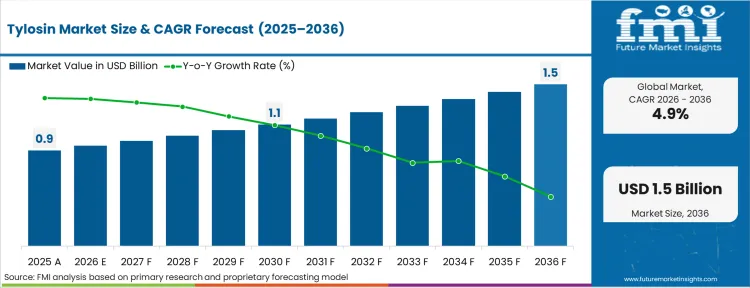
The Tylosin market progressed from USD 0.8 billion in 2025, propelling it to cross USD 0.9 billion in 2026 at a CAGR of 4.9% during the forecast period. Predicting a significant boost in the assessed market value to USD 1.3 billion through 2036, as intensive livestock integrators optimize mortality rates against tightening antimicrobial resistance frameworks.
Integrators are abandoning continuous prophylactic feed inclusion in favor of targeted therapeutic intervention. This shift forces farm managers to balance transient mortality spikes against the commercial risk of failing export residue protocols. Operations delaying this operational pivot face disqualification from tier-one global quick-service restaurant supply chains. Implementing targeted water-soluble macrolides requires a fundamental redesign of farm dosing infrastructure.
The transition to digital veterinary prescriptions acts as the primary inflection point. Once farm management software integrates directly with oversight bodies, the friction of securing approvals for veterinary antibiotics drops significantly. This digital gating transforms procurement from a bulk commodity purchase to a precise, data-backed medical intervention.
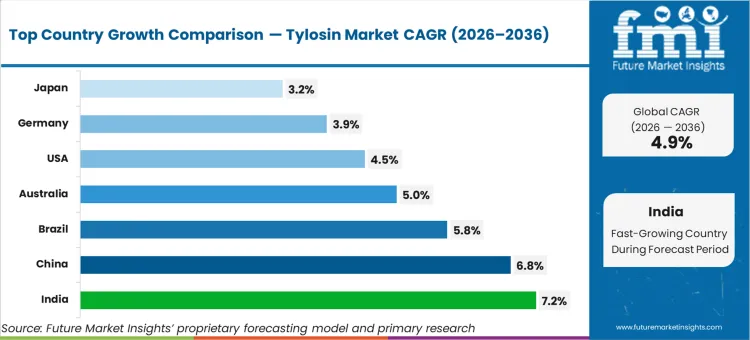
India leads geographic expansion, poised to advance at a CAGR of 7.2%, followed closely by China at 6.8%. Brazil is estimated to track at 5.8%, while Australia and the USA are expected to record 5.0% and 4.5%, respectively. Germany and Japan follow at 3.9% and 3.2%. This divergence reflects the operational gap between markets formalizing their intensive farming infrastructure and those actively suppressing baseline antibiotic volumes through legislative mandates.
Tylosin is a macrolide antibiotic produced via fermentation, specifically indicated for veterinary use to combat Gram-positive bacteria and specific mycoplasma strains in livestock. The boundary of this market restricts itself to veterinary formulations deployed in intensive protein production, excluding human-grade macrolides. This specific categorization allows precise animal feed additives share analysis by isolating agricultural procurement from broad pharmaceutical supply chains.
The scope encompasses all commercial salt forms of the active pharmaceutical ingredient, primarily tartrate and phosphate variations. It includes bulk premixes designed for milling integration, water-soluble powders for acute herd dosing, and injectable formulations for individual clinical treatment. Revenue tracking incorporates medicated feed additives explicitly labeled for growth promotion where legally permitted, alongside strictly therapeutic veterinary drugs.
The analysis explicitly excludes off-label human antibiotic compounds diverted into agricultural use. It also omits baseline nutritional feed supplements, vitamins, and non-medicated growth promoters that do not rely on antimicrobial mechanisms. These categories are excluded because their procurement cycles, regulatory oversight, and supplier ecosystems function entirely independent of veterinary oversight frameworks.
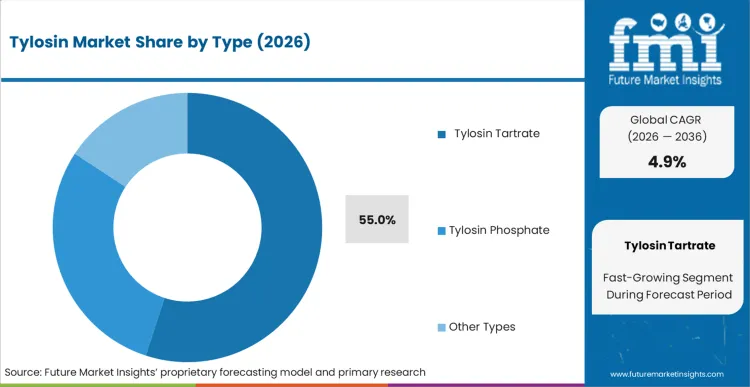
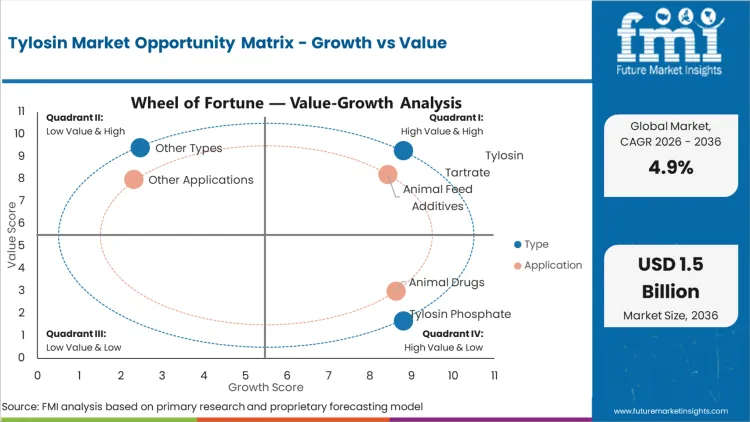
Legacy phosphate premixes fail to deliver the rapid systemic absorption required when a respiratory outbreak hits a high-density broiler house. This operational reality explains why Tylosin Tartrate holds 55.0% share in 2026. Farm operators cannot wait 48 hours for a new batch of medicated feed to be milled, transported, and consumed by sick animals with depressed appetites. According to FMI's estimates, the tartrate salt form solves this bottleneck via drinking water systems, providing immediate therapeutic loading doses. The shift away from continuous feed integration toward acute intervention heavily favors highly soluble animal feed antibiotics. Integrators attempting to manage acute outbreaks with insoluble formulations suffer severe mortality spikes that destroy batch profitability.
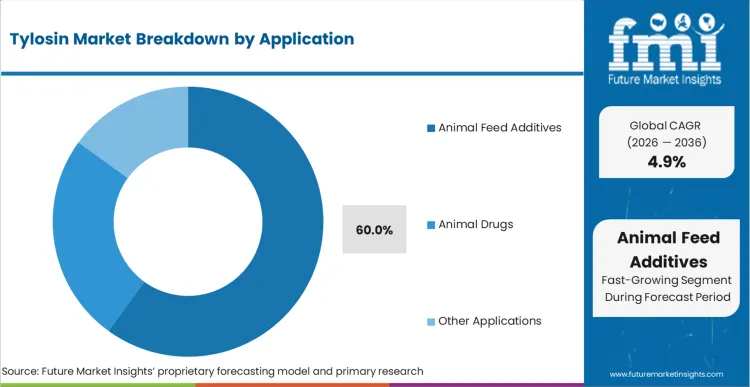
FMI analysts opine that blending the active ingredient at the central mill essentially outsources the dosing labor to the feed manufacturer. This structural advantage makes animal feed additives the default operational standard where strict prescription mandates have not yet materialized. The dominance of Animal Feed Additives, holding 60.0% share in 2026, stems directly from the labor economics of intensive farming in emerging regions. Administering individual injections or constantly managing water line concentrations requires specialized farm labor that thin-margin integrators lack. As regulatory scrutiny tightens, the reliance on automated mill mixing provides a centralized point of audit for compliance officers.
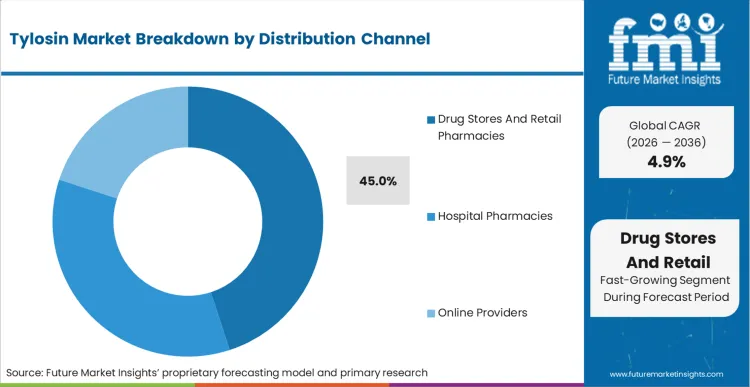
Independent contract growers face a distinct cash flow reality that forces them toward drug stores and retail pharmacies, which account for 45.0% share in 2026. The local network serves as both supplier and informal diagnostic advisor for small-scale operators. Navigating the procurement landscape for animal antimicrobials and antibiotics through centralized corporate channels introduces unacceptable lead times for acute farm-level crises. Unlike massive vertically integrated producers who buy API directly via annual contracts, independent operators purchase inputs on a flock-by-flock basis. Based on FMI's assessment, these localized retail channels provide the essential credit terms and immediate inventory access required when an unexpected bacterial challenge emerges.
Compliance officers at major processing plants no longer evaluate veterinary inputs based on animal growth rates. They are optimizing entirely for zero-delay export clearance. Because a single flock rejected for tissue residues can erase an integrator's quarterly margin, these officers impose rigid standardizations across their entire contract farming networks. This absolute intolerance for residual detection pulls procurement sharply away from generic, long-lasting agricultural compounds. Instead, buyers mandate specific macrolide salts that carry documented, predictable degradation profiles, guaranteeing the active ingredient clears the animal's system exactly when scheduled.
The heaviest drag on market adoption stems directly from the administrative choke point of mandatory veterinary prescriptions. By demanding a unique clinical authorization for every single medicated batch, oversight bodies essentially break the just-in-time procurement model that modern feed mills rely upon to maintain margins. This administrative friction is not an accidental byproduct of poorly written policy; it operates as an intentional regulatory mechanism engineered specifically to suppress overall antimicrobial volume. The integrated digital prescription networks although are beginning to streamline this paperwork, they remain the exclusive domain of highly capitalized corporate operations. Independent producers, meanwhile, are left stranded in an authorization backlog, critically delaying their access to therapeutic inputs during acute outbreaks.
Opportunities in the Tylosin Market
The global landscape for Tylosin reveals a stark geographic divide, while Western geographies aggressively suppress baseline volumes through legislative mandates, emerging Asian hubs rely on massive prophylactic scale to secure their booming protein production. This geographical recalibration forces pharmaceutical providers to continuously adapt their regional supply chain strategies. Based on the regional analysis, the Tylosin market is segmented into North America, South America, Asia Pacific, and Europe across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 7.2% |
| China | 6.8% |
| Brazil | 5.8% |
| Australia | 5.0% |
| USA | 4.5% |
| Germany | 3.9% |
| Japan | 3.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
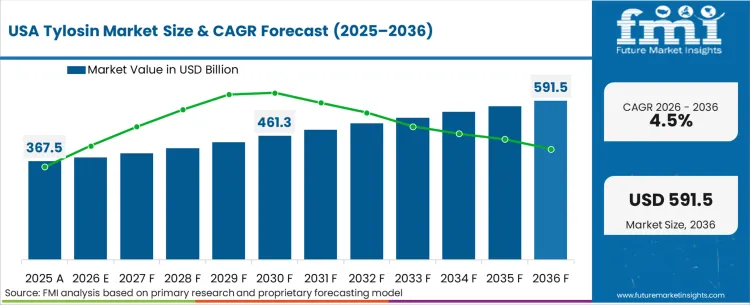
In FMI's view, the enforcement calendar for veterinary directives across the Americas is actively fracturing the commercial landscape into strictly controlled and rapidly realigning zones. Resulting with the procurement priorities to have completely decoupled from baseline nutritional metrics; these compounds are now utilized strictly as managed disease countermeasures. This compliance-led climate elevates the prescribing veterinarian above the nutritionist in the buying hierarchy, forcing stakeholders involved in United States animal healthcare to target clinical decision-makers rather than bulk agricultural buyers.
FMI's report includes extensive analysis of neighboring agricultural hubs modernizing their compliance frameworks. The gradual harmonization of residue testing standards across the broader Mercosur trading bloc steadily eliminates the commercial viability of low-grade, undocumented agricultural pharmaceuticals.
Across the Asia Pacific, cost models and farm consolidation dynamics dictate the foundational baseline. As unorganized backyard farming rapidly evolves into centralized, high-density operations, the sheer magnitude of volume acquisition redirects the global center of gravity. Integrators focus almost exclusively on securing massive quantities of active ingredients to maintain conversion ratios across exponential bird populations, as per FMI's projection. Mirroring this push for raw scale and operational standardization, the regional trajectory highlights an economics-led climate that prioritizes supply chain reliability and bulk pricing over advanced formulation engineering.
FMI's report includes secondary Asian markets currently formalizing their intensive farming sectors. The data clearly indicates that as foreign direct investment modernizes regional feed milling, procurement immediately pivots from fragmented local distributors to direct corporate API contracts.
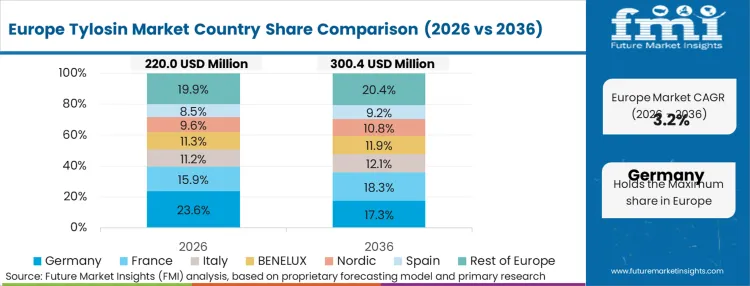
The legislative climate in Europe inherently limits overall volume through aggressive, mandate-driven reduction targets. By penalizing baseline agricultural antibiotic use, European policies compel farm operators to reserve macrolides exclusively for documented clinical emergencies. According to FMI's estimates, this infrastructure-led restriction transforms the supply chain; inventory must sit idle for longer periods, requiring highly stable formulations that survive extended warehousing without degrading. Suppliers operating in this ecosystem are pivoting from volume-based revenue models to high-margin, acute-response delivery systems.
FMI's report includes broader European Union member states navigating identical antimicrobial surveillance frameworks. The systemic pattern across these nations reveals a permanent decoupling of agricultural output growth from veterinary pharmaceutical procurement volumes.
The capacity to ferment high-purity macrolide active pharmaceutical ingredients at a global scale inherently restricts this arena to a concentrated tier of primary manufacturers. Procurement directors at massive poultry integrators do not select their core vendors based on fractional price differences; they evaluate them almost entirely on raw supply chain resilience. When a localized respiratory outbreak hits a high-density facility, it requires an immediate, massive therapeutic intervention, and relying on a supplier with a fragile logistics network risks catastrophic flock mortality. The dominant entities like Zoetis Inc. and Elanco Animal Health Inc., as a result, secure their market positions by physically guaranteeing uninterrupted global delivery across multiple continents. Ceva Santé Animale reinforces this tier by aligning its production output directly with the stringent documentation and residue tracking requirements demanded by top-tier export processing facilities.
The defensive moat protecting these incumbents is built on regulatory capital rather than raw chemical efficacy. Clearing the contradictory approval matrices of North American, European, and Asian agricultural ministries requires a specialized administrative apparatus that regional chemical blenders simply cannot afford. A challenger attempting to replicate the operational footprint of Phibro Animal Health Corporation or Huvepharma AD must first construct a dedicated global compliance team capable of executing simultaneous, cross-border veterinary filings. Beyond administrative hurdles, mastering the specific particle engineering required to produce highly soluble tartrate formulations remains a severe technical barrier. These formulations must remain completely stable without degrading inside automated animal feed antioxidants and continuous-dosing water lines, a manufacturing reality that bulk generic producers consistently fail to achieve.
Large integrated buyers actively attempt to fracture this vendor concentration by qualifying secondary supply sources, yet their ability to diversify is severely capped by rigid export residue limits. This creates a persistent structural tension inside major farming operations: procurement teams are heavily incentivized to source cheaper baseline alternatives, while compliance officers outright refuse to authorize any compound that introduces even a fractional risk of a processing plant rejection. As digital prescription frameworks become the mandatory global standard by 2036, specialized suppliers like Vetoquinol S.A. will capture premium contracts based on how accurately their internal traceability data integrates directly with farm management software. Simultaneously, massive volume producers such as Shandong Lukang Pharmaceutical Co. Ltd. will dictate baseline API pricing across scale-driven Asian hubs, forcing Western competitors to abandon bulk cost strategies and compete entirely on purity guarantees and digital compliance integration.
| Metric | Value |
|---|---|
| Quantitative Units | USD 0.9 billion to USD 1.3 billion, at a CAGR of 4.9% |
| Market Definition | Tylosin is a veterinary-specific macrolide antibiotic utilized in intensive livestock operations. The market captures formulations ranging from prophylactic feed integration to acute therapeutic delivery systems, strictly separated from human pharmaceutical supply channels. |
| Type Segmentation | Tylosin Tartrate, Tylosin Phosphate, Other Types |
| Application Segmentation | Animal Feed Additives, Animal Drugs, Other Applications |
| Distribution Channel Segmentation | Drug Stores And Retail Pharmacies, Hospital Pharmacies, Online Providers |
| Regions Covered | North America, South America, Asia Pacific, Europe |
| Countries Covered | USA, China, India, Brazil, Germany, Japan, Australia, and 40 plus countries |
| Key Companies Profiled | Elanco Animal Health Inc., Zoetis Inc., Ceva Santé Animale, Phibro Animal Health Corporation, Huvepharma AD, Vetoquinol S.A., Shandong Lukang Pharmaceutical Co. Ltd. |
| Forecast Period | 2026 to 2036 |
| Approach | Procurement directors and veterinary compliance officers provided intelligence on formulation switching. Independent feed production indices and registered veterinary output volumes anchored the baseline data. Forecasts were cross-validated using API shipment disclosures and regulatory enforcement timelines. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the Tylosin Market in 2026?
The market reaches USD 0.9 billion in 2026, reflecting the baseline volume needed to sustain intensive livestock operations ahead of strict prescription mandates.
What will it be valued at by 2036?
It is poised to reach USD 1.3 billion by 2036, driven by the premium pricing of high-purity, soluble formulations replacing bulk premixes.
What CAGR is projected?
A 4.9% CAGR is projected, balancing Western volume suppression against scale-driven expansion in emerging Asian hubs.
Which Type segment leads?
Tylosin Tartrate leads with a 55.0% share due to its high solubility, enabling immediate herd-wide water dosing during acute outbreaks.
Which Application segment leads?
Animal Feed Additives capture a 60.0% share because central mill integration efficiently outsources farm-level dosing labor.
Which Distribution Channel segment leads?
Drug Stores And Retail Pharmacies hold a 45.0% share by providing independent growers with immediate inventory and flexible credit during sudden outbreaks.
What drives rapid growth?
Strict export residue protocols compel integrators to adopt premium, rapid-clearance formulations to avoid catastrophic processing plant rejections.
What is the primary restraint?
Mandatory veterinary prescription frameworks create administrative bottlenecks that intentionally disrupt the just-in-time procurement model of modern feed milling.
Which country grows fastest?
India leads at a 7.2% CAGR by rapidly formalizing its unorganized poultry networks, outpacing a heavily capitalized Chinese market.
How do extended producer responsibility and antimicrobial limits affect European procurement?
Aggressive European surveillance eliminates prophylactic baselining, forcing operators to reserve highly specific formulations exclusively for emergency clinical use.
What forces the shift from phosphate premixes to tartrate powders?
Depressed herd appetites during outbreaks make feed-based phosphates ineffective, requiring highly soluble tartrate powders for immediate water-line dosing.
What structural advantage defines the leading API manufacturers?
The capacity to guarantee global supply chain resilience and navigate complex cross-border compliance separates top-tier manufacturers from regional blenders.
Why do independent farmers resist direct corporate API contracts?
Independent farmers lack the capital and storage for direct contracts, relying instead on retail pharmacies to supply short-dated inventory for acute crises.
How does water-line engineering dictate formulation choice?
Integrators mandate highly soluble salts to prevent insoluble powders from causing biofilm buildup and clogging automated water-dosing lines.
What role does software play in this chemical market by 2036?
Cloud-based digital prescription platforms will gatekeep all procurement by automating the compliance paperwork between veterinarians, feed mills, and farms.
Why does Brazil outpace the USA in adoption growth?
Brazil's export-driven integrators rapidly adopt high-purity formulations to meet strict international residue limits, outpacing slower USA regulatory transitions.
How do processing plants enforce formulation standardization?
Processors enforce standardizations through zero-tolerance tissue residue tests at slaughter, where a single failure financially ruins the contract flock.
What hidden costs emerge during acute respiratory outbreaks?
Managing acute outbreaks requires specialized manual labor for water-line dosing, severely spiking farm operating expenses beyond standard mortality losses.
Why do feed mills prefer automated additive blending?
Centralized blending removes farm-level dosing variance and generates automated mixing logs essential for passing rigorous regulatory compliance audits.
How do changing formulation profiles impact market value?
The shift toward premium, highly engineered soluble salts drives revenue expansion even as overall bulk tonnage faces strict regulatory suppression.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.