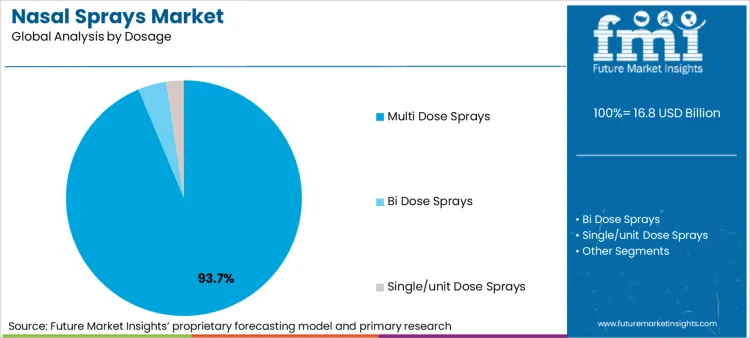
- Multi-dose formats are expected to contribute 74.0% of dosage demand in 2026 as routine allergy and congestion care centers on repeat household use.
- Refill continuity and lower pack cost per actuation keep multi-dose packs ahead in pharmacy shelves and home medicine cabinets.
Insights into the Single-Unit Dose segment
- Single-unit dose formats should record faster value growth due to hygiene control and travel convenience influencing selected prescription and pediatric uses.
- This channel also benefits from rescue therapy use and specialist supervision in selected indications outside routine self-care.
Nasal Sprays Market analysis by Distribution Channel
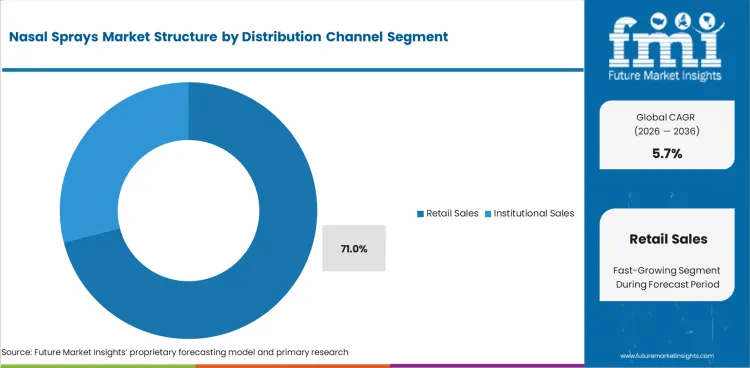
- Retail sales are expected to represent 71.0% of distribution demand in 2026 since most congestion and allergy sprays expand in demand through pharmacy-led self-care.
- Drug stores and retail pharmacy chains sustains this lead by influencing impulse purchase and regular repeat purchase.
Insights into the Institutional Sales segment
- Nasal congestion remains a volume driver as short-duration relief purchases support decongestant sales in retail outlets.
- Unit demand stays high in this sales type, even though treatment duration is often shorter than allergy control therapy in long-use products.
Nasal Sprays Market analysis by Indication
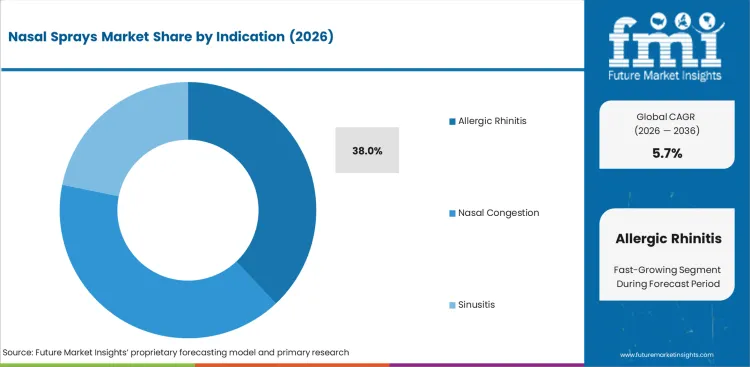
- Allergic rhinitis is expected to account for 38.0% of indication demand in 2026 due to recurrent symptom control supporting repeat use over longer periods.
- Steroid and antihistamine sprays strengthen this lead since they are aligned to the ongoing allergy management.
Insights into the Nasal Congestion segment
- Nasal congestion remains a major volume driver because short-duration relief purchases still support strong decongestant movement in retail outlets.
- Demand for the product stays high in this sector, even though treatment duration is often shorter than allergy control therapy.
Nasal Sprays Market Drivers, Restraints, and Opportunities
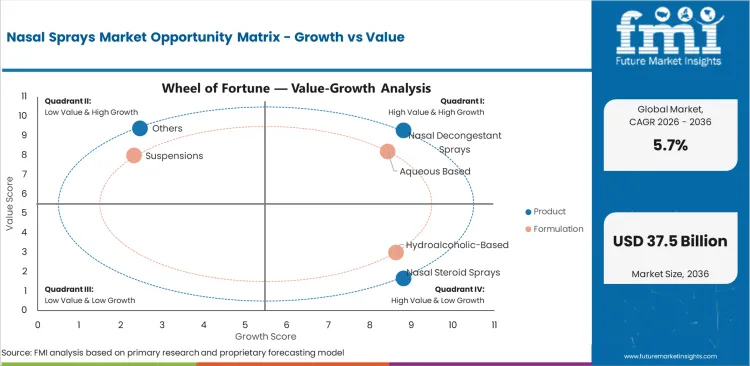
- Repeat allergy burden and routine congestion relief still provide the main demand base for nasal sprays.
- Mature OTC lines face price pressure because generic products and shelf comparison keep brand premiums in check.
- Specialty intranasal launches are opening higher-value uses in emergency allergy care, and they are also expanding prescription nasal therapy.
Repeat Allergy and Congestion Care
Community pharmacy demand still starts with products that solve recurring nasal symptoms in a simple way. Allergic rhinitis and congestion episodes send households back to the same spray formats several times across a year. That repeat use pattern influences stable shelf movement for decongestants and steroids, while antihistamine sprays also benefit from recurring symptom control. Revenue growth in this market starts from everyday symptom management.
Price Comparison in Mature OTC Lines
Retail shelves place many nasal sprays side by side, and that setup keeps price comparison active in mature product lines. Generic competition is especially strong in standard congestion and rhinitis products with familiar active ingredients. Pharmacy staff can also direct households toward lower priced options that deliver similar symptom relief. That pressure limits pricing power for branded suppliers across established spray lines in mature OTC markets.
Higher Value Intranasal Expansion
Intranasal delivery is gaining commercial growth in prescription products that target acute events or specialty care use. ARS Pharma and Milestone Pharmaceuticals added direct proof of that shift, and Lupin added another example through a prescription nasal launch in the United States. That opens a wider revenue pool for suppliers that can pair regulatory execution with dependable spray device performance in drug delivery technology.
Regional Market Analysis
.webp)
| Country | CAGR |
|---|---|
| United States | 4.2% |
| Canada | 5.8% |
| Germany | 4.7% |
| France | 5.5% |
| Spain | 5.8% |
| China | 6.7% |
| India | 7.5% |
Source. Future Market Insights analysis based on the supplied market model and covered country narratives.
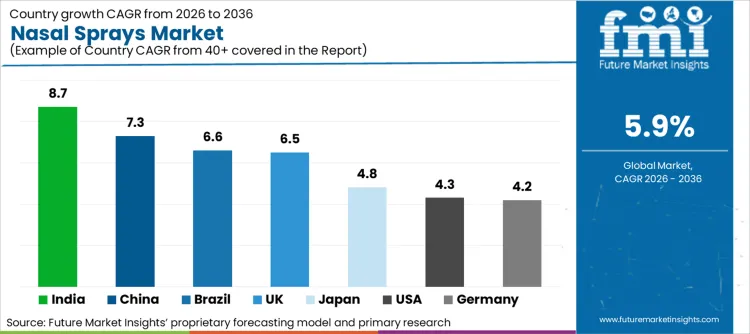
Analysis of Nasal Sprays Market by key countries
- India is expected to record the fastest growth at 7.5% CAGR through 2036 as branded nasal sprays are reaching more urban and semi-urban pharmacies.
- China follows at 6.7% CAGR through 2036 due to city pharmacy networks and respiratory care demand expanding at a healthy pace.
- Spain and Canada are each expected to post 5.8% CAGR through 2036 since seasonal allergy treatment is well embedded in pharmacy demand and repeat household use.
- Germany at 4.7% CAGR and the United States at 4.2% CAGR stay closer to mature market levels as repeat use is strong even though pricing stays disciplined.
Country differences reflect pharmacy access and branded intranasal familiarity, along with the mix between everyday self-care and structured prescription use. India and China stay above the global growth rate because distribution reach and product awareness still have room to improve. Spain and Canada hold a middle position because allergy treatment is well established and seasonal demand stays active.
Demand outlook for Nasal Sprays in the United States
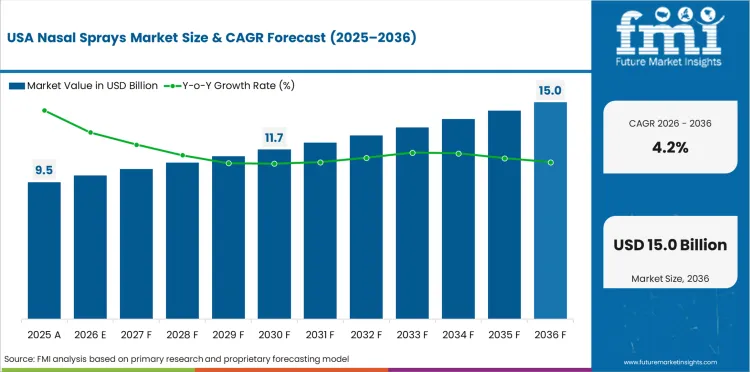
OTC replacement demand remains central in the United States, and mature pharmacy access influences steady spray demand growth. Demand for nasal sprays in the United States is expected to rise at a 4.2% CAGR through 2036 as branded and generic products both stay easy to access. Allergy awareness and chronic rhinitis treatment keep repeat purchase activity present across the year in retail self-care.
- Large pharmacy chains keep shelf access wide for both branded and generic sprays across allergy and congestion care.
- Allergy treatment awareness supports steady refill demand in recurrent rhinitis care across adult and pediatric use.
- Price comparison remains active in mature OTC lines that carry similar active ingredients across major outlets.
Future outlook for Nasal Sprays in Canada
Retail pharmacy access is strong in Canada, and seasonal allergy demand supports stable spray use through the year. Canada is projected to record a 5.8% CAGR in nasal sprays during the forecast period because local symptom relief products are widely available in community outlets. Households also continue to favor simple intranasal options for congestion and allergy care in daily self-care use. That pattern supports a healthier pace than the United States and keeps Canada in the upper middle tier of growth.
- Retail pharmacy base supports repeat household replenishment across common allergy and congestion treatment lines in this country.
- Seasonal allergy treatment sustains a stable demand in peak pollen periods across community pharmacy channels in major provinces.
- Simple symptom relief products stay well aligned with self-care use in this market across routine household treatment.
Opportunity analysis of Nasal Sprays in Germany
Germany has a well established pharmacy base, and structured allergy treatment keeps nasal sprays in routine use. The market for nasal sprays in Germany is set to expand at a 4.7% CAGR through 2036 because mature branded and generic lines are already well distributed. Steroid and antihistamine sprays hold a steady place in recurrent rhinitis care across routine pharmacy treatment.
- Organized pharmacy access supports a stable replenishment cycle across standard nasal spray products in this country.
- Allergy management remains well embedded in routine intranasal care across physician and household use in Germany.
- Mature product familiarity limits a sharper value rise in this established market over the forecast period.
Future outlook for Nasal Sprays in France
Persistent rhinitis treatment supports a dependable demand base in France, especially in retail pharmacy channels. France is likely to expand at a 5.5% CAGR through 2036 ashouseholds and prescribers both continue to use nasal therapies in regular allergy care. Retail access and repeat use of intranasal products keep refill frequency healthy across everyday symptom control. This creates a stable medium-term outlook for spray suppliers serving both branded and generic pharmacy channels.
- Recurring rhinitis care supports regular refill activity across branded and generic nasal spray products in France.
- Retail pharmacy access keeps product availability strong across both urban and smaller local outlets in this country.
- Intranasal treatment familiarity supports steady household use through the full allergy cycle in French pharmacy care.
In-depth analysis of Nasal Sprays in Spain
High seasonality in allergy complaints promotes nasal spray demand in Spain, and pharmacy buying habits remain well established. Spain is expected to register a 5.8% CAGR in nasal sprays through 2036 sincelocal delivery products still align closely with seasonal symptom relief needs. Decongestant and allergy sprays both benefit from this recurring demand cycle during high pollen periods.
- Seasonal allergy burden keeps spray turnover high in peak periods across retail pharmacy channels in Spain.
- Pharmacy purchase habits remain favorable for intranasal relief products in both branded and generic lines across the country.
- Local delivery still suits quick symptom management in routine household care across common seasonal allergy episodes.
Sales analysis of Nasal Sprays in China
China is adding stronger urban pharmacy reach, and respiratory care products are gaining wider branded presence in city channels. China is likely to see nasal sprays advance at a 6.7% CAGR through 2036 because household access to proven nasal antihistamines and other intranasal therapies is still improving. Allergy and congestion treatment demand is also rising with expanding retail health access in urban settings.
- Urban pharmacy expansion supports stronger product reach across allergy and congestion spray lines in major Chinese cities.
- Branded intranasal use is still widening in major cities through retail health outlets and growing pharmacy access.
- Respiratory care demand continues to support spray adoption across a large urban user base in this market.
In-depth analysis of Nasal Sprays in India
India still offers room for deeper branded nasal spray penetration, especially beyond large metro markets. India is expected to post a 7.5% CAGR in nasal sprays through 2036 due to urban and semi-urban pharmacy networks carrying more allergy treatment products and congestion products. Growing awareness of intranasal relief is also helping repeat purchase in everyday respiratory care across expanding self-care channels.
- Semi-urban pharmacy reach is improving branded spray availability across a wider retail footprint in India.
- Allergy and congestion care demand is rising in daily self-care channels with stronger product access across major states.
- Repeat purchase potential remains high in underpenetrated markets across this country during the forecast period.
Competitive landscape and strategic positioning
- Competition is moderately concentrated around branded suppliers with strong pharmacy reach and established intranasal product portfolios.
- Large companies hold an advantage as shelf access and clinician familiarity support repeat purchase, while refill visibility also helps them defend mature spray lines.
- Smaller or regional suppliers can win in generics and focused prescription products, yet entry still needs regulatory execution and steady channel supply.
Brand familiarity plays a major role in nasal sprays as households often repurchase names they already know from prior allergy or congestion treatment. Large suppliers benefit from long pharmacy relationships and a strong OTC presence, which helps them protect refill volume in mature spray lines. That advantage is strongest in decongestant and allergy control products that rely on shelf recognition more than on complex clinical education.
Route innovation is adding a second layer of competition in nasal delivery acrosshigher-value therapy areas. ARS Pharma used intranasal epinephrine to show that nasal delivery can move into emergency allergy care, while Milestone Pharmaceuticals used CARDAMYST to enter acute prescription therapy through a self-administered spray format.
Generic and focused prescription suppliers still have room to compete in selected product lines across nasal care. Lupin showed that path in 2025 with its United States launch of ipratropium bromide nasal solution for rhinorrhea relief. Regional challengers can also gain ground through lower priced allergy products or by focusing on hospital and clinic use linked to migraine nasal sprayand other specialty lines. Even so, steady channel supply and shelf continuity remain hard entry barriers in this market.
Key companies in the Nasal Sprays Market
Competition in nasal sprays depends on channel depth and brand familiarity, along with intranasal product breadth.
- Global leaders: IncludesGlaxoSmithKline and Bayer AG, while Sanofi and Pfizer also carry broad international reach in allergy and respiratory care. Their strength comes from branded product equity and large pharmacy relationships, while financial depth supports line extensions and regulatory work in new nasal indications.
Established intranasal and consumer-health portfolios: Include Janssen Pharmaceuticals and Merck & Co., while Boehringer Ingelheim maintains market presence by respiratory franchise depth. These companies benefit from physician awareness and channel continuity, while their portfolio mix supports intranasal products in routine care and selected specialty uses.
Value based players: Include Viatris and Sandoz, and both support price-led competition through mature spray lines and wide generic access. Their route to share is strongest in established nasal products that face high shelf comparison and require dependable supply rather than large promotional spending.
Competitive benchmarking: Nasal Sprays Market
| Company | Brand Strength | Pharmacy Reach | Intranasal Depth | Geographic Footprint |
|---|---|---|---|---|
| GlaxoSmithKline | High | High | High | Global |
| Bayer AG | High | High | Medium | Global |
| Sanofi | High | High | Medium | Global |
| Pfizer Inc. | Medium | High | Medium | Global |
| AstraZeneca | Medium | Medium | Medium | Global |
| Viatris Inc. | Medium | High | Low | Global |
| Sandoz | Medium | High | Low | Global |
Source. Future Market Insights analysis based on the supplied market model and covered country narratives.
Key developments in the Nasal Sprays Market
- In July 2025, Lupin launched ipratropium bromide nasal solution in the United States for rhinorrhea linked to allergic and nonallergic perennial rhinitis.
- In November 2025, Mucinex introduced Clear and Cool Saline Nasal Spray to target drug-free congestion relief in retail self-care channels.
- In December 2025, Milestone Pharmaceuticals announced FDA approval of CARDAMYST nasal spray for acute symptomatic PSVT conversion in adults.
Key players in the Nasal Sprays Market
Major global players
- Janssen Pharmaceuticals, Inc.
- GlaxoSmithKline
- AstraZeneca
- Pfizer Inc.
- Bayer AG
- Sanofi
- Merck & Co., Inc.
Branded and generic portfolio participants
- Novartis AG (Sandoz)
- Boehringer Ingelheim International GmbH
- Viatris Inc. (Mylan N.V)
Report Scope and Coverage
| Metric | Value |
|---|---|
| Quantitative Units | USD 19.3 billion in 2025 to USD 35.6 billion in 2036 at a 5.7% CAGR. |
| Market Definition | Intranasal pharmaceutical spray products used for local or systemic therapeutic delivery in congestion relief, allergy care, inflammatory nasal conditions, and selected specialty indications. |
| Product Segmentation | Nasal Decongestant Sprays, Nasal Steroid Sprays, Nasal Antihistamine Sprays, Nasal Cromolyn Sodium Sprays, Nasal Anticholinergic Sprays, Nasal Triamcinolone Sprays, Others. |
| Formulation Segmentation | Aqueous-Based, Hydroalcoholic-Based, Non-Aqueous-Based Solutions, Suspensions, Emulsions, Dry Powder or Microparticles. |
| Packaging, Dosage, and Channel | Pressurized Canisters, Pump Bottles, Multi-Dose, Bi-Dose, Single-Unit Dose, Institutional Sales, and Retail Sales. |
| Indication Coverage | Allergic Rhinitis, Nasal Congestion, Sinusitis, Nonallergic Rhinitis, and Other Inflammatory Nasal Conditions. |
| Countries Covered | United States, Canada, Germany, France, Spain, China, India, Japan, Poland, and 40 plus countries. |
| Approach | Hybrid top-down and bottom-up modelling validated through physician input, pharmacy movement, refill patterns, formulation mix, and packaging norms. |
Source. Future Market Insights analysis based on the supplied market model and covered country narratives.
Nasal Sprays Market breakdown by Product, Formulation, Packaging, Dosage, Distribution Channel, and Indication
Nasal Sprays Market Segmented by Product:
- Nasal Decongestant Sprays
- Nasal Steroid Sprays
- Nasal Antihistamine Sprays
- Nasal Cromolyn Sodium Sprays
- Nasal Anticholinergic Sprays
- Nasal Triamcinolone Sprays
- Others
Nasal Sprays Market Segmented by Formulation:
- Aqueous-Based
- Hydroalcoholic-Based
- Non-Aqueous-Based Solutions
- Suspensions
- Emulsions
- Dry Powder or Microparticles
Nasal Sprays Market Segmented by Packaging:
- Pressurized Canisters
- Pump Bottles
Nasal Sprays Market Segmented by Dosage:
- Multi-Dose
- Bi-Dose
- Single-Unit Dose
Nasal Sprays Market Segmented by Distribution Channel:
- Institutional Sales
- Retail Sales
Nasal Sprays Market Segmented by Indication:
- Allergic Rhinitis
- Nasal Congestion
- Sinusitis
- Nonallergic Rhinitis
- Other Inflammatory Nasal Conditions
Nasal Sprays Market by Region:
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN Countries
- Australia and New Zealand
- Rest of South Asia
- Western Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- BENELUX
- Nordic Countries
- Rest of Western Europe
- Eastern Europe
- Russia
- Hungary
- Poland
- Rest of Eastern Europe
- Middle East and Africa
- Saudi Arabia
- Türkiye
- South Africa
- Other African Union
- Rest of Middle East and Africa
Research Sources and Bibliography
- ARS Pharmaceuticals, Inc. (2025, December 29). neffy® (epinephrine nasal spray) approved in China as the first and only community use epinephrine product for the treatment of allergic reactions (anaphylaxis).
- USA Food and Drug Administration. (2024, November 4). Scientific review supporting proposed administrative order OTC000036: Amending over-the-counter monograph M012: Cold, cough, allergy, bronchodilator, and antiasthmatic drug products for over-the-counter human use.
- Alkalash, S. H., Alsokani, A. M., Alrezqi, A. A. A., & Alfaqih, K. A. M. (2024). Utilization pattern and related knowledge of nasal decongestants among the general population in Al-Qunfudah Governorate, Saudi Arabia: A community-based cross-sectional study. Cureus, 16(1), e53006.
- Forbes, B., Goodacre, L., Lansley, A. B., Martin, A. R., Palmer, H., Patterson, C., Roe, C., & Scherließ, R. (2025). Advances in nasal biopharmaceutics to support product development and therapeutic needs. Pharmaceutics, 17(10), 1321.
- Milestone Pharmaceuticals Inc. (2025, December 12). Milestone receives FDA approval of CARDAMYST™ (etripamil) as first and only self-administered nasal spray for adults with paroxysmal supraventricular tachycardia (PSVT).
- Reckitt. (2025, November 20). Mucinex® introduces new Clear & Cool sinus nasal spray, delivering instant cooling relief for congestion.
- Lupin Limited. (2025, July 4). Lupin launches ipratropium bromide nasal solution (nasal spray), 0.03% and 0.06% in the United States.
The bibliography is provided for reader reference.
This Report Answers
- What size is the Nasal Sprays Market in 2026 and how large can it become by 2036?
- How fast is Nasal Sprays Market demand expected to expand from 2026 through 2036 under the baseline forecast case?
- Which product and formulation segments and which packaging and dosage and sales channel and indication segments lead Nasal Sprays Market demand in 2026?
- Why do decongestant sprays and aqueous formulations retain the largest 2026 share in the Nasal Sprays Market?
- How are pharmacy access and refill behavior shaping volume and value in the Nasal Sprays Market?
- Which countries are expected to post the fastest growth in the Nasal Sprays Market through 2036?
- How are new intranasal launches changing the longer-term commercial outlook of the Nasal Sprays Market?
- Which companies hold the clearest brand and channel advantages within the Nasal Sprays Market during the forecast period?
Frequently Asked Questions
How large is the nasal sprays market in 2026?
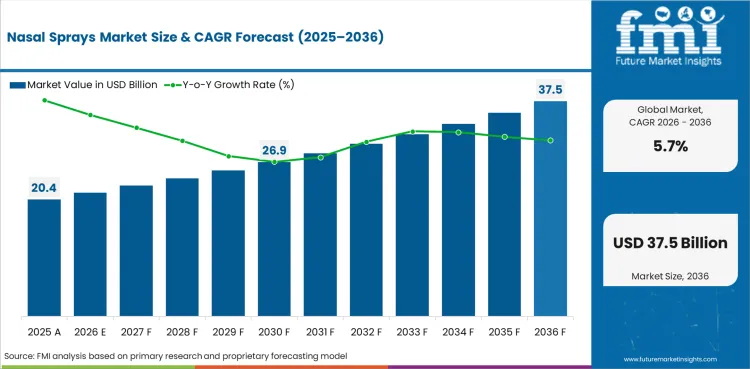
The nasal sprays market is expected to reach USD 20.4 billion in 2026, supported by strong allergy treatment demand and repeat congestion care purchases.
How large can the nasal sprays market become by 2036?
The nasal sprays market is projected to reach USD 35.6 billion by 2036 as refill demand and new intranasal launches keep expansion steady.
What growth rate is expected in the nasal sprays market through 2036?
The nasal sprays market is projected to expand at a 5.7% CAGR from 2026 to 2036, reflecting stable OTC demand and route expansion.
Which product segment leads the nasal sprays market by 2026?
Decongestant sprays are expected to lead the nasal sprays market in 2026 with a 46.1% share because quick relief still drives routine purchases.
Which formulation segment leads the nasal sprays market by 2026?
Aqueous formulations are expected to lead the nasal sprays market in 2026 with a 54.1% share because pump-ready liquids still dominate approvals.
Which packaging segment leads the nasal sprays market by 2026?
Pump bottles are expected to lead the nasal sprays market in 2026 with a 78.0% share because standard metered delivery still dominates.
Which country records the fastest growth in the nasal sprays market?
India is expected to record the fastest nasal sprays market growth with a 7.5% CAGR through 2036 as pharmacy reach expands further.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2026 to 2036
- Nasal Decongestant Sprays
- Nasal Steroid Sprays
- Others
- Nasal Decongestant Sprays
- Y to o to Y Growth Trend Analysis By Product , 2021 to 2025
- Absolute $ Opportunity Analysis By Product , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Formulation
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Formulation, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Formulation, 2026 to 2036
- Aqueous Based
- Hydroalcoholic-Based
- Suspensions
- Aqueous Based
- Y to o to Y Growth Trend Analysis By Formulation, 2021 to 2025
- Absolute $ Opportunity Analysis By Formulation, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Packaging
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Packaging, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Packaging, 2026 to 2036
- Pump Bottles
- Pressurized Canisters
- Pump Bottles
- Y to o to Y Growth Trend Analysis By Packaging, 2021 to 2025
- Absolute $ Opportunity Analysis By Packaging, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Dosage
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Dosage, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Dosage, 2026 to 2036
- Multi-Dose
- Bi-Dose
- Single-Value (USD Million) Dose
- Multi-Dose
- Y to o to Y Growth Trend Analysis By Dosage, 2021 to 2025
- Absolute $ Opportunity Analysis By Dosage, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Retail Sales
- Institutional Sales
- Retail Sales
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Indication, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Indication, 2026 to 2036
- Allergic Rhinitis
- Nasal Congestion
- Sinusitis
- Allergic Rhinitis
- Y to o to Y Growth Trend Analysis By Indication, 2021 to 2025
- Absolute $ Opportunity Analysis By Indication, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Formulation
- By Packaging
- By Dosage
- By Distribution Channel
- By Indication
- Competition Analysis
- Competition Deep Dive
- Janssen Pharmaceuticals, Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- GlaxoSmithKline
- AstraZeneca
- Pfizer Inc.
- Bayer AG
- Sanofi
- Merck & Co., Inc.
- Janssen Pharmaceuticals, Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Formulation, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Dosage, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 7: Global Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Formulation, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Dosage, 2021 to 2036
- Table 13: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 14: North America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Formulation, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 19: Latin America Market Value (USD Million) Forecast by Dosage, 2021 to 2036
- Table 20: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 21: Latin America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Formulation, 2021 to 2036
- Table 25: Western Europe Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 26: Western Europe Market Value (USD Million) Forecast by Dosage, 2021 to 2036
- Table 27: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 28: Western Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 31: Eastern Europe Market Value (USD Million) Forecast by Formulation, 2021 to 2036
- Table 32: Eastern Europe Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 33: Eastern Europe Market Value (USD Million) Forecast by Dosage, 2021 to 2036
- Table 34: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 35: Eastern Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: East Asia Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 38: East Asia Market Value (USD Million) Forecast by Formulation, 2021 to 2036
- Table 39: East Asia Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 40: East Asia Market Value (USD Million) Forecast by Dosage, 2021 to 2036
- Table 41: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 42: East Asia Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 43: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: South Asia and Pacific Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 45: South Asia and Pacific Market Value (USD Million) Forecast by Formulation, 2021 to 2036
- Table 46: South Asia and Pacific Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 47: South Asia and Pacific Market Value (USD Million) Forecast by Dosage, 2021 to 2036
- Table 48: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 49: South Asia and Pacific Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 50: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 51: Middle East & Africa Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 52: Middle East & Africa Market Value (USD Million) Forecast by Formulation, 2021 to 2036
- Table 53: Middle East & Africa Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 54: Middle East & Africa Market Value (USD Million) Forecast by Dosage, 2021 to 2036
- Table 55: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 56: Middle East & Africa Market Value (USD Million) Forecast by Indication, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by Formulation, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Formulation, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Formulation
- Figure 9: Global Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Packaging, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Packaging
- Figure 12: Global Market Value Share and BPS Analysis by Dosage, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Dosage, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Dosage
- Figure 15: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Distribution Channel
- Figure 18: Global Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Indication
- Figure 21: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 22: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 23: Global Market Attractiveness Analysis by Region
- Figure 24: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 29: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 30: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 31: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 32: North America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Product
- Figure 35: North America Market Value Share and BPS Analysis by Formulation, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Formulation, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Formulation
- Figure 38: North America Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Packaging, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Packaging
- Figure 41: North America Market Value Share and BPS Analysis by Dosage, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Dosage, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Dosage
- Figure 44: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 45: North America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 46: North America Market Attractiveness Analysis by Distribution Channel
- Figure 47: North America Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 48: North America Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 49: North America Market Attractiveness Analysis by Indication
- Figure 50: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 51: Latin America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Product
- Figure 54: Latin America Market Value Share and BPS Analysis by Formulation, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Formulation, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Formulation
- Figure 57: Latin America Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Packaging, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Packaging
- Figure 60: Latin America Market Value Share and BPS Analysis by Dosage, 2026 and 2036
- Figure 61: Latin America Market Y-o-Y Growth Comparison by Dosage, 2026-2036
- Figure 62: Latin America Market Attractiveness Analysis by Dosage
- Figure 63: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 64: Latin America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 65: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 66: Latin America Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 67: Latin America Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 68: Latin America Market Attractiveness Analysis by Indication
- Figure 69: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 70: Western Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Product
- Figure 73: Western Europe Market Value Share and BPS Analysis by Formulation, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Formulation, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Formulation
- Figure 76: Western Europe Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 77: Western Europe Market Y-o-Y Growth Comparison by Packaging, 2026-2036
- Figure 78: Western Europe Market Attractiveness Analysis by Packaging
- Figure 79: Western Europe Market Value Share and BPS Analysis by Dosage, 2026 and 2036
- Figure 80: Western Europe Market Y-o-Y Growth Comparison by Dosage, 2026-2036
- Figure 81: Western Europe Market Attractiveness Analysis by Dosage
- Figure 82: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 83: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 84: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 85: Western Europe Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 86: Western Europe Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 87: Western Europe Market Attractiveness Analysis by Indication
- Figure 88: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Product
- Figure 92: Eastern Europe Market Value Share and BPS Analysis by Formulation, 2026 and 2036
- Figure 93: Eastern Europe Market Y-o-Y Growth Comparison by Formulation, 2026-2036
- Figure 94: Eastern Europe Market Attractiveness Analysis by Formulation
- Figure 95: Eastern Europe Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 96: Eastern Europe Market Y-o-Y Growth Comparison by Packaging, 2026-2036
- Figure 97: Eastern Europe Market Attractiveness Analysis by Packaging
- Figure 98: Eastern Europe Market Value Share and BPS Analysis by Dosage, 2026 and 2036
- Figure 99: Eastern Europe Market Y-o-Y Growth Comparison by Dosage, 2026-2036
- Figure 100: Eastern Europe Market Attractiveness Analysis by Dosage
- Figure 101: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 102: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 103: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 104: Eastern Europe Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 105: Eastern Europe Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 106: Eastern Europe Market Attractiveness Analysis by Indication
- Figure 107: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 108: East Asia Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 109: East Asia Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 110: East Asia Market Attractiveness Analysis by Product
- Figure 111: East Asia Market Value Share and BPS Analysis by Formulation, 2026 and 2036
- Figure 112: East Asia Market Y-o-Y Growth Comparison by Formulation, 2026-2036
- Figure 113: East Asia Market Attractiveness Analysis by Formulation
- Figure 114: East Asia Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 115: East Asia Market Y-o-Y Growth Comparison by Packaging, 2026-2036
- Figure 116: East Asia Market Attractiveness Analysis by Packaging
- Figure 117: East Asia Market Value Share and BPS Analysis by Dosage, 2026 and 2036
- Figure 118: East Asia Market Y-o-Y Growth Comparison by Dosage, 2026-2036
- Figure 119: East Asia Market Attractiveness Analysis by Dosage
- Figure 120: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 121: East Asia Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 122: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 123: East Asia Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 124: East Asia Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 125: East Asia Market Attractiveness Analysis by Indication
- Figure 126: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 127: South Asia and Pacific Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 128: South Asia and Pacific Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 129: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 130: South Asia and Pacific Market Value Share and BPS Analysis by Formulation, 2026 and 2036
- Figure 131: South Asia and Pacific Market Y-o-Y Growth Comparison by Formulation, 2026-2036
- Figure 132: South Asia and Pacific Market Attractiveness Analysis by Formulation
- Figure 133: South Asia and Pacific Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 134: South Asia and Pacific Market Y-o-Y Growth Comparison by Packaging, 2026-2036
- Figure 135: South Asia and Pacific Market Attractiveness Analysis by Packaging
- Figure 136: South Asia and Pacific Market Value Share and BPS Analysis by Dosage, 2026 and 2036
- Figure 137: South Asia and Pacific Market Y-o-Y Growth Comparison by Dosage, 2026-2036
- Figure 138: South Asia and Pacific Market Attractiveness Analysis by Dosage
- Figure 139: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 140: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 141: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 142: South Asia and Pacific Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 143: South Asia and Pacific Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 144: South Asia and Pacific Market Attractiveness Analysis by Indication
- Figure 145: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 146: Middle East & Africa Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 147: Middle East & Africa Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 148: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 149: Middle East & Africa Market Value Share and BPS Analysis by Formulation, 2026 and 2036
- Figure 150: Middle East & Africa Market Y-o-Y Growth Comparison by Formulation, 2026-2036
- Figure 151: Middle East & Africa Market Attractiveness Analysis by Formulation
- Figure 152: Middle East & Africa Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 153: Middle East & Africa Market Y-o-Y Growth Comparison by Packaging, 2026-2036
- Figure 154: Middle East & Africa Market Attractiveness Analysis by Packaging
- Figure 155: Middle East & Africa Market Value Share and BPS Analysis by Dosage, 2026 and 2036
- Figure 156: Middle East & Africa Market Y-o-Y Growth Comparison by Dosage, 2026-2036
- Figure 157: Middle East & Africa Market Attractiveness Analysis by Dosage
- Figure 158: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 159: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 160: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 161: Middle East & Africa Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 162: Middle East & Africa Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 163: Middle East & Africa Market Attractiveness Analysis by Indication
- Figure 164: Global Market - Tier Structure Analysis
- Figure 165: Global Market - Company Share Analysis

