Sterile Packaging Market
Sterile Packaging Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2015 to 2023 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2026 to 2035
Sterile Packaging Market Forecast and Outlook 2026 to 2036
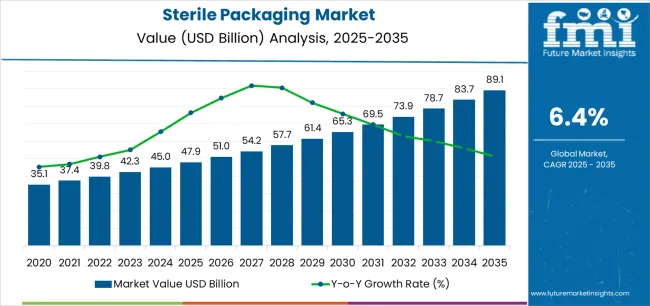
The sterile packaging market is valued at USD 51.0 billion in 2026 and is forecasted to reach USD 94.8 billion by 2036, registering a CAGR of 6.4%. Demand expansion reflects rising intolerance for contamination risk across pharmaceuticals, medical devices, diagnostics, and aseptic food systems, where packaging failure directly translates into regulatory exposure and product loss. Sterile barriers function as risk-control infrastructure rather than secondary packaging, concentrating value in materials and formats that maintain integrity across sterilization, transport, and storage cycles. Adoption favors solutions with proven seal reliability, material consistency, and compatibility with validated sterilization methods, as failure costs often exceed incremental packaging spend. Commercial relevance is tied to assurance rather than volume, positioning sterile packaging as a compliance-linked operating requirement.
Quick Stats for Sterile Packaging Market
- Sterile Packaging Market Value (2026): USD 51.0 billion
- Sterile Packaging Market Forecast Value (2036): USD 94.8 billion
- Sterile Packaging Market Forecast CAGR 2026 to 2036: 6.4%
- Leading Material Type: Plastic Films
- Fastest-Growing Countries: China, India, USA, Germany, Japan
- Key Players: Berry Global Group, Amcor, WestRock, Huhtamäki, Sealed Air
Structural shifts in manufacturing and distribution further reinforce demand quality. Growth of biologics, injectable, and combination products increases reliance on packaging that tolerates aggressive sterilization without compromising barrier performance. Cross-border supply chains, contract manufacturing, and cold-chain logistics expose products to repeated handling, elevating importance of mechanical strength, traceability, and tamper evidence. Automation within pharmaceutical and device packaging lines rewards substrates with predictable sealing behavior and low defect tolerance. Sustainability pressure influences material selection, though sterility assurance remains primary constraint, limiting substitution flexibility. Food and beverage applications using aseptic processing add incremental demand where shelf-life extension justifies sterile formats. Expansion is anchored in regulatory intensity, product value protection, and operational risk reduction, not discretionary packaging upgrades or short-term consumption cycles.
Sterile Packaging Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 51.0 billion |
| Market Forecast Value (2036) | USD 94.8 billion |
| Forecast CAGR 2026 to 2036 | 6.4% |
Why is the Demand for Sterile Packaging Market Growing?
Demand for sterile packaging is growing globally due to increasing requirements for product safety, contamination control, and extended shelf life across healthcare, pharmaceutical, and food sectors. Sterile packaging solutions protect medical devices, surgical instruments, diagnostic kits, and implantables from microbial contamination during storage and transport, which aligns with stringent regulatory frameworks enforced in multiple regions. Regulatory authorities require validated sterile barriers, tamper evidence, and sterilization compatibility to ensure patient safety and support quality management systems within hospitals and clinics.
Growth in biologics, vaccines, and advanced therapies increases need for sterile packaging formats that maintain product integrity under controlled environments. In food and beverage markets, sterile packaging extends freshness, reduces spoilage, and supports aseptic distribution models, which improves product quality across long supply chains. Consumer expectations for hygienic packaging, clear labeling, and minimized contamination risk reinforce adoption of certified solutions. Advances in barrier materials, multi-layer films, and sterilization-friendly substrates improve performance, durability, and process compatibility with automated packaging lines. Expansion of global manufacturing, e-commerce, and cold chain logistics increases exposure to variable conditions that demand robust sterile packaging. Continued emphasis on product safety, compliance, and quality assurance supports sustained growth of sterile packaging across international markets.
How Is the Sterile Packaging Market Structured by Material Type and Application?
Sterile packaging demand is driven by infection prevention standards, regulatory compliance requirements, and expansion of pharmaceutical and medical device manufacturing. Packaging systems must maintain sterility throughout storage, transport, and handling while ensuring compatibility with sterilization methods such as gamma irradiation, ethylene oxide, and steam. Market structure reflects risk management priorities, material performance under sterilization stress, and cost-to-volume efficiency across healthcare supply chains.
Which Material Type Holds the Largest Share in the Sterile Packaging Market?
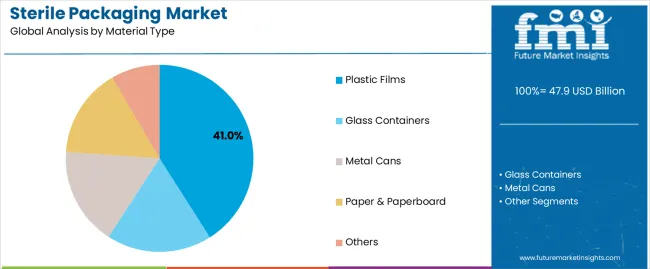
Plastic films account for the largest material share at 41.0%, reflecting their versatility, seal integrity, and compatibility with multiple sterilization technologies. Plastic films are widely used for blister packs, pouches, wraps, and sachets due to controlled permeability, high tensile strength, and consistent barrier performance. Their lightweight nature also supports logistics efficiency and reduced transportation costs for high-volume sterile products.
Glass containers remain essential for applications requiring high chemical inertness and long-term stability, particularly for injectable pharmaceuticals and biologics. Metal cans are used selectively where mechanical protection and hermetic sealing are critical. Paper and paperboard materials support breathable sterile barrier systems, especially for surgical instruments and medical device trays, while other materials include composites and specialty laminates engineered for niche sterilization and handling requirements.
Key Observations
- Plastic films dominate due to flexibility and sterilization compatibility
- Glass remains critical for injectable and sensitive formulations
- Paper-based systems support breathable sterile barriers
Which Application Segment Represents the Highest Demand?
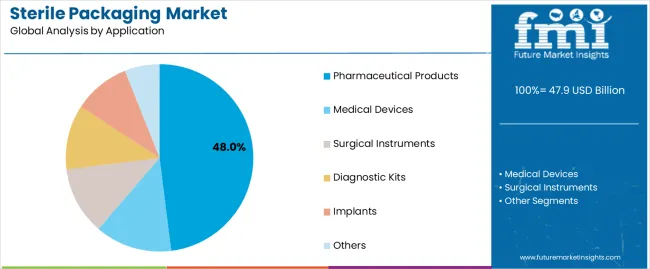
Pharmaceutical products represent the largest application segment at 48.0%, driven by high-volume production of sterile injectable, biologics, vaccines, and ophthalmic formulations. Regulatory scrutiny around drug sterility and patient safety reinforces demand for validated sterile packaging solutions that ensure integrity across global distribution networks.
Medical devices, surgical instruments, diagnostic kits, and implants form significant secondary segments. These applications require packaging systems that protect against microbial ingress while allowing sterilization and aseptic presentation in clinical environments. Growth in minimally invasive procedures and diagnostic testing expands demand for single-use sterile packaging formats across these categories. Other applications include laboratory consumables and specialized healthcare components.
Key Observations
- Pharmaceuticals lead due to volume and regulatory intensity
- Medical devices and instruments drive diverse packaging needs
- Single-use healthcare products support steady demand expansion
What are the Key Dynamics in the Sterile Packaging Market?
Global demand rises as medical device manufacturers, pharmaceutical companies, and healthcare providers adopt sterile packaging solutions to protect products from microbial contamination, ensure patient safety, and extend shelf life. Sterile packaging includes barrier films, pouches, rigid trays, and overwraps designed to maintain sterility through distribution, storage, and point of use. Adoption aligns with rising surgical procedures, biologics production, outpatient care growth, and stringent hygiene expectations across North America, Europe, Asia-Pacific, and Latin America. Usage spans single-use medical devices, injectable drugs, implantable products, and diagnostics requiring validated sterile barriers.
How do regulatory requirements and clinical safety priorities shape adoption?
Regulatory agencies impose strict sterile packaging standards and validation protocols to ensure integrity of barrier systems, sterility maintenance, and contamination prevention. Manufacturers integrate sterile packaging into quality management systems with documented sterilization methods, barrier performance testing, and traceability from production to patient use. Adoption increases where healthcare-acquired infection prevention is a priority and regulators mandate sterility assurance levels for devices and pharmaceuticals. Retailers and healthcare networks emphasize packaging that supports aseptic presentation, tamper evidence, and compatibility with automated dispensing systems. Performance criteria include barrier resistance to microbes, mechanical protection, and compatibility with sterilization technologies such as gamma irradiation or ethylene oxide.
How do cost pressures, material complexity, and sustainability expectations influence market scalability?
Sterile packaging systems often involve advanced materials and multi-layer constructions that increase production cost relative to non-sterile options. Material selection must balance barrier performance, mechanical strength, and seal integrity with compatibility across sterilization methods. Rising cost pressures drive interest in optimized designs that reduce material use without compromising protection. Healthcare providers and regulators increasingly emphasize sustainability, prompting development of recyclable or reduced-material solutions that address environmental impact. Complexity in supply chains for specialized films, trays, and pouches affects planning and delivery timelines. Variability in regional regulatory frameworks for medical packaging labeling and disposal practices shapes compliance strategies. Long-term growth depends on innovations that improve cost-efficiency, maintain stringent safety performance, and align sterile packaging practices with global sustainability and regulatory expectations across diverse healthcare and pharmaceutical markets.
How Is Demand for Sterile Packaging Evolving Globally?
Demand for sterile packaging is increasing globally due to rising pharmaceutical production, growth of medical devices, and stricter regulatory requirements for contamination control. China leads with a 7.8% CAGR, supported by large-scale pharmaceutical manufacturing and export-oriented sterile drug production. India follows at 7.5%, driven by expansion of injectable drugs and contract manufacturing. USA records a 7.1% CAGR, shaped by regulatory compliance and growth of biologics and combination products. Germany posts 6.3%, reflecting precision packaging standards and strong medical device output. Japan records 5.9%, supported by stable healthcare demand and quality-focused packaging practices. Growth reflects sustained need for validated sterile barrier systems across healthcare and life science supply chains.
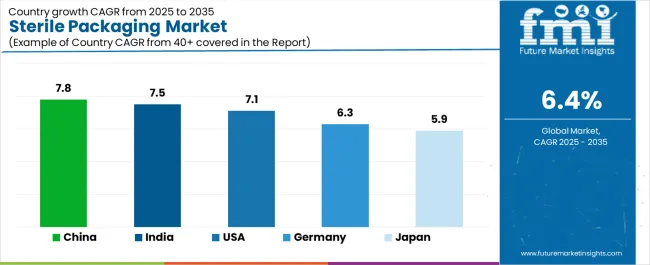
| Country | CAGR (%) |
|---|---|
| China | 7.8% |
| India | 7.5% |
| USA | 7.1% |
| Germany | 6.3% |
| Japan | 5.9% |
What is driving strong growth of sterile packaging demand in China?
China demand is driven by rapid expansion of pharmaceutical manufacturing, vaccine production, and medical device exports. CAGR of 7.8% reflects increasing use of sterile packaging for injectable, IV solutions, and diagnostic devices. Regulatory authorities strengthen requirements for sterile barrier integrity and traceability. High-volume production necessitates scalable packaging solutions with consistent quality. Domestic packaging manufacturers invest in cleanroom capacity and automation. Export compliance further accelerates adoption of validated sterile packaging formats. Growth remains scale-driven and compliance-aligned, supported by healthcare industrialization and global supply participation.
- Expansion of pharmaceutical and vaccine manufacturing
- Export-driven compliance with sterile packaging standards
- Investment in cleanroom and automated packaging lines
- High demand for injectable and diagnostic packaging
How is India sustaining growth in sterile packaging adoption?
India demand reflects growth of injectable pharmaceuticals, biosimilar, and contract manufacturing services. CAGR of 7.5% is supported by rising output of sterile formulations for domestic use and export markets. Regulatory alignment with international standards drives packaging upgrades. Manufacturers require cost-effective yet compliant sterile barrier systems. Expansion of hospital infrastructure supports additional demand. Domestic packaging suppliers scale production of blister packs, vials, and pouches. Growth remains manufacturing-led and cost-aligned, supported by pharmaceutical exports and healthcare capacity expansion.
- Growth of injectable and biosimilar production
- Export-driven regulatory compliance requirements
- Need for cost-effective sterile barrier solutions
- Expansion of domestic packaging manufacturing
What factors are shaping sterile packaging demand growth in the USA?
USA demand is shaped by growth of biologics, cell and gene therapies, and complex medical devices. CAGR of 7.1% reflects increasing need for high-integrity sterile packaging systems with traceability and validation. Regulatory oversight emphasizes sterility assurance and material compatibility. Pharmaceutical and device manufacturers adopt advanced barrier films and rigid sterile packs. Single-use medical products contribute steady volume demand. Growth remains quality-driven and compliance-focused, supported by innovation in healthcare products and strict regulatory frameworks.
- Expansion of biologics and advanced therapies
- Strong regulatory emphasis on sterility assurance
- Adoption of advanced sterile barrier materials
- Stable demand from single-use medical products
Why is Germany showing steady expansion in sterile packaging demand?
Germany demand reflects strong medical device manufacturing, pharmaceutical quality standards, and regulatory discipline. CAGR of 6.3% is supported by use of sterile packaging for implants, diagnostics, and injectable drugs. Manufacturers prioritize precision, documentation, and validated processes. Packaging solutions emphasize material consistency and barrier reliability. Collaboration between packaging suppliers and device manufacturers supports innovation. Growth remains technology-led and quality-focused, aligned with stringent compliance and export requirements.
- Strong medical device and pharma manufacturing base
- Emphasis on validated and documented packaging systems
- Focus on barrier reliability and material precision
- Collaboration across healthcare supply chains
What is shaping more measured growth of sterile packaging demand in Japan?
Japan demand reflects mature healthcare infrastructure and high standards for product safety. CAGR of 5.9% is supported by stable demand from pharmaceuticals, diagnostics, and medical devices. Manufacturers emphasize reliability, precision, and long-term supplier relationships. Regulatory frameworks ensure consistent sterility requirements. Growth remains incremental, driven by replacement cycles and gradual product innovation rather than capacity expansion. Domestic suppliers focus on quality consistency and compliance. Growth remains quality-driven and stability-focused, aligned with disciplined healthcare manufacturing practices.
- Mature healthcare and pharmaceutical markets
- Emphasis on reliability and long-term quality
- Incremental growth through replacement demand
- Strong regulatory oversight and compliance
What is the competitive landscape of demand for the sterile packaging market globally?
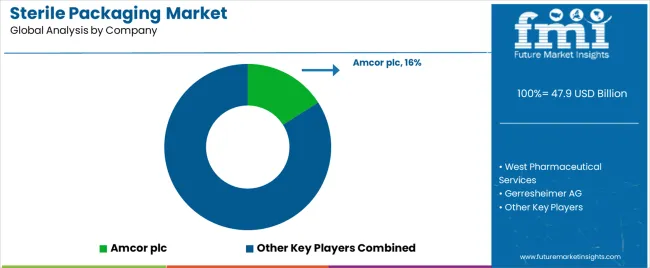
Demand for sterile packaging is driven by growth in pharmaceuticals, medical devices, biologics, and advanced therapies requiring contamination-controlled environments. Buyers evaluate barrier performance, microbial ingress resistance, compatibility with sterilization methods such as E-beam, gamma, and EtO, and supply chain reliability under strict regulatory frameworks. Procurement teams prioritize suppliers able to deliver validated sterile barrier systems, traceability, regulatory documentation, and global logistics support. Trend in the global market reflects increasing adoption of complex primary packaging for injectables, prefilled syringes, and combination products, alongside stringent quality expectations across developed and emerging markets.
Berry Global Group holds leading positioning through extensive sterile barrier packaging solutions, medical pouches, and trays backed by a broad global footprint. Amcor participates with sterile medical and pharma packaging systems designed to meet regulatory standards and commingling controls. WestRock supports demand with sterile board and paper-based systems used in device and healthcare packaging. Huhtamäki supplies sterile pharma packaging formats and containment solutions aligned with driving patient safety. Sealed Air contributes sterile packaging films and systems enabling high-barrier performance. Competitive differentiation depends on barrier quality, sterilization method compatibility, documentation support, and ability to scale validated solutions across multilocation operations.
Key Players in the Sterile Packaging Market
- Berry Global Group
- Amcor
- WestRock
- Huhtamäki
- Sealed Air
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Material Type | Plastic Films; Glass Containers; Metal Cans; Paper & Paperboard; Others |
| Application | Pharmaceutical Products; Medical Devices; Surgical Instruments; Diagnostic Kits; Implants; Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | China, India, USA, Germany, Japan, and 40+ countries |
| Key Companies Profiled | Berry Global Group; Amcor; WestRock; Huhtamäki; Sealed Air |
| Additional Attributes | Dollar sales by material type and application; adoption trends for sterile barrier systems across pharmaceutical and medical device supply chains; material integrity, microbial barrier performance, and seal reliability metrics; compatibility with aseptic processing, sterilization methods, and regulatory validation protocols; shelf-life assurance, transport durability, and traceability considerations; regulatory compliance with global sterile packaging standards influencing material and format selection. |
Sterile Packaging Market by Segment
Material Type:
- Plastic Films
- Glass Containers
- Metal Cans
- Paper & Paperboard
- Others
Application:
- Pharmaceutical Products
- Medical Devices
- Surgical Instruments
- Diagnostic Kits
- Implants
- Others
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Frequently Asked Questions
How big is the sterile packaging market in 2026?
The global sterile packaging market is estimated to be valued at USD 51.0 billion in 2026.
What will be the size of sterile packaging market in 2036?
The market size for the sterile packaging market is projected to reach USD 94.8 billion by 2036.
How much will be the sterile packaging market growth between 2026 and 2036?
The sterile packaging market is expected to grow at a 6.4% CAGR between 2026 and 2036.
What are the key product types in the sterile packaging market?
The key product types in sterile packaging market are plastic films, glass containers, metal cans, paper & paperboard and others.
Which application segment to contribute significant share in the sterile packaging market in 2026?
In terms of application, pharmaceutical products segment to command 48.0% share in the sterile packaging market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Material Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Material Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Material Type , 2026 to 2036
- Plastic Films
- Glass Containers
- Metal Cans
- Paper & Paperboard
- Others
- Plastic Films
- Y to o to Y Growth Trend Analysis By Material Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Material Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Pharmaceutical Products
- Medical Devices
- Surgical Instruments
- Diagnostic Kits
- Implants
- Others
- Pharmaceutical Products
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Material Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Material Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Material Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Material Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Material Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Material Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Material Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Material Type
- By Application
- Competition Analysis
- Competition Deep Dive
- Berry Global Group
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Amcor
- WestRock
- Huhtamäki
- Sealed Air
- Berry Global Group
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Material Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Material Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Material Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Material Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Material Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Material Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Material Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Material Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Material Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Material Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Material Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Material Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Material Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Material Type
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Material Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Material Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Material Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Material Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Material Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Material Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Material Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Material Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Material Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Material Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Material Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Material Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Material Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Material Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Material Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Material Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Material Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Material Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

