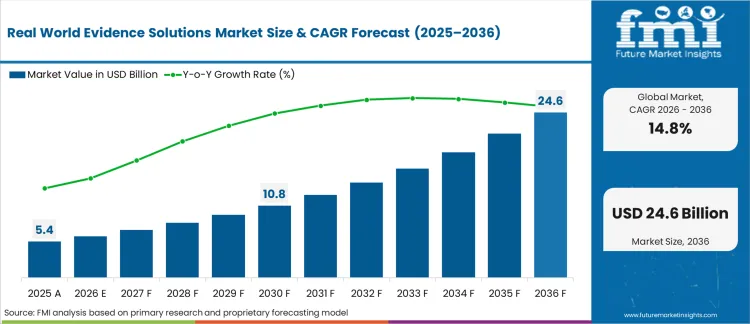
Regulatory agencies rejecting single-arm trials without synthetic control arms derived from longitudinal health records have driven the real-world evidence solutions market to USD 5.4 billion in 2025. Chief medical officers executing accelerated approval pathways now mandate multi-modal dataset integration to prove long-term safety profiles to scrutinizing health authorities. Compound expansion at 14.80% through 2036 carries the market to USD 24.6 billion as national health technology assessment bodies permanently transition from evaluating static clinical trial results to demanding continuous post-market surveillance streams.
The transition from episodic data gathering to continuous patient tracking forces pharmaceutical sponsors to abandon isolated trial databases and commit significant capital to federated networks. Health economics directors entering drug reimbursement negotiations without comprehensive observational registries face a massive evidentiary gap they cannot bridge with conventional trial data alone. Delaying this architectural pivot directly results in restricted payer coverage formulas and severely delayed regional market access.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 6.2 billion |
| Industry Value (2036) | USD 24.6 billion |
| CAGR (2026-2036) | 14.80% |
Procurement leads who continue relying on fragmented claims data find their regulatory submissions rejected outright by compliance committees demanding fully tokenized patient journeys. Interoperability directors must deploy these cryptographic matching standards before the European cross-border safety mandates take effect to retain critical market access. Securing this native analytical layer allows life science researchers to continuously map rare disease progressions without ever triggering devastating data residency violations.
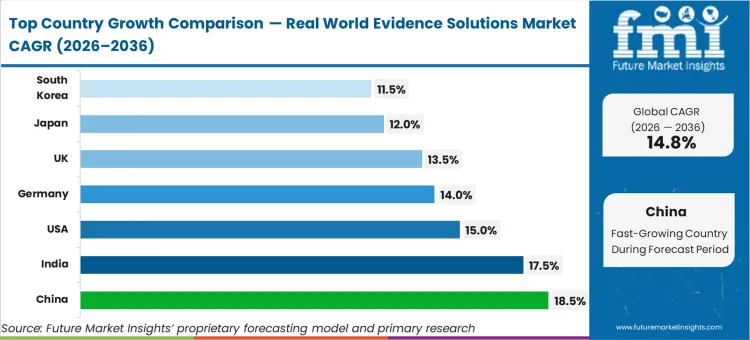
As regional healthcare systems transition toward federated data models, China sets the pace with an 18.5% CAGR, followed closely by India at 17.5% as both nations leverage greenfield digital health construction to bypass legacy retrofit hurdles. The United States advances at a 15.0% rate, supported by infrastructure modernization mandates from federal health agencies. Germany registers a 14.0% expansion while the UK tracks at 13.5%, both driven by precision engineering and genomic registry standards. Japan expands at a 12.0% trajectory anchored by its robust geriatric research sector. South Korea grows at 11.5% as localized bioinformatics funding matures, confirming that national privacy policies permanently dictate geographic adoption velocities.
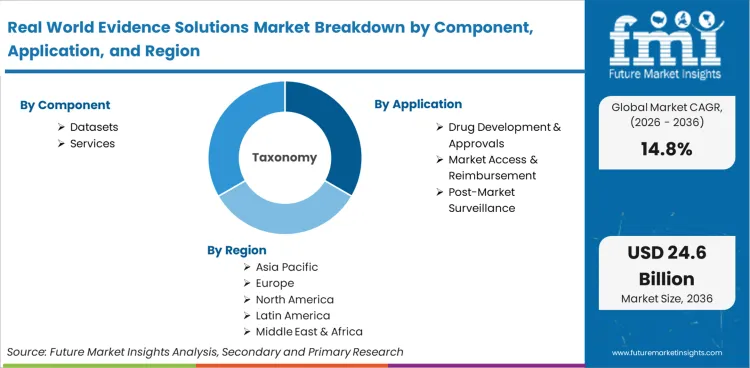
Real world evidence solutions represent the integrated datasets, analytical platforms, and consulting frameworks designed specifically to generate actionable clinical insights from observational patient data. The ecosystem includes de-identified electronic health records, medical claims databases, disease registries, and the epidemiological methodologies required to transform raw information into regulatory-grade evidence, enabling continuous therapeutic tracking outside controlled settings.
The market scope includes commercialized clinical datasets, federated data networks, tokenization software, and specialized analytical services tailored for life science research. Retrospective cohort study platforms, synthetic control arm generation algorithms, and applications supporting electronic health records interoperability fall entirely within the boundaries. Products utilizing privacy-preserving record linkage mechanisms for critical post-market safety tracking are fully incorporated into the valuation.
Standard commercial data warehousing solutions lacking healthcare-specific cryptographic tokenization are excluded. Pure hospital billing software without external epidemiological analysis capabilities falls outside the defined parameters. Legacy localized clinical trial management systems without multi-modal real-world interoperability features are explicitly omitted from the scope.
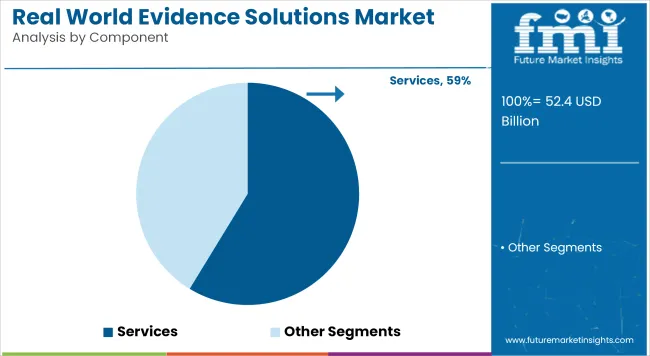
With dominant databases providing the absolute foundation for health outcomes research, procurement teams execute massive acquisition strategies to capture longitudinal value. Datasets command a striking 62.5% share in 2026, fusing raw clinical notes with comprehensive billing histories to track definitive patient journeys. Health economics directors deploying these combined data streams directly into their analytical engines establish the core evidence required for value-based pricing negotiations.
Based on FMI's assessment, this integration eliminates the fatal blind spots inherent in single-source observational studies. Procurement leads specifying these dual-source linkages streamline their entire evidentiary pipeline and significantly reduce the time required to answer regulatory inquiries. Health authorities facing complex safety signals now mandate comprehensive record alignment to authorize continued market access.
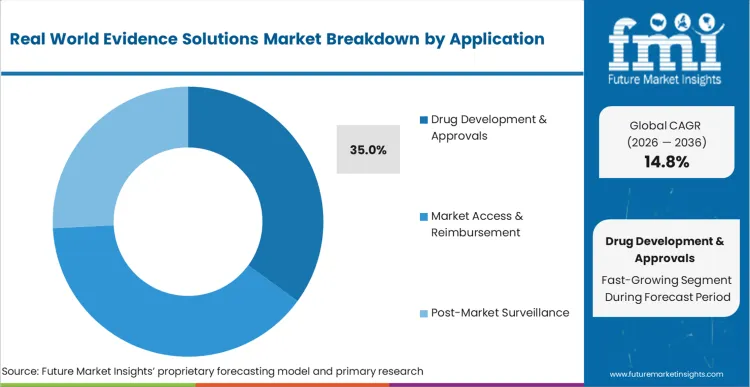
Traditional clinical pipelines face severe bottlenecks when evaluating therapeutics designed for exceptionally small patient populations. Drug Development & Approvals secures a 35.0% share in 2026 as late-stage development teams scramble to enrich their traditional trial dossiers with extended real-world observations.
FMI analysts opine that incorporating precise clinical trial management augmentation techniques allows sponsors to confidently present combined safety profiles to health authorities. Trial designers specifying these workflows bypass the immense costs of launching entirely new observational registries from scratch. Transforming static trial results into dynamic living datasets enables medical affairs teams to rapidly secure accelerated approval pathways. Value-based contracting directors utilizing robust observational evidence bypass standard reimbursement delays.
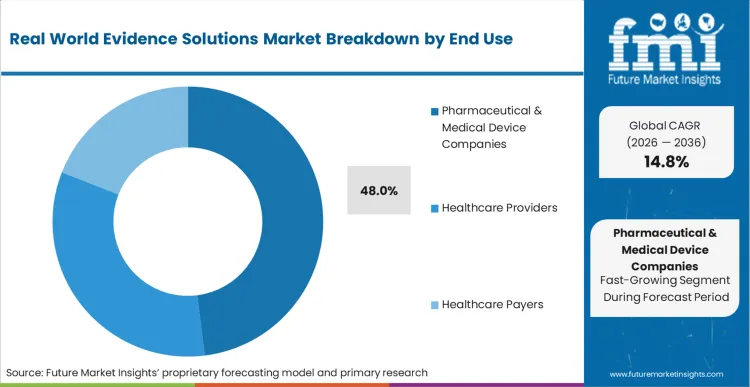
Sponsor organizations face mounting pressure to demonstrate therapeutic value across highly diverse patient populations over extended timelines. Pharmaceutical & Medical Device Companies represent a 48.0% share in 2026, driven directly by regulatory bodies imposing strict decade-long tracking requirements on advanced biologics.
In FMI's view, the convergence of comprehensive pharmaceutical contract research paradigms with massive claims databases amplifies the demand for continuous safety signal detection. Safety officers monitoring newly launched formulations reject delayed manual reporting workflows that introduce dangerous blind spots into the risk management plan. Automating the linkage between pharmacy dispensing records and subsequent hospital admissions provides immediate visibility into rare adverse events. Pharmacovigilance directors failing to deploy continuous automated linkage workflows face catastrophic regulatory warnings and potential product withdrawals.
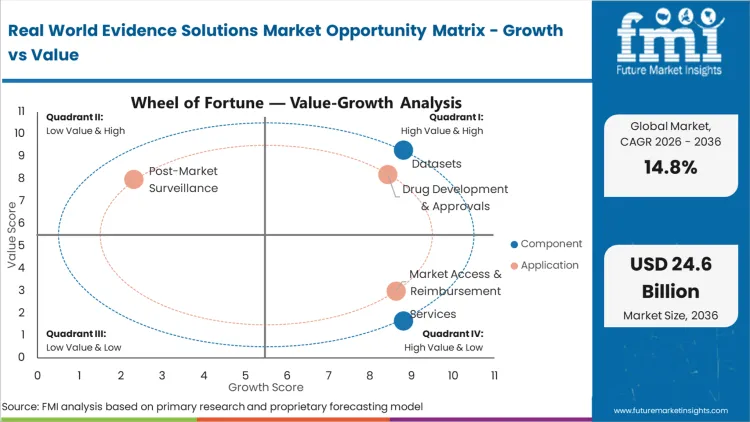
The convergence of value-based healthcare models and sophisticated precision medicine therapeutics forces health economics directors to extract actionable longitudinal data directly from fragmented provider networks. This commercial requirement renders siloed, single-source clinical registries virtually obsolete. Life sciences companies upgrading their evidentiary pipelines face a strict binary choice between abandoning complex post-market studies or deploying advanced observational networks. Transitioning to a unified federated matching backbone simplifies the entire regulatory submission process and enables instant cohort validation. Data aggregators that fail to modernize their core cryptographic linkage layers risk permanent exclusion from lucrative top-tier pharmaceutical procurement cycles.
The intricate legal parameters required to optimize cross-border data linkage create steep compliance learning curves for traditional clinical informaticians. Designing conflict-free data sharing schemas across multi-national registries demands specialized privacy legislation expertise that most biotechnology firms simply lack internally. To mitigate this skill gap, clinical operations directors increasingly rely on managed integration services that completely automate the token generation process and validate compliance frameworks before any physical data matching occurs.
Based on the regional analysis, the Real World Evidence Solutions market is segmented into North America, Latin America, Europe, Asia Pacific, and Middle East & Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
| China | 18.5% |
| India | 17.5% |
| USA | 15.0% |
| Germany | 14.0% |
| UK | 13.5% |
| Japan | 12.0% |
| South Korea | 11.5% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
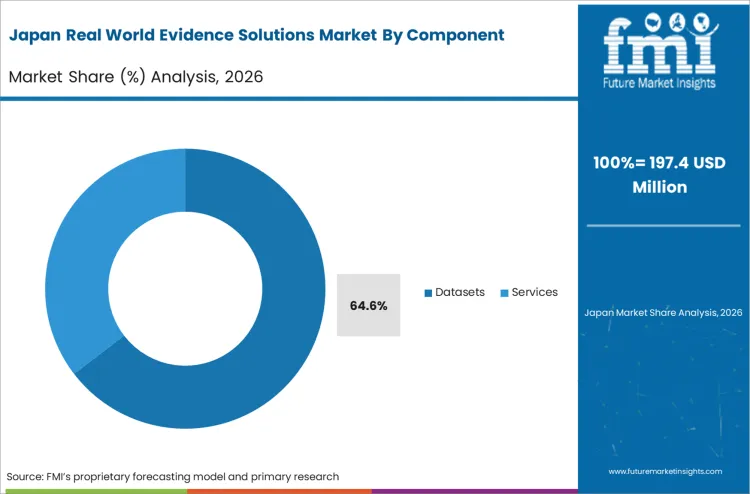
Rapid greenfield digital health adoption across the Asia Pacific region accelerates the bypass of archaic paper-based clinical trial constraints. Principal bioinformaticians constructing massive new genomic sequencing repositories specify unified tokenized networking in their initial architectural blueprints. FMI's analysis indicates that this clean-slate approach eliminates the costly and time-consuming data remediation phase that plagues older Western healthcare systems. By building native cryptographic linkage capabilities into the foundation, regional health authorities establish highly flexible data environments capable of rapid epidemiological analysis. The integration of robust network architecture forms the critical prerequisite for deploying advanced diagnostic machine learning applications across the region.
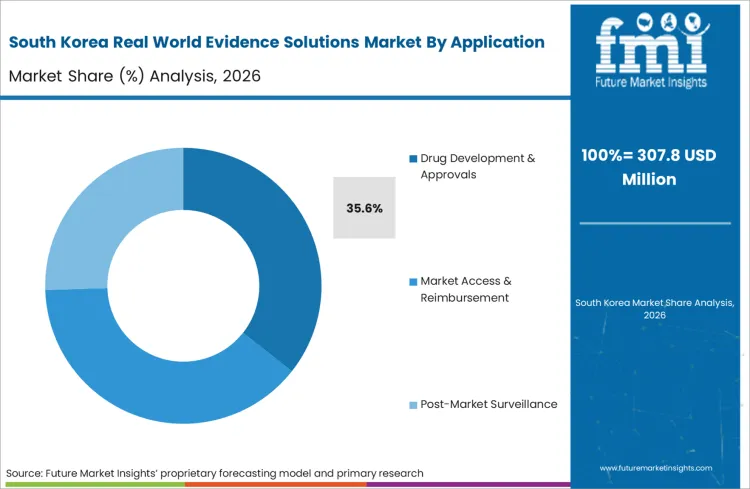
FMI's report encompasses a holistic evaluation of the broader Asia Pacific digital health sector. The analysis investigates the emerging data linkage frameworks across Taiwan and the broader ASEAN region. A defining dynamic involves the integration of cross-border clinical trial datasets, which requires highly standardized tokenization protocols to coordinate multi-national regulatory submissions and maintain synchronized safety tracking schedules across completely different regional healthcare systems.
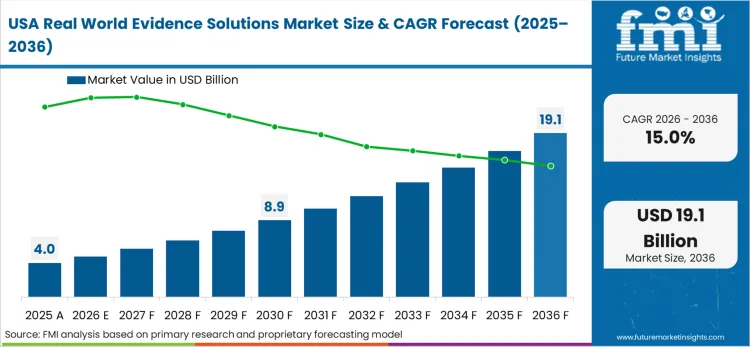
Sophisticated commercial claims ecosystems across North America target the systematic eradication of isolated health data domains. Health economics leaders executing critical value-based pricing strategies face strict mandates to unify diverse payer datasets under a single secure tokenization architecture. Implementing a standardized, privacy-preserving backbone enables pharmaceutical sponsors to deploy advanced predictive modelling algorithms without compromising patient anonymity.
FMI's research confirms that this strategic shift requires immense capital allocation toward robust cryptographic software capable of processing millions of records simultaneously. The success of these modernization initiatives depends entirely on the availability of certified interoperable networking components.
FMI's report includes extensive coverage of the North American real world evidence market situation. It incorporates a detailed analysis of Canadian provincial data repositories and their specific integration challenges. A primary trend shaping these additional markets involves the rapid localization of data residency laws, forcing regional service providers to deploy entirely localized analytical engines to satisfy stringent federal health data sovereignty requirements.
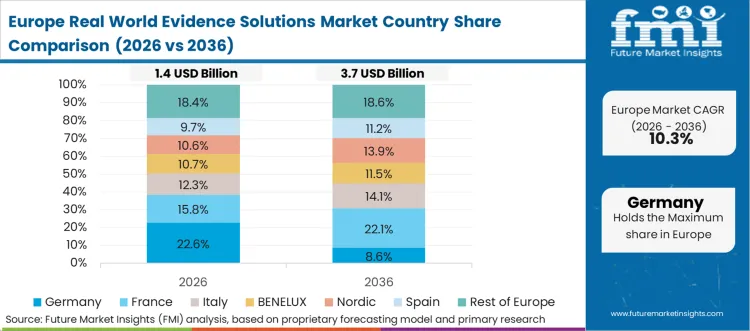
Strict European data protection policies actively penalize the commercialization of unsecured or poorly anonymized patient information. Chief privacy officers redesigning legacy clinical trial databases must integrate continuous compliance monitoring mechanisms alongside core cohort matching engines. This intense regulatory pressure forces the rapid adoption of zero-knowledge proof protocols to guarantee absolute patient anonymity while extracting highly valuable diagnostic trends. Upgrading the core linkage software provides the necessary mathematical certainty to support advanced cross-border epidemiological studies. As per FMI's projection, facility directors recognize that maintaining outdated network standards severely limits their ability to compete in highly regulated European markets.
FMI's report includes thorough investigation of the broader European health informatics framework. The analysis encompasses Italy, France, Spain, the Nordics, and the Benelux region. A prevailing structural condition across these nations is the mandatory compliance with the impending European Health Data Space directives, forcing pharmaceutical sponsors to specify federated tokenization networks that can reliably transport anonymized safety signals without ever moving the underlying raw clinical data across sovereign borders.
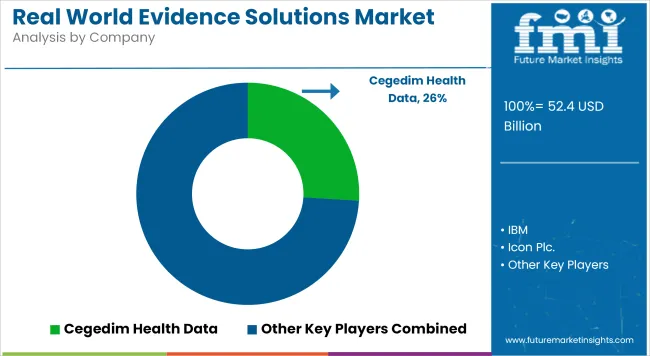
The rapid enforcement of stringent global data privacy frameworks is entirely reshaping how pharmaceutical buyers evaluate clinical data integration vendors. Instead of accepting vulnerable deterministic matching tools, procurement teams now demand certified, cryptographically secure analytical platforms in all new enterprise software tenders. This absolute shift has raised the privacy threshold across the entire ecosystem, meaning data brokers that continue to rely on centralized, unencrypted data pooling models risk being completely excluded from massive real-world evidence modernization programs. Industry leaders utilizing sophisticated artificial intelligence have already begun adapting rapidly to these structural mandates.
Prominent aggregators such as IQVIA, ICON plc, and Optum have strategically aligned their platform capabilities with highly advanced federated learning protocols, setting an entirely new competitive baseline for mid-tier challengers to follow. As a direct result, data integration vendors are severely pushed to integrate native zero-knowledge proof capabilities and advanced hashing algorithms directly into their core service offerings instead of utilizing vulnerable third-party bridging applications. Embedding robust healthcare analytical testing capabilities specifically for token matching grants a massive architectural advantage that streamlines procurement for large life science sponsors.
Growth of open, interoperable tokenization standards is permanently disrupting long-established proprietary data strongholds. With more health information exchanges mandating open application programming interfaces, system integrators designing converged data architectures can now select de-identification engines from multiple competing suppliers without worrying about incompatible hashing formats or massive data loss. This expanded operational flexibility empowers clinical trial sponsors to fully optimize their observational networks for raw evidentiary yield instead of being permanently constrained by legacy, closed-loop analytics platforms, structurally shifting pricing leverage back to the pharmaceutical buyers.
| Metric | Value |
| Quantitative Units | USD 6.2 billion to USD 24.6 billion, at a CAGR of 14.80% |
| Market Definition | Real world evidence solutions encompass the integrated datasets and analytical services required to translate fragmented observational health data into validated clinical proof. |
| Component Segmentation | Datasets, Services |
| Application Segmentation | Drug Development & Approvals, Market Access & Reimbursement, Post-Market Surveillance, Clinical Trial Design |
| End Use Segmentation | Pharmaceutical & Medical Device Companies, Healthcare Providers, Healthcare Payers, Regulatory Agencies |
| Regions Covered | North America, Latin America, Europe, Asia Pacific, Middle East & Africa |
| Countries Covered | China, India, USA, Germany, UK, Japan, South Korea, and 40+ other countries |
| Key Companies Profiled | IQVIA, ICON plc, Thermo Fisher Scientific (PPD), Syneos Health, Optum, IBM (Merative), Oracle (Cerner), Cognizant |
| Forecast Period | 2026 to 2036 |
| Approach | The baseline value derives from a bottom-up aggregation of commercial tokenization software deployments and dataset subscription contracts, applying region-specific digitalization curves to project future adoption velocity. Forecasts are validated against capital expenditure guidance from major health data aggregators. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the Real World Evidence Solutions Market in 2026?
The market is estimated to be valued at USD 6.2 billion in 2026. This absolute necessity for regulatory-grade post-market safety validation drives continuous analytical investments.
What will the Real World Evidence Solutions Market be valued at by 2036?
The industry value is projected to reach USD 24.6 billion by 2036. The architectural pivot to federated data networks forces pharmaceutical sponsors to commit significant capital to observational registries.
What CAGR is projected for the Real World Evidence Solutions Market from 2026 to 2036?
Demand is expected to grow at a CAGR of 14.80% from 2026 to 2036. This compound expansion is sustained by health technology assessment bodies demanding continuous longitudinal tracking streams.
Which Component is poised to lead global sales by 2026?
Datasets accounts for 62.5% share in 2026. Health economics directors deploying these combined data streams directly into their analytical engines establish the core evidence required for value-based pricing.
How significant is the role of Drug Development & Approvals in driving market adoption in 2026?
Drug Development & Approvals represents 35.0% share in 2026. Trial designers specifying these observational workflows bypass the immense costs of launching entirely new clinical registries from scratch.
What specific requirement forces health economics directors to abandon traditional clinical trial data?
Standard trial data represents only a brief snapshot of highly controlled patient interactions. Health economics teams require decade-long real-world adherence records to justify premium formulary positioning to aggressive national payers.
Why do Pharmaceutical & Medical Device Companies dominate the end use segments?
This segment captures 48.0% share in 2026, driven directly by regulatory bodies imposing strict decade-long tracking requirements on advanced biologics. Incorporating precise observational techniques allows sponsors to confidently present combined safety dossiers.
What is the core tension driving the rapid adoption of tokenization engines?
The sheer volume of available health data clashes directly with strict global privacy laws. Clinical informatics leads attempting to generate longitudinal evidence must use cryptographic hashing to match patients without exposing protected health information.
What compliance standard fundamentally alters how clinical linkage is performed?
The impending European Health Data Space directives and massive HIPAA enforcement actions force the abandonment of centralized data pooling. Sponsors must deploy federated networks that process matches securely at the local hospital source.
What structural limitation plagues single-source observational studies?
Unifying fragmented care episodes prevents patients from artificially dropping out of the observational cohort when they change insurance providers. This eliminates the fatal flaws inherent in single-source claims analysis.
What is driving the massive real world evidence demand in the United States?
The United States commercial insurance sector enforces incredibly complex data usage frameworks across its fragmented provider ecosystem. Health informatics directors must deploy probabilistic algorithms capable of reconciling billing discrepancies across multiple state exchanges.
What is the growth outlook for the United States in this report?
The USA advances at a 15.0% compound rate from 2026 to 2036.
Why is China's digital health strategy unique regarding observational data?
China's state-sponsored initiatives deploy highly centralized regional health exchanges that bypass legacy deterministic matching entirely. Greenfield infrastructure architects specify advanced neural network linkage algorithms for all new population health contracts.
What is the China growth outlook in this report?
China is projected to expand at a CAGR of 18.5% during the forecast period.
How do German privacy regulations impact the local adoption of analytical services?
German regulations mandate completely localized tokenization layers for all commercial observational studies. System integrators must demonstrate flawless federated query execution before connecting disease registries to the national telematics infrastructure.
What is Germany's growth outlook in this report?
Germany tracks a 14.0% CAGR as the federated standard proliferates across all major university hospital networks.
Why does South Korea heavily invest in advanced data linkage?
South Korea's advanced bio-manufacturing complex operates under mandates demanding direct linkage to national health insurance claims. Medical affairs leads mandate zero-loss architectures to ensure complex advanced therapy outcomes are tracked flawlessly.
What sustains Japan's continuous investment in interoperable data registries?
Japan's rapidly aging demographic profile enforces strict post-market tracking requirements for long-term geriatric safety. Every new observational architecture must validate longitudinal capability to satisfy the Pharmaceuticals and Medical Devices Agency.
What specific application captures massive value in the regulatory pipeline?
Synthetic control generation enables trial designers to construct robust external control arms from existing electronic health records. This eliminates the need for massive placebo groups in specialized rare disease trials.
What is Real World Evidence Solutions and what is it mainly used for?
It represents the integrated datasets, analytical platforms, and consulting frameworks designed to generate actionable clinical insights from observational patient data. It is primarily used to map long-term therapeutic safety and efficacy profiles outside of controlled clinical trials.
What does the Real World Evidence Solutions market encompass in this report?
The scope includes commercialized clinical datasets, federated data networks, tokenization software, and specialized analytical services tailored for life science research.
What is explicitly excluded from the scope of this report?
Standard commercial data warehousing solutions lacking healthcare-specific cryptographic tokenization are excluded. Pure hospital billing software without external epidemiological analysis capabilities is also omitted.
What does the market forecast mean in the context of this report?
The forecast represents a model-based projection built on bottom-up tokenization software deployments and dataset subscription contracts for strategic planning purposes.
How does FMI build and validate this real world evidence solutions forecast?
The model applies regional digitalization curves to commercial tokenization deployments. Segment forecasts are cross-validated against quarterly software subscription revenues from major life science technology providers.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.