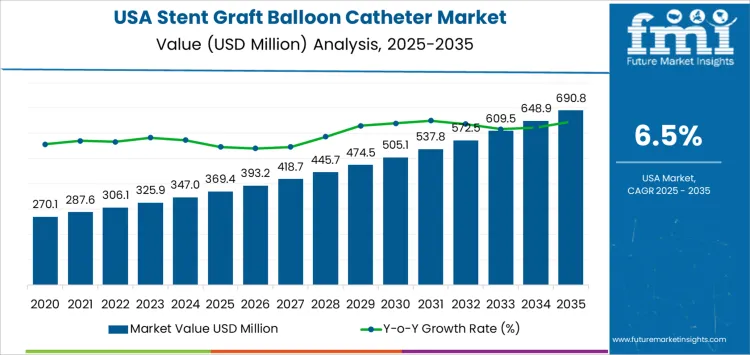
Demand for stent graft balloon catheters in the USA is expected to grow from USD 369.4 million in 2025 to USD 690.8 million by 2035, reflecting a compound annual growth rate (CAGR) of 6.5%. This growth is driven by the increasing prevalence of cardiovascular diseases, particularly in the aging population, and the rising adoption of minimally invasive procedures.
Stent graft balloon catheters are vital in treating complex vascular conditions, such as aneurysms and peripheral artery disease, by providing a dual function of expanding and sealing arteries, reducing the need for open surgeries. As healthcare providers increasingly prefer minimally invasive options, the demand for these devices is expected to rise.
The market is further supported by advancements in catheter technology, including improved balloon designs, biocompatible materials, and more precise delivery systems. The increasing focus on reducing recovery times and improving patient outcomes also drives the demand for stent graft balloon catheters. With continued research and development in the field of vascular therapies and the growing adoption of advanced catheter-based interventions, the market for these devices is expected to expand steadily over the next decade.
The compound absolute growth analysis for the stent graft balloon catheter market in the USA reflects a steady and predictable increase in market value over the forecast period. Starting at USD 369.4 million in 2025, the market grows to USD 393.2 million in 2026, and USD 418.7 million in 2027. This early growth reflects the gradual adoption of stent graft balloon catheters as part of the broader shift towards minimally invasive vascular interventions.
As the market continues, the absolute growth accelerates slightly, with the market reaching USD 445.7 million in 2028 and USD 474.5 million in 2029. This acceleration is driven by growing demand for vascular treatments, particularly in older populations with a higher incidence of cardiovascular diseases. By 2030, the market reaches USD 505.1 million, and further growth continues with the market reaching USD 537.8 million in 2031, USD 572.5 million in 2032, and USD 609.5 million in 2033.
In the final years of the forecast period, the market sees continued growth, reaching USD 648.9 million in 2034 and USD 690.8 million by 2035. The compound absolute growth analysis highlights a strong upward trajectory in market value, driven by increasing adoption rates, technological advancements, and a rising number of patients requiring vascular treatments. This steady expansion reflects the growing role of stent graft balloon catheters in modern vascular care.
| Metric | Value |
|---|---|
| Industry Sales Value (2025) | USD 369.4 million |
| Industry Forecast Value (2035) | USD 690.8 million |
| Industry Forecast CAGR (2025-2035) | 6.5% |
Demand for stent-graft balloon catheters in the USA is rising as more patients are treated for aortic aneurysms and other vascular conditions using minimally invasive endovascular procedures. Many cases that previously required open surgery are now addressed via endovascular repair - where a stent graft is delivered through a catheter and expanded inside the blood vessel. This method reduces hospital stay, lowers risk of complications, and speeds up recovery compared with open surgery.
At the same time, advances in stent-graft and catheter technology are fueling further adoption. Modern balloon-catheter delivery systems enable precise placement and reliable expansion of stent grafts even in complex vascular anatomies. These tools make it feasible for more patients - including older individuals and high-risk surgical candidates - to receive treatment. As awareness grows about benefits of endovascular repair and as screening for aneurysms and vascular disease improves, demand for stent-graft balloon catheters in hospitals and vascular centers across the USA is expected to continue rising.
The stent graft balloon catheter market in the USA is segmented by product and end user. Non-compliant balloon stent catheters dominate the product segment, holding 85% of the market share. In terms of end users, hospitals account for 60% of the market. The demand for stent graft balloon catheters is driven by the increasing number of patients requiring vascular interventions, including those with peripheral artery disease and coronary artery disease.
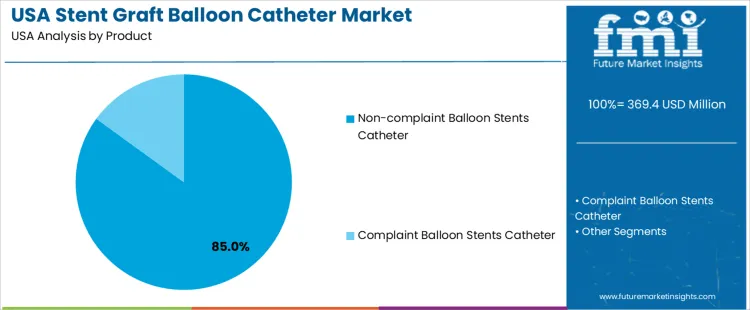
Non-compliant balloon stent catheters lead the market, holding 85% of the total demand in the USA. Non-compliant balloons are designed to maintain their shape and diameter at high inflation pressures, making them ideal for precise stent deployment in the treatment of vascular blockages. These catheters are preferred in cases where greater radial force is required to open narrowed or blocked arteries. The ability to withstand high pressures without changing size is essential for ensuring optimal stent placement, which contributes to the dominance of non-compliant balloon catheters in the market. Compliant balloon stent catheters, which expand more easily and are typically used in less rigid vascular regions, make up a smaller portion of the market but are still important in certain clinical scenarios.
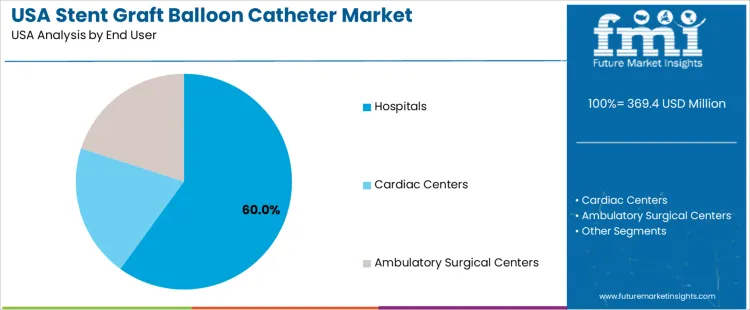
Hospitals are the primary end users of stent graft balloon catheters in the USA, accounting for 60% of the market share. Hospitals, with their advanced medical equipment and specialized staff, are the primary settings for complex vascular procedures such as angioplasty, stent placement, and graft surgeries. These procedures often require non-compliant balloon catheters for accurate and effective stent deployment in the arteries. Cardiac centers and ambulatory surgical centers also contribute to the demand, particularly for less invasive, outpatient procedures, but hospitals remain the dominant end user due to the complexity of cases and the need for comprehensive medical care. The growing adoption of minimally invasive surgical techniques in hospitals continues to drive the demand for stent graft balloon catheters.
The demand for stent graft balloon catheters in the USA is increasing as they play a crucial role in the treatment of vascular diseases, particularly in procedures like endovascular aneurysm repair (EVAR) and peripheral artery disease (PAD) interventions. These devices, which combine balloon catheter technology with stent grafts, are essential for repairing blood vessels and arteries that are narrowed, blocked, or weakened. As the prevalence of cardiovascular diseases and vascular conditions continues to rise, the need for stent graft balloon catheters is expected to grow, particularly in aging populations and patients with chronic conditions requiring vascular intervention.
Several factors are driving the demand for stent graft balloon catheters in the USA. First, the rising incidence of cardiovascular diseases, including peripheral artery disease, aortic aneurysms, and other vascular conditions, is increasing the need for interventions that use stent graft balloon catheters. Second, the growing preference for minimally invasive procedures, which offer reduced recovery times and lower complication rates compared to traditional open surgery, is boosting the adoption of these devices.
Third, advancements in medical technology, including improved stent graft materials and balloon catheter designs, are making these devices more effective and reliable, encouraging healthcare providers to use them in various vascular treatments. Finally, the increasing aging population and the rising prevalence of risk factors such as hypertension, diabetes, and smoking are contributing to the higher demand for vascular interventions, which drives the need for stent graft balloon catheters.
Despite the growing demand, several challenges may limit the widespread adoption of stent graft balloon catheters in the USA. One major restraint is the high cost of these devices, which can be a barrier for some healthcare providers, particularly smaller hospitals or those with limited budgets. Additionally, the complexity of stent graft balloon catheter procedures requires skilled specialists and advanced medical infrastructure, which may not be readily available in all healthcare settings.
Another challenge is the risk of complications, such as restenosis or device migration, which may reduce the overall effectiveness of the procedure and require additional interventions. Finally, the limited reimbursement for some endovascular procedures, especially in certain insurance plans, may restrict patient access to stent graft balloon catheter treatments.
Several key trends are shaping the demand for stent graft balloon catheters in the USA. One significant trend is the increasing focus on minimally invasive vascular procedures, which are preferred for their ability to reduce hospital stays and recovery times. Another trend is the development of advanced materials and designs for stent grafts and balloon catheters, which enhance performance and reduce the risk of complications.
Additionally, the growing number of clinical trials and research in endovascular therapies is leading to new innovations in stent graft balloon catheter technology, improving treatment options for patients. The rising prevalence of cardiovascular risk factors, such as obesity, diabetes, and smoking, is also driving the demand for these devices as healthcare providers seek effective solutions for treating vascular diseases. Lastly, the increasing emphasis on patient outcomes and cost-effectiveness in healthcare is encouraging the use of advanced, reliable, and minimally invasive interventions like stent graft balloon catheters.
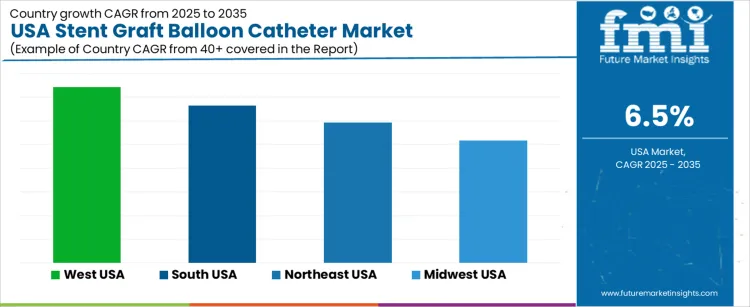
The Stent Graft Balloon Catheter market in the USA is projected to grow steadily, with the West USA leading the way at a projected CAGR of 7.4%. South USA follows closely with a growth rate of 6.7%, while the Northeast USA is expected to grow at 5.9%. The Midwest USA shows moderate growth with a CAGR of 5.2%. Stent graft balloon catheters, which are used in endovascular procedures to treat vascular diseases such as aneurysms and arterial blockages, are gaining traction as healthcare providers increasingly adopt minimally invasive procedures. As the demand for advanced medical devices rises, particularly in cardiovascular treatments, the market for stent graft balloon catheters is expected to expand across the United States.
| Region | CAGR (2025-2035) |
|---|---|
| West USA | 7.4% |
| South USA | 6.7% |
| Northeast USA | 5.9% |
| Midwest USA | 5.2% |
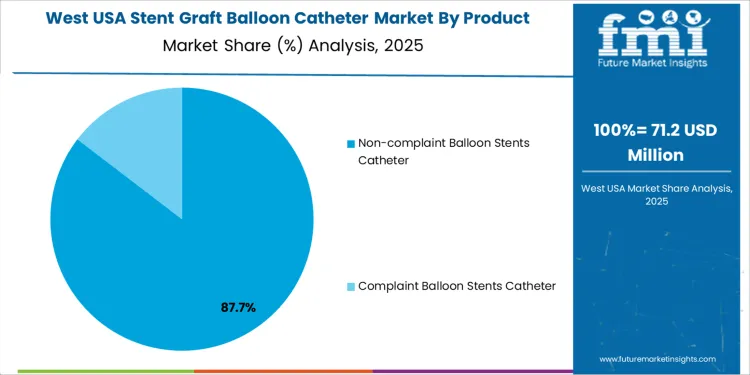
The West USA is projected to experience the highest growth in the Stent Graft Balloon Catheter market, with a projected CAGR of 7.4%. The region’s strong healthcare infrastructure, combined with a high prevalence of cardiovascular diseases, is a significant driver of this growth. States like California, Washington, and Oregon have a high rate of cardiovascular procedures, with healthcare providers increasingly turning to minimally invasive endovascular treatments, including stent graft balloon catheters, for vascular repair and treatment.
The West’s emphasis on technological innovation, coupled with advancements in catheter design and materials, is further fueling the demand for these devices. Additionally, the region’s large population of aging individuals, who are at greater risk for vascular diseases, ensures a continued demand for effective and less invasive treatment options like stent graft balloon catheters.
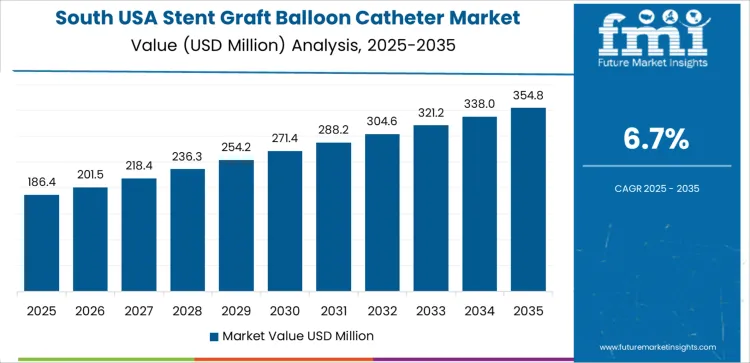
In South USA, the demand for stent graft balloon catheters is projected to grow at a CAGR of 6.7%. The region’s expanding healthcare sector and increasing incidence of vascular diseases, particularly among older populations, are contributing to the adoption of advanced medical devices like stent graft balloon catheters. States such as Texas, Florida, and Georgia are seeing rising demand for endovascular treatments as the healthcare infrastructure in these areas improves.
With a growing focus on reducing recovery times and minimizing surgical risks, healthcare providers in the South are increasingly using stent graft balloon catheters in procedures that require vessel repair or support. As the region continues to emphasize accessibility to advanced healthcare and effective treatment solutions, the market for stent graft balloon catheters is expected to expand.
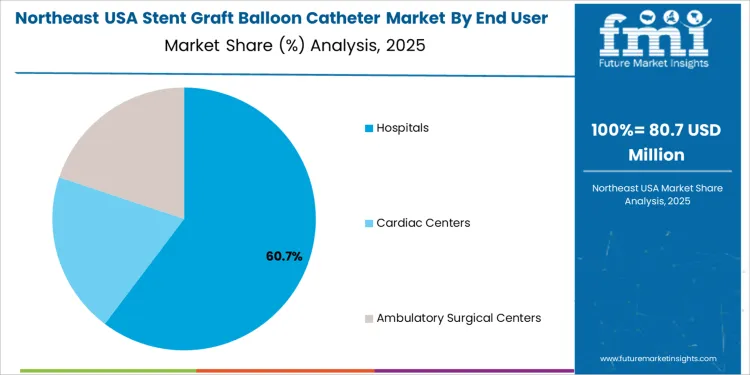
In Northeast USA, the Stent Graft Balloon Catheter market is projected to grow at a CAGR of 5.9%. The region’s well-established healthcare systems and high concentration of advanced medical centers contribute to the growing adoption of stent graft balloon catheters. Major cities like New York, Boston, and Philadelphia are home to leading hospitals and research institutions that specialize in cardiovascular treatments. The Northeast’s aging population, combined with an increased focus on minimally invasive procedures, is driving the demand for stent graft balloon catheters.
These devices are being used in a variety of vascular interventions to treat conditions like aneurysms, arterial blockages, and other cardiovascular diseases. As healthcare providers in the region continue to prioritize patient safety and quicker recovery times, stent graft balloon catheters are expected to become more widely used in clinical practice.
In Midwest USA, the Stent Graft Balloon Catheter market is expected to grow at a more moderate rate of 5.2%. The region’s healthcare systems are increasingly adopting advanced technologies for the treatment of vascular diseases, and stent graft balloon catheters are becoming an integral part of endovascular procedures. States like Illinois, Michigan, and Ohio are seeing increasing adoption of minimally invasive treatments for cardiovascular conditions.
While the Midwest’s growth rate is slower compared to other regions, the rising incidence of cardiovascular diseases, particularly in the aging population, is driving demand for these devices. Healthcare providers are increasingly seeking efficient and less invasive treatment options, and as stent graft balloon catheters offer improved recovery times and reduced complications compared to traditional surgical methods, the market is expected to continue growing steadily in the Midwest.
Demand for stent graft balloon catheters in the USA remains substantial as vascular and endovascular procedures grow in number. The increasing prevalence of cardiovascular diseases, aortic aneurysms, peripheral artery disease and other vascular disorders drives demand for endovascular repair devices. Minimally invasive interventions using stent grafts and balloon catheters offer patients shorter recovery times, reduced surgical risk, and better long-term outcomes compared with open-surgery alternatives. As the population ages and awareness of less-invasive vascular therapies expands, demand for stent graft balloon catheter systems continues to grow.
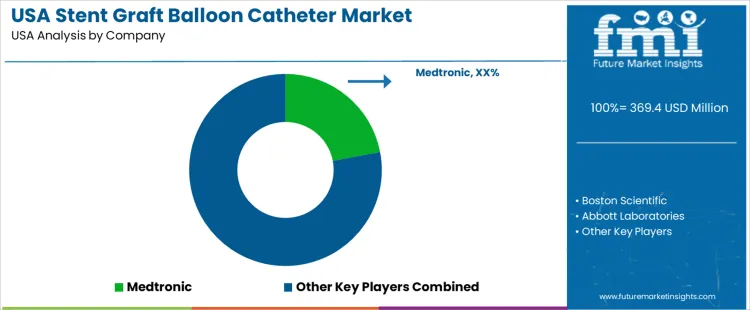
On the supply side, a handful of major medical device companies dominate the market. According to your list, Medtronic holds a leading share. Other key suppliers include Boston Scientific, Abbott Laboratories, Cordis, and Atrium Medical. These firms provide a range of stent graft balloon-catheter systems - including devices for aortic repair, peripheral vascular interventions, and peripheral-artery disease management. Competition among these suppliers centers on factors such as device safety, delivery accuracy, stent graft compatibility, catheter flexibility and sizing options, regulatory compliance, and hospital/clinician support. Suppliers that offer a broad product portfolio, robust clinical data, reliable supply, and strong after-sales support are best positioned to capture growth as endovascular procedures continue to rise in the USA.
| Items | Details |
|---|---|
| Quantitative Units | USD Million |
| Regions Covered | USA |
| Product | Non-Compliant Balloon Stents Catheter, Compliant Balloon Stents Catheter |
| End User | Hospitals; Cardiac Centers; Ambulatory Surgical Centers (ASCs) |
| Key Companies Profiled | Medtronic; Boston Scientific; Abbott Laboratories; Cordis; Atrium Medical |
| Additional Attributes | Dollar sales by product type and end-user show strong demand for both compliant and non-compliant balloon stent graft catheters - driven by increasing prevalence of vascular diseases, aortic aneurysms and growing adoption of minimally invasive endovascular procedures. Hospitals remain the primary end users. |
How big is the demand for stent graft balloon catheter in USA in 2025?
The demand for stent graft balloon catheter in USA is estimated to be valued at USD 369.4 million in 2025.
What will be the size of stent graft balloon catheter in USA in 2035?
The market size for the stent graft balloon catheter in USA is projected to reach USD 690.8 million by 2035.
How much will be the demand for stent graft balloon catheter in USA growth between 2025 and 2035?
The demand for stent graft balloon catheter in USA is expected to grow at a 6.5% CAGR between 2025 and 2035.
What are the key product types in the stent graft balloon catheter in USA?
The key product types in stent graft balloon catheter in USA are non-complaint balloon stents catheter and complaint balloon stents catheter.
Which end user segment is expected to contribute significant share in the stent graft balloon catheter in USA in 2025?
In terms of end user, hospitals segment is expected to command 60.0% share in the stent graft balloon catheter in USA in 2025.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.