SGLT2 Inhibitors Market
The SGLT2 Inhibitors Market is segmented by Drug (Jardiance (empagliflozin), Farxiga (dapagliflozin), Invokana (canagliflozin), Inpefa (sotagliflozin), Qtern (dapagliflozin/saxagliptin), and Other SGLT2 Inhibitors), Indication (Type 2 Diabetes, Cardiovascular Diseases, Chronic Kidney Disease (CKD), and Others), Distribution Channel (Hospital Pharmacies, Retail Pharmacies, and Online Pharmacies) and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
SGLT2 Inhibitors Market Forecast and Outlook By FMI
In 2025, the SGLT2 inhibitors market was valued at USD 17.8 billion. Based on Future Market Insights’ analysis, demand for SGLT2 inhibitors is estimated to grow to USD 18.9 billion in 2026 and USD 34.5 billion by 2036. FMI projects a CAGR of 6.2% during the forecast period.
Absolute dollar growth of USD 15.6 billion over 2026 to 2036 signals steady expansion driven by guideline-backed cardiorenal uptake and broader payer coverage rather than a single breakthrough molecule. Growth remains capped by class-wide safety monitoring requirements, step-therapy controls, and the mid-term impact of generic entry in select markets, which compresses net pricing even as treated patient pools expand.
Summary of the SGLT2 Inhibitors Market
- SGLT2 Inhibitors Market Definition
- The industry tracks prescription SGLT2 inhibitor revenues across diabetes and cardiorenal indications influenced by label breadth, guideline placement, and reimbursement access structures.
- Demand drivers
- Rising type 2 diabetes prevalence expands the eligible base, especially where earlier-line combination therapy is adopted. [2]
- Cardiovascular and heart-failure positioning increases use beyond endocrinology into cardiology-led pathways. [2] [4]
- CKD progression-slowing evidence supports earlier initiation in high-risk renal cohorts under updated care standards. [3]
- Key segments analyzed
- Drug: Jardiance (empagliflozin) leads with 55.3% share in 2025 on breadth of outcomes positioning and physician familiarity. [4]
- Indication: Type 2 Diabetes remains largest at 71.8% share in 2025, sustaining base volumes while cardiorenal adds incremental growth. [2]
- Geography: Asia Pacific drives volume expansion, while North America and Europe sustain value through higher treatment intensity and broader specialist prescribing.
- Analyst opinion at FMI
- Sabyasachi Ghosh, Senior Consultant at Future Market Insights, opines, “Over 2026 to 2036, the winning SGLT2 portfolios will be those that prove downstream cost offsets in heart failure and CKD in real-world payer datasets, because access will tighten as generics widen class availability.”
- Strategic implications / executive takeaways
- Tighten evidence strategy around hospitalisation avoidance and CKD delay to defend reimbursement as generics increase price pressure.
- Build channel programs with hospital pharmacies and discharge pathways to capture cardiology and nephrology initiations.
- Prioritise differentiated patient support and adherence services where cost-sharing limits persistence.
- Methodology
- Forecasts are triangulated against guideline placement, label expansions, and payer coverage rules. [2] [3] [4]
- Validation incorporates company reporting, tender/procurement signals, and physician adoption checks. [1]
- Scenario testing includes generic entry timing, step-therapy tightening, and persistence-rate sensitivity.
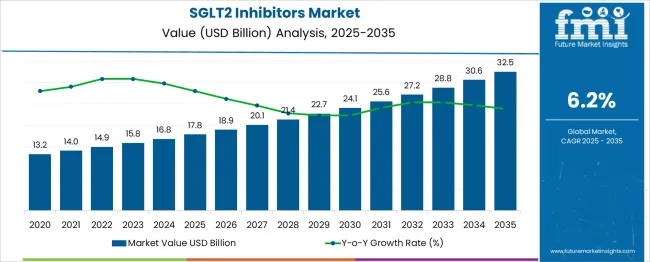
Major pharmaceutical manufacturers report that SGLT2 inhibitors increasingly compete on cardiorenal outcomes evidence and real-world hospitalization avoidance data rather than glycemic control metrics alone. Updated professional diabetes care standards embed SGLT2 inhibitors as foundational options for patients with cardiovascular and renal comorbidity profiles, widening prescriber segments beyond endocrinologists into cardiology and nephrology specialists where therapy initiation occurs through hospital-linked pathways and structured discharge protocols supporting systematic medication optimization.
China (8.4% CAGR) and India (7.7% CAGR) drive new opportunities through rising type 2 diabetes prevalence and faster specialist adoption of cardiorenal protocols within expanding urban hospital networks. Germany (7.1% CAGR) and France (6.5% CAGR) contribute via outcomes-based reimbursement scrutiny and structured care pathways integrating SGLT2 inhibitors for high-risk cohorts. Mature markets such as the USA (5.3%), UK (5.9%), and Brazil (4.6%) generate replacement demand and indication expansion rather than pure volume growth, with adoption shaped by payer utilization management controls and public reimbursement procurement cycles.
Market Definition
SGLT2 inhibitors are prescription medicines that lower blood glucose by blocking sodium-glucose co-transporter 2 in the kidney, increasing urinary glucose excretion. While originally adopted for type 2 diabetes, the market now spans cardiorenal care, with usage expanding in heart failure and chronic kidney disease cohorts based on outcomes evidence and guideline integration. Commercial demand is measured through branded and authorised-generic sales across hospital and retail channels, shaped by reimbursement status, prescribing restrictions, and the breadth of labelled indications.
Market inclusions
The report includes comprehensive analysis of market dynamics, featuring Global and Regional Market Sizes (Revenue) and a 10-year Forecast (2026-2036). It covers segmental breakdowns by Drug (Jardiance, Farxiga, Invokana, Inpefa, Qtern, Others), Indication (Type 2 Diabetes, Cardiovascular Diseases, Chronic Kidney Disease, Others), Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), and competitive benchmarking with pricing and access dynamics tied to guideline updates and reimbursement decisions.
Market exclusions
The scope excludes non-SGLT2 glucose-lowering drug classes (GLP-1 receptor agonists, DPP-4 inhibitors, insulin, sulfonylureas) except where referenced for treatment sequencing context. It also omits downstream diabetes devices (CGMs, pumps), telehealth platforms as standalone markets, and non-prescription supplements. Combination products outside the listed drug set are excluded unless explicitly captured under “Other SGLT2 inhibitors”.
SGLT2 Inhibitors Market Research methodology
- Primary research: FMI conducted interviews with endocrinologists, cardiologists, nephrologists, hospital pharmacists, payer/formulary stakeholders, and distribution partners to validate adoption and access constraints.
- Desk research: Public clinical guidance, reimbursement decisions, regulator labelling updates, and corporate filings/press releases were reviewed to anchor indication expansion and channel dynamics. [2] [3] [4]
- Market-sizing and forecasting: A hybrid approach was used, combining bottom-up revenue build by molecule and geography with top-down epidemiology-linked treatment penetration, stress-tested against access rules and switching behaviour.
- Data validation and update cycle: Outputs were cross-checked using multi-source triangulation (guidelines, reimbursement documents, label changes, and company disclosures), with periodic refresh based on policy updates and quarterly company reporting. [1] [4]
Segmental Analysis
SGLT2 Inhibitors Market Analysis by Indication
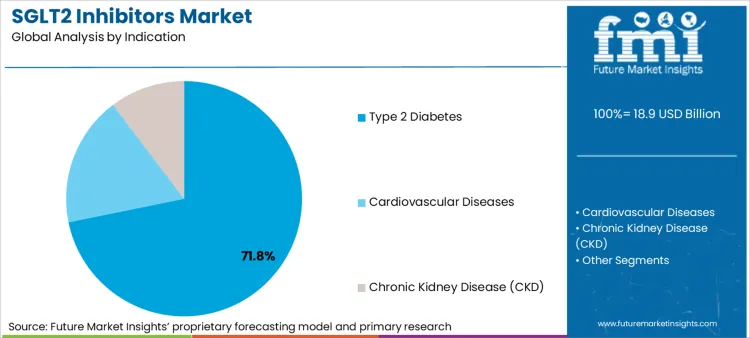
Based on FMI’s SGLT2 inhibitors market report, consumption of Type 2 Diabetes is estimated to hold 71.8% share in 2025. This dominance persists because T2D remains the primary diagnosed population and the base indication where SGLT2 inhibitors are embedded in long-term therapy stacks, with add-on use accelerating in patients with obesity, hypertension, and early renal risk. [2]
- Access expansion: American Diabetes Association Standards of Care published in 2025 increasingly place SGLT2 inhibitors earlier for high-risk Type 2 Diabetes patients, widening treated cohorts beyond late-stage add-on therapy into earlier treatment lines where cardiovascular and renal protection benefits justify initiation [1].
- Cardiorenal crossover: FDA prescribing information for Farxiga updated to include heart failure and chronic kidney disease indications in 2023-2024 period, increasing prescribing by cardiologists and nephrologists raising initiation points outside traditional endocrinology clinics into specialist-led pathways [2].
- Channel shift: Scottish Medicines Consortium empagliflozin appraisal completed in July 2024 demonstrating hospital-driven initiations for heart failure and CKD patients increasing downstream fulfillment through hospital pharmacies and discharge-linked dispensing pathways supporting systematic medication optimization [3].
SGLT2 Inhibitors Market Analysis by Drug
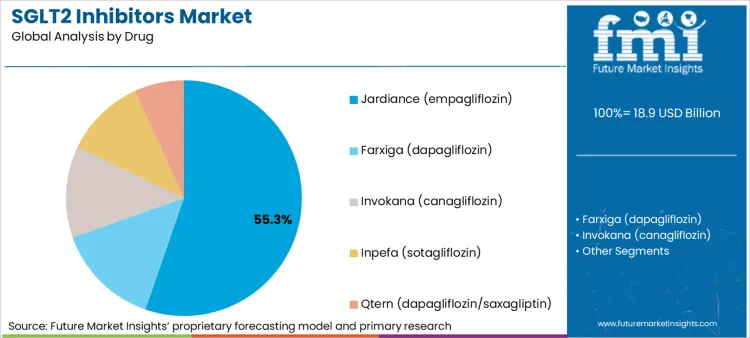
Jardiance (empagliflozin)'s 55.3% market share in 2025 stems from its role as molecule with durable prescriber familiarity built through extensive cardiovascular outcomes trial dissemination and strong positioning in specialist-led pathways, yet this concentration creates structural dependencies on maintaining payer preferred status and managing generic competition pressure that compound during periods of formulary repositioning or step-therapy tightening. The segment's reliance on outcomes evidence differentiation and specialist adoption transforms any erosion in comparative effectiveness perception or loss of preferred formulary tier placement into immediate market share vulnerability, while patent expiration timelines offer minimal buffer against biosimilar or generic entry reducing pricing power even when clinical preference persists among prescriber segments.
- Company Investment: AstraZeneca reported in February 2026 continued growth momentum positioning medicines portfolio for long-term delivery with emphasis on high-value therapy areas including cardiorenal disease management, demonstrating sustained manufacturer investment in outcomes datasets and care-pathway programs supporting payer discussions on avoidable hospitalizations [4].
- Product Differentiation: KDIGO 2024 Clinical Practice Guideline for chronic kidney disease evaluation and management published in March 2024 references SGLT2 inhibitor evidence and labeling around heart failure and renal protection influencing molecule selection where formularies allow choice inside the drug class [5].
- Supply and Contracting: NICE Type 2 Diabetes Management guidance implementation materials published in October 2024 demonstrate large health systems prefer predictable supply, rebates, and adherence support for chronic therapies, increasing importance of contracting capabilities and patient services in maintaining systematic prescribing [6].
SGLT2 Inhibitors Market Drivers, Restraints, Opportunities
Future Market Insights analysis indicates the market’s structural base was built in diabetes care, but the value expansion now follows “risk-based prescribing” in which therapy is prioritised for patients with heart failure, CKD, and high cardiovascular risk. Updated professional standards increasingly place SGLT2 inhibitors as foundational options for patients with cardiorenal comorbidity profiles, widening prescriber segments and increasing initiation in hospital-linked settings. [2] [3]
The market’s central tension is access versus outcomes. As class utilisation grows, payers tighten utilisation management (prior authorisation, step edits), while manufacturers push broader adoption through outcomes evidence and pathway integration. Net price compression from competition and future generics is partially offset by higher treated counts and longer duration in CKD and heart-failure cohorts where therapy persistence can be stronger when monitored through structured clinics. [2] [4]
- Guideline-Led Expansion: Diabetes and kidney disease professional guidance increasingly recommend SGLT2 inhibitors for high-risk patients based on cardiovascular outcomes trials, converting clinical evidence into routine prescribing rules and quality metrics in mature healthcare systems where guideline adherence is monitored.
- Reimbursement Tightening: As prescription volumes rise creating budget impact, payers respond with step-therapy requirements and narrower eligibility criteria, shifting growth toward healthcare systems where outcomes-based justification is strongest and clinical documentation supporting high-risk patient identification is standardized through electronic health record integration.
- Emerging Market Access: Faster diagnosis coverage and specialist network expansion in Asia increases therapy initiations, but sustained growth depends on patient affordability programs and public reimbursement listings that reduce out-of-pocket payment barriers preventing treatment persistence in cost-sensitive populations.
Regional Analysis
Based on the regional analysis, SGLT2 inhibitors market is segmented into North America, Europe, Asia Pacific, Latin America, and Middle East & Africa across 40+ countries. Growth is shaped by type 2 diabetes diagnosis rates, clinical guideline uptake velocity, and reimbursement breadth determining systematic prescribing integration. The full report also offers market attractiveness analysis based on regional trends.
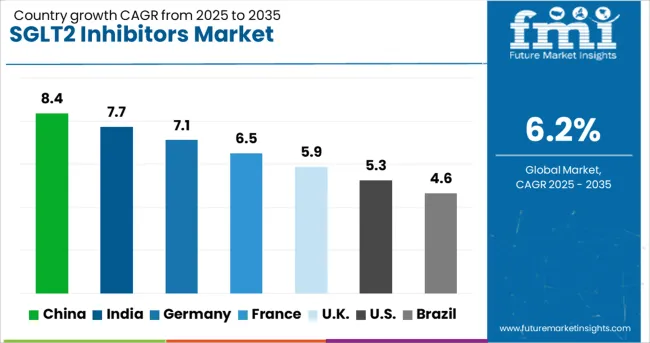
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 8.4% |
| India | 7.7% |
| Germany | 7.1% |
| France | 6.5% |
| United Kingdom | 5.9% |
| United States | 5.3% |
| Brazil | 4.6% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America market analysis
North America functions as outcomes-evidence monetization region, characterized by payer coverage and step-therapy protocols determining net pricing dynamics and manufacturer access defense through real-world hospitalization avoidance data. Key players in North America include AstraZeneca, leading Farxiga brand commercialization with cardiorenal positioning; Boehringer Ingelheim, maintaining Jardiance market leadership through cardiovascular outcomes evidence; and pharmacy benefit managers shaping formulary preferred status through rebate contracting negotiations.
- United States: Demand for SGLT2 inhibitors in the United States is projected to rise at 5.3% CAGR through 2036. Growth is tied to guideline-backed positioning for patients with diabetes plus heart failure or CKD, which increases initiation in cardiology and nephrology settings rather than purely endocrinology. The ADA Standards of Care continues to embed SGLT2 inhibitors in risk-based treatment selection, supporting broader prescribing in high-risk cohorts. [2] Health systems also build discharge pathways for heart failure patients that standardise medication optimisation, increasing hospital-originated starts and improving follow-up adherence when paired with structured clinics. [6]
FMI’s analysis of SGLT2 inhibitors market in North America consists of country-wise assessment that includes the United States, Canada, and Mexico. Readers can find channel-wise demand, payer control intensity, and adoption scenarios by risk cohort.
Europe market analysis
Europe acts as health technology assessment and pathway governance region, where NICE, HAS, and G-BA-style decision frameworks and clinical pathway structures govern systematic access expansion based on cost-effectiveness analysis and budget impact assessment. Boehringer Ingelheim, AstraZeneca, and other class participants compete within structured reimbursement rules rewarding demonstrated outcomes and documented cost-offset logic through formal health economic submissions.
- United Kingdom: Demand for SGLT2 inhibitors in the United Kingdom is projected to rise at 5.9% CAGR through 2036. Uptake is driven by pathway integration under NICE-aligned diabetes guidance that supports use of SGLT2 inhibitors in appropriate high-risk patients, particularly where heart failure and CKD risks are material to long-term cost and hospitalisation avoidance. [5] Access is broad in many areas, but growth is moderated by prescribing governance and structured initiation criteria that prioritise patients most likely to gain outcomes benefit. Hospital-linked initiation pathways, especially for cardiovascular patients, increasingly influence volume, which supports hospital pharmacy and specialist clinic channels over purely retail-driven growth.
- Germany: Demand for SGLT2 inhibitors in Germany is rising at 7.1% CAGR through 2036, driven by structured care pathways integrating cardiorenal protection agents for diabetes patients with elevated cardiovascular risk profiles. German Institute for Quality and Efficiency in Health Care evaluations supporting reimbursement decisions create formal evidence requirements that manufacturers address through outcomes data submissions demonstrating hospitalization reduction and quality-adjusted life year improvements justifying premium pricing maintenance despite generic competition pressure in related drug classes.
FMI’s analysis of SGLT2 inhibitors market in Europe consists of country-wise assessment that includes Germany, the United Kingdom, France, Italy, Spain, Nordic, BENELUX, and Rest of Europe. Readers can find HTA-linked adoption drivers and pricing/access scenarios by country.
Asia Pacific market analysis
Asia Pacific is volume expansion engine, where rising diagnosis rates, expanding insurance coverage breadth, and faster specialist adoption of evidence-based protocols drive treated patient population growth at pace exceeding mature markets. Global pharmaceutical leaders and regional manufacturers compete on pricing strategies, patient access programs, and hospital channel reach, with national reimbursement listing decisions shaping affordability and systematic uptake velocity.
- China: Demand for SGLT2 inhibitors in China is projected to rise at 8.4% CAGR through 2036. Growth is primarily powered by rising diabetes prevalence, rapid expansion of specialist care in large urban hospital networks, and widening affordability through reimbursement listings that reduce patient out-of-pocket burden. National reimbursement list updates influence which molecules scale fastest, and price negotiations can accelerate uptake while compressing margins, shifting competition toward volume, distribution reach, and patient support programs. [7]
- India: Demand for SGLT2 inhibitors in India is projected to rise at 7.7% CAGR through 2036. Growth is linked to an expanding diagnosed base, increased use of modern diabetes algorithms among urban specialists, and wider availability of branded and lower-cost options that improve access in private care. Clinical adoption also rises as physicians address cardiovascular and renal risk alongside glycaemic control, increasing the share of patients placed on SGLT2 inhibitors earlier in therapy lines. Constraints remain affordability and uneven access: outside major metros, follow-up monitoring and patient persistence can be weaker, which slows net demand despite high disease prevalence.
The full report analyzes the SGLT2 inhibitors market across East and South Asia from 2021-2036, covering reimbursement impacts, distribution channel structure shifts, and disease-burden-linked demand forecasts in China, Japan, South Korea, India, ASEAN, Australia and New Zealand. The assessment highlights pricing and access trends that dictate regional adoption velocity and therapy persistence patterns.
Latin America market analysis
Latin America is reimbursement-and-procurement paced region where growth velocity depends on public healthcare system adoption decisions and government procurement cycle timing rather than pure clinical demand. Multinational pharmaceutical companies compete with localized access strategies addressing affordability barriers, but public coverage breadth and patient out-of-pocket payment capacity govern therapy persistence and systematic uptake beyond initial prescription.
- Brazil: A projected 4.6% CAGR through 2036 is supported by public reimbursement decisions and pace at which therapies are incorporated into guideline-based care protocols within Sistema Único de Saúde-linked healthcare settings. Brazil Ministry of Health CONITEC access decisions reviewed in February 2026 demonstrate formal health technology assessment processes governing inclusion of newer diabetes and cardiovascular medications in public reimbursement formularies [8]. As Brazilian clinicians adopt cardiorenal risk management frameworks aligned with international professional society guidance, SGLT2 inhibitors expand beyond narrow diabetes-only use case into heart failure and chronic kidney disease patient populations, but diffusion speed depends on procurement inclusion timelines and regional budget allocation decisions.
The report consists of detailed analysis for the market in Brazil, Chile, and Rest of Latin America. Readers can find detailed information about several factors, such as access scenarios, procurement-linked growth sensitivity, and private-versus-public channel dynamics, which are impacting growth in the Latin America region.
Competitive Aligners for Market Players
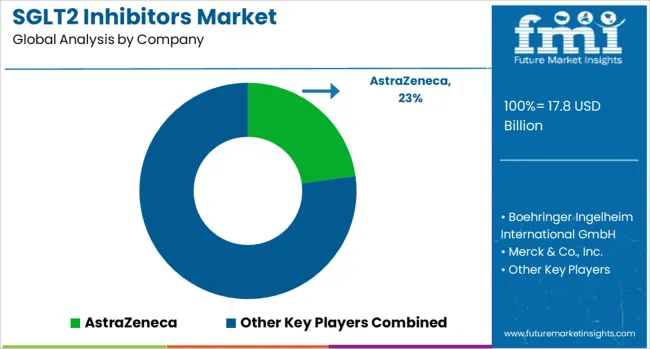
The SGLT2 inhibitors market remains moderately concentrated, with leading branded products capturing the majority of global value, while smaller brands and “other SGLT2” contributors fill regional and niche positions. Competitive advantage is built on label breadth, outcomes evidence credibility, and payer contracting capability, since class differentiation is evaluated through hard endpoints and reimbursement logic rather than symptomatic relief.
Structural advantage sits with companies that can defend premium positioning through multi-indication narratives (T2D plus HF plus CKD), invest in outcomes data generation, and support specialist pathway integration at hospital level. Firms with strong cardiology and nephrology commercial footprint convert non-endocrine initiations into repeat dispensing, improving persistence and share stability even under step therapy. [2] [4]
Buyer behaviour is increasingly payer-led and protocol-driven. Large health systems seek budget predictability and therapy optimisation through formularies, which compresses pricing power but rewards suppliers that can demonstrate hospitalisation reduction and renal progression delay in real-world cohorts. This pushes companies toward evidence packages, adherence programs, and pathway partnerships rather than pure promotional spend. [2]
Recent Developments:
- In February 2026, AstraZeneca reported continued growth momentum entering 2026 positioning medicines portfolio and pipeline for long-term delivery with emphasis on high-value therapy areas including cardiorenal disease management [4].
- In October 2024, NICE published implementation-focused materials supporting roll-out of SGLT2 inhibitor recommendations in Type 2 Diabetes care pathways across National Health Service settings [6].
Key Players in the SGLT2 Inhibitors Market
- AstraZeneca PLC
- Boehringer Ingelheim International GmbH
- Merck & Co., Inc.
- Johnson & Johnson Services, Inc.
- Lexicon Pharmaceuticals, Inc.
- Eli Lilly and Company
- Bristol-Myers Squibb Company
- Glenmark Pharmaceuticals Limited
- TheracosBio, LLC
Scope of the Report
| Items | Values |
|---|---|
| Quantitative units (Base year and forecast) | USD 17.8 billion (2025), USD 18.9 billion (2026), USD 34.5 billion (2036); CAGR 6.2% (2026 to 2036) |
| Drug coverage | Jardiance (empagliflozin); Farxiga (dapagliflozin); Invokana (canagliflozin); Inpefa (sotagliflozin); Qtern (dapagliflozin/saxagliptin); Other SGLT2 inhibitors |
| Indication coverage | Type 2 Diabetes; Cardiovascular Diseases; Chronic Kidney Disease (CKD); Others |
| Distribution channel coverage | Hospital Pharmacies; Retail Pharmacies; Online Pharmacies |
| Regions covered | North America; Europe; Asia Pacific; Latin America; Middle East and Africa |
| Countries covered | United States; Canada; Mexico; Germany; United Kingdom; France; Italy; Spain; Nordic; BENELUX; Rest of Europe; China; Japan; South Korea; India; ASEAN; Australia and New Zealand; Rest of Asia Pacific; Brazil; Chile; Rest of Latin America; Kingdom of Saudi Arabia; Other GCC Countries; Turkey; South Africa; Other African Union; Rest of Middle East and Africa (40+ countries in total) |
| Key companies profiled | AstraZeneca PLC; Boehringer Ingelheim International GmbH; Merck & Co., Inc.; Johnson & Johnson Services, Inc.; Lexicon Pharmaceuticals, Inc.; Eli Lilly and Company; Bristol-Myers Squibb Company; Glenmark Pharmaceuticals Limited; TheracosBio, LLC |
| Approach | Hybrid top down and bottom up market modeling combining epidemiology-linked treatment penetration with molecule revenue build, validated through primary interviews with specialists and payer stakeholders, supported by guideline placement analysis and reimbursement policy benchmarking |
SGLT2 Inhibitors Market Analysis by Segments
Drug:
- Jardiance (empagliflozin)
- Farxiga (dapagliflozin)
- Invokana (canagliflozin)
- Inpefa (sotagliflozin)
- Qtern (dapagliflozin/saxagliptin)
- Other SGLT2 Inhibitors
Indication:
- Type 2 Diabetes
- Cardiovascular Diseases
- Chronic Kidney Disease (CKD)
- Others
Distribution Channel:
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
Region:
- North America
- United States
- Canada
- Mexico
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- Asia Pacific
- China
- Japan
- South Korea
- India
- ASEAN
- Australia and New Zealand
- Rest of Asia Pacific
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East and Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East and Africa
Bibliography
- [1] American Diabetes Association. (2025). Standards of Care in Diabetes. Professional Practice Committee, January 2025.
- [2] U.S. Food and Drug Administration. (2023). FARXIGA (dapagliflozin) Prescribing Information. FDA Label Repository, Updated 2023.
- [3] Scottish Medicines Consortium. (2024). Empagliflozin (Jardiance) Final Advice (Amended). SMC Detailed Advice Document, July 2024.
- [4] AstraZeneca PLC. (2026). Full-year and Q4 2025 Results. Corporate Financial Results Press Release, February 2026.
- [5] Kidney Disease: Improving Global Outcomes (KDIGO). (2024). KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney International, March 2024.
- [6] National Institute for Health and Care Excellence (NICE). (2024). Type 2 Diabetes in Adults: Management (NG28) Resource Impact Report. NICE Implementation Materials, October 2024.
- [7] Center for Drug Evaluation, NMPA & China Hospital Pharmaceutical Affairs Association (CIRS & CHPA). (2024). China Adds 91 Drugs to 2024 National Reimbursement Drug List (NRDL). NRDL Update Announcement, December 2024.
- [8] Brazil Ministry of Health, National Commission for Health Technology Incorporation (CONITEC). (2026). CONITEC Publications and Access Decisions Repository. Ministry of Health Official Publications, February 2026.
- [9] European Medicines Agency (EMA). (2024). SGLT2 Inhibitors Assessment Reports and Product Information. EMA Medicine Database, Various 2024 Updates.
- [10] World Health Organization (WHO). (2024). Global Report on Diabetes and Cardiovascular Disease. WHO Technical Report Series, November 2024.
This Report Addresses
- Market intelligence to enable strategic decision making across pharmaceutical manufacturers, payer organizations, and healthcare system formulary committees
- Market size estimation and 10-year revenue forecasts from 2026 to 2036 supported by epidemiology modeling and reimbursement benchmarks
- Growth opportunity mapping across branded drugs, indication expansion, and distribution channels with cardiorenal positioning emphasis
- Segment and regional revenue forecasts covering Type 2 Diabetes, cardiovascular diseases, and chronic kidney disease applications
- Competition strategy assessment including outcomes evidence positioning, payer contracting dynamics, and specialist pathway integration
- Indication mix evolution tracking from diabetes-only to multi-indication cardiorenal protection positioning across drug portfolio
- Regulatory and reimbursement impact analysis covering guideline updates, health technology assessment decisions, and utilization management
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive and operational use
Frequently Asked Questions
How large is the SGLT2 inhibitors market in 2025?
The global SGLT2 inhibitors market is valued at USD 17.8 billion in 2025.
What is the projected market size for 2026?
The market is estimated to reach USD 18.9 billion in 2026.
What will the SGLT2 inhibitors market size be by 2036?
FMI projects the market will reach USD 34.5 billion by 2036.
What is the expected CAGR for 2026 to 2036?
The market is forecast to expand at a 6.2% CAGR from 2026 to 2036.
How much absolute growth is expected between 2026 and 2036?
The market is expected to add about USD 15.6 billion in value over 2026 to 2036.
Which indication leads the market today?
Type 2 Diabetes leads, holding 71.8% share in 2025.
Which drug is projected to remain the leading revenue contributor?
Jardiance (empagliflozin) leads with 55.3% share in 2025.
Which distribution channel is most significant?
Hospital pharmacies lead with 50.4% share in 2025 due to specialist-led initiations.
Which country is expected to grow fastest?
China is projected to expand at 8.4% CAGR through 2036.
What is India’s projected growth rate?
India is projected to grow at 7.7% CAGR through 2036.
How does Germany compare on growth?
Germany is projected at 7.1% CAGR through 2036, supported by structured care pathways.
What is the projected CAGR for France?
France is projected to grow at 6.5% CAGR through 2036.
What is the UK’s expected CAGR?
The United Kingdom is forecast at 5.9% CAGR through 2036.
What is the USA’s expected CAGR?
The United States is projected to expand at 5.3% CAGR through 2036.
How fast is Brazil expected to grow?
Brazil is forecast at 4.6% CAGR through 2036.
What is the biggest near-term growth lever for manufacturers?
Growth is most sensitive to cardiorenal pathway integration that shifts initiation into cardiology and nephrology.
What is the largest commercial constraint in mature markets?
The main constraint is payer utilisation management (step therapy, prior authorisation) that limits net expansion.
Which segment should new entrants prioritise?
New entrants should prioritise hospital-originated starts and high-risk CKD/HF cohorts where persistence tends to be stronger.
How important are generics to the 2026 to 2036 outlook?
Generics are expected to compress net pricing, making volume growth and adherence programs more important to defend value.
What is the headline 2036 number executives should plan around?
The market is projected to reach USD 34.5 billion by 2036 at 6.2% CAGR from 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Indication , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Indication , 2026 to 2036
- Type 2 Diabetes
- Cardiovascular Diseases
- Chronic Kidney Disease (CKD)
- Type 2 Diabetes
- Y to o to Y Growth Trend Analysis By Indication , 2021 to 2025
- Absolute $ Opportunity Analysis By Indication , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Drug
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Drug, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Drug, 2026 to 2036
- Jardiance (empagliflozin)
- Farxiga (dapagliflozin)
- Invokana (canagliflozin)
- Inpefa (sotagliflozin)
- Qtern (dapagliflozin/saxagliptin)
- Jardiance (empagliflozin)
- Y to o to Y Growth Trend Analysis By Drug, 2021 to 2025
- Absolute $ Opportunity Analysis By Drug, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Hospital Pharmacies
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Indication
- By Drug
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By Drug
- By Distribution Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Indication
- By Drug
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By Drug
- By Distribution Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Indication
- By Drug
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By Drug
- By Distribution Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Indication
- By Drug
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By Drug
- By Distribution Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Indication
- By Drug
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By Drug
- By Distribution Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Indication
- By Drug
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By Drug
- By Distribution Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Indication
- By Drug
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By Drug
- By Distribution Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Drug
- By Distribution Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Drug
- By Distribution Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Drug
- By Distribution Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Drug
- By Distribution Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Drug
- By Distribution Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Drug
- By Distribution Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Drug
- By Distribution Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Drug
- By Distribution Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Drug
- By Distribution Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Drug
- By Distribution Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Drug
- By Distribution Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Drug
- By Distribution Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Drug
- By Distribution Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Drug
- By Distribution Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Drug
- By Distribution Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Drug
- By Distribution Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Drug
- By Distribution Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Drug
- By Distribution Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Drug
- By Distribution Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Drug
- By Distribution Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Drug
- By Distribution Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Drug
- By Distribution Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Indication
- By Drug
- By Distribution Channel
- Competition Analysis
- Competition Deep Dive
- AstraZeneca PLC
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Boehringer Ingelheim International GmbH
- Merck & Co., Inc.
- Johnson & Johnson Services, Inc.
- Lexicon Pharmaceuticals, Inc.
- Eli Lilly and Company
- Bristol-Myers Squibb Company
- AstraZeneca PLC
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Indication , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Drug, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Indication , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Drug, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Indication , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Drug, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Indication , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Drug, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Indication , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Drug, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Indication , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Drug, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Indication , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Drug, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Indication , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Drug, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Indication , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Indication , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Indication
- Figure 6: Global Market Value Share and BPS Analysis by Drug, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Drug, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Drug
- Figure 9: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Distribution Channel
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Indication , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Indication , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Indication
- Figure 26: North America Market Value Share and BPS Analysis by Drug, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Drug, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Drug
- Figure 29: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Distribution Channel
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Indication , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Indication , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Indication
- Figure 36: Latin America Market Value Share and BPS Analysis by Drug, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Drug, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Drug
- Figure 39: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Indication , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Indication , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Indication
- Figure 46: Western Europe Market Value Share and BPS Analysis by Drug, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Drug, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Drug
- Figure 49: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Indication , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Indication , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Indication
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Drug, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Drug, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Drug
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Indication , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Indication , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Indication
- Figure 66: East Asia Market Value Share and BPS Analysis by Drug, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Drug, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Drug
- Figure 69: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Indication , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Indication , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Indication
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Drug, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Drug, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Drug
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Indication , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Indication , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Indication
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Drug, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Drug, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Drug
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

