Vitamin and Mineral Supplement Market
Vitamin and Mineral Supplement Market Insights - Trends & Forecast 2026 to 2036
Vitamin and Mineral Supplement Market Forecast and Outlook 2026 to 2036
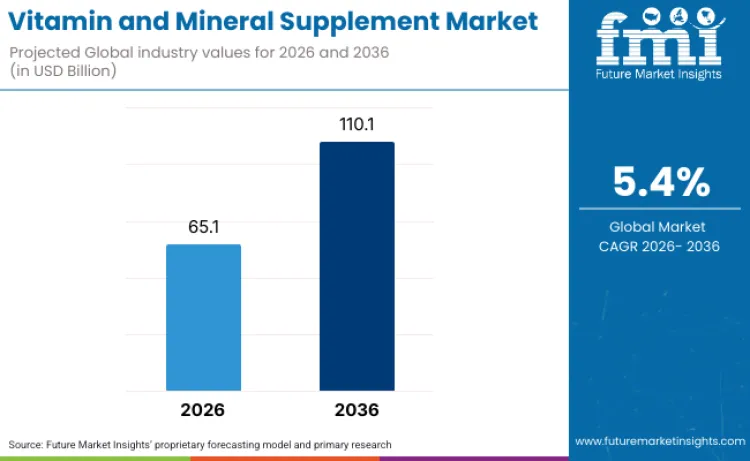
The global vitamin and mineral supplement sector is on track to achieve a valuation of USD 110.1 billion by 2036, accelerating from USD 65.1 billion in 2026 at a CAGR of 5.4%. As per Future Market Insights, expansion is structurally underpinned by aging population demographics where individuals aged 60 and above exhibit higher micronutrient deficiency prevalence requiring supplementation, combined with preventive healthcare awareness driving proactive nutrient optimization before clinical deficiency manifestation.
Nestlé Health Science noted in its 2023 reporting that the company's consumer care business, which includes vitamins, minerals, and supplements, continues experiencing growth driven by consumer focus on preventive health and immunity support, with management highlighting portfolio expansion in science-backed formulations addressing specific health needs, though precise vitamin and mineral segment revenue was not disclosed separately within the broader nutritional health portfolio.
This preventive health momentum compels manufacturers to develop evidence-based formulations supported by clinical research rather than relying solely on nutritional adequacy claims, creating differentiation through therapeutic positioning validated by randomized controlled trials. Simultaneously the regulatory landscape is tightening as authorities including FDA in the United States and EFSA in Europe increase scrutiny of health claims, forcing brands to substantiate efficacy assertions through human clinical studies rather than theoretical nutrient function or animal research extrapolation.
The operational reality for supplement manufacturers is defined by quality control complexity where raw material sourcing, manufacturing processes, and finished product testing must ensure label claim accuracy for micronutrient content while preventing contamination with heavy metals, microbial pathogens, and adulterants. Third-party certification programs including USP Verified, NSF International, and ConsumerLab provide independent validation that builds consumer trust, creating competitive advantages for brands investing in quality assurance beyond minimum regulatory requirements.
Bioavailability optimization through chelation, lipid encapsulation, and co-factor inclusion represents formulation frontiers where manufacturers develop proprietary delivery systems claiming superior absorption compared to generic mineral salts and synthetic vitamins.
Summary of Vitamin and Mineral Supplement Market
What Is Growth Outlook for Vitamin and Mineral Supplement Market as per Future Market Insights Projection?
Future Market Insights projects the vitamin and mineral supplement market to expand at a CAGR of 5.4% from 2026 to 2036, increasing from USD 65.1 Billion in 2026 to USD 110.1 Billion by 2036.
FMI Research Approach: FMI proprietary forecasting model based on aging population demographics, preventive healthcare adoption trajectories, micronutrient deficiency prevalence data, and chronic disease burden requiring therapeutic supplementation support.
Which Country Holds Largest Share in Global Vitamin and Mineral Supplement Market?
The United States holds a significant share supported by dietary supplement industry maturity, widespread physician recommendations for specific deficiencies, and consumer health consciousness driving proactive supplementation beyond minimum nutritional requirements. Dietary supplements market analysis informs micronutrient category growth modeling.
FMI Research Approach: FMI country-level revenue modeling by supplement usage prevalence, aging population demographics, healthcare expenditure patterns, and preventive health awareness indicators.
What Are Globally Unique Trends Shaping Vitamin and Mineral Supplement Market?
Globally unique trends include genetic testing-guided personalized supplement protocols where DNA polymorphism analysis identifies individual methylation deficiencies requiring activated B-vitamin forms, gut microbiome testing informing probiotic-vitamin combinations optimizing nutrient synthesis by beneficial bacteria, and wearable device data integration where continuous glucose monitoring and activity tracking inform dynamic vitamin D and magnesium dosing adjusted based on metabolic demand fluctuations. Nutraceuticals market personalization represents next-generation supplementation approaches.
FMI Research Approach: Personalized nutrition technology tracking and precision supplementation trend analysis examining genetic testing, microbiome assessment, and biomarker-guided dosing adoption.
Vitamin and Mineral Supplement Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 65.1 Billion |
| Industry Value (2036) | USD 110.1 Billion |
| CAGR (2026 to 2036) | 5.4% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Which Factors Propel Adoption of Vitamin and Mineral Supplements?
Population aging serves as the primary catalyst, as individuals aged 60 and above experience reduced nutrient absorption from decreased gastric acid production, diminished intrinsic factor synthesis affecting B12 uptake, and lower vitamin D cutaneous synthesis from reduced skin 7-dehydrocholesterol conversion capacity. World Health Organization data indicates that the global population aged 60 years and older will reach 2.1 billion by 2050, double the 2020 level, creating expanding demographic cohorts requiring supplementation. This structural aging creates persistent demand growth independent of economic cycles or discretionary spending fluctuations, as micronutrient supplementation transitions from optional to medically necessary for maintaining functional capacity and preventing deficiency-related complications including osteoporosis, anemia, and cognitive decline.
Chronic disease burden amplifies adoption as conditions including diabetes, cardiovascular disease, and osteoporosis require nutrient optimization beyond dietary intake alone. Clinical practice guidelines from medical societies including American Diabetes Association and American Heart Association incorporate specific micronutrient recommendations, with vitamin D for bone health in diabetics, magnesium for cardiovascular function, and B-vitamins for homocysteine metabolism receiving professional endorsement. This medical integration converts supplementation from consumer-initiated wellness practice to physician-directed therapeutic adjunct, expanding addressable markets beyond health-conscious individuals to patient populations following clinical protocols.
Dietary pattern insufficiency creates supplementation needs as modern food systems emphasizing processed convenience foods over nutrient-dense whole foods result in inadequate micronutrient intake despite caloric sufficiency. Functional foods market fortification addresses gaps but cannot fully compensate for diverse micronutrient requirements, particularly for minerals including magnesium, zinc, and selenium where soil depletion reduces food content independent of processing.
How is the Vitamin and Mineral Supplement Market Segmented?
The vitamin and mineral supplement market is segmented by product type, form, distribution channel, and region to reflect the evolving pathways through which preventive nutrition and therapeutic supplementation are delivered. By product type, the market is divided into vitamin supplements and mineral say supplements, with vitamin products forming the primary interface for consumers due to higher awareness and clearer deficiency narratives around vitamin D, B12, and folate. By form, demand spans tablets, capsules, powders, liquids, and gummies, capturing both traditional pharmaceutical-style dosing and consumer-friendly formats designed to improve adherence across age groups.
By distribution channel, adoption is led by pharmacies and drug stores, where professional guidance reinforces supplements as health interventions rather than lifestyle add-ons, while online retail and direct-to-consumer platforms are emerging as key access points for personalized and subscription-based regimens. Regionally, growth patterns vary between mature markets focused on regulatory compliance and clinical substantiation, and emerging markets driven by expanding healthcare access and preventive awareness.
FMI analysis suggests that while personalization technologies are accelerating, the market structure remains anchored in standardized, trusted supplement formats, highlighting a dual-track landscape where mass-market products provide scale while bioavailability-optimized and condition-specific formulations provide depth and premium value.
Why Do Vitamin Products Hold Dominant Product Type Share?
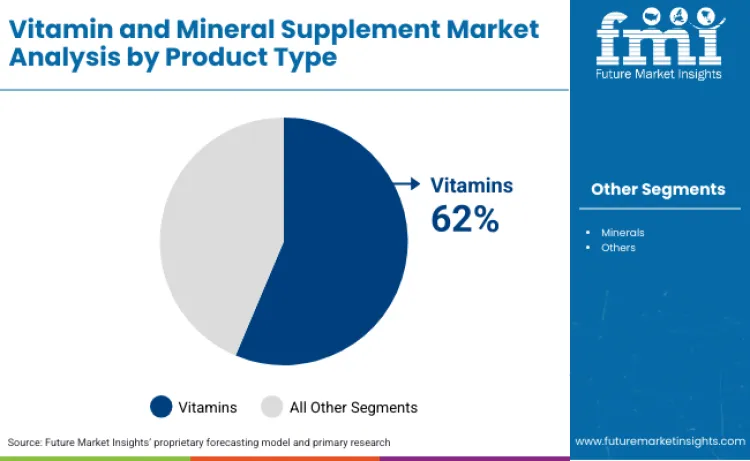
Vitamins maintain 62% product type share due to established medical guidelines recommending supplementation for specific populations, with vitamin D for bone health, B12 for vegetarians and elderly, and folate for pregnant women receiving explicit clinical practice recommendations that drive physician-initiated supplementation. Research published in JAMA demonstrating vitamin D's role in immune function and B-vitamin benefits for cardiovascular health provides clinical evidence supporting supplementation beyond deficiency treatment, expanding indications to preventive health optimization. Consumer awareness of vitamin deficiency symptoms including fatigue, bone pain, and cognitive impairment creates self-initiated supplementation demand, whereas mineral deficiencies often present with non-specific symptoms requiring laboratory testing for identification, reducing consumer-driven purchase without medical guidance.
Why Do Tablets Lead Form Segments?
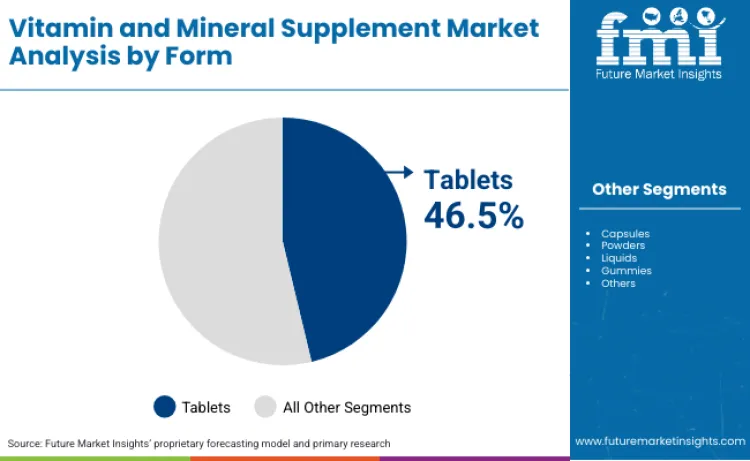
Tablets hold 46.5% form share as the most cost-effective manufacturing format where compression technology enables high-volume production compared to capsules and soft gels, creating retail price advantages important for price-sensitive consumers purchasing daily supplements. The format's stability advantages where compressed tablets resist moisture and oxidation better than capsules and powders extend shelf life to 24-36 months, reducing waste and enabling bulk purchasing through warehouse clubs and online subscriptions offering quantity discounts. Consumer familiarity from pharmaceutical tablet experience reduces hesitation versus newer delivery formats including sublingual strips or liposomal liquids requiring usage instruction and format adaptation.
Why Do Pharmacies Dominate Distribution Channels?
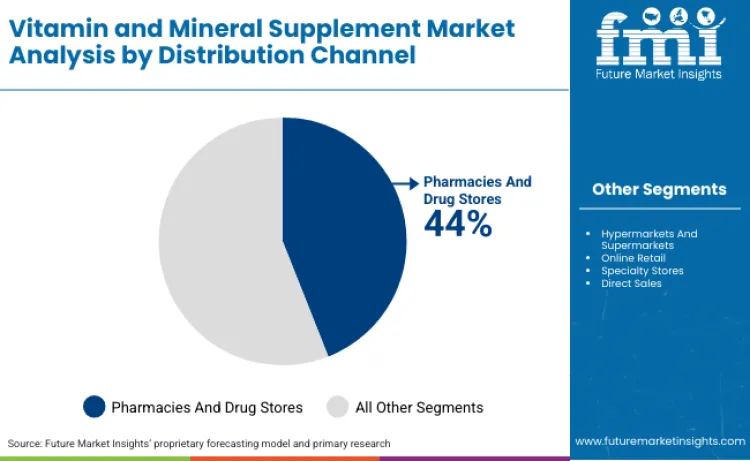
Pharmacies and drug stores hold 44% distribution share as trusted healthcare settings where consumers seek supplement recommendations from pharmacists providing professional guidance on product selection, dosing, and drug-nutrient interaction screening. This consultation environment creates value beyond transactional product sale, with pharmacist expertise addressing consumer uncertainty about appropriate formulation selection among hundreds of options presenting conflicting marketing claims. Pharmacy placement adjacent to prescription medications reinforces supplement perception as health intervention rather than lifestyle product, supporting premium pricing and clinical positioning. Integration with prescription fulfillment enables cross-promotion where pharmacists recommend supplements addressing known deficiency risks from specific medications, including magnesium for diuretic users and vitamin K2 for anticoagulant therapy patients.
Why Is Targeted, Condition-Specific Supplementation Reshaping the Market?
Consumer behavior is shifting decisively away from broad multivitamins toward targeted single-nutrient and condition-specific formulations that address clearly defined health needs. Supplement users increasingly prefer personalized stacks focused on nutrients they actively require, rather than one-size-fits-all products containing unnecessary components. This disaggregation is creating space for specialized brands that compete on bioavailability, dosing precision, and therapeutic relevance instead of marketing scale. As a result, differentiation is moving from label breadth to formulation depth, repositioning supplements from general nutritional insurance into focused health interventions designed to support immunity, cardiovascular health, cognition, or metabolic function.
How Is Clinical Evidence Investment Changing Competitive Dynamics?
The industry is pivoting toward evidence-backed positioning as manufacturers invest heavily in clinical programs that validate supplement efficacy beyond basic nutritional adequacy. Brands are prioritizing human data to support claims around specific health outcomes, which is reshaping recommendation behavior among healthcare professionals. Supplements supported by rigorous clinical validation are increasingly favored by physicians, pharmacists, and integrative practitioners over products relying on theoretical nutrient functions. This emphasis on substantiation is opening institutional distribution pathways, including hospital pharmacies and wellness clinics, and raising entry barriers for brands that lack the resources or commitment to generate high-quality human evidence.
What Role Does Digital Integration Play in Sustaining Adoption?
Technology integration is emerging as a critical enabler of long-term supplement adherence. Smartphone applications that track intake, monitor symptom changes, and deliver reminder prompts are addressing the industry’s core challenge of inconsistent usage. By improving adherence, these tools increase the likelihood that consumers experience tangible benefits, reinforcing repeat purchasing behavior. Digital platforms also support subscription-based models that convert episodic supplement use into sustained regimens, strengthening customer lifetime value. As health and wellness digitalization advances, technology is becoming a central mechanism through which supplement brands drive engagement, consistency, and recurring revenue.
How Will Vitamin and Mineral Supplement Market Expansion Unfold Across Key Global Regions?
The vitamin and mineral supplement market is poised for significant expansion with varying growth rates projected from 2026 to 2036. India leads with 8.6% CAGR driven by expanding middle-class healthcare access, rising chronic disease awareness, and increasing physician recommendations for nutritional supplementation. China follows at 6.6% benefiting from aging population, urban lifestyle dietary inadequacies, and government health initiatives promoting preventive nutrition. Brazil records 6.3% reflecting healthcare system improvements and growing consumer health consciousness. The United Kingdom anticipates 5.5% growth from National Health Service nutritional guidance and pharmacy-channel professional recommendations. The United States at 5.4% maintains substantial market scale through established supplement culture and insurance coverage for specific deficiencies. Germany at 4.4% shows moderate growth from mature market saturation and stringent regulatory requirements limiting new product innovation velocity.
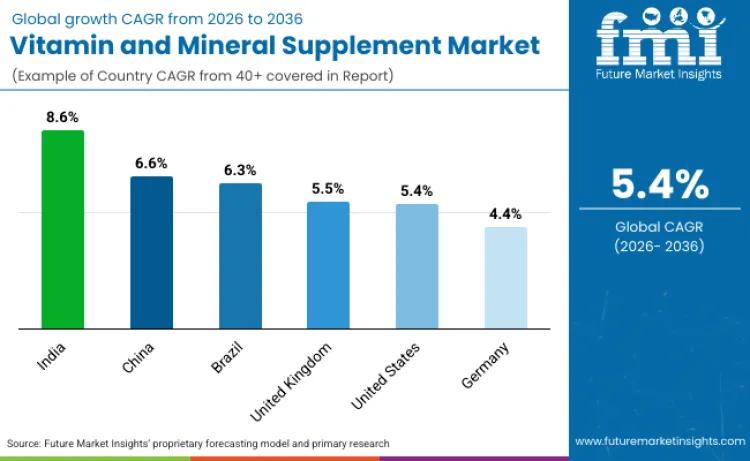
| Country | CAGR (2026-2036) |
|---|---|
| India | 8.6% |
| China | 6.6% |
| Brazil | 6.3% |
| United Kingdom | 5.5% |
| United States | 5.4% |
| Germany | 4.4% |
Source: Future Market Insights (FMI) analysis
Why is India Prioritizing Micronutrient Certification?
India is expanding at an 8.6% CAGR, driven by the formalization of nutritional standards and large-scale public health mandates that are converting supplementation from an informal household habit into a regulated health intervention. The Food Safety and Standards Authority of India has highlighted that nearly 80% of the population remains deficient in vitamin D, creating urgency for nationwide fortification and supplementation programs. This structural push is reinforced by the Anemia Mukt Bharat strategy, under which the Ministry of Health reported that 15.4 crore children and adolescents received iron and folic acid supplements in a single quarter of FY 2024 to 2025. Digital health platforms now track real-time anemia screening, supply-chain movement, and compliance metrics, ensuring supplementation outcomes are measurable and auditable. FMI analysis indicates that this shift is redefining micronutrient intake as a state-monitored public health requirement rather than a discretionary wellness choice, with certified volumes expected to rise steadily as rural coverage expands and preventive nutrition becomes embedded into national health delivery systems.
How is China Scaling Preventive Nutrition for an Aging Population?
China is growing at a 6.6% CAGR, anchored in the Healthy China 2030 framework that shifts the healthcare system from reactive treatment toward preventive nutrition and lifestyle management. With near-universal health insurance coverage achieved, the state is incentivizing early nutritional intervention to manage the long-term fiscal burden of population aging. The population aged 60 and above continues to expand rapidly, increasing demand for calcium, vitamin D, and B-vitamin supplementation as tools to prevent osteoporosis, cognitive decline, and metabolic disorders. Government and multilateral projections warn that without preventive reforms, national health spending could approach USD 2.5 trillion by 2035, creating strong policy motivation to scale supplementation. FMI analysis suggests that micronutrients in China are increasingly positioned as cost-containment instruments within primary care pathways, embedding supplements into standardized preventive protocols rather than framing them as optional consumer wellness products.
Why is Brazil Overhauling Supplement Safety Standards?
Brazil’s market is rising at a 6.3% CAGR, shaped by a decisive regulatory overhaul focused on transparency, safety, and consumer protection. In December 2024, the national health surveillance authority updated permitted active substance lists, tightening controls on formulation composition and forcing manufacturers to align products with clearly defined safety parameters. This reform is reinforced by broader revisions to nutritional and front-of-pack labeling rules during 2024 to 2025, requiring clearer disclosure to support informed and safe consumption. The result is a market environment where clinical validation and regulatory compliance are becoming prerequisites for scale. FMI analysis indicates that this clarity is positioning Brazil as a hub for evidence-led supplementation in Latin America, attracting brands willing to invest in documentation, testing, and professional endorsement. As trust improves, supplementation is moving beyond niche wellness use toward mainstream preventive healthcare adoption across urban and semi-urban populations.
How is the United States Market Shifting Toward Clinical Stewardship?
The United States is advancing at a 5.4% CAGR, characterized by a transition from consumer-driven supplement choice toward scientific accountability and clinical stewardship. The NIH Office of Dietary Supplements’ 2025-2029 strategic plan emphasizes funding, coordination, and dissemination of high-quality scientific research to ensure public confidence in supplement efficacy and safety. This policy direction elevates evidence generation as a competitive requirement, with brands increasingly expected to substantiate claims through human clinical data rather than marketing narratives alone. Physician recommendations, insurance-linked deficiency management, and integration with preventive care protocols are becoming central demand drivers. FMI analysis suggests that supplementation in the U.S. is being repositioned as a clinically aligned intervention, where quality assurance, third-party testing, and research-backed formulations determine long-term success in an increasingly scrutinized market.
Why is the United Kingdom Focusing on Targeted “Pharmacy-First” Guidance?
The United Kingdom is projected to grow at a 5.5% CAGR, driven by a pharmacy-first model that channels supplementation through professional guidance rather than mass retail promotion. Updated NHS protocols in 2025 explicitly recommend year-round vitamin D supplementation for adults with defined risk factors, embedding supplements into standardized clinical advice. Pharmacies act as the primary gatekeepers, translating national guidance into targeted recommendations based on age, lifestyle, and health status. This structure reinforces supplements as preventive health tools rather than discretionary consumer goods. FMI analysis indicates that this protocol-driven approach is sustaining steady growth by aligning supplementation with public health messaging, professional trust, and repeat purchasing through pharmacy networks rather than relying on aggressive consumer marketing.
Why Does Germany Show Moderate but Stable Growth?
Germany is expanding at a 4.4% CAGR, reflecting a mature supplement market shaped by high baseline usage and stringent regulatory oversight. Strict EU health-claim substantiation requirements limit rapid product proliferation, favoring established, pharmacy-distributed brands with strong quality credentials and documented efficacy. German consumers demonstrate high trust in pharmacist-led recommendations, reinforcing premium positioning but constraining volume-led expansion. Reimbursement restrictions for non-prescription supplementation further moderate growth compared to less regulated markets. FMI analysis indicates that Germany’s trajectory will remain steady rather than rapid, supported by demographic aging and persistent demand for regulated, evidence-backed micronutrient solutions rather than experimental or trend-driven formulations.
What Strategic Moves Are Defining Competitive Landscape?
The competitive landscape experiences consolidation as pharmaceutical companies acquire supplement brands to expand consumer health portfolios, with recent transactions including GSK's split creating Haleon focusing on consumer healthcare including supplements and vitamins. These integrations leverage pharmaceutical companies' clinical research capabilities, regulatory expertise, and healthcare professional relationships to elevate supplement credibility through evidence-based positioning validated by medical community endorsement rather than purely consumer marketing.
Personalization platforms are emerging where brands offer customized formulations based on health questionnaires, genetic testing, or blood biomarker analysis, creating tailored supplement regimens addressing individual needs rather than standardized multivitamins. Personalized nutrition market represents next-generation supplementation moving beyond one-size-fits-all toward precision micronutrient delivery optimized for individual physiology.
Quality differentiation strategies focus on third-party testing verification where brands pursue USP Verified, NSF Certified, or Informed-Sport approval to substantiate label claim accuracy and contaminant absence, creating competitive advantages over untested products where quality remains unverified.
Recent Developments:
- In January 2024, Nestlé Health Science expanded its Garden of Life brand with bioavailability-enhanced mineral formulations using chelation technology for improved absorption.
- In September 2024, Bayer AG introduced vitamin D3-K2 combination specifically formulated for cardiovascular health based on clinical research published in cardiovascular journals.
Key Players in Vitamin and Mineral Supplement Market
- Nestlé Health Science S.A.
- Abbott Laboratories
- Haleon plc
- Bayer AG
- Amway (Nutrilite)
- Herbalife Nutrition Ltd.
- Nature's Bounty Co.
- DSM-Firmenich AG
- Blackmores Limited
- Pfizer Inc. (Consumer Health)
Market Definition
The vitamin and mineral supplement market represents revenue generated from over-the-counter dietary supplements containing essential micronutrients including fat-soluble vitamins (A, D, E, K), water-soluble vitamins (B-complex, C), and minerals (calcium, magnesium, iron, zinc, selenium) formulated as single-nutrient or combination products for nutritional supplementation addressing dietary inadequacy, clinical deficiency, or therapeutic health optimization. The market measures retail sales value of supplements sold through pharmacies, supermarkets, online channels, and specialty stores to consumers seeking nutritional support beyond dietary intake.
Inclusions cover vitamin products including vitamin D for bone health, B-complex for energy metabolism, vitamin C for immune support, and multivitamins combining multiple nutrients. Mineral supplements encompass calcium for bone density, magnesium for cardiovascular function, iron for anemia prevention, zinc for immune health, and multimineral formulations. Form categories include tablets, capsules, powders, liquids, and gummies. Distribution channels encompass pharmacies and drug stores providing professional consultation, hypermarkets and supermarkets offering convenience, and online platforms enabling subscription services. End users include elderly populations, pregnant women, vegetarians and vegans, athletes, and general wellness-focused consumers.
Exclusions include prescription vitamin formulations requiring medical oversight. Herbal supplements, amino acids, and specialty nutrients beyond vitamins and minerals fall outside scope. Functional foods and fortified beverages containing added vitamins represent separate categories. Medical foods for disease-specific management are excluded. Infant formula and pediatric-specific supplements constitute distinct markets. Veterinary supplements for animal health are not included.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Product Type | Vitamins (Vitamin D, B-Complex, Vitamin C, Multivitamins); Minerals (Calcium, Magnesium, Iron, Zinc, Multiminerals) |
| Form | Tablets; Capsules; Powders; Liquids; Gummies; Others |
| Distribution Channel | Pharmacies & Drug Stores; Hypermarkets & Supermarkets; Online Retail; Specialty Stores; Direct Sales |
| Target Population Segments | Elderly Population; Pregnant & Lactating Women; Vegetarians & Vegans; Athletes; Chronic Disease Patients; General Wellness Consumers |
| Formulation & Technology Focus | Bioavailability-Optimized Mineral Chelates; Liposomal & Encapsulated Vitamins; Co-Factor Enhanced Absorption Systems; Personalized Micronutrient Stacks; Clinically Validated Therapeutic Dosing Protocols |
| Regions Covered | North America; Europe; East Asia; South Asia; Latin America; Middle East & Africa |
| Countries Covered | United States; China; India; Brazil; United Kingdom; Germany; Japan; South Korea; France; and 40+ additional countries |
| Key Companies Profiled | Nestlé Health Science; Abbott Laboratories; Haleon plc; Bayer AG; Amway (Nutrilite); Herbalife Nutrition; Nature’s Bounty; DSM-Firmenich; Blackmores Limited; Pfizer Consumer Health |
| Additional Attributes | Dollar sales by product type, form, and channel; clinical substantiation trends for micronutrient efficacy; bioavailability performance benchmarking; quality certification impact (USP, NSF, third-party verification); personalization platform adoption; subscription-based regimen growth; regulatory influence on health claims and formulation standards |
Vitamin and Mineral Supplement Market by Segments
By Product Type:
- Vitamins
- Minerals
By Form:
- Tablets
- Capsules
- Powders
- Liquids
- Gummies
- Others
By Distribution Channel:
- Pharmacies And Drug Stores
- Hypermarkets And Supermarkets
- Online Retail
- Specialty Stores
- Direct Sales
By Region:
- North America
- Europe
- East Asia
- South Asia
- Latin America
- Middle East & Africa
Bibliography
- Department of Empowerment of Persons with Disabilities. (2024, March 14). Latest notified guidelines for assessing the extent of specified disabilities. Ministry of Social Justice and Empowerment, Government of India.
- Food and Drug Administration. (2024). 510(k) Premarket notification: EarliPoint evaluation for autism spectrum disorder. USA Department of Health and Human Services.
- Instituto Brasileiro de Geografia e Estatística. (2024). Censo Demográfico 2022: Questionários e manuais. Ministério do Planejamento e Orçamento.
Frequently Asked Questions
What is the current global market size for Vitamin and Mineral Supplements?
The global market is valued at USD 65.1 billion in 2026, driven by aging populations, preventive healthcare awareness, and rising chronic disease-related nutrient deficiencies.
What is the projected CAGR for the Vitamin and Mineral Supplement market?
Sales are projected to increase at a 5.4% CAGR from 2026 to 2036, reflecting steady integration of supplements into therapeutic and preventive health routines.
Which segment currently dominates the industry?
Vitamin products lead with a 62% share, while tablets dominate formulation formats with 46.5% of total consumption.
What are the primary drivers for adoption in emerging markets?
Expanding healthcare access, rising chronic disease prevalence, and growing physician-recommended supplementation in India, China, and Brazil are the main growth engines.
Who are the leading players in the Vitamin and Mineral Supplement space?
Nestlé Health Science, Abbott Laboratories, Haleon plc, Bayer AG, Amway, Herbalife Nutrition, and Nature’s Bounty lead the global competitive landscape.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand Side Trends
- Supply Side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Knowledge Base and Historical Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter's Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Y-o-Y Growth Trend Analysis 2021 to 2025
- Absolute $ Opportunity Analysis 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type, 2026 to 2036
- Vitamin Supplements
- Mineral Supplements
- Y-o-Y Growth Trend Analysis By Product Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Form
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Form, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Form, 2026 to 2036
- Tablets
- Capsules
- Powders
- Liquids
- Gummies
- Others
- Y-o-Y Growth Trend Analysis By Form, 2021 to 2025
- Absolute $ Opportunity Analysis By Form, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Pharmacies and Drug Stores
- Hypermarkets and Supermarkets
- Online Retail
- Specialty Stores
- Direct Sales
- Y-o-Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Form
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Form
- By Distribution Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Argentina
- Chile
- Rest of Latin America
- By Product Type
- By Form
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Form
- By Distribution Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- France
- Italy
- Spain
- BENELUX
- Rest of Western Europe
- By Product Type
- By Form
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Form
- By Distribution Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Form
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Form
- By Distribution Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Form
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Form
- By Distribution Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- Australia
- Southeast Asia
- Rest of South Asia and Pacific
- By Product Type
- By Form
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Form
- By Distribution Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Form
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Form
- By Distribution Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Distribution Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Distribution Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Distribution Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Distribution Channel
- Argentina
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Distribution Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Distribution Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Distribution Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Distribution Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Distribution Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Distribution Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Distribution Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Distribution Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Distribution Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Distribution Channel
- Australia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Distribution Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Distribution Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Distribution Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Distribution Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Distribution Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Form
- By Distribution Channel
- Competition Analysis
- Competition Deep Dive
- Nestlé Health Science S.A.
- Abbott Laboratories
- Haleon plc
- Bayer AG
- Amway (Nutrilite)
- Herbalife Nutrition Ltd.
- Nature's Bounty Co.
- DSM-Firmenich AG
- Blackmores Limited
- Pfizer Inc. (Consumer Health)
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Vitamin and Mineral Supplement Market Key Takeaways
- Global Vitamin and Mineral Supplement Market Size (USD Million), 2021-2036
- Global Vitamin and Mineral Supplement Market Y-o-Y Growth (%), 2021-2036
- Global Vitamin and Mineral Supplement Market Absolute $ Opportunity Analysis (USD Million), 2026-2036
- Global Vitamin and Mineral Supplement Market Pricing Analysis (USD/Unit), 2021-2036
- Global Vitamin and Mineral Supplement Market Size (USD Million) By Product Type, 2021-2036
- Global Vitamin and Mineral Supplement Market Size (USD Million) By Form, 2021-2036
- Global Vitamin and Mineral Supplement Market Size (USD Million) By Distribution Channel, 2021-2036
- Global Vitamin and Mineral Supplement Market Size (USD Million) By Region, 2021-2036
- North America Vitamin and Mineral Supplement Market Size (USD Million) By Country, 2021-2036
- North America Vitamin and Mineral Supplement Market Size (USD Million) By Product Type, 2021-2036
- North America Vitamin and Mineral Supplement Market Size (USD Million) By Form, 2021-2036
- North America Vitamin and Mineral Supplement Market Size (USD Million) By Distribution Channel, 2021-2036
- North America Vitamin and Mineral Supplement Market Attractiveness Analysis By Country
- North America Vitamin and Mineral Supplement Market Attractiveness Analysis By Product Type
- North America Vitamin and Mineral Supplement Market Attractiveness Analysis By Form
- North America Vitamin and Mineral Supplement Market Attractiveness Analysis By Distribution Channel
- Latin America Vitamin and Mineral Supplement Market Size (USD Million) By Country, 2021-2036
- Latin America Vitamin and Mineral Supplement Market Size (USD Million) By Product Type, 2021-2036
- Latin America Vitamin and Mineral Supplement Market Size (USD Million) By Form, 2021-2036
- Latin America Vitamin and Mineral Supplement Market Size (USD Million) By Distribution Channel, 2021-2036
- Latin America Vitamin and Mineral Supplement Market Attractiveness Analysis By Country
- Latin America Vitamin and Mineral Supplement Market Attractiveness Analysis By Product Type
- Latin America Vitamin and Mineral Supplement Market Attractiveness Analysis By Form
- Latin America Vitamin and Mineral Supplement Market Attractiveness Analysis By Distribution Channel
- Western Europe Vitamin and Mineral Supplement Market Size (USD Million) By Country, 2021-2036
- Western Europe Vitamin and Mineral Supplement Market Size (USD Million) By Product Type, 2021-2036
- Western Europe Vitamin and Mineral Supplement Market Size (USD Million) By Form, 2021-2036
- Western Europe Vitamin and Mineral Supplement Market Size (USD Million) By Distribution Channel, 2021-2036
- Western Europe Vitamin and Mineral Supplement Market Attractiveness Analysis By Country
- Western Europe Vitamin and Mineral Supplement Market Attractiveness Analysis By Product Type
- Western Europe Vitamin and Mineral Supplement Market Attractiveness Analysis By Form
- Western Europe Vitamin and Mineral Supplement Market Attractiveness Analysis By Distribution Channel
- Eastern Europe Vitamin and Mineral Supplement Market Size (USD Million) By Country, 2021-2036
- Eastern Europe Vitamin and Mineral Supplement Market Size (USD Million) By Product Type, 2021-2036
- Eastern Europe Vitamin and Mineral Supplement Market Size (USD Million) By Form, 2021-2036
- Eastern Europe Vitamin and Mineral Supplement Market Size (USD Million) By Distribution Channel, 2021-2036
- Eastern Europe Vitamin and Mineral Supplement Market Attractiveness Analysis By Country
- Eastern Europe Vitamin and Mineral Supplement Market Attractiveness Analysis By Product Type
- Eastern Europe Vitamin and Mineral Supplement Market Attractiveness Analysis By Form
- Eastern Europe Vitamin and Mineral Supplement Market Attractiveness Analysis By Distribution Channel
- East Asia Vitamin and Mineral Supplement Market Size (USD Million) By Country, 2021-2036
- East Asia Vitamin and Mineral Supplement Market Size (USD Million) By Product Type, 2021-2036
- East Asia Vitamin and Mineral Supplement Market Size (USD Million) By Form, 2021-2036
- East Asia Vitamin and Mineral Supplement Market Size (USD Million) By Distribution Channel, 2021-2036
- East Asia Vitamin and Mineral Supplement Market Attractiveness Analysis By Country
- East Asia Vitamin and Mineral Supplement Market Attractiveness Analysis By Product Type
- East Asia Vitamin and Mineral Supplement Market Attractiveness Analysis By Form
- East Asia Vitamin and Mineral Supplement Market Attractiveness Analysis By Distribution Channel
- South Asia and Pacific Vitamin and Mineral Supplement Market Size (USD Million) By Country, 2021-2036
- South Asia and Pacific Vitamin and Mineral Supplement Market Size (USD Million) By Product Type, 2021-2036
- South Asia and Pacific Vitamin and Mineral Supplement Market Size (USD Million) By Form, 2021-2036
- South Asia and Pacific Vitamin and Mineral Supplement Market Size (USD Million) By Distribution Channel, 2021-2036
- South Asia and Pacific Vitamin and Mineral Supplement Market Attractiveness Analysis By Country
- South Asia and Pacific Vitamin and Mineral Supplement Market Attractiveness Analysis By Product Type
- South Asia and Pacific Vitamin and Mineral Supplement Market Attractiveness Analysis By Form
- South Asia and Pacific Vitamin and Mineral Supplement Market Attractiveness Analysis By Distribution Channel
- Middle East & Africa Vitamin and Mineral Supplement Market Size (USD Million) By Country, 2021-2036
- Middle East & Africa Vitamin and Mineral Supplement Market Size (USD Million) By Product Type, 2021-2036
- Middle East & Africa Vitamin and Mineral Supplement Market Size (USD Million) By Form, 2021-2036
- Middle East & Africa Vitamin and Mineral Supplement Market Size (USD Million) By Distribution Channel, 2021-2036
- Middle East & Africa Vitamin and Mineral Supplement Market Attractiveness Analysis By Country
- Middle East & Africa Vitamin and Mineral Supplement Market Attractiveness Analysis By Product Type
- Middle East & Africa Vitamin and Mineral Supplement Market Attractiveness Analysis By Form
- Middle East & Africa Vitamin and Mineral Supplement Market Attractiveness Analysis By Distribution Channel
- USA Vitamin and Mineral Supplement Pricing Analysis (USD/Unit), 2021-2036
- USA Vitamin and Mineral Supplement Market Share Analysis (%) By Product Type, 2025
- USA Vitamin and Mineral Supplement Market Share Analysis (%) By Form, 2025
- USA Vitamin and Mineral Supplement Market Share Analysis (%) By Distribution Channel, 2025
- Canada Vitamin and Mineral Supplement Pricing Analysis (USD/Unit), 2021-2036
- Canada Vitamin and Mineral Supplement Market Share Analysis (%) By Product Type, 2025
- Canada Vitamin and Mineral Supplement Market Share Analysis (%) By Form, 2025
- Canada Vitamin and Mineral Supplement Market Share Analysis (%) By Distribution Channel, 2025
- Mexico Vitamin and Mineral Supplement Pricing Analysis (USD/Unit), 2021-2036
- Mexico Vitamin and Mineral Supplement Market Share Analysis (%) By Product Type, 2025
- Mexico Vitamin and Mineral Supplement Market Share Analysis (%) By Form, 2025
- Mexico Vitamin and Mineral Supplement Market Share Analysis (%) By Distribution Channel, 2025
- Brazil Vitamin and Mineral Supplement Pricing Analysis (USD/Unit), 2021-2036
- Brazil Vitamin and Mineral Supplement Market Share Analysis (%) By Product Type, 2025
- Brazil Vitamin and Mineral Supplement Market Share Analysis (%) By Form, 2025
- Brazil Vitamin and Mineral Supplement Market Share Analysis (%) By Distribution Channel, 2025
- Argentina Vitamin and Mineral Supplement Pricing Analysis (USD/Unit), 2021-2036
- Argentina Vitamin and Mineral Supplement Market Share Analysis (%) By Product Type, 2025
- Argentina Vitamin and Mineral Supplement Market Share Analysis (%) By Form, 2025
- Argentina Vitamin and Mineral Supplement Market Share Analysis (%) By Distribution Channel, 2025
- Germany Vitamin and Mineral Supplement Pricing Analysis (USD/Unit), 2021-2036
- Germany Vitamin and Mineral Supplement Market Share Analysis (%) By Product Type, 2025
- Germany Vitamin and Mineral Supplement Market Share Analysis (%) By Form, 2025
- Germany Vitamin and Mineral Supplement Market Share Analysis (%) By Distribution Channel, 2025
- UK Vitamin and Mineral Supplement Pricing Analysis (USD/Unit), 2021-2036
- UK Vitamin and Mineral Supplement Market Share Analysis (%) By Product Type, 2025
- UK Vitamin and Mineral Supplement Market Share Analysis (%) By Form, 2025
- UK Vitamin and Mineral Supplement Market Share Analysis (%) By Distribution Channel, 2025
- France Vitamin and Mineral Supplement Pricing Analysis (USD/Unit), 2021-2036
- France Vitamin and Mineral Supplement Market Share Analysis (%) By Product Type, 2025
- France Vitamin and Mineral Supplement Market Share Analysis (%) By Form, 2025
- France Vitamin and Mineral Supplement Market Share Analysis (%) By Distribution Channel, 2025
- Italy Vitamin and Mineral Supplement Pricing Analysis (USD/Unit), 2021-2036
- Italy Vitamin and Mineral Supplement Market Share Analysis (%) By Product Type, 2025
- Italy Vitamin and Mineral Supplement Market Share Analysis (%) By Form, 2025
- Italy Vitamin and Mineral Supplement Market Share Analysis (%) By Distribution Channel, 2025
- Spain Vitamin and Mineral Supplement Pricing Analysis (USD/Unit), 2021-2036
- Spain Vitamin and Mineral Supplement Market Share Analysis (%) By Product Type, 2025
- Spain Vitamin and Mineral Supplement Market Share Analysis (%) By Form, 2025
- Spain Vitamin and Mineral Supplement Market Share Analysis (%) By Distribution Channel, 2025
- China Vitamin and Mineral Supplement Pricing Analysis (USD/Unit), 2021-2036
- China Vitamin and Mineral Supplement Market Share Analysis (%) By Product Type, 2025
- China Vitamin and Mineral Supplement Market Share Analysis (%) By Form, 2025
- China Vitamin and Mineral Supplement Market Share Analysis (%) By Distribution Channel, 2025
- Japan Vitamin and Mineral Supplement Pricing Analysis (USD/Unit), 2021-2036
- Japan Vitamin and Mineral Supplement Market Share Analysis (%) By Product Type, 2025
- Japan Vitamin and Mineral Supplement Market Share Analysis (%) By Form, 2025
- Japan Vitamin and Mineral Supplement Market Share Analysis (%) By Distribution Channel, 2025
- South Korea Vitamin and Mineral Supplement Pricing Analysis (USD/Unit), 2021-2036
- South Korea Vitamin and Mineral Supplement Market Share Analysis (%) By Product Type, 2025
- South Korea Vitamin and Mineral Supplement Market Share Analysis (%) By Form, 2025
- South Korea Vitamin and Mineral Supplement Market Share Analysis (%) By Distribution Channel, 2025
- India Vitamin and Mineral Supplement Pricing Analysis (USD/Unit), 2021-2036
- India Vitamin and Mineral Supplement Market Share Analysis (%) By Product Type, 2025
- India Vitamin and Mineral Supplement Market Share Analysis (%) By Form, 2025
- India Vitamin and Mineral Supplement Market Share Analysis (%) By Distribution Channel, 2025
- Australia Vitamin and Mineral Supplement Pricing Analysis (USD/Unit), 2021-2036
- Australia Vitamin and Mineral Supplement Market Share Analysis (%) By Product Type, 2025
- Australia Vitamin and Mineral Supplement Market Share Analysis (%) By Form, 2025
- Australia Vitamin and Mineral Supplement Market Share Analysis (%) By Distribution Channel, 2025
- Russia Vitamin and Mineral Supplement Pricing Analysis (USD/Unit), 2021-2036
- Russia Vitamin and Mineral Supplement Market Share Analysis (%) By Product Type, 2025
- Russia Vitamin and Mineral Supplement Market Share Analysis (%) By Form, 2025
- Russia Vitamin and Mineral Supplement Market Share Analysis (%) By Distribution Channel, 2025
- Poland Vitamin and Mineral Supplement Pricing Analysis (USD/Unit), 2021-2036
- Poland Vitamin and Mineral Supplement Market Share Analysis (%) By Product Type, 2025
- Poland Vitamin and Mineral Supplement Market Share Analysis (%) By Form, 2025
- Poland Vitamin and Mineral Supplement Market Share Analysis (%) By Distribution Channel, 2025
- Kingdom of Saudi Arabia Vitamin and Mineral Supplement Pricing Analysis (USD/Unit), 2021-2036
- Kingdom of Saudi Arabia Vitamin and Mineral Supplement Market Share Analysis (%) By Product Type, 2025
- Kingdom of Saudi Arabia Vitamin and Mineral Supplement Market Share Analysis (%) By Form, 2025
- Kingdom of Saudi Arabia Vitamin and Mineral Supplement Market Share Analysis (%) By Distribution Channel, 2025
- South Africa Vitamin and Mineral Supplement Pricing Analysis (USD/Unit), 2021-2036
- South Africa Vitamin and Mineral Supplement Market Share Analysis (%) By Product Type, 2025
- South Africa Vitamin and Mineral Supplement Market Share Analysis (%) By Form, 2025
- South Africa Vitamin and Mineral Supplement Market Share Analysis (%) By Distribution Channel, 2025
- Vitamin and Mineral Supplement Market Competition Dashboard
- Vitamin and Mineral Supplement Market Competition Benchmarking Analysis
- Vitamin and Mineral Supplement Market Share Analysis of Top Players By Regional, 2025
- Vitamin and Mineral Supplement Market Share Analysis of Top Players By Product Type, 2025
- Vitamin and Mineral Supplement Market Share Analysis of Top Players By Form, 2025
- Vitamin and Mineral Supplement Market Share Analysis of Top Players By Distribution Channel, 2025
- Nestlé Health Science S.A. - Company Profile
- Abbott Laboratories - Company Profile
- Haleon plc - Company Profile
- Bayer AG - Company Profile
- Amway (Nutrilite) - Company Profile
- Herbalife Nutrition Ltd. - Company Profile
- Nature's Bounty Co. - Company Profile
- DSM-Firmenich AG - Company Profile
- Blackmores Limited - Company Profile
- Pfizer Inc. (Consumer Health) - Company Profile
- Assumptions & Acronyms Used in Vitamin and Mineral Supplement Market Report
List of Figures
- Vitamin and Mineral Supplement Market Size (USD Million), 2021-2036
- Vitamin and Mineral Supplement Market Y-o-Y Growth (%), 2021-2036
- Vitamin and Mineral Supplement Market Absolute $ Opportunity Analysis (USD Million), 2026-2036
- Vitamin and Mineral Supplement Market Value Chain Analysis
- Vitamin and Mineral Supplement Market Supply Chain Analysis
- Vitamin and Mineral Supplement Market Investment Feasibility Matrix
- Vitamin and Mineral Supplement Market PESTLE Analysis
- Vitamin and Mineral Supplement Market Porter's Five Forces Analysis
- Vitamin and Mineral Supplement Market Product Life Cycle Analysis
- Vitamin and Mineral Supplement Market Opportunity Map Analysis
- Vitamin and Mineral Supplement Market Scenario Forecast Analysis
- Vitamin and Mineral Supplement Market Production and Consumption Statistics
- Vitamin and Mineral Supplement Market Import and Export Statistics
- Global Vitamin and Mineral Supplement Market Share (%) By Product Type, 2025
- Global Vitamin and Mineral Supplement Market Share (%) By Product Type, 2036
- Global Vitamin and Mineral Supplement Market Share (%) By Form, 2025
- Global Vitamin and Mineral Supplement Market Share (%) By Form, 2036
- Global Vitamin and Mineral Supplement Market Share (%) By Distribution Channel, 2025
- Global Vitamin and Mineral Supplement Market Share (%) By Distribution Channel, 2036
- Global Vitamin and Mineral Supplement Market Share (%) By Region, 2025
- Global Vitamin and Mineral Supplement Market Share (%) By Region, 2036
- Global Vitamin and Mineral Supplement Market Attractiveness Analysis By Region
- North America Vitamin and Mineral Supplement Market Size (USD Million), 2021-2036
- North America Vitamin and Mineral Supplement Market Share (%) By Country, 2025
- North America Vitamin and Mineral Supplement Market Share (%) By Country, 2036
- North America Vitamin and Mineral Supplement Market Share (%) By Product Type, 2025
- North America Vitamin and Mineral Supplement Market Share (%) By Product Type, 2036
- North America Vitamin and Mineral Supplement Market Share (%) By Form, 2025
- North America Vitamin and Mineral Supplement Market Share (%) By Form, 2036
- North America Vitamin and Mineral Supplement Market Share (%) By Distribution Channel, 2025
- North America Vitamin and Mineral Supplement Market Share (%) By Distribution Channel, 2036
- North America Vitamin and Mineral Supplement Market Attractiveness Analysis By Country
- North America Vitamin and Mineral Supplement Market Attractiveness Analysis By Product Type
- North America Vitamin and Mineral Supplement Market Attractiveness Analysis By Form
- North America Vitamin and Mineral Supplement Market Attractiveness Analysis By Distribution Channel
- Latin America Vitamin and Mineral Supplement Market Size (USD Million), 2021-2036
- Latin America Vitamin and Mineral Supplement Market Share (%) By Country, 2025
- Latin America Vitamin and Mineral Supplement Market Share (%) By Country, 2036
- Latin America Vitamin and Mineral Supplement Market Share (%) By Product Type, 2025
- Latin America Vitamin and Mineral Supplement Market Share (%) By Product Type, 2036
- Latin America Vitamin and Mineral Supplement Market Share (%) By Form, 2025
- Latin America Vitamin and Mineral Supplement Market Share (%) By Form, 2036
- Latin America Vitamin and Mineral Supplement Market Share (%) By Distribution Channel, 2025
- Latin America Vitamin and Mineral Supplement Market Share (%) By Distribution Channel, 2036
- Latin America Vitamin and Mineral Supplement Market Attractiveness Analysis By Country
- Latin America Vitamin and Mineral Supplement Market Attractiveness Analysis By Product Type
- Latin America Vitamin and Mineral Supplement Market Attractiveness Analysis By Form
- Latin America Vitamin and Mineral Supplement Market Attractiveness Analysis By Distribution Channel
- Western Europe Vitamin and Mineral Supplement Market Size (USD Million), 2021-2036
- Western Europe Vitamin and Mineral Supplement Market Share (%) By Country, 2025
- Western Europe Vitamin and Mineral Supplement Market Share (%) By Country, 2036
- Western Europe Vitamin and Mineral Supplement Market Share (%) By Product Type, 2025
- Western Europe Vitamin and Mineral Supplement Market Share (%) By Product Type, 2036
- Western Europe Vitamin and Mineral Supplement Market Share (%) By Form, 2025
- Western Europe Vitamin and Mineral Supplement Market Share (%) By Form, 2036
- Western Europe Vitamin and Mineral Supplement Market Share (%) By Distribution Channel, 2025
- Western Europe Vitamin and Mineral Supplement Market Share (%) By Distribution Channel, 2036
- Western Europe Vitamin and Mineral Supplement Market Attractiveness Analysis By Country
- Western Europe Vitamin and Mineral Supplement Market Attractiveness Analysis By Product Type
- Western Europe Vitamin and Mineral Supplement Market Attractiveness Analysis By Form
- Western Europe Vitamin and Mineral Supplement Market Attractiveness Analysis By Distribution Channel
- Eastern Europe Vitamin and Mineral Supplement Market Size (USD Million), 2021-2036
- Eastern Europe Vitamin and Mineral Supplement Market Share (%) By Country, 2025
- Eastern Europe Vitamin and Mineral Supplement Market Share (%) By Country, 2036
- Eastern Europe Vitamin and Mineral Supplement Market Share (%) By Product Type, 2025
- Eastern Europe Vitamin and Mineral Supplement Market Share (%) By Product Type, 2036
- Eastern Europe Vitamin and Mineral Supplement Market Share (%) By Form, 2025
- Eastern Europe Vitamin and Mineral Supplement Market Share (%) By Form, 2036
- Eastern Europe Vitamin and Mineral Supplement Market Share (%) By Distribution Channel, 2025
- Eastern Europe Vitamin and Mineral Supplement Market Share (%) By Distribution Channel, 2036
- Eastern Europe Vitamin and Mineral Supplement Market Attractiveness Analysis By Country
- Eastern Europe Vitamin and Mineral Supplement Market Attractiveness Analysis By Product Type
- Eastern Europe Vitamin and Mineral Supplement Market Attractiveness Analysis By Form
- Eastern Europe Vitamin and Mineral Supplement Market Attractiveness Analysis By Distribution Channel
- East Asia Vitamin and Mineral Supplement Market Size (USD Million), 2021-2036
- East Asia Vitamin and Mineral Supplement Market Share (%) By Country, 2025
- East Asia Vitamin and Mineral Supplement Market Share (%) By Country, 2036
- East Asia Vitamin and Mineral Supplement Market Share (%) By Product Type, 2025
- East Asia Vitamin and Mineral Supplement Market Share (%) By Product Type, 2036
- East Asia Vitamin and Mineral Supplement Market Share (%) By Form, 2025
- East Asia Vitamin and Mineral Supplement Market Share (%) By Form, 2036
- East Asia Vitamin and Mineral Supplement Market Share (%) By Distribution Channel, 2025
- East Asia Vitamin and Mineral Supplement Market Share (%) By Distribution Channel, 2036
- East Asia Vitamin and Mineral Supplement Market Attractiveness Analysis By Country
- East Asia Vitamin and Mineral Supplement Market Attractiveness Analysis By Product Type
- East Asia Vitamin and Mineral Supplement Market Attractiveness Analysis By Form
- East Asia Vitamin and Mineral Supplement Market Attractiveness Analysis By Distribution Channel
- South Asia and Pacific Vitamin and Mineral Supplement Market Size (USD Million), 2021-2036
- South Asia and Pacific Vitamin and Mineral Supplement Market Share (%) By Country, 2025
- South Asia and Pacific Vitamin and Mineral Supplement Market Share (%) By Country, 2036
- South Asia and Pacific Vitamin and Mineral Supplement Market Share (%) By Product Type, 2025
- South Asia and Pacific Vitamin and Mineral Supplement Market Share (%) By Product Type, 2036
- South Asia and Pacific Vitamin and Mineral Supplement Market Share (%) By Form, 2025
- South Asia and Pacific Vitamin and Mineral Supplement Market Share (%) By Form, 2036
- South Asia and Pacific Vitamin and Mineral Supplement Market Share (%) By Distribution Channel, 2025
- South Asia and Pacific Vitamin and Mineral Supplement Market Share (%) By Distribution Channel, 2036
- South Asia and Pacific Vitamin and Mineral Supplement Market Attractiveness Analysis By Country
- South Asia and Pacific Vitamin and Mineral Supplement Market Attractiveness Analysis By Product Type
- South Asia and Pacific Vitamin and Mineral Supplement Market Attractiveness Analysis By Form
- South Asia and Pacific Vitamin and Mineral Supplement Market Attractiveness Analysis By Distribution Channel
- Middle East & Africa Vitamin and Mineral Supplement Market Size (USD Million), 2021-2036
- Middle East & Africa Vitamin and Mineral Supplement Market Share (%) By Country, 2025
- Middle East & Africa Vitamin and Mineral Supplement Market Share (%) By Country, 2036
- Middle East & Africa Vitamin and Mineral Supplement Market Share (%) By Product Type, 2025
- Middle East & Africa Vitamin and Mineral Supplement Market Share (%) By Product Type, 2036
- Middle East & Africa Vitamin and Mineral Supplement Market Share (%) By Form, 2025
- Middle East & Africa Vitamin and Mineral Supplement Market Share (%) By Form, 2036
- Middle East & Africa Vitamin and Mineral Supplement Market Share (%) By Distribution Channel, 2025
- Middle East & Africa Vitamin and Mineral Supplement Market Share (%) By Distribution Channel, 2036
- Middle East & Africa Vitamin and Mineral Supplement Market Attractiveness Analysis By Country
- Middle East & Africa Vitamin and Mineral Supplement Market Attractiveness Analysis By Product Type
- Middle East & Africa Vitamin and Mineral Supplement Market Attractiveness Analysis By Form
- Middle East & Africa Vitamin and Mineral Supplement Market Attractiveness Analysis By Distribution Channel
- USA Vitamin and Mineral Supplement Pricing Trend Analysis (USD/Unit), 2021-2036
- Canada Vitamin and Mineral Supplement Pricing Trend Analysis (USD/Unit), 2021-2036
- Mexico Vitamin and Mineral Supplement Pricing Trend Analysis (USD/Unit), 2021-2036
- Brazil Vitamin and Mineral Supplement Pricing Trend Analysis (USD/Unit), 2021-2036
- Argentina Vitamin and Mineral Supplement Pricing Trend Analysis (USD/Unit), 2021-2036
- Germany Vitamin and Mineral Supplement Pricing Trend Analysis (USD/Unit), 2021-2036
- UK Vitamin and Mineral Supplement Pricing Trend Analysis (USD/Unit), 2021-2036
- France Vitamin and Mineral Supplement Pricing Trend Analysis (USD/Unit), 2021-2036
- Italy Vitamin and Mineral Supplement Pricing Trend Analysis (USD/Unit), 2021-2036
- Spain Vitamin and Mineral Supplement Pricing Trend Analysis (USD/Unit), 2021-2036
- China Vitamin and Mineral Supplement Pricing Trend Analysis (USD/Unit), 2021-2036
- Japan Vitamin and Mineral Supplement Pricing Trend Analysis (USD/Unit), 2021-2036
- South Korea Vitamin and Mineral Supplement Pricing Trend Analysis (USD/Unit), 2021-2036
- India Vitamin and Mineral Supplement Pricing Trend Analysis (USD/Unit), 2021-2036
- Australia Vitamin and Mineral Supplement Pricing Trend Analysis (USD/Unit), 2021-2036
- Russia Vitamin and Mineral Supplement Pricing Trend Analysis (USD/Unit), 2021-2036
- Poland Vitamin and Mineral Supplement Pricing Trend Analysis (USD/Unit), 2021-2036
- Kingdom of Saudi Arabia Vitamin and Mineral Supplement Pricing Trend Analysis (USD/Unit), 2021-2036
- South Africa Vitamin and Mineral Supplement Pricing Trend Analysis (USD/Unit), 2021-2036
- Vitamin and Mineral Supplement Market Competitive Landscape
- Vitamin and Mineral Supplement Market Structure Analysis
- Vitamin and Mineral Supplement Market Competition Dashboard
- Vitamin and Mineral Supplement Market Competition Benchmarking Analysis
- Vitamin and Mineral Supplement Market Share Analysis of Top Players By Regional
- Vitamin and Mineral Supplement Market Share Analysis of Top Players By Product Type
- Vitamin and Mineral Supplement Market Share Analysis of Top Players By Form
- Vitamin and Mineral Supplement Market Share Analysis of Top Players By Distribution Channel
- Nestlé Health Science S.A. - Revenue Analysis (USD Million), 2019-2025
- Abbott Laboratories - Revenue Analysis (USD Million), 2019-2025
- Haleon plc - Revenue Analysis (USD Million), 2019-2025
- Bayer AG - Revenue Analysis (USD Million), 2019-2025
- Amway (Nutrilite) - Revenue Analysis (USD Million), 2019-2025
- Herbalife Nutrition Ltd. - Revenue Analysis (USD Million), 2019-2025
- Nestlé Health Science S.A. - Market Position Analysis
- Abbott Laboratories - Market Position Analysis
- Haleon plc - Market Position Analysis
- Bayer AG - Market Position Analysis
- Amway (Nutrilite) - Market Position Analysis
- Herbalife Nutrition Ltd. - Market Position Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

