Vaporized Hydrogen Peroxide Sterilization System Market
The Vaporized Hydrogen Peroxide Sterilization System Market is segmented by System Type (Fixed Type, Mobile Type, and Others), Application (Pharmaceutical and Biotech, Food and Beverage, and Others) and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Vaporized Hydrogen Peroxide Sterilization System Market Forecast and Outlook By FMI
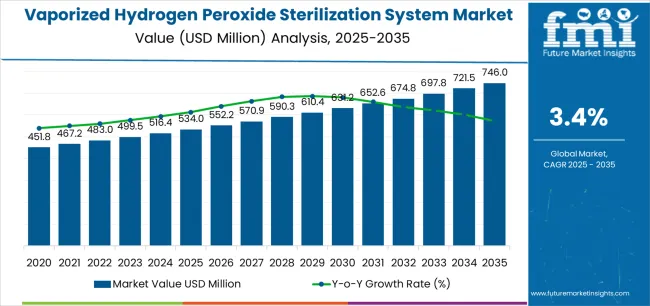
In 2025, vaporized hydrogen peroxide sterilization system market was valued at USD 0.53 billion. Based on Future Market Insights’ analysis, demand for vaporized hydrogen peroxide sterilization systems is estimated to grow to USD 0.55 billion in 2026 and USD 0.77 billion by 2036. FMI projects a CAGR of 3.4% during the forecast period.
Absolute dollar growth of USD 0.22 billion over decade signals steady infrastructure-linked expansion rather than a fast-cycle replacement boom. Growth stays anchored in aseptic manufacturing upgrades, tighter sterile manufacturing expectations, and infection control programs, while capex intensity, qualification lead times, and operator capability requirements keep adoption disciplined across smaller sites.
As Glenn Pennington, Director of Central Services at University Hospitals Coventry and Warwickshire NHS Trust, noted regarding room bio-decontamination outcomes, ‘we found that VHP gives a consistent, repeatable process with clear audit trails that supports infection prevention objectives’ [2].
Summary of Vaporized Hydrogen Peroxide Sterilization System Market
- Market Definition
- Market covers validated low-temperature bio-decontamination systems used for cleanroom, isolator, and enclosed space sterilization workflows where documentation and repeatability are procurement-grade requirements.
- Demand Drivers in the Market
- Sterile manufacturing expectations under Annex 1 style contamination control frameworks increase demand for repeatable decontamination cycles [4].
- PIC/S alignment accelerates harmonised sterile product expectations across member and partner regulators, shaping validation spend [8].
- Infection prevention programs in hospitals sustain demand for room and equipment bio-decontamination with audit-ready documentation [2].
- Key Segments Analyzed in the FMI Report
- System Type: Fixed Type leads due to facility integration and throughput-driven repeatability.
- Application: Pharmaceutical and Biotech dominates through GMP-driven validation and routine cleanroom requalification.
- Geography: Asia Pacific drives net additions, while Europe is shaped by sterile guidance driven upgrades [4].
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant at Future Market Insights, opines, ‘CXOs will find that validation capability and service depth are now purchase criteria, since sterile compliance is won through repeatable cycles, documentation discipline, and fast requalification support rather than equipment spec sheets alone.’
- Strategic Implications/Executive Takeaways
- Prioritise service networks and qualification toolkits as revenue engines, not optional add-ons.
- Design offerings around Annex 1 style contamination control expectations and audit trail readiness [4].
- Target multi-site frameworks with standardised cycle libraries to reduce customer revalidation cost.
- Methodology
- Validated through primary interviews with QA and sterile engineering buyers.
- Triangulated using regulator and standards body publications on sterile expectations [8].
- Benchmarked against OEM disclosures and documented deployments where available.
China (4.6% CAGR) and India (4.3% CAGR) lead due to sterile capacity additions and export-aligned GMP upgrades, while Germany (3.9%) advances through Annex 1 driven contamination control modernization. Brazil (3.6%) grows on regulatory cooperation and PIC/S alignment momentum. Mature markets such as USA (3.2%), UK (2.9%), and Japan (2.6%) generate replacement and compliance-driven demand, constrained by long validation cycles, procurement scrutiny, and competing low-temperature modalities.
Market Definition
Vaporized hydrogen peroxide sterilization systems are low-temperature bio-decontamination platforms that generate and distribute hydrogen peroxide vapour across controlled spaces such as cleanrooms, isolators, pass-through chambers, and enclosed processing zones. Market covers system supply, installation, and validation-oriented deployment that enables microbial reduction in pharma, biotech, healthcare, and sterile processing environments where heat-sensitive equipment, electronics, and polymer components limit steam usage. Core purchase decision is tied to contamination control assurance, cycle documentation, and repeatability required for GMP, cleanroom requalification, and infection prevention programs across manufacturing and clinical facilities.
Market Inclusions
Report includes global and regional market sizing in value terms with a 10-year forecast (2026 to 2036). Coverage spans segmentation by system type, application, and region, supported by country-level demand outlook, adoption drivers, and regulatory or standards references shaping validation practice. Analysis incorporates buyer requirements across lifecycle service, qualification documentation, and integration constraints, alongside competitive benchmarking of equipment suppliers and their recent corporate actions relevant to sterilization and bio-decontamination.
Market Exclusions
Scope excludes consumable hydrogen peroxide chemical supply as a standalone market unless bundled as part of validated system operation contracts. It also omits unrelated sterilization modalities such as ethylene oxide, radiation, or steam autoclaves unless used in facility-level comparison context. Downstream sterile finished goods, hospital outcomes analytics products, and general infection prevention services not tied to VHP system deployment remain outside coverage, with focus held on equipment-centric bio-decontamination systems and immediate validation workflows.
Vaporized Hydrogen Peroxide Sterilization System Market Research Methodology
Primary Research: Interviews were conducted with sterile facility engineers, validation leads, QA heads, hospital sterile services managers, OEM service teams, and cleanroom consultants across major regions.
Desk Research: Public sources included GMP guidance, regulator updates, PIC/S and Annex 1 related publications, and OEM disclosures on product platforms and service capabilities.
Market-Sizing and Forecasting: A hybrid model was built using installed-base logic, facility expansion indicators, and replacement cycles, triangulated with application mix and region-level compliance intensity.
Data Validation and Update Cycle: Outputs undergo variance checks across regulatory timelines, OEM disclosure screening, and structured review to remove inconsistencies between forecast totals, country CAGRs, and segment shares.
Segmental Analysis
Vaporized Hydrogen Peroxide Sterilization System Market Analysis by System Type
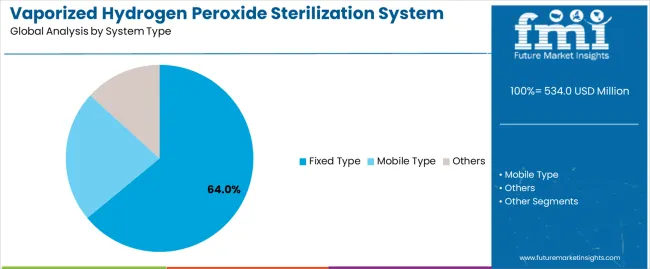
Based on FMI’s vaporized hydrogen peroxide sterilization system market report, consumption of Fixed Type systems is estimated to hold 64% share in 2026. Dominance comes from permanent integration with facility HVAC and controlled distribution paths that support repeatable cycles, predictable turnaround, and documentation continuity across scheduled decontamination routines.
- Facility Integration: Getinge expanded decontamination portfolio positioning around faster turnaround and validated bio-decontamination workflows, reflecting buyer preference for integrated, repeatable solutions in high-grade environments [1].
- Cycle Documentation: NHS sterile services commentary highlights procurement preference for audit-ready trails and repeatability, reinforcing why fixed installations win in validation-driven programs [2].
- Throughput Discipline: Annex 1 style sterile expectations increase pressure to run decontamination as a scheduled, standardised operation, which structurally favours fixed systems built for routine cadence [4].
Vaporized Hydrogen Peroxide Sterilization System Market Analysis by Application
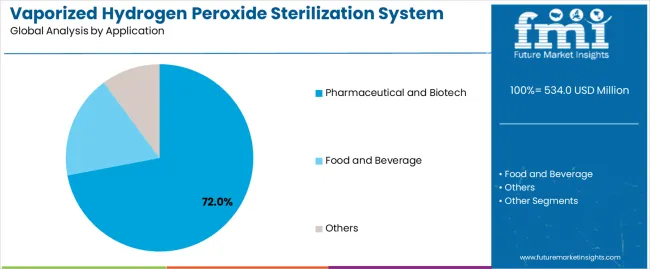
Based on FMI’s vaporized hydrogen peroxide sterilization system market report, consumption of Pharmaceutical and Biotech applications is estimated to hold 72% share in 2026. Leadership stems from GMP-driven sterility assurance needs, frequent cleanroom requalification, isolator use in aseptic fill-finish, and a requirement for full cycle records that support inspections and batch release readiness.
- Aseptic Guidance Pull: FDA draft guidance reinforces contamination control expectations in aseptic processing, strengthening demand for validated decontamination workflows in sterile drug manufacture [3].
- Sterile Annex Alignment: PIC/S Annex 1 implementation timeline supports broader adoption of structured sterile manufacture expectations, reinforcing pharma-led demand [8].
- Export Quality Goals: China’s sterile GMP drafting and alignment work indicates compliance-driven facility upgrades that intensify pharma demand for bio-decontamination and validation tooling [6].
Vaporized Hydrogen Peroxide Sterilization System Market Drivers, Restraints, and Opportunities
Future Market Insights analysis observes a compliance-shaped market where demand tracks cleanroom build-outs, sterile line expansions, and periodic requalification cycles rather than consumer-like replacement behaviour. Market size reflects essential spend for contamination control in GMP and healthcare environments, with adoption rising when audit expectations harden or when new sterile capacity comes online under export-driven quality goals.
Core tension sits between high assurance outcomes and operational burden. VHP systems reduce thermal risk and can deliver strong biological efficacy, yet sites must invest in facility readiness, safety controls, and documentation capability, while some applications face compatibility limits and competing low-temperature modalities.
- Annex 1 Execution: EU-aligned sterile guidance increases demand for documented contamination control strategies and repeatable decontamination validation, favouring suppliers that provide qualification templates and cycle development support [4].
- PIC/S Convergence: PIC/S Annex 1 alignment expands common expectations for sterile manufacture, pushing multinational firms toward standardised decontamination approaches across sites and countries [8].
- Asia Capacity Build: China and India expand sterile and biologics capacity, creating demand for integrated cleanroom and isolator bio-decontamination systems that can be validated on accelerated project schedules [6].
Regional Analysis
Coverage spans Asia Pacific, Europe, North America, Latin America, and Middle East & Africa across 40+ countries. Regional assessment includes country-wise demand outlook, adoption constraints, and policy or standards effects, while full report offers market attractiveness analysis based on compliance intensity and installed-base expansion.
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 4.6% |
| India | 4.3% |
| Germany | 3.9% |
| Brazil | 3.6% |
| United States | 3.2% |
| United Kingdom | 2.9% |
| Japan | 2.6% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia Pacific Vaporized Hydrogen Peroxide Sterilization System Market Analysis
Asia Pacific acts as capacity build epicentre, where sterile injectables, biologics, and biosimilars expansions drive demand for cleanroom and isolator bio-decontamination. Key suppliers such as Shinva and global OEMs expand coverage through regional service partners, with buyer selection often anchored in validation support availability and project commissioning speed.
China: Demand for vaporized hydrogen peroxide sterilization systems in China is projected to rise at 4.6% CAGR through 2036. Momentum links to sterile capacity builds and regulator alignment work, where sterile GMP drafting signals tighter expectation setting for controlled manufacturing environments [6]. PIC/S Annex 1 timing also shapes multinational site harmonisation strategies that influence Chinese export-facing plants and engineering standards selection [8].
India: Demand for vaporized hydrogen peroxide sterilization systems in India is projected to rise at 4.3% CAGR through 2036. Adoption is supported by GMP upgrade pressure and export-facing documentation requirements, with revised Schedule M references signalling stepped compliance expectations that lift validation and contamination control investment [11]. Alignment with global sterile manufacture expectations also increases focus on structured decontamination evidence and audit readiness [8].
FMI's analysis of vaporized hydrogen peroxide sterilization system market in Asia Pacific consists of country-wise assessment that includes China, India, Japan, South Korea, Australia & New Zealand, ASEAN, and Rest of Asia Pacific. Readers can find compliance-linked demand patterns, application shifts, and supplier positioning tied to commissioning speed and validation readiness.
Europe Vaporized Hydrogen Peroxide Sterilization System Market Analysis
Europe operates as sterile compliance laboratory, where guidance-driven upgrades shape spending even in mature installed bases. Suppliers such as Bioquell and Getinge compete through validation libraries, service responsiveness, and integration capability for isolators, pass-throughs, and cleanrooms supporting aseptic operations.
Germany: Demand for vaporized hydrogen peroxide sterilization systems in Germany is projected to rise at 3.9% CAGR through 2036. EU Annex 1 expectations influence contamination control strategy design and requalification cadence, pushing investments into documented decontamination approaches that can withstand inspection scrutiny [4]. PIC/S Annex 1 alignment further reinforces harmonised sterile expectations that standardise validation disciplines across multi-site operators [8].
United Kingdom: Demand for vaporized hydrogen peroxide sterilization systems in United Kingdom is projected to rise at 2.9% CAGR through 2036. MHRA GMP guidance and inspection expectations increase focus on documented contamination control and validated decontamination programs, strengthening demand for systems with audit-friendly records and service support [5]. NHS infection prevention programs reinforce room bio-decontamination use cases where repeatability and traceability remain valued procurement attributes [2].
FMI's analysis of vaporized hydrogen peroxide sterilization system market in Europe consists of country-wise assessment that includes Germany, United Kingdom, France, Italy, Spain, Netherlands, Nordic, BENELUX, and Rest of Europe. Readers can find regulatory alignment effects, validation practice trends, and application-level adoption tied to aseptic manufacturing footprints.
North America Vaporized Hydrogen Peroxide Sterilization System Market Analysis
North America is a validation-first market where aseptic guidance, quality systems maturity, and established sterile manufacturing footprints support steady demand. Suppliers such as STERIS and ASP compete on reliability, documentation tooling, and service coverage that reduces downtime and accelerates requalification.
United States: Demand for vaporized hydrogen peroxide sterilization systems in United States is projected to rise at 3.2% CAGR through 2036. FDA draft aseptic processing guidance reinforces contamination control expectations that lift demand for validated decontamination, documentation, and cycle control for sterile manufacture and cleanroom operations [3]. Hospital-grade adoption remains supported by infection prevention outcomes and audit trail discipline emphasised by sterile services leadership in NHS-style deployments, shaping buyer expectations for documentation even in healthcare settings [2].
FMI's analysis of vaporized hydrogen peroxide sterilization system market in North America consists of country-wise assessment that includes United States, Canada, and Mexico. Readers can find regulatory driven purchasing patterns, application splits, and competitive benchmarking tied to service capability and documentation readiness.
Latin America Vaporized Hydrogen Peroxide Sterilization System Market Analysis
Latin America is modernisation-driven, where regulator cooperation and sterile manufacture capability expansion raise demand for validated bio-decontamination. Regional procurement often depends on distributor capability and validation service availability, making OEM partner networks a decisive factor.
Brazil: Demand for vaporized hydrogen peroxide sterilization systems in Brazil is projected to rise at 3.6% CAGR through 2036. ANVISA engagement on PIC/S-focused sterile manufacturing topics highlights ongoing attention to sterile product guidance and cross-border regulatory cooperation, supporting upgraded contamination control expectations [9]. PIC/S Annex 1 implementation timing strengthens harmonised sterile manufacture norms that influence multinational and export-aligned facilities operating in Brazil [8].
FMI's analysis of vaporized hydrogen peroxide sterilization system market in Latin America consists of country-wise assessment that includes Brazil, Argentina, and Rest of Latin America. Readers can find regulatory cooperation signals, adoption constraints, and distributor-led deployment dynamics across pharma and healthcare applications.
Competitive Aligners for Market Players
Market structure is moderately concentrated, with a limited set of global suppliers and specialist bio-decontamination players shaping procurement shortlists for GMP-grade environments. Practical competition concentrates in vendors that can provide validated cycles, documentation packages, and global service reach, since buyer risk sits in inspection exposure and operational downtime rather than initial purchase price.
Structural advantage comes from lifecycle capability. Suppliers with strong commissioning teams, validation toolkits, and field service networks reduce customer burden for qualification, periodic revalidation, and troubleshooting. Portfolio breadth across isolators, chambers, and room systems supports cross-sell into multi-site frameworks where standardised cycle libraries reduce long-term compliance cost.
Buyer behaviour remains risk-managed. Large pharma manufacturers and hospital groups standardise approved vendor lists, require documented performance evidence, and negotiate service SLAs that protect uptime. Multi-sourcing is used to manage dependency, yet switching cost remains high due to revalidation effort, giving incumbents leverage when documentation quality and service responsiveness remain strong.
Recent Developments
- Getinge introduced a next-generation bio-decontamination solution and positioned faster, validated turnaround as a core buyer outcome (March 2024) [1].
- ANVISA hosted PIC/S-focused sterile manufacturing discussions, signalling regulatory attention to sterile guidance alignment that supports validation-led investment (November 2024) [9].
Key Players in Vaporized Hydrogen Peroxide Sterilization System Market
- Advanced Sterilization Products (ASP)
- Bioquell
- Fedegari Group
- Getinge
- Steelco S.p.A.
- STERIS Life Sciences
- Shinva
- Van der Stahl Scientific
- STERIS AST
- Stryker
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 0.55 billion (2026) to USD 0.77 billion (2036), at a CAGR of 3.4% |
| Market Definition | Market covers VHP bio-decontamination systems used for low-temperature sterilization of cleanrooms, isolators, chambers, and enclosed controlled environments requiring validation and documented cycle control. |
| System Type | Fixed Type, Mobile Type, Others |
| Application | Pharmaceutical and Biotech, Food and Beverage, Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | China, India, Germany, Brazil, United States, United Kingdom, Japan, and 40+ countries |
| Key Companies Profiled | Advanced Sterilization Products (ASP), Bioquell, Fedegari Group, Getinge, Steelco S.p.A., STERIS Life Sciences, Shinva, Van der Stahl Scientific, STERIS AST, Stryker |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid modelling validated through primary interviews and triangulated with policy, standards, and OEM disclosures |
Key Segments
System Type:
- Fixed Type
- Mobile Type
- Others
Application:
- Pharmaceutical and Biotech
- Food and Beverage
- Others
Region:
- Asia Pacific
- China
- India
- Japan
- South Korea
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Netherlands
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- [1] Getinge. (2024, March). Getinge launches a new bio-decontamination solution for faster validated turnaround.
- [2] STERIS. (2024). Interview feature on NHS deployment of VHP room bio-decontamination and audit trail expectations.
- [3] USA Food and Drug Administration. (2023, April). Sterile drug products produced by aseptic processing: Current Good Manufacturing Practice (Draft guidance for industry).
- [4] European Commission. (2022). EudraLex Volume 4: EU guidelines for Good Manufacturing Practice, Annex 1: Manufacture of sterile medicinal products.
- [5] Medicines and Healthcare products Regulatory Agency. (2024, May). GMP guidance and inspection expectations.
- [6] GMP Publishing. (2025). China NMPA draft guideline for manufacture of sterile drugs: Publication notice.
- [8] Pharmaceutical Inspection Co-operation Scheme. (2023). Publication of revised PIC/S Annex 1 and entry into force timeline.
- [9] ANVISA. (2024, November). PIC/S seminar in Brazil focused on sterile manufacture Annex 1 discussions.
- [11] Indian Pharmaceutical Alliance. (2024). Note on revised Schedule M notification and implementation timeline (G.S.R. 922(E)).
This Report Addresses
- Market intelligence to support sterile manufacturing and healthcare contamination control planning
- Market size estimation and 10-year revenue forecasts from 2026 to 2036 aligned to a 3.4% CAGR
- Growth opportunity mapping across system type and application segments with buyer requirement context
- Regional and country outlook for major adoption markets including China, India, Germany, Brazil, USA, UK, and Japan
- Competition strategy assessment focused on validation tooling, service coverage, and lifecycle support models
- Compliance tracking linked to aseptic guidance and sterile manufacture expectations affecting procurement [3]
- Policy and standards influence assessment including Annex 1 and PIC/S alignment effects [8]
- Report delivery formats for executive use including PDF, Excel, PPT, and dashboard-ready structures
Frequently Asked Questions
How large is demand for vaporized hydrogen peroxide sterilization systems in 2026?
Demand is estimated to be valued at USD 0.55 billion in 2026.
What will market size be by 2036?
Market size is projected to reach USD 0.77 billion by 2036.
What is expected demand growth between 2026 and 2036?
Demand is expected to grow at a CAGR of 3.4% between 2026 and 2036.
Which system type is poised to lead global sales in 2026?
Fixed Type is expected to lead due to permanent integration and repeatable validation cycles aligned to sterile facility routines.
How significant is Pharmaceutical and Biotech application in 2026?
Pharmaceutical and Biotech remains dominant at an estimated 72% share due to GMP-driven documentation and requalification needs.
Which countries lead growth outlook through 2036?
China and India lead growth outlook with projected CAGRs of 4.6% and 4.3% through 2036.
What keeps growth steady rather than exponential?
Capital intensity, validation lead times, and operator capability requirements keep adoption paced and procurement disciplined.
Why do buyers prioritise documentation readiness?
Audit trail completeness reduces inspection risk and shortens requalification timelines, improving facility uptime.
What is main procurement risk buyers manage?
Risk centres on qualification failure or cycle variability that can disrupt sterile operations and trigger revalidation cost.
How does Annex 1 influence adoption decisions?
Annex 1 aligned contamination control expectations reinforce structured decontamination strategies and documentation discipline [4].
How does PIC/S alignment influence multinational deployments?
PIC/S Annex 1 alignment supports harmonised sterile expectations, enabling standardised cycle libraries across sites [8].
What role does service coverage play in vendor selection?
Service responsiveness reduces downtime and supports periodic revalidation, creating lifecycle advantage for suppliers.
What is primary value proposition versus steam sterilization?
Low-temperature processing enables bio-decontamination where heat-sensitive assets and electronics limit steam use.
What limits adoption in smaller facilities?
Upfront capex plus safety controls, training, and validation workloads can exceed smaller site capability.
Why do fixed systems retain advantage in high-throughput pharma sites?
Permanent integration supports repeatability and predictable turnaround across scheduled decontamination cadence.
What drives mobile system demand?
Mobile systems fit multi-room facilities seeking flexible deployment without multiple fixed installations.
What is typical buyer segmentation in healthcare?
Hospitals prioritise environmental decontamination outcomes, repeatability, and traceability for infection prevention programs [2].
What drives demand in China?
Sterile GMP alignment and capacity additions raise need for validated contamination control in export-facing facilities [6].
What drives demand in India?
GMP upgrades and export documentation expectations raise investment in validation-grade decontamination programs [11].
What is central competitive variable in this market?
Validation capability plus lifecycle support depth is primary competitive variable, since switching cost is driven by revalidation effort.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By System Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By System Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By System Type , 2026 to 2036
- Fixed
- Mobile
- Type
- Fixed
- Y to o to Y Growth Trend Analysis By System Type , 2021 to 2025
- Absolute $ Opportunity Analysis By System Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Pharmaceutical and Biotech
- Food and Beverage
- Others
- Pharmaceutical and Biotech
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By System Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By System Type
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By System Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By System Type
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By System Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By System Type
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By System Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By System Type
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By System Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By System Type
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By System Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By System Type
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By System Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By System Type
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By System Type
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By System Type
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By System Type
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By System Type
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By System Type
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By System Type
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By System Type
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By System Type
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By System Type
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By System Type
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By System Type
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By System Type
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By System Type
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By System Type
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By System Type
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By System Type
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By System Type
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By System Type
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By System Type
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By System Type
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By System Type
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By System Type
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By System Type
- By Application
- Competition Analysis
- Competition Deep Dive
- Advanced Sterilization Products (ASP)
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Bioquell
- Fedegari Group
- Getinge
- Steelco S.p.A.
- STERIS Life Sciences
- Shinva
- Advanced Sterilization Products (ASP)
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by System Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by System Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by System Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by System Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by System Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by System Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by System Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by System Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by System Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by System Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by System Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by System Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by System Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by System Type
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by System Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by System Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by System Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by System Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by System Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by System Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by System Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by System Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by System Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by System Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by System Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by System Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by System Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by System Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by System Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by System Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by System Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by System Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

